Growth and characterization of semi-organic third order nonlinear optical (NLO) potassium 3,5-dinitrobenzoate (KDNB) single crystals
Abstract
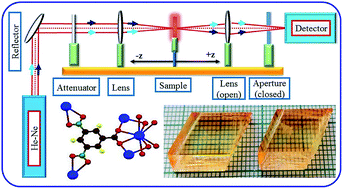
The semi-organic single crystal of potassium 3,5-dinitrobenzoate (KDNB) was successfully grown by slow evaporation solution technique (SEST) at room temperature. The lattice parameters of the grown KDNB crystal were confirmed by single crystal X-ray diffraction. The functional groups of the KDNB crystal were confirmed by Fourier transform infrared (FTIR) spectroscopy analysis. The optical quality of the grown crystal was identified by UV-Vis NIR spectral analysis. The grown crystal has good optical transparency in the range of 410–1100 nm. In the photoluminescence spectrum, sharp broad emission peaks were observed, which indicate violet and blue emission. The photoconductivity study reveals that the grown crystal has a negative photoconductive nature. The thermal behaviour of the crystal has been investigated by thermogravimetric and differential thermal analysis (TG-DTA). Vickers microhardness analysis was carried out to identify the mechanical stability of the grown crystal and the indentation size effect (ISE) was explained satisfactorily by Hays–Kendall's approach and proportional specimen resistance model [PSRM]. A chemical etching study was carried out and the etch pit density (EPD) was calculated. Laser damage threshold (LDT) energy has been measured by using a Nd:YAG laser (1064 nm). The dielectric permittivity and dielectric loss as a function of frequency were measured for the grown crystal. The solid state properties such as plasma energy, Penn gap and Fermi energy were evaluated for the KDNB crystal using the empirical relation. These estimated values were utilized to report the electronic polarizability. This matches well to the value calculated from Clausius–Mossotti relation, Lorentz–Lorentz equation, optical band gap energy and coupled dipole method (CDM). The third-order nonlinear optical properties such as refractive index (n2), absorption co-efficient (β) and susceptibility (χ(3)) were studied using Z-scan technique at 632.8 nm of He–Ne laser.

 Please wait while we load your content...
Please wait while we load your content...