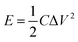Mn0.5Co2.5O4 nanofibers sandwiched in graphene sheets for efficient supercapacitor electrode materials†
Jinzuan Wanga,
Jun Yang*b,
Tao Huangb and
Wenyan Yin*ac
aSchool of Electronic Information and Electrical Engineering, Shanghai Jiao Tong University, Shanghai 200240, China. E-mail: wyyin@sjtu.edu.cn; wyyin@zju.edu.cn
bSchool of Chemistry and Chemical Engineering, Shanghai Jiao Tong University, Shanghai 200240, China. E-mail: yangj723@sjtu.edu.cn
cInnovative Institute of Electromagnetic Information and Electric Integration, College of Information Science and Electronic Engineering, Zhejiang University, Hangzhou 310058, China
First published on 24th October 2016
Abstract
Self-assembled manganese cobalt oxide (Mn0.5Co2.5O4) nanofibers sandwiched in graphene sheets (Mn0.5Co2.5O4@G) are successfully synthesized by a hydrothermal treatment and annealing process. Owning to the unique sandwich-like structures, rich active sites, and boosted electrical conductivity, Mn0.5Co2.5O4@G composite demonstrates superior electrochemical performance for potential supercapacitors. In a three-electrode system, it exhibits excellent cycling stability with a specific capacitance retention of 93.8% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at a current density of 10 A g−1, and achieves specific capacitances of 617 and 434 F g−1 at the ultrahigh current densities of 30 and 40 A g−1, respectively. For an asymmetric supercapacitor of Mn0.5Co2.5O4@G//AC, high energy densities of 36.8 and 13.6 W h kg−1 are also achieved at power densities of 147.3 and 3755.8 W kg−1, respectively. It is believed that the synthesized Mn0.5Co2.5O4@G composite can be a promising electrode material for high-performance supercapacitors, and the present synthetic strategy can be further extended for designing many other functional composites with desirable nanostructures and performance.
000 cycles at a current density of 10 A g−1, and achieves specific capacitances of 617 and 434 F g−1 at the ultrahigh current densities of 30 and 40 A g−1, respectively. For an asymmetric supercapacitor of Mn0.5Co2.5O4@G//AC, high energy densities of 36.8 and 13.6 W h kg−1 are also achieved at power densities of 147.3 and 3755.8 W kg−1, respectively. It is believed that the synthesized Mn0.5Co2.5O4@G composite can be a promising electrode material for high-performance supercapacitors, and the present synthetic strategy can be further extended for designing many other functional composites with desirable nanostructures and performance.
1. Introduction
Owning to their high power densities, fast charge/discharge rates, low maintenance cost, long cycling stability, and superior operational safety, supercapacitors have drawn much attention as auxiliary energy storage devices with fuel cells and lithium ion batteries (LIBs) for portable electronic devices, and hybrid electric vehicles, etc.1–5 There are two major types of electrode materials reported for making supercapacitors. One stores energy via fast reversible adsorption of ion onto both negative and positive electrode surfaces, such as carbon-based material, and it is named as electrical double layer capacitance (EDLC). The other stores energy via fast redox reactions or faradaic processes on the electrode surfaces, such as transition metal oxide or conducting polymer, and it is named as pseudocapacitance. Comparing these two types of materials, transition metal oxides, such as RuO2,6,7 MnO2,8,9 Co3O4,10,11 V2O5,12 and NiO,13 can achieve higher energy density. However, RuO2 and V2O5 are toxic, and MnO2 shows low electrical conductivity, which limit their applications.3,4 Furthermore, these transition metal oxides often tend to form big agglomerates, which may significantly degrade their electrochemical redox process due to incomplete reaction of agglomerated particles.4,6Ternary metal oxides with two different metal cations have received considerable attention due to their superior properties such as rich oxidation states, high theoretical capacitance, improved electronic transportation, environmental friendliness, and low cost compared to single-component oxides for supercapacitors.2,14 Their typical representatives are MnCo2O4,2 CoMn2O4,15 NiCo2O4,16–20 CoMoO4,21 FeCo2O4,22 and ZnCo2O4.23,24 Especially, one-dimensional (1D) nanofibers own a large surface area and small particle size which can produce more active interfacial sites.2,25 However, the reduced particle size with high surface energy also undergoes obvious agglomeration during its electrochemical cycling.14,25 Therefore, how to make full use of active materials at the nanoscale still remains a great challenge.
Graphene,26,27 well known as a two dimensional (2D) “aromatic” monolayer of carbon material with high surface area, excellent electrical conductivity, outstanding mechanical flexibility, and high chemical stability, has been regarded as superior electrode materials for the development of supercapacitors. However, due to van der Waals interactions between neighbouring sheets, graphene sheets usually suffer from serious agglomeration and restacking during electrochemical process, which reduce its effective surface area and decrease its EDLC energy storage. Fortunately, graphene oxide (GO), produced by chemically exfoliated method, is rich in oxygen-containing functional groups for the growth and anchoring of nanomaterials, such as metal oxide nanoparticles and electrochemically active polymers, etc.3,6,28–30 The strong coupling between metal oxide nanoparticles and graphene sheets in a confined 2D manner creates a composite with unique structural features, synergistic physical and electrochemical properties. As for supercapacitors, graphene sheets can suppress agglomeration of metal oxide nanoparticles, and thus ensure their complete reaction during the electrochemical redox process for enhancing pseudocapacitance.6,14 At the same time, to load metal oxide nanoparticles can also decrease the agglomeration of graphene sheets, and consequently provides a higher electrochemical active surface area for increasing EDLC energy storage. The pseudocapacitance from metal oxides and the EDLC energy storage from graphene sheets contribute to the total capacitance, and therefore, graphene-based materials have a great potential for supercapacitor application.
In this study, we have prepared a new type of graphene sandwiched Mn0.5Co2.5O4 nanofibers (Mn0.5Co2.5O4@G) composite. The method involves hydrothermal approach to synthesize of uniform manganese cobalt oxide nanofibers on graphene sheets, followed by a low thermal annealing in air to form Mn0.5Co2.5O4@G composite. The unique sandwich-like structure can effectively prevent the agglomeration of Mn0.5Co2.5O4 nanofibers and restacking of graphene sheets, therefore enhancing the electrode performances and activities. As a result, our realized Mn0.5Co2.5O4@G shows an excellent capacitive behaviour used as an electrode material for supercapacitors.
2. Experimental
2.1 Materials
The basic method of preparation for GO sheets from natural graphite flakes is a modified Hummers method.31,32 Manganese acetate tetrahydrate (Mn(CH3COO)2·4H2O), cobalt acetate tetrahydrate (Co(CH3COO)2·4H2O), activated carbon, graphene and ethanol were bought from the Aladdin Chemistry Co., Ltd. Unless stated elsewhere, all reagents were of analytical grade and used as received. The aqueous solutions were prepared with distilled water (DI water).2.2 Preparation of Mn0.5Co2.5O4@G composite
The formation processes of manganese cobalt oxide nanofibers are based on a hydrothermal treatment, followed by a thermal decomposition process. In a typical procedure, 2.5 mmol Mn(CH3COO)2·4H2O and 5.0 mmol Co(CH3COO)2·4H2O were added to a mixed solvent of 20 mL graphene oxide (GO, 6 mg mL−1) aqueous dispersion and 20 mL ethanol, then ultrasonicated for 30 min to form a homogeneous suspension. The mixture was transferred into a Teflon-lined autoclave and hydrothermally treated at 180 °C for 12 h. After cooling to room temperature, the resulting stable black product was collected by centrifugation and washed with ethanol and DI water for several times. Subsequently, the precursor product was freeze-dried for 24 h, followed by annealing at 200 °C in air for 30 min with a heating rate of 5.0 °C min−1 to obtain a composite structure with crystalline Mn0.5Co2.5O4 nanofibers on graphene, as denoted by Mn0.5Co2.5O4@G. It is really difficult to synthesize Mn0.5Co2.5O4 without graphene to compare with the Mn0.5Co2.5O4@G composite. The functional groups (e.g. –COOH, –OH) on the GO surfaces may effect on the formation of Mn0.5Co2.5O4. For comparison, MnCo2O4 compound was synthesized using the same synthesized procedures without adding GO, and changing the annealing temperature to 400 °C for 2 h.2.3 Characterization
The morphologies of as-prepared samples were observed using a FEI SIRION 200 field emission scanning electron microscope (FESEM) at an acceleration voltage of 5 kV. Their fine structures were characterized by a high resolution transmission electron microscopy (HRTEM), energy-dispersive X-ray spectrum (EDS) mapping and selected area electron diffraction (SAED) patterns on a JEOL 2100 microscopy at an acceleration voltage of 200 kV. X-ray diffraction (XRD) was measured by a Rigaku Bruker D8 advance with Cu Kα radiation (λ = 1.5418 Å) at a generator voltage of 40 kV and a generator current of 50 mA with a scanning speed of 5° min−1. The elemental composition was determined by an inductively coupled plasma atomic emission spectroscopy (ICP-AES, iCAP 6000 Radial), an elemental analysis (EA, Vario-EL Cube) and X-ray photoelectron spectroscopy (XPS, Kratos AXIS Ultra DLD). For XPS measurements, all of the binding energies (BEs) in this XPS analysis were corrected for specimen charging by referencing them to the C 1s peak (set at 284.6 eV). The thermal behaviour of the samples was investigated by the technique of thermogravimetric analyzer-derivative thermogravimetry (TGA, Pyris 1) measured at a heating rate of 5 °C min−1 in air.2.4 Electrochemical measurements
For the three-electrode electrochemical measurements, the working electrode was prepared by mixing the electroactive material, Super-P, and polytetrafluoroethylene (PTFE) binder in a weight ratio of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10. The slurry was pasted to nickel foam, and dried at 80 °C overnight under vacuum. The area of the active material on nickel foam for each electrode was about 1.0 cm × 1.0 cm. The supercapacitor behaviour was tested with the CHI 760D electrochemical workstation in an aqueous KOH electrolyte (6.0 M) with a three-electrode cell where Pt foil serves as the counter electrode and Ag/AgCl acts as the reference electrode. Cyclic voltammetry (CV), galvanostatic charge/discharge and electrochemical impedance spectroscopy (EIS) were measured on the CHI 760D electrochemical workstation. For EIS measurements a sine wave with amplitude of 5.0 mV was applied from the frequency of 100 kHz to 0.01 Hz. Then, the code Z view was used to fit the impedance spectra to the proposed equivalent circuit.
10. The slurry was pasted to nickel foam, and dried at 80 °C overnight under vacuum. The area of the active material on nickel foam for each electrode was about 1.0 cm × 1.0 cm. The supercapacitor behaviour was tested with the CHI 760D electrochemical workstation in an aqueous KOH electrolyte (6.0 M) with a three-electrode cell where Pt foil serves as the counter electrode and Ag/AgCl acts as the reference electrode. Cyclic voltammetry (CV), galvanostatic charge/discharge and electrochemical impedance spectroscopy (EIS) were measured on the CHI 760D electrochemical workstation. For EIS measurements a sine wave with amplitude of 5.0 mV was applied from the frequency of 100 kHz to 0.01 Hz. Then, the code Z view was used to fit the impedance spectra to the proposed equivalent circuit.
For the asymmetric supercapacitor test, an activated carbon (AC) electrode was prepared by mixing the activated carbon, carbon black (Super-P), and polytetrafluoroethylene (PTFE) binder in a weight ratio of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 and pasting onto the nickel foam in 1.0 cm × 1.0 cm. Then an aqueous asymmetric supercapacitor device was fabricated in a commercial coin cell (LIR 2032) by using Mn0.5Co2.5O4@G positive and AC negative electrodes, where one slice of cellulose paper was used as the separator, and 6 M KOH solution was used as the electrolyte. The potential window and loading mass ratio of active materials (Mn0.5Co2.5O4@G
10 and pasting onto the nickel foam in 1.0 cm × 1.0 cm. Then an aqueous asymmetric supercapacitor device was fabricated in a commercial coin cell (LIR 2032) by using Mn0.5Co2.5O4@G positive and AC negative electrodes, where one slice of cellulose paper was used as the separator, and 6 M KOH solution was used as the electrolyte. The potential window and loading mass ratio of active materials (Mn0.5Co2.5O4@G![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AC) were optimized by a set of CV curves, and all electrochemical experiments were performed at room temperature.
AC) were optimized by a set of CV curves, and all electrochemical experiments were performed at room temperature.
3. Results and discussion
The overall synthetic strategy of the Mn0.5Co2.5O4@G composite is illustrated in Fig. 1. First, graphene oxide (GO) sheets aqueous suspension was dispersed into an ethanol solution containing metal acetate (Mn(CH3COO)2·4H2O and Co(CH3COO)2·4H2O) as the precursor of manganese cobalt/reduced graphene oxide (MnCo@rGO precursor), followed by a hydrothermal treatment. Because the metal acetate has a strong tendency to the polycondensation reaction during hydrothermal process, hexacoordinated metal ions (Mn2+ and Co2+) can form manganese cobalt oxide nanofibers.33,34 Further, abundant carboxyl groups from metal acetate and strong coordination between metal ions and functional groups (e.g. –COOH, –OH) on the GO surfaces can direct the formation of manganese cobalt oxide nanofibers with a uniform morphology.35,36 In addition, as reported, because of the shielding effect of ethanol, the OH− or H+ activity in the reverse reaction was reduced, which facilitated the formation of highly aligned nanofibers on and between graphene sheets.2,37,38 Finally, the obtained MnCo@rGO precursor was converted into Mn0.5Co2.5O4@G composite by a low temperature heat treatment (200 °C) so as to achieve a complete morphology retention. | ||
| Fig. 1 Schematic illustration of the formation process of the sandwich-like Mn0.5Co2.5O4@G composite. | ||
Thermogravimetric analysis (TGA) was first carried out to examine the thermal decomposition behaviour of the MnCo@rGO precursor (Fig. S1, ESI†). A major weight loss was observed in the TGA curves between 220 and 350 °C, and we chose a low temperature of 200 °C to produce our sample in here. The X-ray diffraction (XRD) pattern of the final composite is similar to the MnCo2O4 (JCPDS no. 23-1237) as we synthesized, and also similar to the standard patterns of Co3O4 and Mn2O3, indicating that the mixed manganese cobalt oxide composite grows on the graphene with the similar lattice constants (Fig. 2). The Co/Mn atomic ratio for the manganese cobalt oxide composite of about 5, was confirmed by the inductively coupled plasma atomic emission spectroscopy (ICP-AES) confirms (Table S1†). In order to obtain more detailed information about elemental composition and oxidation state of the composite, the corresponding results measured by X-ray photoelectron spectroscopy (XPS) were presented in Fig. S2.† It is obvious that the peaks in our full survey reveal the existence of Mn 2p, Co 2p, O 1s, and C 1s (Fig. S2a†). The de-convolution of the Mn 2p peaks shows the atoms in 2p1/2 and 2p3/2 electronic configurations at 653.6 and 641.8 eV, respectively, indicating the existence of Mn3+ and Mn2+ cations (Fig. S2b†).25,39,40 Similarly, the Co 2p spectra for the composite also consist of two spin–orbit doublets, i.e. 2p1/2 and 2p3/2 states at 797.3 and 780.8 eV, respectively. The two shakeup satellites denoted as “Sat.”, characteristic of Co3+ and Co2+ cations (Fig. S2c†).25,39–41 The O 1s shows a large M–O–M peak at 529.8 eV which is indicative of most of the oxygen in the lattice (M refers to Mn or Co, Fig. S2d†).39 The C 1s of GO shows two peaks at 283.9 and 286.2 eV, corresponding to C–C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and C–OX (epoxy (X = C) and hydroxyl groups (X = H)), respectively. In comparison with the C 1s of Mn0.5Co2.5O4@G, it is obvious that the signal attributed to the C–OH groups is decreased significantly in the heat-treated sample.42,43 This observation indicates that oxygen-containing functional groups on the GO sheets are partially removed upon thermal treatment, which is beneficial for the electrical conductivity (Fig. S2e and f†). The Co/Mn atomic ratio analyzed by XPS basically accords with the ICP-AES result (Table S1†). Therefore, it is concluded that the manganese cobalt oxide composite fabricated on the graphene has a chemical composition of Mn0.5Co2.5O4. Further, the mass fraction of graphene sheets in the Mn0.5Co2.5O4@G composite is about 43.5 wt% which is determined by an elemental analysis (EA) measurement (Table S1†).
C and C–OX (epoxy (X = C) and hydroxyl groups (X = H)), respectively. In comparison with the C 1s of Mn0.5Co2.5O4@G, it is obvious that the signal attributed to the C–OH groups is decreased significantly in the heat-treated sample.42,43 This observation indicates that oxygen-containing functional groups on the GO sheets are partially removed upon thermal treatment, which is beneficial for the electrical conductivity (Fig. S2e and f†). The Co/Mn atomic ratio analyzed by XPS basically accords with the ICP-AES result (Table S1†). Therefore, it is concluded that the manganese cobalt oxide composite fabricated on the graphene has a chemical composition of Mn0.5Co2.5O4. Further, the mass fraction of graphene sheets in the Mn0.5Co2.5O4@G composite is about 43.5 wt% which is determined by an elemental analysis (EA) measurement (Table S1†).
 | ||
| Fig. 2 XRD patterns of the MnCo@rGO precursor, Mn0.5Co2.5O4@G, MnCo2O4, and standard patterns of Mn2O3 and Co3O4. | ||
Scanning electron microscope (SEM) images of the MnCo@rGO precursor and Mn0.5Co2.5O4@G composite are shown in Fig. 3. Similar to the hydrothermal MnCo@rGO precursor (Fig. 3a), the nanofibers are uniformly deposited on graphene sheets after an annealing process in air (Fig. 3c). The cross-section SEM images in Fig. 3b and d reveal that well-distributed nanofibers are homogenously sandwiched in graphene sheets, forming a sandwiched composite structure, in which one-dimensional nanofibers and two-dimensional graphene are integrated together.
To further characterize the morphologies, high resolution transmission electron microscopy (HRTEM) and energy-dispersive X-ray spectrum (EDS) mapping analysis are performed and the results are shown in Fig. 4. The Mn0.5Co2.5O4 nanofibers are characterized by a few micrometers in length with their diameters ranging from 15 to 17 nm (Fig. 4a and b). A distinct set of visible lattice fringes with an inter-planar spacing of 0.46 nm is clearly observed from Fig. 4c, corresponding to the (111) crystal planes, which is consistent with the XRD result. The selected-area electron diffraction (SAED) pattern also shows as the inset, and further indicates the polycrystallinity of cobalt manganese oxides. The HRTEM image further manifests the intimate contact and intertwined array of the Mn0.5Co2.5O4 nanofibers on graphene sheets. Fig. 4d shows the elemental mapping results for the distribution of Mn, Co, O and C elements based on an individual Mn0.5Co2.5O4@G composite sheet. The sandwiched composite demonstrates a uniform elemental distribution.
The electrochemical performance of Mn0.5Co2.5O4@G as an electrode material of supercapacitor was evaluated using a conventional three-electrode system with Ag/AgCl as the reference electrode, Pt foil as the counter electrode, and 6 M KOH solution as the electrolyte.28,44 Fig. 5a shows the CV curves with various scan rates of 5, 10, 20, and 50 mV s−1 in the potential window of 0 to 0.45 V vs. Ag/AgCl electrode. The asymmetrical CV curves can be attributed to the combined EDLC and pseudocapacitance contributions to the total capacitance.3,45,46 At the low scan rate (5 mV s−1), Mn0.5Co2.5O4@G exhibits slight redox peaks, mainly resulted from the faradaic redox reactions related to M–O/M–O–OH, where M refers to the Mn or Co ions.47–49 With increasing the sweep rates, the voltage difference between anodic and cathodic peak positions becomes slightly larger, but the shape of CV curve remains almost unchanged, suggesting that both outstanding electrochemical reversibility and excellent rate capability. Our galvanostatic discharge measurements at various current densities were also performed as plotted in Fig. 5b. The specific capacitance during the galvanostatic discharge processes is calculated by2,14
The specific capacitance derived from the galvanostatic discharge processes, as shown in Fig. 5c, is given by 1213, 897, 829, and 755 F g−1 at current densities of 5, 10, 15, and 20 A g−1, respectively. Even at the ultrahigh current densities of 30 and 40 A g−1, the specific capacitances 617 and 434 F g−1 are still maintained. The improved electrochemical activity, especially at high current densities, could be ascribed to the excellent electrical conductivity of Mn0.5Co2.5O4@G composite.2,25,50 The cycling performance of Mn0.5Co2.5O4@G electrode is further evaluated by continuous charge/discharge test at a current density of 10 A g−1, with the result plotted in Fig. 5d. It is seen that the specific capacitance increased during the first 1000 cycles and then stayed stable afterward. During the initial cycles, the electrode undergoes an electrochemical activation process to enhance the contact between active material and electrolyte, thus increasing the specific capacitance.2,44,47 The specific capacitance reaches a high value of 1044 F g−1 at the 1000th cycle and 980 F g−1 after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, corresponding to high capacitance retention of 93.8%. In order to demonstrate the advantage of the sandwiched structure, we prepared MnCo2O4 as electrode material for supercapacitors. The galvanostatic discharge curves and specific capacitances at various current densities are shown in Fig. S3a and b,† respectively. It is clear that the specific capacitances of MnCo2O4 electrode are lower than Mn0.5Co2.5O4@G electrode. The cycling performance of the MnCo2O4 is also shown in Fig. S3c.† With the discharge–charge current density of 10 A g−1, the specific capacitance of MnCo2O4 electrode is 370 F g−1 after 1000 cycles, in contrast, Mn0.5Co2.5O4@G reaches 1044 F g−1. The specific capacitance of Mn0.5Co2.5O4@G electrode is also superior to the previously reported hierarchical NiCo2O4 nanosheets (835 F g−1 at 2 A g−1),5 NiCo2O4 hollow spheres (914 F g−1 at 5 A g−1),46 graphene/polyaniline/MnO2 nanorods (512 F g−1 at 0.25 A g−1),3 MnO2/carbon nanotubes (MnO2/CNTs, 164 F g−1 at 10 A g−1),51 Co3O4 nanowire (106.7 F g−1 at 10 A g−1),4 and needlelike Co3O4/graphene (60 F g−1 at 2 A g−1).52 The EDLC from the graphene and the pseudocapacitance from the Mn0.5Co2.5O4 contribute to the total capacitance. All these results suggest that the sandwiched structure is a suitable architecture for supercapacitor electrode materials.
000 cycles, corresponding to high capacitance retention of 93.8%. In order to demonstrate the advantage of the sandwiched structure, we prepared MnCo2O4 as electrode material for supercapacitors. The galvanostatic discharge curves and specific capacitances at various current densities are shown in Fig. S3a and b,† respectively. It is clear that the specific capacitances of MnCo2O4 electrode are lower than Mn0.5Co2.5O4@G electrode. The cycling performance of the MnCo2O4 is also shown in Fig. S3c.† With the discharge–charge current density of 10 A g−1, the specific capacitance of MnCo2O4 electrode is 370 F g−1 after 1000 cycles, in contrast, Mn0.5Co2.5O4@G reaches 1044 F g−1. The specific capacitance of Mn0.5Co2.5O4@G electrode is also superior to the previously reported hierarchical NiCo2O4 nanosheets (835 F g−1 at 2 A g−1),5 NiCo2O4 hollow spheres (914 F g−1 at 5 A g−1),46 graphene/polyaniline/MnO2 nanorods (512 F g−1 at 0.25 A g−1),3 MnO2/carbon nanotubes (MnO2/CNTs, 164 F g−1 at 10 A g−1),51 Co3O4 nanowire (106.7 F g−1 at 10 A g−1),4 and needlelike Co3O4/graphene (60 F g−1 at 2 A g−1).52 The EDLC from the graphene and the pseudocapacitance from the Mn0.5Co2.5O4 contribute to the total capacitance. All these results suggest that the sandwiched structure is a suitable architecture for supercapacitor electrode materials.
To further understand the excellent electrochemical performance of the supercapacitor, the morphology of Mn0.5Co2.5O4@G composite after 5000 charge/discharge cycles was investigated (Fig. S4†). The SEM image clearly shows that the Mn0.5Co2.5O4 nanofibers remain highly dispersive on the graphene sheets and the sandwich-like structure is almost the same as before. The interfacial property of the composite electrode was also investigated by an electrochemical impedance spectroscopy (EIS) measurements after 1st, 1000th and 5000th cycle from the frequency of 100 kHz to 0.1 Hz at an open circuit potential with 5 mV ac perturbation. As shown in Fig. S5,† the impedance spectra of the Mn0.5Co2.5O4@G electrode are nearly identical in form with the semicircle at high frequency range and the straight line at low frequency range, indicating that superior long-term electrochemical stability is achieved. The corresponding equivalent circuit model is further supported to analyze the measured impedance spectra according to the literature.53 The solution resistance (Rs) and charge-transfer resistance (Rct) for the Mn0.5Co2.5O4@G electrode after 1st cycle are 1.45 and 10.36 Ω, respectively. After 1000th cycle, the charge-transfer resistance is 1.5 Ω, validating the electrode underwent an electrochemical activation process during the initial cycles. After 5000th cycle, it increases to 3.1 Ω. This low resistance brings an outstanding cycle stability.
To estimate the Mn0.5Co2.5O4@G electrode for practical application, an asymmetric supercapacitor device was fabricated in a commercial coin cell using the Mn0.5Co2.5O4@G as the positive electrode and the AC on nickel foam as the negative electrode with one slice of cellulose paper as the separator soaked in 6 M KOH solution electrolyte (denoted as Mn0.5Co2.5O4@G//AC). In order to obtain its maximum effectiveness, the potential window and loading mass ratio of active materials (Mn0.5Co2.5O4@G![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AC) are optimized via CV measurements. The CV curves at different potential windows at a scan rate of 10 mV s−1 are shown in Fig. S6a.† The asymmetric supercapacitor can afford a stable electrochemical window from 0.0 to 1.5 V, which is higher than the conventional AC based symmetric supercapacitors from 0.0 to 0.8 V in the aqueous electrolyte.54 The different loading mass ratios of Mn0.5Co2.5O4@G and AC for the asymmetric supercapacitors were also measured by the CV tests at a scan rate of 10 mV s−1 within the potential range of 0 to 1.5 V (Fig. S6b†). Based on a series of comparative tests, the loading mass ratio of 1
AC) are optimized via CV measurements. The CV curves at different potential windows at a scan rate of 10 mV s−1 are shown in Fig. S6a.† The asymmetric supercapacitor can afford a stable electrochemical window from 0.0 to 1.5 V, which is higher than the conventional AC based symmetric supercapacitors from 0.0 to 0.8 V in the aqueous electrolyte.54 The different loading mass ratios of Mn0.5Co2.5O4@G and AC for the asymmetric supercapacitors were also measured by the CV tests at a scan rate of 10 mV s−1 within the potential range of 0 to 1.5 V (Fig. S6b†). Based on a series of comparative tests, the loading mass ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 was chosen to balance the two electrodes' capacitance. In Fig. 6a, the CV curves at different scan rates exhibit pseudocapacitance, which arises from the Mn0.5Co2.5O4 nanofibers, and EDLC which supplied from graphene and AC, respectively.45,46 Galvanostatic discharge measurements (Fig. 6b) at different current densities were employed to calculate the specific capacitance of the asymmetric supercapacitor (Fig. 6c). The specific capacitance of the Mn0.5Co2.5O4@G//AC device can achieve 117.6, 86.4, 67.1, 54, 48 and 43.4 F g−1 at current densities of 0.2, 0.5, 1.0, 1.5, 2.0 and 5.0 A g−1, respectively. The energy and power densities (E and P), which are the two key factors for the practical applications of supercapacitors, are further calculated by14
2.5 was chosen to balance the two electrodes' capacitance. In Fig. 6a, the CV curves at different scan rates exhibit pseudocapacitance, which arises from the Mn0.5Co2.5O4 nanofibers, and EDLC which supplied from graphene and AC, respectively.45,46 Galvanostatic discharge measurements (Fig. 6b) at different current densities were employed to calculate the specific capacitance of the asymmetric supercapacitor (Fig. 6c). The specific capacitance of the Mn0.5Co2.5O4@G//AC device can achieve 117.6, 86.4, 67.1, 54, 48 and 43.4 F g−1 at current densities of 0.2, 0.5, 1.0, 1.5, 2.0 and 5.0 A g−1, respectively. The energy and power densities (E and P), which are the two key factors for the practical applications of supercapacitors, are further calculated by14
where C is the specific capacitance, ΔV is the voltage range, and Δt is the discharge time.
The results are presented as Ragone plots in Fig. 6d. The Mn0.5Co2.5O4@G//AC supercapacitor can achieve a high energy density of 36.8 W h kg−1 at a power density of 147.3 W kg−1. Even at a high power density of 3755.8 W kg−1, such device still remains a high energy density of 13.6 W h kg−1. The maximum energy density of the Mn0.5Co2.5O4@G//AC asymmetric supercapacitor is much higher than symmetrical AC//AC supercapacitors (2.8 W h kg−1), graphene//graphene supercapacitors (4.9 W h kg−1). (The information on the AC//AC and graphene//graphene symmetric supercapacitors are provided in the ESI Fig. S7.†). Also, it is much higher than symmetrical graphene/MnO2//graphene/MnO2 supercapacitors (8.1 W h kg−1),55 and MnO2//MnO2 supercapacitors (1.9 W h kg−1).56 In addition, it is much higher than those of asymmetric supercapacitors, such as NiCo2O4@MnO2//AC supercapacitors (35 W h kg−1),47 MnO2/CNTs//AC (13.3 W h kg−1),51 and Co3O4@Ni(OH)2//AC supercapacitors (30 W h kg−1).57 A long-term cycling test of the Mn0.5Co2.5O4@G//AC supercapacitors was conducted at a constant current density of 2 A g−1. As shown in Fig. 6e, the overall specific capacitance can remain about 75% of its initial value after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, demonstrating its outstanding cycling stability. When two supercapacitors are assembled in series and used for powering a 3 mm diameter red round light-emitting diode (LED), the device is lighted after charging to 1.5 V (Fig. 6f).
000 cycles, demonstrating its outstanding cycling stability. When two supercapacitors are assembled in series and used for powering a 3 mm diameter red round light-emitting diode (LED), the device is lighted after charging to 1.5 V (Fig. 6f).
The excellent electrochemical performance of the Mn0.5Co2.5O4@G composite can be attributed to the following aspects: first, the strong coupling between Mn0.5Co2.5O4 nanofibers and graphene in a confined sandwich-like structure can effectively prevent the agglomeration of Mn0.5Co2.5O4 nanofibers and restacking of graphene sheets, and consequently providing the active materials efficiently utilized. Second, highly conductive and flexible graphene benefits the stabilization of the composite structure and electrical conductivity during the volume-changeable cycling. Third, the nanodispersed Mn0.5Co2.5O4 can extend electrochemical reaction zone and improve the electrode kinetics. The excellent capability of delivering high power and energy densities makes the realized Mn0.5Co2.5O4@G composite very appealing as electrode materials for advanced supercapacitors.
4. Conclusions
In summary, we have developed a simple hydrothermal strategy for synthesis of uniform Mn0.5Co2.5O4@G composite, in which one-dimensional Mn0.5Co2.5O4 nanofibers are sandwiched in two-dimensional graphene. The composite manifests superior electrochemical performance as electrode materials for supercapacitors. The Mn0.5Co2.5O4@G electrodes demonstrate high specific capacitances of 1213 and 829 F g−1 at current densities of 5 and 15 A g−1, respectively. Even at the ultrahigh current densities of 30 and 40 A g−1, the specific capacitances 617 and 434 F g−1 have been also retained. Moreover, the Mn0.5Co2.5O4@G electrode retains 93.8% of its initial specific capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at the current density of 10 A g−1. For the device verification, an asymmetric supercapacitor of Mn0.5Co2.5O4@G//AC achieves high energy densities of 36.8 and 13.6 W h kg−1 at power densities of 147.3 and 3755.8 W kg−1, respectively. The proposed Mn0.5Co2.5O4@G composite proves very promising electrode material for the advanced supercapacitors.
000 cycles at the current density of 10 A g−1. For the device verification, an asymmetric supercapacitor of Mn0.5Co2.5O4@G//AC achieves high energy densities of 36.8 and 13.6 W h kg−1 at power densities of 147.3 and 3755.8 W kg−1, respectively. The proposed Mn0.5Co2.5O4@G composite proves very promising electrode material for the advanced supercapacitors.
Acknowledgements
This work was supported by the SJTU-UM project, the National Natural Science Foundation of China (No. 61171037 and 61431014). The authors also thank the Instrumental Analysis Center of Shanghai Jiao Tong University for the characterization of the samples.Notes and references
- X. Wang, C. X. Lu, H. F. Peng, X. Zhang, Z. K. Wang and G. K. Wang, J. Power Sources, 2016, 324, 188–198 CrossRef CAS.
- Y. A. Xu, X. F. Wang, C. H. An, Y. J. Wang, L. F. Jiao and H. T. Yuan, J. Mater. Chem. A, 2014, 2, 16480–16488 CAS.
- G. Q. Han, Y. Liu, L. L. Zhang, E. J. Kan, S. P. Zhang, J. Tang and W. H. Tang, Sci. Rep., 2014, 4, 4824–4831 Search PubMed.
- Z. X. Gu, R. F. Wang, H. H. Nan, B. Y. Geng and X. J. Zhang, J. Mater. Chem. A, 2015, 3, 14578–14584 CAS.
- G. X. Gao, H. B. Wu, S. J. Ding, L. M. Liu and X. W. Lou, Small, 2015, 11, 804–808 CrossRef CAS PubMed.
- Z. S. Wu, D. W. Wang, W. Ren, J. Zhao, G. Zhou and F. Li, Adv. Funct. Mater., 2010, 20, 3595–3602 CrossRef CAS.
- J. T. Zhang, J. Z. Ma, L. L. Zhang, P. Z. Guo, J. W. Jiang and X. S. Zhao, J. Phys. Chem. C, 2010, 114, 13608–13613 CAS.
- X. Long, Z. G. Zeng, E. J. Guo, X. B. Shi, H. J. Zhou and X. H. Wang, J. Power Sources, 2016, 325, 264–272 CrossRef CAS.
- C. P. Mao, S. G. Liu, L. Pang, Q. Sun, Y. Liu, M. W. Xu and Z. S. Lu, RSC Adv., 2016, 6, 5184–5191 RSC.
- W. Du, R. M. Liu, Y. W. Jiang, Q. Y. Lu, Y. Z. Fan and F. Gao, J. Power Sources, 2013, 227, 101–105 CrossRef CAS.
- Y. H. Xiao, S. J. Liu, F. Li, A. Q. Zhang, J. H. Zhao, S. M. Fang and D. Z. Jia, Adv. Funct. Mater., 2012, 22, 4052–4059 CrossRef CAS.
- C. Y. Foo, A. Sumboja, D. J. H. Tan, J. X. Wang and P. S. Lee, Adv. Energy Mater., 2014, 4, 1400236 CrossRef.
- Q. Li, Q. Wei, L. J. Xie, C. M. Chen, C. X. Lu, F. Y. Su and P. C. Zhou, RSC Adv., 2016, 6, 46548–46557 RSC.
- H. B. Wu, H. Pang and X. W. Lou, Energy Environ. Sci., 2013, 6, 3619–3626 CAS.
- J. Bhagwan, V. Sivasankaran, K. L. Yadav and Y. Sharma, J. Power Sources, 2016, 327, 29–37 CrossRef CAS.
- C. Z. Yuan, J. Y. Li, L. R. Hou, X. G. Zhang, L. F. Shen and X. W. Lou, Adv. Funct. Mater., 2012, 22, 4592–4597 CrossRef CAS.
- H. C. Chien, W. Y. Cheng, Y. H. Wang and S. Y. Lu, Adv. Funct. Mater., 2012, 22, 5038–5043 CrossRef CAS.
- R. J. Zou, K. B. Xu, T. Wang, G. J. He, Q. Liu, X. J. Liu, Z. Y. Zhang and J. Q. Hu, J. Mater. Chem. A, 2013, 1, 8560–8566 CAS.
- G. Q. Zhang and X. W. Lou, Adv. Mater., 2013, 25, 976–979 CrossRef CAS PubMed.
- Q. Lu, Y. P. Chen, W. F. Li, J. G. Chen, J. Q. Xiao and F. Jiao, J. Mater. Chem. A, 2013, 1, 2331–2336 CAS.
- S. H. Kazemi, M. Tabibpour, M. A. Kiani and H. Kazemi, RSC Adv., 2016, 6, 71156–71164 RSC.
- B. G. Zhu, S. C. Tang, S. Vongehr, H. Xie and X. K. Meng, ACS Appl. Mater. Interfaces, 2016, 8, 4762–4770 CAS.
- F. X. Bao, Z. Q. Zhang, X. Y. Liu and X. D. Zhao, RSC Adv., 2014, 4, 38073–38077 RSC.
- B. Liu, B. Y. Liu, Q. F. Wang, X. F. Wang, Q. Y. Xiang, D. Chen and G. Z. Shen, ACS Appl. Mater. Interfaces, 2013, 20, 10011–10017 Search PubMed.
- L. Yu, L. Zhang, H. B. Wu, G. Q. Zhang and X. W. Lou, Energy Environ. Sci., 2013, 6, 2664–2671 CAS.
- J. X. Zhu, T. Zhu, X. Z. Zhou, Y. Y. Zhang, X. W. Lou, X. D. Chen, H. Zhang, H. H. Hng and Q. Y. Yan, Nanoscale, 2011, 3, 1084–1089 RSC.
- Y. W. Zhu, S. Murali, W. W. Cai, X. S. Li, J. W. Suk, J. R. Potts and R. S. Ruoff, Adv. Mater., 2010, 22, 3906–3924 CrossRef CAS PubMed.
- S. Li, D. Q. Wu, C. Cheng, J. Z. Wang, F. Zhang, Y. Z. Su and X. L. Feng, Angew. Chem., Int. Ed., 2013, 52, 12105–12109 CrossRef CAS PubMed.
- J. Z. Wang, P. Liu, Y. S. Huang, J. J. Jiang, S. Han, D. Q. Wu and X. L. Feng, RSC Adv., 2014, 4, 57869–57874 RSC.
- Z. H. Pan, Y. C. Qiu, J. Yang, F. M. Ye, Y. J. Xu, X. Y. Zhang, M. N. Liu and Y. G. Zhang, Nano Energy, 2016, 26, 610–619 CrossRef CAS.
- W. S. Hummers and R. E. Offeman, J. Am. Chem. Soc., 1958, 80, 1339 CrossRef CAS.
- S. B. Yang, X. L. Feng and K. Müllen, Adv. Mater., 2011, 23, 3575–3579 CrossRef CAS PubMed.
- N. A. Barakat, M. A. Abdelkareem, A. Yousef, S. S. Al-Deyab, M. El-Newehy and H. Y. Kim, Int. J. Hydrogen Energy, 2013, 38, 3387–3394 CrossRef CAS.
- A. Yousef, N. A. Barakat, T. Amna, A. R. Unnithan, S. S. Al-Deyab and H. Y. Kim, J. Lumin., 2012, 132, 1668–1677 CrossRef CAS.
- G. X. Gao, H. B. Wu and X. W. Lou, Adv. Energy Mater., 2014, 4, 1400422–1400427 CrossRef.
- H. B. Lu, S. M. Wang, L. Zhao, J. C. Li, B. H. Dong and Z. X. Xu, J. Mater. Chem., 2011, 21, 4228–4234 RSC.
- C. C. Li, L. Mei, L. B. Chen, Q. H. Li and T. H. Wang, J. Mater. Chem., 2012, 22, 4982–4988 RSC.
- S. H. Liu, H. P. Jia, L. Han, J. L. Wang, P. F. Gao, D. D. Xu, J. Yang and S. A. Che, Adv. Mater., 2012, 24, 3201–3204 CrossRef CAS PubMed.
- D. U. Lee, B. J. Kim and Z. W. Chen, J. Mater. Chem. A, 2013, 1, 4754–4762 CAS.
- J. F. Li, S. L. Xiong, X. W. Li and Y. T. Qian, Nanoscale, 2013, 5, 2045–2054 RSC.
- S. L. Xiong, J. S. Chen, X. W. Lou and H. C. Zeng, Adv. Funct. Mater., 2012, 22, 861–871 CrossRef CAS.
- G. B. Zeng, N. Shi, M. Hess, X. Chen, W. Cheng, T. X. Fan and M. Niederberger, ACS Nano, 2015, 9, 4227–4235 CrossRef CAS PubMed.
- I. K. Moon, J. Lee, R. S. Ruoff and H. Lee, Nat. Commun., 2010, 1, 73–78 Search PubMed.
- B. G. Choi, M. H. Yang, W. H. Hong, J. W. Choi and Y. S. Huh, ACS Nano, 2012, 6, 4020–4028 CrossRef CAS PubMed.
- H. Y. Xian, T. J. Peng, H. J. Sun and J. D. Wang, Nano-Micro Lett., 2015, 7, 17–26 CrossRef CAS.
- L. F. Shen, L. Yu, X. Y. Yu, X. G. Zhang and X. W. Lou, Angew. Chem., Int. Ed., 2015, 54, 1868–1872 CrossRef CAS PubMed.
- K. B. Xu, W. Y. Li, Q. Liu, B. Li, X. J. Liu, L. An, Z. G. Chen, R. J. Zou and J. Q. Hu, J. Mater. Chem. A, 2014, 2, 4795–4802 CAS.
- Y. H. Li, L. J. Cao, L. Qiao, M. Zhou, Y. Yang, P. Xiao and Y. H. Zhang, J. Mater. Chem. A, 2014, 2, 6540–6548 CAS.
- C. Lamiel, V. H. Nguyen, C. Roh, C. K. Kang and J. J. Shim, Electrochim. Acta, 2016, 210, 240–250 CrossRef CAS.
- S. Han, J. Z. Wang, S. Li, D. Q. Wu and X. L. Feng, J. Mater. Chem. A, 2014, 2, 6174–6179 CAS.
- L. Li, Z. A. Hu, N. An, Y. Y. Yang, Z. M. Li and H. Y. Wu, J. Phys. Chem. C, 2014, 118, 22865–22872 CAS.
- Q. Guan, J. L. Cheng, B. Wang, W. Ni, G. F. Gu, X. D. Li, L. Huang, G. C. Yang and F. D. Nie, ACS Appl. Mater. Interfaces, 2014, 6, 7626–7632 CAS.
- J. W. Xiao and S. H. Yang, J. Mater. Chem., 2012, 22, 12253–12262 RSC.
- X. Z. Yu, B. A. Lu and Z. Xu, Adv. Mater., 2014, 26, 1044–1051 CrossRef CAS PubMed.
- Z. J. Fan, J. Yan, T. Wei, L. J. Zhi, G. Q. Ning, T. Y. Li and F. Wei, Adv. Funct. Mater., 2011, 21, 2366–2375 CrossRef CAS.
- V. Khomenko, E. Raymundo-Pinero and F. Beguin, J. Power Sources, 2006, 153, 183–190 CrossRef CAS.
- C. H. Tang, X. S. Yin and H. Gong, ACS Appl. Mater. Interfaces, 2013, 5, 10574–10582 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra21281f |
| This journal is © The Royal Society of Chemistry 2016 |