Magnetic field induced push–pull motility of liquibots†
Abstract
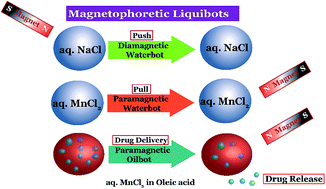
Liquid droplets loaded with paramagnetic or diamagnetic salts, namely liquibots, showed controlled migration inside a fluid medium and on slippery solid surfaces under remote magnetic guidance. The water or oilbots of size ranging from a millimetre to a few microns showed facile attraction, repulsion, division, and coalescence when guided by a magnetic field. The speed of the liquibots could be tuned by varying the size, salt-loading, and magnetic field strength. While the paramagnetic liquibot migrated towards a magnet with a velocity as high as ∼8 body length per s, the diamagnetic one migrated away from the field with a maximum velocity of ∼1 body length per s. The liquibots transported and delivered commercially available drugs to targeted locations showing their potential as drug-delivery vehicles. Remarkably, the experiments showed the utility of the liquibots in digital microfluidics because they moved easily on slippery solid surfaces. For example, a waterbot was split into many droplets on an oil coated solid surface before forming the patterns resembling polygons under magnetic guidance. Further, the liquibot based Packman™ game could also be played with the help of magnetic guidance. The extent of control demonstrated on the motions of the remotely guided liquibots could be useful in diverse futuristic applications including drug-transport, digital-microfluidics, and droplet-electronics.

 Please wait while we load your content...
Please wait while we load your content...