DOI:
10.1039/C6RA21713C
(Paper)
RSC Adv., 2016,
6, 115222-115237
Preparation and characterization of a series of porous anion-exchanger chelating fibers and their adsorption behavior with respect to removal of cadmium(II)†
Received
31st August 2016
, Accepted 1st December 2016
First published on 2nd December 2016
Abstract
The aim of the present study was to investigate a series of porous anion-exchanger chelating fibers (PP-g-AA-Am), prepared using polypropylene (PP) for the removal of Cd(II) in non-salt systems and in high-salt complex systems. FTIR, BET, FESEM, and XPS spectra indicated that AA was grafted onto the surface of PP fibers and, subsequently, amino functional groups and amide bonds were introduced by the amide reaction. A batch experimental study indicated that the adsorption of Cd(II) could be well-fitted with the pseudo-second-order kinetic model and the Freundlich isotherm. Thermodynamic studies indicated that the adsorption reaction was a spontaneous and endothermic process. Because of the electrostatic interactions and coordination bonds between Cd(II) and PP-g-AA-Am fibers, the resulting adsorption capacities are as follows: PP-g-AA-EDA (125.34 mg g−1), PP-g-AA-DETA (116.15 mg g−1), PP-g-AA-TETA (93.02 mg g−1) and PP-g-AA-TEPA (81.01 mg g−1). The presence of high-salt NaCl solution, as well as CaCl2, KCl and MgCl2, did not significantly affect the Cd(II) adsorption process, whereas strongly acidic or strongly alkaline conditions were unfavorable. In addition, the reusability of the obtained fibers, without a significant deterioration in performance, was demonstrated with at least five repeated cycles.
1. Introduction
As is well known, water pollution caused by heavy metal ions has resulted in rising worldwide environmental concern.1 These toxic metal ions cannot be biodegraded and tend to accumulate in human bodies once ingested, causing serious health disorders, even at very low concentrations.2 Among the heavy metals, cadmium (Cd), lead (Pb), mercury (Hg), nickel (Ni), copper (Cu), and zinc (Zn) are the most hazardous and are included on the U.S. Environmental Protection Agency's (EPA) list of priority pollutants. Particularly for Cd(II), long-term drinking of water contaminated by Cd(II) can cause serious diseases, such as diarrhea, nausea, muscular cramps, and even cancer.3 Thus, the efficient removal of heavy metal ions released in water has become an increasingly urgent issue and has attracted extensive research.
The current technologies to remove Cd(II) ions from water/wastewater include adsorption, coagulation, membrane filtration, chemical oxidation, photodegradation and electrodialysis.4–8 Each technique has its own features, advantages and application fields. Among all the traditional techniques for removing Cd(II) ions, adsorption has become one of the most promising methods due to its high reaction efficiency, simple operation, absence of secondary pollution and easy regeneration. Regarding the adsorption technique, the application of effective adsorption is critical to guaranteeing the efficiency of water treatment. Several solid materials have been investigated as adsorbents, including active carbons, graphene oxide (GO), mesoporous silica (MS), magnetic nanoparticles, polymeric adsorbents, and nanobiocomposite adsorbents. Wang et al.9 reported two series of activated carbons prepared from Spartina alterniflora and from its anaerobically digested residue by H3PO4 activation under various processing conditions, which were used as adsorbents for the removal of Cd(II) in aqueous solutions. A study by Zhao et al.10 reported that few-layered graphene oxide nanosheets were synthesized from graphite using a modified Hummers method, and were used as sorbents for the removal of Cd(II) and Co(II) ions from large volumes of aqueous solutions. Liu et al.11 reported alkaline deoxygenated graphene oxide (aGO), prepared through alkaline hydrothermal treatment and used it as an adsorbent to remove Cd(II) ions from aqueous solutions for the first time. Li et al.12 designed and synthesized periodic Cd(II) imprinted mesoporous silica (Cd-IMS) for the effective removal of Cd(II) in water through a one-step hydrothermal process, combining the advantages of MCM-41 mesoporous materials and molecularly imprinted polymers. Shan et al.13 designed poly(1-vinylimidazole)-grafted Fe3O4@SiO2 magnetic nanoparticles (FSPV), which were developed to remove both free Cd(II) and nitrilotriacetic acid (NTA)-complexed Cd(II) from water. M. Soleymanzadeh et al.14 reported the synthesis and characterization of a new adsorbent containing nanoscale zero-valent iron particles (NZVI) supported on sineguelas waste (S-NaOH-NZVI) from agricultural biomass, which was developed as a new nanobioadsorbent for the adsorption of inorganic pollution, such as Cd(II) ions. Although nanoparticulate adsorbents have been widely used in the treatment of Cd(II) pollution, several shortcomings, such as being unwieldy, inflexible, and hard to handle, with a slow adsorption rate, limited their further application. It is important to seek new adsorbent materials to rapidly remove Cd(II) ions from water.
In recent years, there has been increasing interest in the removal of heavy metal ions using modified fibers.15–18 M. Monier et al.19 investigated a new chelating PET fiber functionalized with thiosemicarbazide moieties (PET-TSC), which were prepared for the fast removal of Hg(II), Cu(II) and Co(II) from water. Qu et al.20 prepared two types of chitosan-coated cotton fibers (SCCH and RCCH), and they found that the abovementioned two fibers exhibited good affinity and adsorption selectivity for Hg(II) in aqueous solution. Kong et al.21 prepared a type of quaternary ammonium fiber by radiation-induced grafting of DMAEMA onto PP fiber and modifying with 1-bromoalkanes, and the obtained fiber could rapidly remove Cr(VI) ions. Ma et al.22 reported an aminated chelating fiber (AF) with a high adsorption capacity for mercury ions, which was prepared by grafting copolymerization of acrylonitrile onto polypropylene fiber, followed by aminating with the chelating molecule, diethylenetriamine. Wang et al.23 successfully prepared a new chelating fiber (WJN-1) for the fast removal of Cu(II) and Ni(II) from water, by grafting acrylic acid onto waste PET fibers and modifying with amide/amine groups. An equilibrium adsorption assay indicated that the adsorption capacity of WJN-1 for Cu(II) and Ni(II) was higher than that of a commercial ion-exchange fiber (IEXF), which was attributed to its amide/amine groups. Zhang et al.24 reported that a novel anion-exchange fiber with strong basic groups was prepared by grafting styrene onto poly(tetrafluoroethylene) fibers via irradiation. The experimental results also showed that the anion-exchange fibers had excellent mechanical properties and thermal stability at a temperature up to 420 °C. The fibers were stable in acidic, alkaline, and oxidative solutions. The static ion-exchange capacity of the fibers was as high as 6.08 mmol g−1. However, few studies have been carried out to combine the excellent adsorption performance of porous materials with the braced structures of chelating fibers in the field of heavy metal ion abatement.
As a conventional material, polyethylene (PE), polypropylene (PP) and polyethylene-coated polypropylene (PE/PP) fiber is widely used in adsorption and filtration processes. It is an attractive material because of its low cost, good mechanical strength, short transit distance, and chemical/thermal resistance.16,25 As is well known, PP (or PE or PE/PP) fiber structures contain alkane chains, and these groups are hydrophobic; thus, the PP (or PE or PE/PP) fiber was hydrophobic. It was modified with hydrophilic groups (amino/amide groups), which improved the hydrophilicity of PP (or PE or PE/PP) fiber materials. For example, Wang et al.16 reported that the hydrophilic surface of PE-MA-NN could be beneficial to increasing its compatibility with heavy-metal ions in solution, in addition to enhancing the adsorption rate of PE-MA-NN and its capacity for Pb(II). Thus, PP (or PE or PE/PP) fiber has become a potential supporting material for the uptake or separation of organic liquids, organic vapors, metal ions, and water.
In this study, we reported the design and successful synthesis of a series of porous anion-exchanger chelating fibers (PP-g-AA-Am), consisting of a PP skeleton, with a grafted outer layer of amino-functionalized PP fibers. Because polypropylene fiber (PP) is inexpensive, has high chemical resistibility, and endures harsh conditions, the novel anion-exchange adsorbent based on PP fibers was prepared using an electron beam, and then modified with ethylenediamine (EDA) (or diethylenetriamine (DETA), triethylenetetramine (TETA) or tetraethylenepentamine (TEPA)), for effective adsorption and recognition of Cd(II) in non-salt systems and in high-salt complex systems. The adsorption behavior of Cd(II) ions, including the adsorption kinetics, isotherms, and thermodynamics, as well as the effect of contact time, pH, and high-salt solution, on adsorption was studied. A series of novel porous anion-exchanger chelating fibers was proven to remove Cd(II) ions rapidly from wastewater with a high adsorption capacity.
2. Experimental
2.1 Materials
PP fiber was provided by Shijiazhuang Tobacco Center (Shijiazhuang, China). The fiber was washed with acetone and distilled water and dried at 323 K before use. Acrylic acid and ammonium ferric sulfate (polymerization inhibitor) were purchased from Aldrich. Ethylenediamine (EDA, 60.10 g mol−1, AR, 99%), diethylenetriamine (DETA, 103.17 g mol−1, AR, 99%), triethylenetetramine (TETA, 146.23 g mol−1, AR, 99%) and tetraethylenepentamine (TEPA, 189.3 g mol−1, AR, 99%), aluminum chloride (AlCl3·6H2O, 241.43 g mol−1, AR, >98%), sulfuric acid, and ethanol were purchased from Tianjin Guangfu Fine Chemical Research Institute and used without further purification. Cadmium nitrate was obtained from Beijing Chemical Reagent Factory. All chemicals used in this study were of analytical grade and were used without further purification. All solutions were prepared with ultrapure water purified with a Millipore-Q system (18.2 M cm). Ion-exchange fiber (IEF, total ion-exchange capacity of ≥3.0 mmol g−1) was supplied by Changda Company (China), and this fibrous adsorbent was used to remove heavy-metal ions from wastewater.
2.2 Preparation of PP-g-AA-Am fibers
A series of porous adsorbent fibers (PP-g-AA-Am) was synthesized, as described by the following steps, and is shown in Scheme 1. The PP-g-AA-Am fiber preparation procedure can be divided into two stages. The first stage was the modification of PP fiber with carboxyl groups, using high-energy electrons, as described in our previous paper.26 PP fibers containing AA (–COOH) and Am (–NH2) groups were prepared by photoinduced graft polymerization and subsequent chemical modification. First, AA was grafted onto PP fiber by a conventional photoinduced graft polymerization technique. The grafting solution was prepared using 65/35 (v/v)% water/methanol, and the solution contained 10 (v/v)% AA, 0.2 (w/v)% BP, 0.2 M H2SO4, and 5 × 10−3 M FeSO4·7H2O. Thirty milliliters of grafting solution and PP fiber (0.5 g) were placed in a glass tube, nitrogen gas was passed through the solution to eliminate oxygen, and then the glass tube was sealed. Photopolymerization on the PP surface was carried out using a 400 W high-pressure mercury lamp (Miya Electric Co., Korea) at 60 °C. After the grafting reaction, the PP grafted samples were taken out from the grafting solution and extracted with methanol for 10 h in a Soxhlet apparatus to remove the homopolymer, potentially also formed during the reaction, and then dried at 60 °C to constant weight. The degree of grafting was calculated from the weight gain as follows:| | | Degree of grafting (%) = 100(W1 − W0)/W0 | (1) |
where W0 and W1 are the weights of the original PP fiber and the grafted PP fiber, respectively.
 |
| | Scheme 1 Preparation procedure for PP-g-AA-Am fibers. | |
At the second stage, an amination reaction of the modified fibers and porous anion exchanger was performed as follows: the grafted fibers (PP-g-AA) and AlCl3·6H2O with an equal mass ratio (0.5 g![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.0 g) were soaked in 60 mL of EDA, DETA, TETA and TEPA solution, followed by sealing in three-necked flasks with magnetic agitators in an oil bath for 3 h at 120 °C. The resulting PP-g-AA-Am fiber was washed with ethanol and deionized water more than three times and dried in a vacuum oven at room temperature overnight. The products are named as PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA, respectively. The degree of amination was calculated from the weight gain using the following formula:
2.0 g) were soaked in 60 mL of EDA, DETA, TETA and TEPA solution, followed by sealing in three-necked flasks with magnetic agitators in an oil bath for 3 h at 120 °C. The resulting PP-g-AA-Am fiber was washed with ethanol and deionized water more than three times and dried in a vacuum oven at room temperature overnight. The products are named as PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA, respectively. The degree of amination was calculated from the weight gain using the following formula:
| | | Degree of amination (%) = 100(W2 − W1)/M/(W1 − W0)/MAA | (2) |
where
W2 is the weight after the amination reaction, and the figures
M and
MAA are the molecular weights of EDA (or DETA, TETA or TEPA) and AA, respectively.
2.3 Characterization
Field emission scanning electron microscopy (FESEM) was used to analyze the surface morphologies of fibers. The measurements were performed using a Hitachi S-4800 FESEM (Hitachi, Japan) with 10 kV accelerating voltage. Fourier transform infrared (FTIR) spectroscopy was carried out with a Nicolet 6700 (Thermo Nicolet, USA), and X-ray photoelectron spectroscopy (XPS) analysis was carried out on an AEM PHI 5300X spectrometer with an Al Kα X-ray source to confirm the chemical structures of modified fibers. The specific surface area of an adsorbent was measured with a JW-BK surface area measurement instrument following the BET method. The wettability and hydrophilic–hydrophobic property regulation of the samples was measured with a contact angle analyzer (KRUSS DSA100, Germany). The concentration of metal ions in solution was analyzed using an inductively coupled plasma optical atomic emission spectrometer (ICP-OES, Type Varian 715-ES, USA). The zeta potential was measured with a Zetasizer Nano ZS90 (Malvern, UK) at 25 °C as a function of pH.
2.4 Adsorption experiment
Cd(II) batch adsorption experiments were performed in glass conical flasks. Fifty milligrams of PP-g-AA-Am and IEF fibers were added to 50 mL of Cd(II) aqueous solution. The flasks were shaken at 150 rpm in an orbital shaker for 2 h. The concentration of Cd(II) was determined with an inductively coupled plasma optical atomic emission spectrometer (ICP-OES, Type Varian 715-ES, USA). The removal rate and adsorption capabilities can be calculated using the following equations:| |  | (3) |
| | 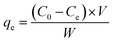 | (4) |
where C0 and Ce are the initial and equilibrium mercury ion concentrations (mg L−1), qe is the equilibrium adsorption capacity (mg g−1), W is the weight of the PP-g-AA-Am fibers (g), and V is the volume of the solution (L). The Cd(II) uptake was investigated as a function of pH by varying the pH from around 1.0–8.0 at an initial Cd(II) concentration of 100 mg L−1. Equilibrium studies were performed as a function of temperature (15 °C, 25 °C, 35 °C and 45 °C). The initial Cd(II) concentration was studied between 0.1 and 250 mg L−1.
3. Results and discussion
3.1 Characterization of PP-g-AA-Am fibers
FESEM images of PP-g-AA and PP-g-AA-Am fibers are shown in Fig. 1. When compared with the surface of the PP fibers as previously reported,27 the surface of the PP-g-AA fiber was evidently rough. Specifically, a heterogeneous grafting layer was formed on the surface of the PP-g-AA fiber, and therefore the fiber became coarse. These findings indicated that AA was grafted onto the fiber during irradiation. The surfaces of the PP-g-AA-Am fibers: (c and d) PP-g-AA-EDA; (e and f) PP-g-AA-DETA; (g and h) PP-g-AA-TETA; and (i and l) PP-g-AA-TEPA fibers were significantly rougher than that of than PP-g-AA fibers, and a porous structure appeared on the surface. This indicated that changes occurred on the fiber surfaces during the amide reaction.
 |
| | Fig. 1 FESEM images of the modified PP fibers: (a and b) PP-g-AA; (c and d) PP-g-AA-EDA; (e and f) PP-g-AA-DETA; (g and h) PP-g-AA-TETA; (i and j) PP-g-AA-TEPA fibers. | |
The nitrogen adsorption and desorption isotherms of fibers at 80 °C are shown in Fig. 2a. In comparison with the original PP, the BET surface areas and pore volumes of PP-g-AA and PP-g-AA-Am fibers increased to some extent. The shape of the adsorption isotherms for PP-g-AA-Am fibers can be considered as a combination of types I and IV (or V) according to the IUPAC classification.28 As shown in Fig. 1, comparison with PP-g-AA shows that the PP-g-AA-Am fiber has a rough surface and a porous structure, indicating that the mesostructure is maintained during the amine-grafting process. Their pore size distribution (Fig. 2b), varying with the surface modifications, revealed a successive decrease in pore volume with a relatively uniform mean pore diameter of 2.5 nm (see Table 1).
 |
| | Fig. 2 (a) N2 adsorption–desorption isotherms of original PP, PP-g-AA, PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA fibers. (b) Pore size distributions from adsorption branches for PP samples calculated by BJH method. | |
Table 1 BET surface areas, pore volumes, and average pore sizes of PP samples
| Sample |
S
BET
a (m2 g−1) |
V
t
b (cm3 g−1) |
D
p
c (nm) |
|
Specific surface area (BET).
Total pore volume.
Average pore size.
|
| Original PP |
4.49 |
0.003 |
22.53 |
| PP-g-AA |
15.78 |
0.055 |
2.87 |
| PP-g-AA-EDA |
35.87 |
0.055 |
2.48 |
| PP-g-AA-DETA |
18.26 |
0.028 |
2.49 |
| PP-g-AA-TETA |
21.03 |
0.038 |
2.51 |
| PP-g-AA-TEPA |
24.26 |
0.033 |
2.53 |
The FT-IR spectra of PP-g-AA and PP-g-AA-Am fibers are shown in Fig. 3a. Compared with the original PP fiber, the appearance of a new band at 1710 cm−1 in PP-g-AA fibers was due to the stretching vibrations of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and the bands at 1308 cm−1 reflected the stretching vibrations of C–N.22,23 These findings indicated that AA had been grafted onto the PP fiber. After the amide reaction, a broad band ranging from 3200 to 3700 cm−1, corresponding to the EDA (or DETA, TETA or TEPA) stretching vibrations of –NH2 and –NH groups, appeared. In addition, the appearance of a new band at 1542 cm−1 was assigned to the stretching vibration of the amide bond, and the bands at 1380 cm−1 reflected the stretching vibration of –CH2 in PP-g-AA-Am fibers. It was evident that after Cd(II) adsorption, as shown in Fig. 3b, the characteristic peaks at 1630 (C
O, and the bands at 1308 cm−1 reflected the stretching vibrations of C–N.22,23 These findings indicated that AA had been grafted onto the PP fiber. After the amide reaction, a broad band ranging from 3200 to 3700 cm−1, corresponding to the EDA (or DETA, TETA or TEPA) stretching vibrations of –NH2 and –NH groups, appeared. In addition, the appearance of a new band at 1542 cm−1 was assigned to the stretching vibration of the amide bond, and the bands at 1380 cm−1 reflected the stretching vibration of –CH2 in PP-g-AA-Am fibers. It was evident that after Cd(II) adsorption, as shown in Fig. 3b, the characteristic peaks at 1630 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in amide) and 1542 cm−1 (N–H group in amine) were shifted to 1706 and 1625 cm−1, respectively,24 and their intensities were significantly decreased. Simultaneously, the characteristic peaks of the amine group bending vibration (N–H group) at 838 cm−1 were replaced by peaks at 802 and 630 cm−1, which could be designated as the adsorbed cadmium species. These changes indicated that the Cd(II) species attached to both amide bonds and amino groups, and certain chemical bonds were formed, thus causing alterations in the vibrational frequencies and intensities of the surface chemical groups.
O in amide) and 1542 cm−1 (N–H group in amine) were shifted to 1706 and 1625 cm−1, respectively,24 and their intensities were significantly decreased. Simultaneously, the characteristic peaks of the amine group bending vibration (N–H group) at 838 cm−1 were replaced by peaks at 802 and 630 cm−1, which could be designated as the adsorbed cadmium species. These changes indicated that the Cd(II) species attached to both amide bonds and amino groups, and certain chemical bonds were formed, thus causing alterations in the vibrational frequencies and intensities of the surface chemical groups.
 |
| | Fig. 3 FTIR spectra of (a) the prepared PP-g-AA-Am fibers, (b) the PP-g-AA-Am fibers after Cd(II) adsorption. | |
The XPS spectra of PP-g-AA, PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA fibers are shown in Fig. 4a. When compared with the PP fibers, as previously reported,27 the new binding energies of ca. 533.4 eV, corresponding to O 1s, appeared in the spectra of PP-g-AA fibers. This indicated that AA had been grafted onto the PP fibers. After the amide reaction, new binding energies of ca. 400.5 eV, corresponding to N 1s, appeared in the spectra of the PP-g-AA-Am fibers. This indicated that EDA (or DETA, TETA or TEPA) had been grafted onto the PP-g-AA fibers.
 |
| | Fig. 4 Survey XPS spectra of the (a) PP-g-AA-Am fibers; HR-XPS spectra of C 1s for (b) PP-g-AA, (c) PP-g-AA-EDA, (d) PP-g-AA-DETA, (e) PP-g-AA-TETA, and (f) PP-g-AA-TEPA. | |
The XPS C 1s spectrum for the modified PP fiber, PP-g-AA, is shown in Fig. 4b; C–C (284.5 eV) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carbonyl, 288.5 eV) of the successfully modified PP indicated efficient high-energy electrons. After chemical modification, C–C (284.5 eV), C–O (hydroxyl and epoxy, 286.4 eV), C–N (285.7 eV) and C
O (carbonyl, 288.5 eV) of the successfully modified PP indicated efficient high-energy electrons. After chemical modification, C–C (284.5 eV), C–O (hydroxyl and epoxy, 286.4 eV), C–N (285.7 eV) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carboxyl, 288.5 eV) were detected in PP-g-AA-Am fibers. The percentage contents of C–N were 5.04% in PP-g-AA-EDA (c), 6.01% in PP-g-AA-DETA (d), 2.86% in PP-g-AA-TETA (e), and 8.97% in PP-g-AA-TEPA (f). This indicated that the order of the reaction activity of the four polyamine compounds was as follows: tetraethylenepentamine (TEPA) > diethylenetriamine (DETA) > ethylenediamine (EDA) > triethylenetetramine (TETA) under the same conditions.
O (carboxyl, 288.5 eV) were detected in PP-g-AA-Am fibers. The percentage contents of C–N were 5.04% in PP-g-AA-EDA (c), 6.01% in PP-g-AA-DETA (d), 2.86% in PP-g-AA-TETA (e), and 8.97% in PP-g-AA-TEPA (f). This indicated that the order of the reaction activity of the four polyamine compounds was as follows: tetraethylenepentamine (TEPA) > diethylenetriamine (DETA) > ethylenediamine (EDA) > triethylenetetramine (TETA) under the same conditions.
The contact angle was an excellent indication of the hydrophobicity/hydrophilicity of the solid adsorbent: the solid adsorbent was classed as “hydrophilic” if the contact angle was <90° and “hydrophobic” if the contact angle was >90°. As shown in Fig. 6B, the contact angles of PP original fiber, PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA fibers are 96.2°, 27.9°, 47.9°, 32.3° and 36.4°, respectively. The values of the static contact angles indicated that PE fiber was a hydrophobic adsorbent, while PP-g-AA-Am fibers were a hydrophilic adsorbent. This could be explained by the following two reasons: first, the PP original fiber structures contained alkane chains, and these groups were hydrophobic; thus, the PP fiber was hydrophobic. Second, PP-g-AA-Am fibers were modified with hydrophilic groups (amino/amide groups), which improved the hydrophilicity of PP-g-AA-Am. The hydrophilic surface of PP-g-AA-Am fibers could be beneficial to increasing its compatibility for heavy-metal ions in solution, in addition to enhancing the adsorption rate of PP-g-AA-Am fibers and their capacity for Cd(II) adsorption.
3.2 Batch adsorption tests
3.2.1. Effect of pH on adsorption.
As is well known, the pH of a solution is commonly an important parameter affecting the adsorption of metal ions on adsorbents because it not only affects metal species in solution, but also influences the surface properties of the adsorbents in terms of dissociation of functional groups and surface charges. Plots of adsorption capacity versus initial pH are shown in Fig. 5a. The upper limit of the studied pH of Cd(II) was set at 10.0, since at a pH higher than this value, Cd(II) precipitates in solution, making the results invalid.10–12 As the solution pH increased, the adsorption capacities first increased as the solution pH ranged from 2.0 to 4.0, reached a plateau at pH 4.0 to 8.0, and then slightly decreased at pH > 8.0. The dependence of Cd(II) adsorption on the solution pH could be explained from the perspectives of the surface charge of the adsorbent and the state of Cd(II) at various pH values. Under acidic conditions (pH < 4.0), as shown in Fig. 6A(a), the amino groups on the surface of PP-g-AA-Am fibers are easily protonated, forming mainly NH3+ without lone-pair electrons, and it was difficult to form coordination bonds with Cd(II).39 At pH values between 4.0 and 8.0, the main surface group formed might be –NH2. The key is the coordination bond between the adsorbent and Cd(II) (as shown in Fig. 6A(b)). When pH > 8.0, OH− ions may be adsorbed to the surface of PP-g-AA-Am fibers (Fig. 6A(c)), which contributed to the negatively charged sites of the PP-g-AA-Am fibers. The scheme of the adsorption mechanism is shown in Fig. 6A. Moreover, in the case of Cd(II), the interaction between the adsorbents and the Cd(II) ions may be described by eqn (5)–(8).| | | –NH2 + Cd2+ ⇌ –NH2Cd2+ | (6) |
| | | –NH2OH− + Cd2+ (or CdOH+) ⇌ –NH2OH−⋯Cd2+ (or –NH2OH−⋯CdOH+) | (8) |
 |
| | Fig. 5 (a) Effect of pH on adsorption on PP-g-AA-Am fibers. Solution concentrations: 10 mg L−1; adsorption dosage: 30 mg; contact time: 2 h. (b) Zeta potentials of PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA. | |
 |
| | Fig. 6 (A) Probable adsorption mechanism of Cd(II) on the PP-g-AA-Am fibers. (B) Contact angle of PP original fiber (a), PP-g-AA-EDA fiber (b), PP-g-AA-DETA fibers (c), PP-g-AA-TETA fibers (d), and PP-g-AA-TEPA fibers (e). | |
Eqn (5) indicates the protonation/deprotonation reactions of the amine groups of the PP-g-AA-Am fibers in the solution; eqn (6) shows the formation of surface complexes of Cd(II) with the amine groups via coordination interactions. At lower solution pH values, the reaction in eqn (5) favored the protonation of the amine groups to form NH3+. Since more NH2 groups were converted to NH3+, there were fewer NH2 sites available on the adsorbents' surface for Cd(II) adsorption through eqn (6). In addition, the formation of more NH3+ sites on the surface increased the electrostatic repulsion between the Cd(II) and the surfaces of the adsorbents.39 All these effects would result in the reduction of Cd(II) adsorption on the adsorbents with decreasing solution pH values. With the increase in the solution pH, the reaction in eqn (5) proceeds to the left, resulting in an increase in the number of NH2 sites on the surface of the adsorbents for Cd(II) adsorption through eqn (6), thus increasing the adsorption capacity.
These results are seen in Fig. 5b, where their surface charge becomes more positive with an increasing number of amine groups on PP-g-AA-Am fibers due to protonation. Moreover, the isoelectric points (pHPZC) of the synthesized samples were found to be in the following order: PP-g-AA-EDA (pHPZC = 5.4) < PP-g-AA-DETA (pHPZC = 6.8) < PP-g-AA-TETA (pHPZC = 9.2) < PP-g-AA-TEPA (pHPZC = 10.0). The large difference in pHPZC between the PP-g-AA-EDA and PP-g-AA-TEPA could be due to the N density, as presented in Fig. 4. Most carboxyl groups on the PP-g-AA fiber surfaces were protonated and positively charged at pH values lower than the pHPZC. Thus, metals such as Cd(II) cannot easily move toward the positively charged surface of amine-grafted PP-g-AA fibers owing to electrokinetic repulsive forces. Under acidic conditions (pH < 4.0), the amine groups at the edge sites are probably protonated to form NH3+, indicating that they would be no longer reactive for cationic metals in the formation of metal complexes. In pH ranges above the pHPZC, on the other hand, the surfaces of PP-g-AA-Am fibers become negatively charged due to deprotonation, suggesting that all the amine groups could be available for complexation with heavy metal cations.
3.2.2. Adsorption kinetics.
To determine the optimum adsorption time, the contact time from 0 to 160 min was studied. The results for PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA, and PP-g-AA-TEPA fibers are shown in Fig. 7a. The equilibrium times for PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA fibers were about 60 min, 60 min, 120 min, and 120 min, respectively. Of all the materials, the qe (86.66 mg g−1) and the removal rate (97%) of PP-g-AA-EDA was the highest. This may be ascribed to the difference in adsorbing groups (amide bond and amino functional group) and the length of the carbon chain in the reagent in the amide reaction. However, with an increase of the length of the carbon chain, the steric hindrance increased, and the qe and removal rate decreased. The qe and removal rate of PP-g-AA-EDA and PP-g-AA-DETA were higher than those of PP-g-AA-TETA and PP-g-AA-TEPA, which may due to PP-g-AA-EDA and PP-g-AA-DETA fibers being easy to ionize, due to small steric hindrance.
 |
| | Fig. 7 (a) Effect of contact time on the adsorption capacity of PP-g-AA-Am and IEF fiber for Cd(II), solution concentrations: 88.56 mg L−1; adsorption dosage: 30 mg; (b) pseudo-first-order kinetic plots for adsorption of PP-g-AA-Am fiber; (c) pseudo-second-order kinetic plots for adsorption by PP-g-AA-Am fiber. | |
The adsorption kinetic study plays a significant role in the analysis of the adsorption process, and can depict the adsorption rate, which in turn controls the residual time of the adsorption process at the solid–solution interface. Investigating the uptake-time curves showed that the maximum uptake follows the order PP-g-AA-Am > IEF for all time intervals. The adsorption kinetics of heavy metal ions onto PP-g-AA-Am fibers (see Fig. 7a) were investigated with the Lagergren pseudo-first-order model (see Fig. 7b) and a pseudo-second-order model (see Fig. 7c), which are represented by the following equations.26,29
The pseudo-first-order model follows:
| | ln(q1e − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) q1e − k1t q1e − k1t | (9) |
The pseudo-second-order model follows:
| |  | (10) |
where
q1e (mg g
−1) and
q2e (mg g
−1) represent the calculated adsorption capacity of PP-
g-AA-Am fibers at equilibrium;
qt is the adsorption amount at
t (min);
k1 (1 min
−1) and
k2 [g mg
−1 min
−1] are the rate constants of the pseudo-first-order and pseudo-second-order kinetics equations, respectively.
The results obtained from pseudo-first-order and pseudo-second-order models of PP-g-AA-Am fibers are shown in Fig. 7 and Table 2. It is obvious that the correlation coefficients (R2) for the pseudo-second-order kinetic (Fig. 7c) model have higher values (>0.99) than those from the pseudo-first-order kinetic model (Fig. 7b), and the theoretical adsorption capacities (q2e) calculated from the pseudo-second-order kinetic equation are much closer to the experimental adsorption capacities (qexp), demonstrating that the pseudo-second-order adsorption model is predominant in depicting the adsorption process.
Table 2 Kinetic parameters for Cd(II) on PP-g-AA-Am fiber
| Adsorbents |
q
exp (mg g−1) |
First-order kinetic model |
Second-order kinetic model |
|
k
1 (1 min−1) |
q
1e (mg g−1) |
R
2
|
k
2 (1 min−1) |
q
2e (mg g−1) |
R
2
|
| PP-g-AA-EDA |
85.1257 |
0.0658 |
14.8854 |
0.9678 |
6.4898 |
86.6551 |
0.9998 |
| PP-g-AA-DETA |
81.2165 |
0.0524 |
18.8821 |
0.9456 |
3.3816 |
83.6120 |
0.9995 |
| PP-g-AA-TETA |
70.5620 |
0.022 |
32.8423 |
0.9527 |
2.3652 |
74.1437 |
0.9915 |
| PP-g-AA-TEPA |
56.7892 |
0.0158 |
49.3143 |
0.9598 |
2.0259 |
57.0918 |
0.9960 |
3.2.3. Adsorption isotherms.
The adsorption isotherms of Cd(II) onto PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA, and PP-g-AA-TEPA fibers were studied at room temperature. It was seen from the results in Fig. 8a, that initially the isotherm rises sharply as the equilibrium concentrations increase, indicating that plenty of readily accessible sites are available for adsorption. However, as the concentration increases, a plateau is reached, indicating that no more sites are available for adsorption. Moreover, the adsorption capacity was higher for PP-g-AA-EDA and PP-g-AA-DETA fibers.
 |
| | Fig. 8 (a) Effect of initial concentration on adsorption by PP-g-AA-Am fiber at pH 3.0; (b) Langmuir adsorption isotherm plots of PP-g-AA-Am fiber; (c) Freundlich adsorption isotherm plots of PP-g-AA-Am fiber. | |
In this study, Langmuir and Freundlich adsorption isotherms, as two types of isotherm equations, are employed to investigate the adsorption processes. The Langmuir isotherm model assumes that adsorption is based on the homogeneous adsorbent surface. The linearized Langmuir equation is given as follows:38,39
| |  | (11) |
where
Ce (mg L
−1) is the equilibrium concentration;
qe (mg g
−1) is the adsorption amount at equilibrium;
qmax (mg g
−1) denotes the maximum monolayer capacity of the adsorbent;
KL (L mg
−1) is the Langmuir constant that represents the energy of the adsorption process. The values of
qmax and
KL were calculated from the slope and intercept of the Langmuir isotherm (see
Fig. 8b), and their values are listed in
Table 3. Another essential parameter,
RL, known as the separation factor, is determined by the relation
| |  | (12) |
where
RL indicates the type of the isotherm to be either unfavorable (
RL > 1), linear (
RL = 1), favorable (0 <
RL < 1), or irreversible (
RL = 0).
Table 3 Isothermal parameters for the adsorption of Cd(II) on PP-g-AA-Am fiber, based on Langmuir and Freundlich models
| Material |
Freundlich models |
Langmuir models |
| ln(KF) |
n
|
R
2
|
q
m, mg g−1 |
K
L, L mg−1 |
R
2
|
| PP-g-AA-EDA |
3.83466 |
1.19966 |
0.99622 |
111.23471 |
0.63265 |
0.95666 |
| PP-g-AA-EDTA |
3.97346 |
0.80750 |
0.99324 |
56.65722 |
1.44909 |
0.97057 |
| PP-g-AA-TETA |
3.37417 |
0.99640 |
0.99725 |
132.45033 |
0.19008 |
0.94142 |
| PP-g-AA-TEPA |
2.94418 |
1.08101 |
0.99705 |
44.01408 |
0.55401 |
0.92584 |
The Freundlich isotherm model is an empirical relationship depicting the adsorption for solutes from a liquid to a solid surface (see Fig. 8c), which presumes that multilayer adsorption occurs on a heterogeneous surface. The linear form of the Freundlich isotherm is
| |  | (13) |
where
KF is the Freundlich constant and
n is the heterogeneity factor, which are determined from the intercept and slope of the linear plot, respectively.
The isotherm models and constants were calculated from the experimental data and are presented in Fig. 8 and Table 3. The Freundlich isotherm model shows better correlation than the Langmuir isotherm model at room temperature, which indicated that the Freundlich adsorption isotherm model represented the adsorption process more ideally, suggesting multilayer adsorption of Cd(II) ions onto the adsorbent surface.
3.2.4. Effect of temperature on Cd(II) adsorption by PP-g-AA-Am fibers.
The influence of temperature on adsorption efficiency was investigated at various temperatures (298, 308, 318, and 328 K). For each temperature point, 30 mg of PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA, and PP-g-AA-TEPA fibers were added to 30 mL of a 92.65 mg L−1 Cd(II) solution and were kept shaking at 150 rpm in an orbital shaker for 2 h. The standard Gibbs free energy change (ΔG), standard enthalpy change (ΔH), and standard entropy change (ΔS) were determined according to the following equations:29| | ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kd Kd | (14) |
| |  | (15) |
where Kd is the distribution coefficient and R is the gas constant [J mol−1 K−1].
The effect of temperature on the adsorption of Cd(II) by PP-g-AA-Am fibers is shown in Fig. 9a. As the temperature was increased from 298 to 328 K, the amount of Cd(II) adsorbed increased from 80.58 to 90.24 mg g−1 for PP-g-AA-EDA, from 74.09 to 88.17 mg g−1 for PP-g-AA-DETA, from 70.93 to 78.81 mg g−1 for PP-g-AA-TETA and from 61.21 to 68.96 mg g−1 for PP-g-AA-TEPA. The probable reason for this behavior is that the diffusion rate of the sorbate within the pores increased and the solution viscosity decreased as the temperature was increased, thereby causing an increase in the frequency of collision between PP-g-AA-Am and Cd(II) ions and thus enhancing the adsorption capacity. From the plots of the distribution coefficient Kdversus temperature in Fig. 9b, it can be seen that Kd increased with increasing temperature, indicating adsorption that is endothermic in nature.30 According to eqn (14), the values of ΔH and ΔS were calculated from the slope and intercept, respectively, of a plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kdversus 1/T. The calculated values of the thermodynamic parameters are presented in Table 4. The overall Gibbs free energy changes during the adsorption processes at 298, 308, 318 and 328 K were found to be negative, indicating spontaneous adsorption of Cd(II) onto PP-g-AA-Am fibers. The increase in the absolute values of ΔG with increasing temperature indicates that the adsorption process becomes more favorable at higher temperatures. The positive value of the enthalpy changes (ΔH) were indicative of an endothermic process in solution,39 and the positive values of entropy changes (ΔS) for Cd(II) showed increased randomness at the solid solution interface during the adsorption of Cd(II) on PP-g-AA-Am fibers. Therefore, it could be inferred that the adsorption reaction was propelled by entropy.
Kdversus 1/T. The calculated values of the thermodynamic parameters are presented in Table 4. The overall Gibbs free energy changes during the adsorption processes at 298, 308, 318 and 328 K were found to be negative, indicating spontaneous adsorption of Cd(II) onto PP-g-AA-Am fibers. The increase in the absolute values of ΔG with increasing temperature indicates that the adsorption process becomes more favorable at higher temperatures. The positive value of the enthalpy changes (ΔH) were indicative of an endothermic process in solution,39 and the positive values of entropy changes (ΔS) for Cd(II) showed increased randomness at the solid solution interface during the adsorption of Cd(II) on PP-g-AA-Am fibers. Therefore, it could be inferred that the adsorption reaction was propelled by entropy.
 |
| | Fig. 9 (a) Effect of temperature on removal capacity of PP-g-AA-Am fibers. (b) Plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kdversus 1/T for the adsorption of Cd(II) by PP-g-AA-Am fibers. Kdversus 1/T for the adsorption of Cd(II) by PP-g-AA-Am fibers. | |
Table 4 Thermodynamic parameters for adsorption of Cd(II) by PP-g-AA-Am fibers
| Adsorbents |
−ΔGads (kJ mol−1) |
ΔHads (kJ mol−1) |
ΔSads (J mol−1 K−1) |
| 298 K |
308 K |
318 K |
328 K |
| PP-g-AA-EDA |
7.832 |
8.983 |
10.168 |
11.309 |
27.120 |
117.191 |
| PP-g-AA-DETA |
6.585 |
7.821 |
8.629 |
9.730 |
24.134 |
103.243 |
| PP-g-AA-TETA |
5.193 |
5.572 |
5.819 |
6.534 |
7.502 |
42.422 |
| PP-g-AA-TEPA |
2.369 |
2.961 |
3.395 |
5.169 |
16.151 |
61.969 |
3.3 Comparison of adsorption of heavy metals by PP-g-AA-Am fibers
Solutions separately containing Hg(II), Pb(II), Cd(II), Cu(II) and Ni(II) (each 10 mg L−1, pH 5) were prepared. To 50 mL of each of these solutions was added 50 mg of PP-g-AA, PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA, and the mixtures were kept shaking at 150 rpm in an orbital shaker for 2 h. After that, the amounts of these metals adsorbed by PP-g-AA and PP-g-AA-Am fibers were calculated. The adsorption results are presented in Fig. 10. It was found that the equilibrium removal rates (%) of Pb(II) and Cd(II) were much higher than those of the other metals, with an ordering of Pb(II) > Cd(II) > Hg(II) > Cu(II) > Ni(II), indicating that PP-g-AA-Am fibers have an evident adsorption selectivity to Pb(II) and Cd(II). However, for Cd(II), Cu(II) and Ni(II), the ordering was PP-g-AA-EDA > PP-g-AA-DETA > PP-g-AA-TETA > PP-g-AA-TEPA > PP-g-AA, indicating that with an increase in the length of the carbon chain, the steric hindrance increased and the removal rate decreased. According to HSAB theory,31 as Lewis soft acids, Pb(II) and Cd(II) have precedence over Hg(II), Cu(II) and Ni(II), which can be regarded as medium-soft acids, in interactions with the amine/imine and amide bond of PP-g-AA-Am fibers, which are Lewis soft bases.32 Therefore, PP-g-AA-Am fibers show a stronger covalent affinity to Pb(II) and Cd(II), compared to weaker interactions with Hg(II), Cu(II) and Ni(II).
 |
| | Fig. 10 Comparative adsorption of Cd(II) and other metals by PP-g-AA-Am fibers; the initial concentration of Cd(II) was set as 10 mg L−1. | |
3.4 Influences of co-existing cations and salinity
For cation exchange, the co-existing cations may reduce the adsorption capacity via a competing effect. Wastewater typically contains Na+, K+, Mg2+, and Ca2+. Therefore, it is important to investigate the influence of co-existing cations on the adsorption. The results are presented in Fig. 11. Since the alkaline-earth metal ion Ca2+ existed widely in natural water and wastewater, it was important to investigate the effect of Ca2+ on Cd(II) adsorption. In Fig. 11a, the Cd(II) adsorption capacity of PP-g-AA-Am fibers was decreased by less than 5% when the Ca2+ concentration increased from 0 to 50 mg L−1. In addition, the Cd(II) adsorption capacities decreased by 37.5%, when the Ca2+ concentration increased from 40 mg L−1 to 275 mg L−1. Generally, the Ca2+ contents of surface freshwater ranged from 15 mg L−1 to 75 mg L−1. Moreover, the Cd(II) adsorption capacity of IEF was reduced by more than 12% when the Ca2+ concentration increased from 0 to 50 mg L−1, and the adsorption capacities of IEF for Cd(II) reduced by 61.24% when the concentration of Ca2+ increased to 275 mg L−1. Thus, the existence of Ca2+ in this concentration range did not result in a marked reduction in the Cd(II) adsorption capacity. This phenomenon might be explained by the functional –NH2 groups of PP-g-AA-Am forming stable five-membered complexes with Cd(II), and the interaction between Ca2+ and PP-g-AA-Am being weaker than that between Cd(II) and PP-g-AA-Am; moreover, Ca2+ could form outer-sphere surface complexes with water molecules “Ca2+–(H2O)n”.33,34 Thus, the Ca2+ and Ca2+–(H2O)n adsorbed on PP-g-AA-Am via weak complexation could be displaced by Cd(II), and these complexes could cover the surfaces of adsorbents, making them less available to PP-g-AA-Am fibers, and Ca2+–(H2O)n was adsorbed onto the surface functional groups of PP-g-AA-Am fibers via weak complexation, which occupied the effective adsorption sites. On the other hand, the adsorption amount dropped by nearly 21% as the concentration of Na+, K+, Ca2+, and Mg2+ reached 200 mg L−1 (see Fig. 11b). The ion-exchange order was affected by the charge and ionic radius. For PP-g-AA-Am fibers, the exchange order generally follows the sequence Cd(II) > Ca2+ > Mg2+ > K+ > Na+ in aqueous solution. Hence, K+, Ca2+, and Mg2+ exerted a stronger influence than Na+ on the adsorption of Cd(II). Therefore, the effect of Mm+ on Cd(II) adsorption by PP-g-AA-EDA, the existence of Mm+ in this concentration range did not make a marked reduction in Cd(II) adsorption capacity. This phenomenon might be explained and shown in Scheme 2. Competitive adsorption of Mm+ and Cd(II) occurred on the surface of PP-g-AA-Am, but the interaction between the Mm+ and PP-g-AA-Am was weaker than that between Cd(II) and PP-g-AA-Am. In addition, Mm+ could form outer-sphere surface complexes with water molecules “Mm+–(H2O)n”. Thus, the Mm+ and Mm+–(H2O)n adsorbed on PP-g-AA-Am via weak complexation could be displaced by Cd(II), so the impact on the adsorption of Cd(II) is not significant (in the case of PP-g-AA-EDA).
 |
| | Fig. 11 (a) The adsorption amounts of PP-g-AA-Am and IEF fibers under various CaCl2 concentrations ranging from 0 mg L−1 to 300 mg L−1; (b) effect of different salt species on the adsorption performance of PP-g-AA-Am fibers for Cd(II); the salt concentration was set at 200 mmol L−1. The initial concentration of Cd(II) was set as 100 mg L−1. | |
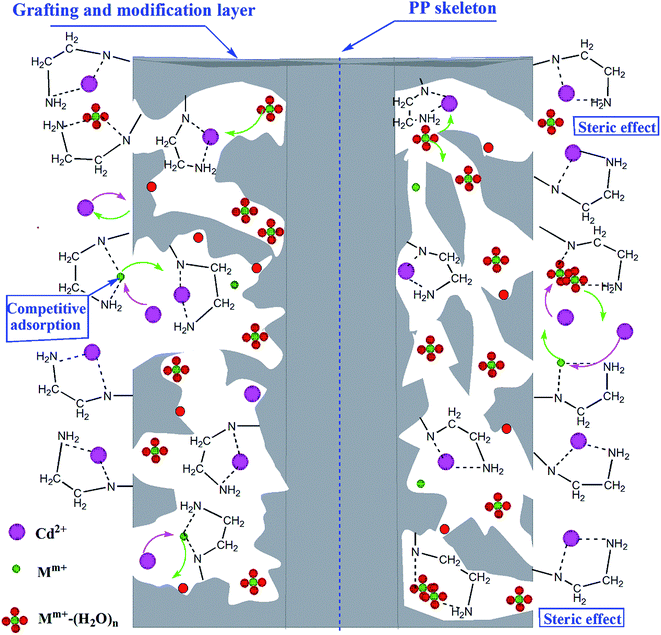 |
| | Scheme 2 Effect of Mm+ on Cd(II) adsorption by PP-g-AA-EDA. | |
3.5 XPS spectra analysis and adsorption mechanism
The adsorption mechanism could be confirmed by XPS (see Fig. 12c) and FTIR (see Fig. 3b) analyses of PP-g-AA-EDA before and after Cd(II) adsorption. From the binding energy of Cd3 d5 and Cd3 d3 after Cd(II) adsorption, as shown in Fig. 12c, the successful adsorption of Cd(II) was confirmed. Two energy bands, at about 404.92 and 411.08 eV, corresponded to the binding energies of Cd3 d5 and Cd3 d3, respectively, and are consistent with Cd(II) (Fig. 12c). The N 1s high-resolution scan of PP-g-AA-EDA could be deconvoluted into one individual peak at a binding energy of 401.08 eV (Fig. 12b), which was assigned to the N atoms in –NH2 and –NH groups.35 A new peak at 403.91 eV was observed after Cd(II) adsorption, which could be attributed to the complexation bonding, R–NH2⋯Cd or –NH⋯Cd.36 In the XPS spectra of C 1s (Fig. 12a), the carbon atoms can be found in two chemically different positions, leading to two differing C 1s binding energies: C–N (285.7 eV) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (288.58 eV). After adsorption, peaks at ∼289.01 eV appeared, which were assigned to C–N groups shifted to shorter binding energies (∼285.7 eV) with a broader band range, which may be attributed to the involvement of the NH and NH2 groups in the adsorption Cd(II). Similar phenomena were observed in the XPS spectra of O 1s, as shown in Fig. 12d, the peaks of O 1s appearing at ∼536.08 eV, which were assigned to C
O (288.58 eV). After adsorption, peaks at ∼289.01 eV appeared, which were assigned to C–N groups shifted to shorter binding energies (∼285.7 eV) with a broader band range, which may be attributed to the involvement of the NH and NH2 groups in the adsorption Cd(II). Similar phenomena were observed in the XPS spectra of O 1s, as shown in Fig. 12d, the peaks of O 1s appearing at ∼536.08 eV, which were assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C–OH groups, broadening with a slight shift of binding energies after adsorption of Cd(II).
O and C–OH groups, broadening with a slight shift of binding energies after adsorption of Cd(II).
 |
| | Fig. 12 XPS spectra of high-resolution scan of: (a) C 1s, (b) N 1s, (c) binding energy of Cd3 d5 and Cd3 d3 after Cd(II) adsorption, (d) O 1s of PP-g-AA-EDA and PP-g-AA-EDA-Cd. | |
In previous reports on the extraction of heavy metal ions using dendrimers, the binding mechanisms of metal ions by multi-amino functional materials were proposed to be: (1) complexation with surface primary amine groups, (2) complexation with interior tertiary amine and amide groups, and (3) physical encapsulation within dendrimer interior cavities or interactions with trapped counterions or water molecules.36–39 However, the high adsorption rate and high adsorption capacity of Cd(II) on PP-g-AA-Am fibers can be clarified as follows: first, as is well-known, ethylenediamine (EDA), diethylenetriamine (DETA), triethylenetetramine (TETA), and tetraethylenepentamine (TEPA) carry large amounts of amine and imine functional groups. After PP-g-AA modification, PP-g-AA-Am has plenty of additional amide functional groups. The –NH2, –NH, and –CONH– coexisting functional groups are expected to have a strong affinity to Cd(II) ions from aqueous solution.12,40 It is reasonable that these functional groups can provide sufficient active adsorption sites to form strong complexes with Cd(II) on the PP-g-AA-Am surface and thereby enhance the adsorption capacity of Cd(II). Second, the grafting of –NH2 on PP-g-AA increases the specific surface areas and total pore volume of PP-g-AA-Am, increasing the interaction areas between the sorbent and adsorbate, and accordingly effectively enhancing the adsorption rate and capacity of Cd(II) ions. However, the order of adsorption capacity is: PP-g-AA-EDA (125.34 mg g−1), PP-g-AA-DETA (116.15 mg g−1), PP-g-AA-TETA (93.02 mg g−1), and PP-g-AA-TEPA (81.01 mg g−1). It can be seen that the more –NH2 and –NH groups there are on adsorbents, the lower the adsorption capacity of Cd(II). In non-salt systems, the possible reasons are as follows: first, with an increase in the molecular chain, although the number of primary amines and secondary amines increases, the adsorption of Cd(II) is influenced by steric hindrance between the molecular chains, as shown in Scheme 3 (right). To observe the hydrodynamic radius during the swelling process of the PP-g-AA-Am fibers with increasing chain length, a microparticle grafting experiment was designed based on the determination of stimuli-responsive materials. First, polypropylene granules were milled into microparticles in a high-speed ball mill. Afterwards, the grafted layer was introduced through the method mentioned in Section 2.2. The median particle diameters (D0.5) of PP-g-AA-Am microparticles in ultrapure water were recorded by dynamic light scattering (Beckman Coulter LS 13-320), and are shown in Fig. S3.† The median particle diameters increased rapidly and attained equilibrium within 20 min, which also corresponded to the sorption kinetics. This experiment confirmed the increasing hydrodynamic radius of the PP-g-AA-Am fibers with increasing chain length.
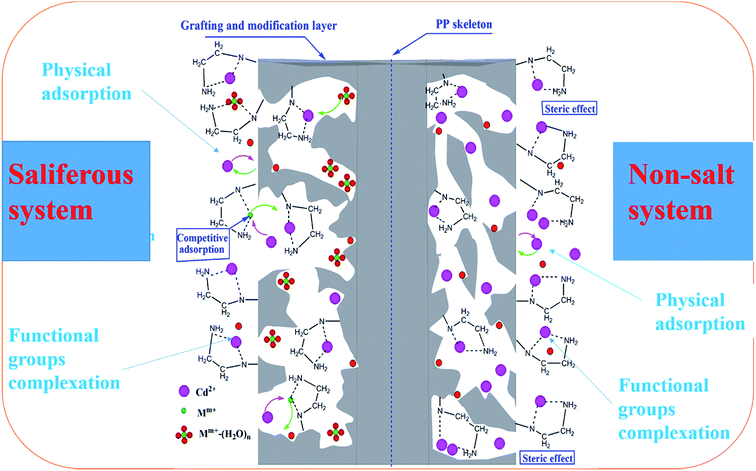 |
| | Scheme 3 Adsorption mechanism of Cd(II) on PP-g-AA-EDA. | |
Second, with an increase in the length of the molecular chain, the number of secondary amine functional groups increased, but due to their basicity order: (CH3)2NH > CH3NH2 > (CH3)3N > NH3. Generally, the greater the number of alkyl groups on the nitrogen atoms in the aliphatic amines of the primary amines and the tertiary amino groups, the easier it is to react with hydrogen ions. In acidic solution, the adsorption agent with positive charges showed the following sequence of formation: [(CH3)2NH+] > [CH3NH2+] > [(CH3)3N+], resulting in the coordination ability decreasing for lead in aqueous solution, while it was not conducive to the adsorption of Cd(II). In high-salt systems, competitive adsorption of Mm+ and Cd(II) on the surface of PP-g-AA-Am occurred, but the interaction between the Mm+ and PP-g-AA-Am was weaker than that between Cd(II) and PP-g-AA-Am, as shown in Scheme 3 (left). In addition, Mm+ could form outer-sphere surface complexes with water molecules “Mm+–(H2O)n”. Thus, the Mm+ and Mm+–(H2O)n adsorbed on PP-g-AA-Am via weak complexation could be displaced by Cd(II), and therefore the impact on the adsorption of Cd(II) is not significant (in the case of PP-g-AA-EDA).
3.6 Desorption and regeneration study
The regeneration of adsorbent is considered a crucial factor in sustainable applications. Acidic solutions and EDTA solutions could desorb the heavy-metal ions from adsorbents. In the present study, both 0.5 M HCl solution and 0.1 M EDTA solution were used to desorb the Cd(II) from PP-g-AA-Am fibers, and PP-g-AA-Am fibers were reused for five sorption–desorption cycles. As shown in Fig. 13, the adsorption capacity of Cd(II) decreased significantly as the PP-g-AA-Am regeneration cycles progressed, which indicates that both 0.5 M HCl solution and 0.1 M EDTA solution can effectively desorb Cd(II) from the PP-g-AA-Am. For 0.1 M EDTA eluent, the regeneration results showed that the removal efficiency decreased by about 12.09% for PP-g-AA-EDA, 9.86% for PP-g-AA-DETA, 10.2% for PP-g-AA-TETA, and 10.34% for PP-g-AA-TEPA after 5 cycles of desorption–adsorption compared with the original removal efficiency. However, for 0.5 M HCl eluent, the removal efficiency decreased by about 19.93% for PP-g-AA-EDA, 18.24% for PP-g-AA-DETA, 20.20% for PP-g-AA-TETA, and 18.15% for PP-g-AA-TEPA fibers. These results indicated that no appreciable loss in activity occurred over at least five cycles. Thus, a good sorption–desorption capacity of PP-g-AA-Am for Cd(II) was obtained in the cycles, and it was expected that PP-g-AA-Am would be a promising adsorbent for the removal of Cd(II) from water, particularly in emergency disposal after Cd(II) water pollution accidents.
 |
| | Fig. 13 Adsorption capacity of regenerated PP-g-AA-Am fibers in different cycles; (a) 0.5 M HCl, (b) 0.1 M EDTA. | |
4. Conclusion
In this study, a series of new porous fibrous adsorbents (PP-g-AA-Am) for the fast removal of Cd(II) from water were successfully prepared by grafting AA onto PP fibers and modifying with amide/amine groups. The equilibrium adsorption assay indicated that the adsorption capacity of PP-g-AA-Am for Cd(II) was higher than that of commercial ion-exchange fiber (IEF), which was attributed to its amide/amine groups, porous structure, and high surface area. The FTIR, FESEM, and XPS spectra indicated that AA was grafted onto the surface of PP fibers, and subsequently the amino functional groups and amide bonds were introduced by the amide reaction. The BET results indicated that PP-g-AA-Am fibers have a specific surface area and pore structure. The kinetics and isotherm data can be well fitted with the pseudo-second-order kinetic model and the Freundlich isotherm, respectively. The large adsorption capacity of PP-g-AA-Am for Cd(II) might be due to its porous structures with amino functional groups and amide bonds, which can form electrostatic interactions and coordination bonds with amino functional groups and amide bonds of Cd(II). Moreover, the effect of PP-g-AA-Am fibers on the adsorption of Cd(II) in salt solution (such as NaCl, KCl, MgCl2 and CaCl2) was not significant. In addition, the obtained fibers can be easily regenerated through 0.5 M HCl and 0.1 M EDTA washing within 2 h. Thus, it was expected that PP-g-AA-Am would be a promising adsorbent for the removal of Cd(II) from water, particularly in the emergency disposal of Cd(II) after water pollution accidents.
Acknowledgements
The financial support from the National Natural Science Foundation of China (Grant No. 30900325) and the Natural Science Foundation of Tianjin (Grant No. 13ZXCXSY14200) is gratefully acknowledged. Innovation Fund for Small Technology-based Firms (Grant No. 14ZXCXGX00724).
References
- P. Nagajyoti, K. Lee and T. Sreekanth, Environ. Chem. Lett., 2010, 8, 199–216 CrossRef CAS.
- M. I. Ansari and A. Malik, Bioresour. Technol., 2007, 98, 3149–3153 CrossRef CAS PubMed.
- D. Mohan and K. P. Singh, Water Res., 2002, 36, 2304–2318 CrossRef CAS PubMed.
- M. Anbia, S. A. Hariri and S. Ashrafizadeh, Appl. Surf. Sci., 2010, 256, 3228–3233 CrossRef CAS.
- S. S. Gupta and K. G. Bhattacharyya, Phys. Chem. Chem. Phys., 2012, 14, 6698–6723 RSC.
- M. Constantin, I. Asmarandei, V. Harabagiu, L. Ghimici, P. Ascenzi and G. Fundueanu, Carbohydr. Polym., 2013, 91, 74–84 CrossRef CAS PubMed.
- G. S. Simate, S. E. Iyuke, S. Ndlovu and M. Heydenrych, Water Res., 2012, 46, 1185–1197 CrossRef CAS PubMed.
- N. Srivastava and C. Majumder, J. Hazard. Mater., 2008, 151, 1–8 CrossRef CAS PubMed.
- Z. Wang, E. Nie, J. Li, Y. Zhao, X. Luo and Z. Zheng, J. Hazard. Mater., 2011, 188, 29–36 CrossRef CAS PubMed.
- G. Zhao, J. Li, X. Ren, C. Chen and X. Wang, Environ. Sci. Technol., 2011, 45, 10454–10462 CrossRef CAS PubMed.
- J. Liu, H. Du, S. Yuan, W. He, P. Yan and Z. Liu, Water Sci. Technol., 2015, 71, 1611–1619 CrossRef CAS PubMed.
- W. Li, R. He, L. Tan, S. Xu, C. Kang, C. Wei and Y. Tang, J. Sol-Gel Sci. Technol., 2016, 78, 632–640 CrossRef CAS.
- C. Shan, Z. Ma and M. Tong, J. Hazard. Mater., 2015, 299, 479–485 CrossRef CAS PubMed.
- M. Soleymanzadeh, M. Arshadi, J. Salvacion and F. SalimiVahid, Chem. Eng. Res. Des., 2015, 93, 696–709 CrossRef CAS.
- Z. Du, T. Zheng, P. Wang, L. Hao and Y. Wang, Bioresour. Technol., 2016, 201, 41–49 CrossRef CAS PubMed.
- J. Wang, C. Cheng, X. Yang, C. Chen and A. Li, Ind. Eng. Chem. Res., 2013, 52, 4072–4082 CrossRef CAS.
- J. K. Bediako, W. Wei, S. Kim and Y.-S. Yun, J. Hazard. Mater., 2015, 299, 550–561 CrossRef CAS PubMed.
- T. B. Mostafa, J. Appl. Polym. Sci., 2009, 111, 11–18 CrossRef CAS.
- M. Monier and D. Abdel-Latif, J. Hazard. Mater., 2013, 250, 122–130 CrossRef PubMed.
- R. Qu, C. Sun, F. Ma, Y. Zhang, C. Ji, Q. Xu, C. Wang and H. Chen, J. Hazard. Mater., 2009, 167, 717–727 CrossRef CAS PubMed.
- Z. Y. Kong, J. F. Wei and Y. H. Li,
et al.
, Chem. Eng. J., 2014, 254, 365–373 CrossRef CAS.
- N. Ma, Y. Yang, S. Chen and Q. Zhang, J. Hazard. Mater., 2009, 171, 288–293 CrossRef CAS PubMed.
- J. Wang, L. Xu, C. Cheng, Y. Meng and A. Li, Chem. Eng. J., 2012, 193, 31–38 CrossRef.
- Q. Zhang, S. Zhang, S. Chen, P. Li, T. Qin and S. Yuan, J. Colloid Interface Sci., 2008, 322, 421–428 CrossRef CAS PubMed.
- T. Li, S. Chen, H. Li, Q. Li and L. Wu, Langmuir, 2011, 27, 6753–6758 CrossRef CAS PubMed.
- H.-J. Park and C.-K. Na, J. Colloid Interface Sci., 2006, 301, 46–54 CrossRef CAS PubMed.
- L. Cui, J. Wei, X. Du and X. Zhou, Ind. Eng. Chem. Res., 2016, 55, 1566–1574 CrossRef CAS.
- T. J. Barton, L. M. Bull, W. G. Klemperer, D. A. Loy, B. McEnaney, M. Misono, P. A. Monson, G. Pez, G. W. Scherer and J. C. Vartuli, Chem. Mater., 1999, 11, 2633–2656 CrossRef CAS.
- Y. Liu, J. Chem. Eng. Data, 2009, 54, 1981–1985 CrossRef CAS.
- Z. Li, Y. Kong and Y. Ge, Chem. Eng. J., 2015, 270, 229–234 CrossRef CAS.
- R. G. Pearson, J. Am. Chem. Soc., 1963, 85, 3533–3539 CrossRef CAS.
- R. G. Pearson, J. Chem. Educ., 1968, 45, 581 CrossRef CAS.
- J. Wang, Y. Zhou, A. Li and L. Xu, J. Hazard. Mater., 2010, 176, 1018–1026 CrossRef CAS PubMed.
- A. Varma, S. Deshpande and J. Kennedy, Carbohydr. Polym., 2004, 55, 77–93 CrossRef CAS.
- Y. Zhang, S. Pan, H. Shen and M. Hu, Anal. Sci., 2012, 28, 887–892 CrossRef CAS PubMed.
- Q. Zhang, N. Wang, L. Zhao, T. Xu and Y. Cheng, ACS Appl. Mater. Interfaces, 2013, 5, 1907–1912 CAS.
- M. S. Diallo, S. Christie, P. Swaminathan, J. H. Johnson and W. A. Goddard, Environ. Sci. Technol., 2005, 39, 1366–1377 CrossRef CAS PubMed.
- T. Chen, F. Liu, C. Ling, J. Gao, C. Xu, L. Li and A. Li, Environ. Sci. Technol., 2013, 47, 13652–13660 CrossRef CAS PubMed.
- J. Tao, J. Xiong, C. Jiao, D. Zhang, H. Lin and Y. Chen, ACS Sustainable Chem. Eng., 2015, 4, 60–68 CrossRef.
- K. N. Han, B. Y. Yu and S.-Y. Kwak, J. Membr. Sci., 2012, 396, 83–91 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra21713c |
|
| This journal is © The Royal Society of Chemistry 2016 |
Click here to see how this site uses Cookies. View our privacy policy here.  *ab,
Fei
Zhao
a and
Qiang
Sun
ab
*ab,
Fei
Zhao
a and
Qiang
Sun
ab
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.0 g) were soaked in 60 mL of EDA, DETA, TETA and TEPA solution, followed by sealing in three-necked flasks with magnetic agitators in an oil bath for 3 h at 120 °C. The resulting PP-g-AA-Am fiber was washed with ethanol and deionized water more than three times and dried in a vacuum oven at room temperature overnight. The products are named as PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA, respectively. The degree of amination was calculated from the weight gain using the following formula:
2.0 g) were soaked in 60 mL of EDA, DETA, TETA and TEPA solution, followed by sealing in three-necked flasks with magnetic agitators in an oil bath for 3 h at 120 °C. The resulting PP-g-AA-Am fiber was washed with ethanol and deionized water more than three times and dried in a vacuum oven at room temperature overnight. The products are named as PP-g-AA-EDA, PP-g-AA-DETA, PP-g-AA-TETA and PP-g-AA-TEPA, respectively. The degree of amination was calculated from the weight gain using the following formula:
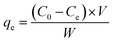

![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and the bands at 1308 cm−1 reflected the stretching vibrations of C–N.22,23 These findings indicated that AA had been grafted onto the PP fiber. After the amide reaction, a broad band ranging from 3200 to 3700 cm−1, corresponding to the EDA (or DETA, TETA or TEPA) stretching vibrations of –NH2 and –NH groups, appeared. In addition, the appearance of a new band at 1542 cm−1 was assigned to the stretching vibration of the amide bond, and the bands at 1380 cm−1 reflected the stretching vibration of –CH2 in PP-g-AA-Am fibers. It was evident that after Cd(II) adsorption, as shown in Fig. 3b, the characteristic peaks at 1630 (C
O, and the bands at 1308 cm−1 reflected the stretching vibrations of C–N.22,23 These findings indicated that AA had been grafted onto the PP fiber. After the amide reaction, a broad band ranging from 3200 to 3700 cm−1, corresponding to the EDA (or DETA, TETA or TEPA) stretching vibrations of –NH2 and –NH groups, appeared. In addition, the appearance of a new band at 1542 cm−1 was assigned to the stretching vibration of the amide bond, and the bands at 1380 cm−1 reflected the stretching vibration of –CH2 in PP-g-AA-Am fibers. It was evident that after Cd(II) adsorption, as shown in Fig. 3b, the characteristic peaks at 1630 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in amide) and 1542 cm−1 (N–H group in amine) were shifted to 1706 and 1625 cm−1, respectively,24 and their intensities were significantly decreased. Simultaneously, the characteristic peaks of the amine group bending vibration (N–H group) at 838 cm−1 were replaced by peaks at 802 and 630 cm−1, which could be designated as the adsorbed cadmium species. These changes indicated that the Cd(II) species attached to both amide bonds and amino groups, and certain chemical bonds were formed, thus causing alterations in the vibrational frequencies and intensities of the surface chemical groups.
O in amide) and 1542 cm−1 (N–H group in amine) were shifted to 1706 and 1625 cm−1, respectively,24 and their intensities were significantly decreased. Simultaneously, the characteristic peaks of the amine group bending vibration (N–H group) at 838 cm−1 were replaced by peaks at 802 and 630 cm−1, which could be designated as the adsorbed cadmium species. These changes indicated that the Cd(II) species attached to both amide bonds and amino groups, and certain chemical bonds were formed, thus causing alterations in the vibrational frequencies and intensities of the surface chemical groups.

![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carbonyl, 288.5 eV) of the successfully modified PP indicated efficient high-energy electrons. After chemical modification, C–C (284.5 eV), C–O (hydroxyl and epoxy, 286.4 eV), C–N (285.7 eV) and C
O (carbonyl, 288.5 eV) of the successfully modified PP indicated efficient high-energy electrons. After chemical modification, C–C (284.5 eV), C–O (hydroxyl and epoxy, 286.4 eV), C–N (285.7 eV) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carboxyl, 288.5 eV) were detected in PP-g-AA-Am fibers. The percentage contents of C–N were 5.04% in PP-g-AA-EDA (c), 6.01% in PP-g-AA-DETA (d), 2.86% in PP-g-AA-TETA (e), and 8.97% in PP-g-AA-TEPA (f). This indicated that the order of the reaction activity of the four polyamine compounds was as follows: tetraethylenepentamine (TEPA) > diethylenetriamine (DETA) > ethylenediamine (EDA) > triethylenetetramine (TETA) under the same conditions.
O (carboxyl, 288.5 eV) were detected in PP-g-AA-Am fibers. The percentage contents of C–N were 5.04% in PP-g-AA-EDA (c), 6.01% in PP-g-AA-DETA (d), 2.86% in PP-g-AA-TETA (e), and 8.97% in PP-g-AA-TEPA (f). This indicated that the order of the reaction activity of the four polyamine compounds was as follows: tetraethylenepentamine (TEPA) > diethylenetriamine (DETA) > ethylenediamine (EDA) > triethylenetetramine (TETA) under the same conditions.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) q1e − k1t
q1e − k1t



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kd
Kd
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kdversus 1/T. The calculated values of the thermodynamic parameters are presented in Table 4. The overall Gibbs free energy changes during the adsorption processes at 298, 308, 318 and 328 K were found to be negative, indicating spontaneous adsorption of Cd(II) onto PP-g-AA-Am fibers. The increase in the absolute values of ΔG with increasing temperature indicates that the adsorption process becomes more favorable at higher temperatures. The positive value of the enthalpy changes (ΔH) were indicative of an endothermic process in solution,39 and the positive values of entropy changes (ΔS) for Cd(II) showed increased randomness at the solid solution interface during the adsorption of Cd(II) on PP-g-AA-Am fibers. Therefore, it could be inferred that the adsorption reaction was propelled by entropy.
Kdversus 1/T. The calculated values of the thermodynamic parameters are presented in Table 4. The overall Gibbs free energy changes during the adsorption processes at 298, 308, 318 and 328 K were found to be negative, indicating spontaneous adsorption of Cd(II) onto PP-g-AA-Am fibers. The increase in the absolute values of ΔG with increasing temperature indicates that the adsorption process becomes more favorable at higher temperatures. The positive value of the enthalpy changes (ΔH) were indicative of an endothermic process in solution,39 and the positive values of entropy changes (ΔS) for Cd(II) showed increased randomness at the solid solution interface during the adsorption of Cd(II) on PP-g-AA-Am fibers. Therefore, it could be inferred that the adsorption reaction was propelled by entropy.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kdversus 1/T for the adsorption of Cd(II) by PP-g-AA-Am fibers.
Kdversus 1/T for the adsorption of Cd(II) by PP-g-AA-Am fibers.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (288.58 eV). After adsorption, peaks at ∼289.01 eV appeared, which were assigned to C–N groups shifted to shorter binding energies (∼285.7 eV) with a broader band range, which may be attributed to the involvement of the NH and NH2 groups in the adsorption Cd(II). Similar phenomena were observed in the XPS spectra of O 1s, as shown in Fig. 12d, the peaks of O 1s appearing at ∼536.08 eV, which were assigned to C
O (288.58 eV). After adsorption, peaks at ∼289.01 eV appeared, which were assigned to C–N groups shifted to shorter binding energies (∼285.7 eV) with a broader band range, which may be attributed to the involvement of the NH and NH2 groups in the adsorption Cd(II). Similar phenomena were observed in the XPS spectra of O 1s, as shown in Fig. 12d, the peaks of O 1s appearing at ∼536.08 eV, which were assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C–OH groups, broadening with a slight shift of binding energies after adsorption of Cd(II).
O and C–OH groups, broadening with a slight shift of binding energies after adsorption of Cd(II).











