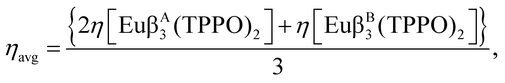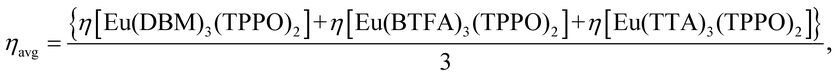Substantial luminescence enhancement in ternary europium complexes by coordination of different ionic ligands†
Anderson I. S. Silva,
Vanessa F. C. Santos,
Nathalia B. D. Lima,
Alfredo M. Simas* and
Simone M. C. Gonçalves*
Departamento de Química Fundamental, CCEN, Universidade Federal de Pernambuco, 50590-470, Recife, Pernambuco, Brazil. E-mail: simas@ufpe.br; simone@ufpe.br
First published on 15th September 2016
Abstract
We demonstrate in a general and comprehensive manner that a substantial enhancement of luminescence in europium complexes can be achieved by increasing ionic ligand diversity. For the complexes studied, the measured boosts in the quantum efficiency of luminescence obtained using this strategy ranged from a minimum of 100% to a maximum of 543%. We formalize this concept by means of mathematical inequalities which describe the fact that either the quantum efficiency η, or the radiative decay rate Arad, of a mixed ligand complex will be larger than the average of the same property for the respective same-ligand complexes. We further introduce the concept of structure hardening by ligands, by linking it quantitatively for the first time with the value of the non-radiative decay rate Anrad, and interpret it in terms of the lowest vibrational frequency of the complex. In light of all these concepts and results, we conclude that luminescence can be boosted in europium complexes by increasing ligand diversity, which, in turn, should be ideally obtained with the help of hardener ligands. This general and comprehensive strategy aims at increasing Arad while simultaneously decreasing Anrad.
1. Introduction
Highly luminescent europium complexes can be used in several applications. For example: a LED was obtained from the complex [Eu2(2,7-BTFDBC)(DBM)4(phen)2] which acts as a red phosphor;1 europium carbon nanotubes were used as luminophores in an electrochemiluminescent aptasensor for thrombin;2 the formation of complexes was monitored by recording luminescence information from the prepared solid compounds.3 Moreover, complexes can be used as efficient red organic light-emitting devices, OLEDs;4 as portable luminescent chemosensors for naked eye Cu2+ detection;5 as sensors for the determination of pH;6 in studies of interactions with DNA;7 in accurate tumor-target bioimaging through specific in vivo biosynthesis;8 for bioimaging;9 and in in situ monitoring via europium emission of the photorelease of the antitumor drug cisplatin.10 In particular, ternary β-diketonate europium complexes can be used, for example, as efficient red organic electroluminescent devices;11 as luminescent pure-red-emitting complexes for full solution-processed OLEDs;12 in mitochondria, as targetable time-gated luminescence probes for singlet oxygen;13 as temperature sensors, in UV light detection, and as laser devices.14 Recently, water-soluble luminescent hybrid composites consisting of oligosilsesquioxanes and lanthanide complexes were obtained.15 These composites exhibited an effective switch-off fluorescence response with high sensing selectivity and sensitivity toward Cu2+ over other common metal ions in aqueous media.15Theoretical tools can predict important aspects of the europium species involved, such as: geometry optimization,16 thermodynamic properties,16 and a treatment of luminescence properties.17 The geometry optimization of europium complexes can be performed, for example, with the Sparkle/RM1 model,18 using the quantum chemical program MOPAC 2016.19 Luminescence properties such as the quantum efficiency, η, and the Judd–Ofelt parameters Ω2, Ω4, and Ω6, can be calculated by the LUMPAC (LUMinescence PACkage) software.17 Through LUMPAC, it is also possible to perform a chemical partition of the global photophysical property Arad (ref. 20) in terms of ligand effects which allows a chemical interpretation of the role of each ligand coordinated to the europium ion in the luminescence phenomenon.
Temperature affects luminescence of europium complexes significantly, with low temperatures leading to higher luminescence.21 Two competitive mechanisms occur when the excited state of the metal decays: radiative processes and non-radiative processes. The radiative processes are the desired ones and occur when the excited state of the trivalent europium ion, 5D0, decays to the lower electronic states of the europium ion: 7FJ, with J ranging from 0 to 6. The sum of all seven radiative decay rates is known simply as Arad. The other decay pathway is the non-radiative one, characterized by the non-radiative decay rate, Anrad. The total decay rate from the 5D0 excited state, Atot, is equal to Arad + Anrad. Seemingly, Anrad competes with Arad and, therefore, whenever the radiationless processes are hindered, Arad usually tends to increase and more luminescence ensues. For example, when water is directly coordinated to the europium ion, their low energy vibrations tend to resonate with vibrations from the solvent molecules in the first solvation layers, thus robbing energy from the complex leading to low luminescence. Raising the vibration energies of the coordinated water molecules by deuterating them usually enhances luminescence.22
Recently, we introduced a comprehensive strategy to boost the luminescence of europium complexes by increasing the diversity of ligands around the europium trivalent ion.23 The strategy states that a luminescence property P, where P stands for either the quantum yield Φ, the quantum efficiency η, or the radiative decay rate Arad, of a mixed ligand complex, would be larger than the average of the same property for the respective same-ligand complexes. The strategy can be quantified by the following inequality:23
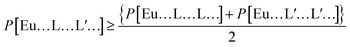 | (1) |
 | (2) |
Previously,23 we showed eqn (1) to be true for mixed non-ionic ligand complexes only, for all combinations of the non-ionic ligands TPPO (triphenylphosphine oxide), DBSO (dibenzyl sulfoxide), and PTSO (p-tolyl sulfoxide), for all ternary complexes of the β-diketonates TTA (1-(2-thenoyl)-3,3,3-trifluoroacetone), and BTFA (4,4,4-trifluoro-1-phenyl-1,3-butanedione). For the cases studied, the observed % BoostΦ in the quantum yield were as high as 81%.23
In this article, we set forth a generalization of eqn (1) and prove it to be also true for ternary mixed ionic ligand europium complexes, and thus valid without exception (at least for all cases studied so far16,23). In order to show the usefulness of eqn (1) for the design of luminescent complexes, we further replace the double TPPO ligands in the mixed ionic ligand complex, by the non-ionic bidentate ligands BIPY (2,2-bipyridyl) and PHEN (1,10-phenanthroline) in order to reduce the non-radiative decay rate due to their increased rigidities, thus enhancing luminescence even more, when compared to the double TPPO complex. We further construe the concept of structure hardening by ligands via their effects on the non-radiative decay rate Anrad, and advance an interpretation of rigidity in terms of the lowest vibrational frequency of the complex. Finally, we use theoretical tools to obtain, for each of the complexes, the optimized geometries; values for the parameters Ω2, Ω4, and Ω6; the quantum efficiency η; Arad and Anrad; and a chemical interpretation of the effect of each ligand coordinated to the europium ion on the global photophysical property Arad.20
2. Results and discussion
2.1. Enhancement of the luminescence properties
The complexes synthesized in this article were Eu(β-diketonate)2(β-diketonate′)(TPPO)2, where β-diketonate can be either DBM, BTFA, or TTA, and Eu(DBM)(BTFA)(TTA)(TPPO)2. Finally, we also synthesized Eu(DBM)(BTFA)(TTA)(BIPY) and Eu(DBM)(BTFA)(TTA)(PHEN). The chemical structures of these ligands are shown in Fig. 1.In order to quantify the effect of mixing ionic ligands on the luminescence of europium complexes, we now present the following generalization of our conjecture, eqn (1), to the case of ternary all mixed ionic ligand complexes:
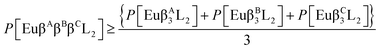 | (3) |
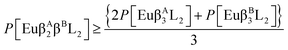 | (4) |
Table 1 shows the luminescence properties of all complexes considered in this article: the first three presented earlier16,23 and the next nine synthesized in this article. The lifetime of complex Eu(DBM)3(TPPO)2 could not be observed in chloroform solution at room temperature, most likely due to the elevated symmetry of both DBM and TPPO ligands, as well as of the complex itself, where the two TPPOs lie opposite to each other as revealed by our Sparkle/RM1 calculation; a result which is in tune with X-ray crystallographic measurements24,25 for complexes Eu(TTA)3(TPPO)2 and Eu(BTFA)3(TPPO)2, when the TPPOs always appear opposite to each other. This dual ligand and coordination symmetry of Eu(DBM)3(TPPO)2 is what probably reduced the luminescence lifetime of Eu(DBM)3(TPPO)2 to an essentially unobservable low value at room temperature. So much so, that a lifetime for this complex could only be measured by other researchers in the solid state and only at liquid nitrogen temperatures.21 For the purposes of this article, therefore, we consider the quantum efficiency of this complex at room temperature to be essentially zero.
| Complex | τ (ms) | Atot (s−1) | Arad (s−1) | Anrad (s−1) | η (%) |
|---|---|---|---|---|---|
| Eu(DBM)3(TPPO)2 (ref. 16) | — | — | 335 | — | — |
| Eu(TTA)3(TPPO)2 (ref. 23) | 0.350 | 2857 | 796 | 2061 | 21 |
| Eu(BTFA)3(TPPO)2 (ref. 23) | 0.367 | 2725 | 919 | 1806 | 21 |
| Eu(DBM)2(TTA)(TPPO)2 | 0.415 | 2410 | 1082 | 1328 | 45 |
| Eu(TTA)2(DBM)(TPPO)2 | 0.424 | 2359 | 1052 | 1307 | 45 |
| Eu(DBM)2(BTFA)(TPPO)2 | 0.423 | 2364 | 1015 | 1349 | 43 |
| Eu(BTFA)2(DBM)(TPPO)2 | 0.473 | 2114 | 1131 | 983 | 53 |
| Eu(TTA)2(BTFA)(TPPO)2 | 0.435 | 2584 | 1093 | 1491 | 42 |
| Eu(BTFA)2(TTA)(TPPO)2 | 0.458 | 2184 | 1058 | 1126 | 48 |
| Eu(DBM)(BTFA)(TTA)(TPPO)2 | 0.434 | 2303 | 1034 | 1269 | 45 |
| Eu(DBM)(BTFA)(TTA)(PHEN) | 0.645 | 1550 | 864 | 686 | 56 |
| Eu(DBM)(BTFA)(TTA)(BIPY) | 0.523 | 1912 | 894 | 1018 | 47 |
Now, we are in position to verify the validity of eqn (3) and (4), a generalization of our previously proposed conjecture to mixed ionic ligand ternary complexes.
The first six lines of Table 2 show the values of the average quantum efficiencies,
| Complex | ηavg (%) | η (%) | % Boostη |
|---|---|---|---|
| Eu(DBM)2(TTA)(TPPO)2 | 7 | 45 | 543% |
| Eu(TTA)2(DBM)(TPPO)2 | 14 | 45 | 221% |
| Eu(DBM)2(BTFA)(TPPO)2 | 7 | 43 | 514% |
| Eu(BTFA)2(DBM)(TPPO)2 | 14 | 53 | 279% |
| Eu(BTFA)2(TTA)(TPPO)2 | 21 | 48 | 129% |
| Eu(TTA)2(BTFA)(TPPO)2 | 21 | 42 | 100% |
| Eu(DBM)(BTFA)(TTA)(TPPO)2 | 14 | 45 | 221% |
The last line on Table 2 shows the value of the average
As a result, the % Boostηs obtained for the mixed ionic ligand complexes were very large, ranging from 100% to 543%; values that are much larger than the % Boostηs obtained in our previous article on mixed non-ionic ligand complexes, in which the maximum % Boostη obtained was 77% for the Eu(TTA)3(PTSO,TPPO) complex.23 Indeed, mixing ionic ligands seems to be an even more effective strategy to enhance luminescence in ternary europium complexes.
Table 3 presents equivalent data proving that eqn (3) and (4) are also valid for Arad, another luminescent property. The % BoostArad values range from 34% to 121%.
| Complex | Arad,avg (s−1) | Arad (s−1) | % BoostArad |
|---|---|---|---|
| Eu(DBM)2(TTA)(TPPO)2 | 489 | 1082 | 121% |
| Eu(TTA)2(DBM)(TPPO)2 | 642 | 1052 | 64% |
| Eu(DBM)2(BTFA)(TPPO)2 | 530 | 1015 | 92% |
| Eu(BTFA)2(DBM)(TPPO)2 | 724 | 1131 | 56% |
| Eu(BTFA)2(TTA)(TPPO)2 | 878 | 1093 | 24% |
| Eu(TTA)2(BTFA)(TPPO)2 | 789 | 1058 | 34% |
| Eu(DBM)(BTFA)(TTA)(TPPO)2 | 683 | 1034 | 51% |
Both TTA and BTFA are already asymmetric ionic ligands, as well as good antennae, and, besides that, somewhat similar to one another. Hence, the ensuing observed % Boosts, both in η and in Arad, obtained by mixing these ionic ligands tend to be smaller than by mixing either of them with DBM. So much so that the largest η of 53% was observed for complex Eu(BTFA)2(DBM)(TPPO)2 (Tables 1 and 2). A final verification of eqn (3) was carried out by synthesizing and measuring the luminescent properties of complex Eu(DBM)(BTFA)(TTA)(TPPO)2. The % Boosts in η and in Arad for this complex, computed as the portion of the property that exceeds the average, as a percentage of the average – the right-hand side of eqn (3) – are 221% and 51%, as shown in the last lines of Tables 2 and 3, respectively.
All these results point in the direction that mixing ligands to boost the luminescent properties of europium complexes is indeed a seemingly comprehensive strategy of general validity.
2.2. The chemical partition of Arad
The boost observed for the all-different ionic ligand complex Eu(β-diketonate)(β-diketonate′)(β-diketonate′′)(TPPO)2, albeit significant, was equivalent to the one obtained for complexes of the type Eu(β-diketonate)2(β-diketonate′)(TPPO)2, seemingly indicating that the presence of at least one different ionic ligand in the complex structure is already enough to obtain a highly luminescent complex. Luminescence properties in Table 1 indicate that the lifetime values of the mixed ionic ligand ternary europium complexes of general formula Eu(β-diketonate)2(β-diketonate′)(TPPO)2 were all similar, averaging 0.438 ms, larger than those of the repeating ionic ligand complexes, which averaged 0.359 ms. Similarly, the Arad values for the mixed ionic ligand ternary europium complexes were much higher than those of the repeating ionic ligand complexes: 1072 s−1 and 683 s−1, respectively, as expected by the inequality in eqn (1). As such, the radiative decay rates, Arad, of mixed ionic ligand complexes involving TPPO are higher than those of the corresponding same ionic ligand complexes. This confirms the positive effect of the diversity of ionic ligands coordinated to the europium ion23 on europium luminescence.The radiative decay rate, Arad, is a global photophysical property of luminescent europium complexes. So, by examining Arad only, it is difficult to grasp the role of each of the ligands on the luminescence phenomenon, and, thus, interpret Arad from a chemical perspective. In order to better understand how each ligand facilitates the decay of the trivalent europium ion from its 5D0 excited state to the 7FJ ones, we recently introduced a partition of a subset of Arad, Arad′,20 which corresponds to the sum of the electric dipole electronic transitions 5D0 → 7FJ, J = 2, 4 and 6, and which constitutes approximately 91% of Arad. Hence, an analysis of Arad′ is essentially an analysis of Arad itself. Fig. 2 shows all three ternary europium complexes with their Arad values, as well as the partitioned values of Arad′ per ligand. Since the effects of the TPPO ligands in Arad′ for all three cases is somewhat equivalent, 102 s−1, 122 s−1 and 160 s−1, the difference in Arad occurs due to the ionic ligands: 190 s−1 for the DBMs, 636 s−1 for the TTAs, and 635 s−1 for the BTFAs. From these data alone, one would be led to the conclusion that the DBM ligand is never a very good ligand for luminescence purposes, whereas TTA and BTFA are.
 | ||
| Fig. 2 Total Arad values, partitioned Arad′ values per ligand for three europium complexes with repeating both ionic and nonionic ligands. The red circle stands for the Eu3+ ion. | ||
However, according to the inequality in eqn (1), adding a different ligand to make a mixed ligand complex, should intensify the Arad value of a more symmetrical complex, regardless of which ligand is chosen. Fig. 3 shows in a pictorial format the effect of removing a DBM ligand, one at a time, from complex Eu(DBM)3(TPPO)2, and replacing it with either a TTA or a BTFA.
 | ||
| Fig. 3 Total Arad values, partitioned Arad′ values per ligand for europium complexes with one, two, and three DBM ligands in their structures. | ||
Clearly, the contribution of DBM to Arad′ increases from an average of 63 s−1 for the complex with three DBMs, to an average of 80 s−1 for both complexes with two DBMs, and to the much larger average value of 338 s−1 for both complexes with one DBM. This trend strongly suggests that, if on one hand DBM does not seem to be a good antenna (and that is why three DBMs make a poorly luminescent complex), a single DBM seems to be important as a coordination polyhedron symmetry breaker – the value of 338 s−1 being a measure of how slightly more allowed the single DBM makes the transition, from the 5D0 excited state of the trivalent europium ion to the 7FJ states, to be. Fig. 3 also shows very clearly that the Arad values of the four mixed ligand complexes are all similar, in average 1051 s−1, indicating that a single good antenna is already enough to guarantee a high quantum efficiency, which can be further boosted by breaking the symmetry of the coordination polyhedron by different ligands – ligands that do not even need to be good antennas.
The first complex with two TPPOs is probably the most asymmetric and distorted of the three complexes in Fig. 4, because it is the one most stuffed with ligands. When the coordination polyhedron is uncongested by replacing both of them with a single ligand, either BIPY and PHEN, Arad diminishes. Since PHEN is a more planar and thus more symmetric ligand than BIPY, not surprisingly its complex displays the lowest value of Arad.
 | ||
| Fig. 4 Total Arad values, partitioned Arad′ values per ligand for the fully mixed ionic ligand complexes. | ||
2.3. Anrad and the rigidity of the non-ionic ligands
Since the radiative and non-radiative decay processes, Arad and Anrad, are competitive kinetic processes, it is plausible that, all other things being equal, the values of Anrad will tend to decrease as the values of Arad increase. Indeed, Anrad competes with Arad for the depopulation of the 5D0 excited state of the trivalent europium ion. Accordingly, consider the data in Table 1. While the average Anrad for the repeating ionic ligand complexes in Table 1 is 1934 s−1, it goes down when symmetry is broken for the more luminescent mixed ligand complexes to the average of 1247 s−1.Anrad is dependent on the ability of the complex to dissipate the 5D0 excited state energy to the ground state via molecular vibrations and/or transient geometric distortions and their interactions with the phonons or solvent molecules in the first solvation layers that surround the complex. As such, by removing the lower frequency molecular vibrations, the probabilities of non-radiative transitions are reduced, thus reducing Anrad and concurrently increasing Arad. Thus, in order to further enhance the luminescence of the completely mixed ionic ligand complex Eu(DBM)(BTFA)(TTA)(TPPO)2, we replaced both TPPO non-ionic ligands by either a 1,10-phenantroline, PHEN or by a 2,2-bipyridine, BIPY. Our expectation was that the quantum efficiencies of these complexes would be larger than those of the double TPPO complex with an Arad of 1034 s−1. From Table 1, however, as we had already discussed in the previous section, the values of Arad decreased for both BIPY and PHEN complexes, with values of 894 s−1 and 864 s−1, respectively. But, based on our previous reasoning, the effect of the rigidity of the non-ionic ligands, on the other hand, should emerge essentially on Anrad. And that was precisely the case, as revealed by the values of Anrad for the BIPY and PHEN complexes of 1018 s−1 and 686 s−1, much lower than the value for the double TPPO complex of 1269 s−1. The impact of the rigidity on Arad going from the double TPPO complex to the PHEN complex is therefore a reduction of 170 s−1 on its value, while the same impact on Anrad is a much larger reduction of 583 s−1. Thus, the effect of the rigidity of the ligands on Anrad overshadows the reduction of Arad, rendering the quantum efficiencies of the BIPY and PHEN complexes, 47% and 56%, respectively; larger than the quantum efficiency of the double TPPO complex of 45%, as originally intended.
From these results, it is possible to qualitatively order the effect of the non-ionic ligands on the rigidity of the complex as a whole, which we propose to call the hardening effect of the ligand, in terms of Anrad, from the more to the less hardener ligand as: PHEN > BIPY > TPPO. This is corroborated by the lowest vibrational frequencies calculated by Sparkle/RM1 for all three mixed ionic ligand complexes, of 2.48 cm−1 for TPPO2; 4.28 cm−1 for BIPY; and 4.48 cm−1 for PHEN. In this sense, we can further say that BTFA is a more hardener ligand than TTA in the complexes of Table 1. In general, whenever TTA is replaced by a BTFA on Table 1, Anrad usually tends to decrease its value. Indeed, while complex Eu(TTA)3(TPPO)2 displays an Anrad of 2061 s−1, Eu(BTFA)3(TPPO)2 displays a smaller Anrad value of 1806 s−1. Likewise, by replacing one TTA in Eu(TTA)2(BTFA)(TPPO)2 with the larger Anrad of 1491 s−1 by a BTFA, we arrive at Eu(BTFA)2(TTA)(TPPO)2 which has a smaller Anrad of 1126 s−1. Finally, replacing TTA in Eu(TTA)2(DBM)(TPPO)2 with a larger Anrad of 1307 s−1, we arrive at Eu(BTFA)2(DBM)(TPPO)2, with the smaller value of Anrad of 983 s−1.
3. Experimental
3.1. Characterization
The mixed ionic ligand complexes synthesized in this article were characterized by MALDI-TOF mass spectrometry (Autoflex 3 Smart Beam Vertical spectrometer); elemental analysis (Perkin-Elmer CHN2400); infrared spectroscopy (samples were prepared as KBr disks, and the spectra were measured in a Bruker model IFS 66 spectrophotometer, 4000–400 cm−1); 1H NMR, 31P NMR and 19F NMR spectroscopy (NMR spectra of all complexes were obtained in CDCl3 solutions by a Varian Unity Plus 400 MHz). The ESI† contains details of the experimental characterizations, data, spectra and all relevant spectral attributions for all complexes synthesized.3.2. Luminescence measurements
Luminescence spectra and quantum efficiency measurements were obtained with a Fluorolog-3 Horiba Jobin Yvon equipped with a Hamamatsu R928P photomultiplier with a SPEX 1934 D phosphorimeter and a 150 W pulsed xenon lamp. The measurements were performed at room temperature, from 10−4 M chloroform solutions at an excitation wavelength, λmax, between 374 nm and 397 nm with a slit width of 1.0 nm for the excitation, and 1.0 nm for the emission. Data were processed by software supplied by Horiba-Jobin-Yvon. The quantum efficiency, η, is defined as η = Arad/(Arad + Anrad), where Arad is the radiative decay rate and Anrad is the nonradiative decay rate. The non-radiative decay rates of the complexes in solution were calculated from the lifetime decay, in which: 1/τobs = Arad + Anrad.3.3. Materials
The reagents and solvents used were 1,3-diphenylpropane-1,3-dione (DBM, Alfa Aesar, 99%); 4,4,4-trifluoro-1-phenyl-1,3-butanedione (BTFA, Alfa Aesar, 99%); 1-(2-thenoyl)-3,3,3-trifluoroacetone (TTA, Alfa Aesar, 99%); triphenylphosphine oxide (TPPO, Sigma Aldrich, 99%); 1,10-phenanthroline monohydrated (PHEN, Panreac, 99%); 2,2′-bipyridyl (BIPY, Aldrich, 99%); ethanol (J.T. Baker); hexane (Sigma Aldrich); acetone (Sigma Aldrich) and chloroform (J.T. Baker).3.4. Syntheses
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H 3090–3015 cm−1; νP
C–H 3090–3015 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1087 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.60–7.31 (m, Ar.), 2.61 (s, OH), and 31P NMR (162 MHz, CDCl3): δ 27 ppm.
O 1087 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.60–7.31 (m, Ar.), 2.61 (s, OH), and 31P NMR (162 MHz, CDCl3): δ 27 ppm.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1685 cm−1; νC
O 1685 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1600 cm−1; νP
O 1600 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1113 cm−1; νC
O 1113 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1179 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.39 (s, CH), 7.85–5.75 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 25 ppm; 19F NMR (376 MHz, CDCl3): δ −83.90 ppm and −84.41 ppm (CF3).
F 1179 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.39 (s, CH), 7.85–5.75 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 25 ppm; 19F NMR (376 MHz, CDCl3): δ −83.90 ppm and −84.41 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1680 cm−1; νC
O 1680 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1608 cm−1; νP
O 1608 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1118 cm−1; νC
O 1118 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1182 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.78 (s, CH),7.82–6.07 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 25 ppm and −73 ppm; 19F NMR (376 MHz, CDCl3): δ −84.06 ppm and −84.64 ppm (CF3).
F 1182 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.78 (s, CH),7.82–6.07 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 25 ppm and −73 ppm; 19F NMR (376 MHz, CDCl3): δ −84.06 ppm and −84.64 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1684 cm−1; νC
O 1684 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1621 cm−1; νP
O 1621 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1118 cm−1; νC
O 1118 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1184 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.63 (s, CH), 7.90–7.54 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 24 ppm and −71 ppm; 19F NMR (376 MHz, CDCl3): δ −83.84 ppm and −84.40 ppm (CF3).
F 1184 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.63 (s, CH), 7.90–7.54 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 24 ppm and −71 ppm; 19F NMR (376 MHz, CDCl3): δ −83.84 ppm and −84.40 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1681 cm−1; νC
O 1681 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1625 cm−1; νP
O 1625 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1117 cm−1; νC
O 1117 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1181 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.44 (s, CH),7.91–7.55 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 28 ppm and −72 ppm; 19F NMR (376 MHz, CDCl3): δ −84.44 ppm (CF3).
F 1181 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.44 (s, CH),7.91–7.55 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 28 ppm and −72 ppm; 19F NMR (376 MHz, CDCl3): δ −84.44 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1595 cm−1; νC
O 1595 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1545 cm−1; νP
O 1545 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1068 cm−1; νC
O 1068 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1177 cm−1; νC
F 1177 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1118 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.97 (s, CH),7.84–6.06 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 25 ppm and −71 ppm; 19F NMR (376 MHz, CDCl3): δ −84.48 ppm and −84.59 ppm (CF3).
F 1118 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.97 (s, CH),7.84–6.06 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 25 ppm and −71 ppm; 19F NMR (376 MHz, CDCl3): δ −84.48 ppm and −84.59 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1684 cm−1; νC
O 1684 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1610 cm−1; νP
O 1610 cm−1; νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1120 cm−1; νC
O 1120 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1188 cm−1; νC
F 1188 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif)
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1168 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.17 (s, CH), 7.85–7.34 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 30 ppm and −76 ppm; 19F NMR (376 MHz, CDCl3): δ −84.65 ppm and −84.73 ppm (CF3).
F 1168 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.17 (s, CH), 7.85–7.34 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 30 ppm and −76 ppm; 19F NMR (376 MHz, CDCl3): δ −84.65 ppm and −84.73 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1619 cm−1, νP
O 1619 cm−1, νP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1115 cm−1; νC
O 1115 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1182 cm−1; νC
F 1182 cm−1; νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) F 1174 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.21 (s, CH), 7.79–6.43 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 28 ppm and 72.43 ppm; 19F NMR (376 MHz, CDCl3): δ –79.38 ppm and −80.00 ppm (CF3).
F 1174 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.21 (s, CH), 7.79–6.43 (m, Ar.); 31P NMR (162 MHz, CDCl3): δ 28 ppm and 72.43 ppm; 19F NMR (376 MHz, CDCl3): δ –79.38 ppm and −80.00 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H 3079–3049 cm−1, νC
C–H 3079–3049 cm−1, νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N 1615 cm−1, νC–F 1179 cm−1, νC
N 1615 cm−1, νC–F 1179 cm−1, νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1694 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.68 (s, CH), 8.27–6.12 (m, Ar.); 19F NMR (376 MHz, CDCl3): δ −79.26 to 74.82 ppm (CF3).
O 1694 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.68 (s, CH), 8.27–6.12 (m, Ar.); 19F NMR (376 MHz, CDCl3): δ −79.26 to 74.82 ppm (CF3).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H 3062–3048 cm−1, νC
C–H 3062–3048 cm−1, νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N 1625 cm−1, νC–F −1, νC
N 1625 cm−1, νC–F −1, νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O 1684 cm−1. 1H NMR (400 MHz, CDCl3): δ 8.62 (s, CH), 7.86–6.03 (m, Ar.); 19F NMR (376 MHz, CDCl3): δ −79.22 to 79.87 ppm (CF3).
O 1684 cm−1. 1H NMR (400 MHz, CDCl3): δ 8.62 (s, CH), 7.86–6.03 (m, Ar.); 19F NMR (376 MHz, CDCl3): δ −79.22 to 79.87 ppm (CF3).3.5. Computational procedure
All geometries of europium complexes were fully optimized using Sparkle/RM1 model18 implemented in MOPAC 2016 software.19 The keywords used in the MOPAC calculations were SPARKLE RM1 GNORM = 0.25 BFGS XYZ T = 10D ALLVEC. The vibrational modes were calculated from the optimized geometries, when we made sure the geometries were true minima with no imaginary frequencies. In this step we used the keywords: SPARKLE RM1 FREQ THERMO XYZ T = 10D ALLVEC. The luminescence parameters and properties were calculated from experimental data of emission spectra and lifetime curves using the LUMPAC software.19 The refractive index used in the calculations was 1.45 because the photophysical experiments were all carried out in chloroform solution.4. Conclusions
We demonstrate that mixing ionic ligands in europium complexes does enhance their luminescence over and above what can be observed for the luminescence of related compounds with repeating ionic ligands. Indeed, this proposition of mixing ionic ligands for the purpose of enhancing luminescence, pairs with the results we obtained previously by mixing non-ionic ligands.23Therefore, our strategy of increasing the diversity of ligands in the coordination polyhedron to enhance the luminescence properties of europium complexes is strengthened as a general and comprehensive one, which can be easily understood and applied to a variety of situations. The strategy is quantified by means of inequalities, as further generalized in eqn (3) and (4).
By means of the chemical partition of Arad′, we discovered that coordination symmetry breaking by a single ionic ligand which can, in itself, even be a poor antenna, does indeed enhance luminescence. This suggests that there is no need for a multitude of antennas around a europium ion – coordination diversity being more important; a result which is in tune with the inequalities present in eqn (3) and (4).
We also introduce the concept of structure hardening by ligands, by linking it for the first time with the values of Anrad. Accordingly, we call ligand L a hardener in comparison with ligand L′, a softener, if the value of Anrad for a complex increases when L′ is replaced by L. Such a concept allows us to order ligands in terms of their hardening effect in a given class of compounds, showing that – for the complexes studied – PHEN is the best non-ionic hardener, followed by BIPY and then by TPPO. Likewise, the ionic ligands can also be ordered, with DBM being the most ionic hardener, followed by both BTFA and TTA, for the class of compounds studied in this article.
In light of these concepts, we conclude that luminescence enhancement can be obtained in europium complexes by increasing ligand diversity, ideally with the help of hardening ligands defined as those capable of lowering Anrad. This general and comprehensive strategy thus boosts the quantum efficiency by increasing Arad while simultaneously decreasing Anrad.
Extension of this rationale of luminescence enhancement to other lanthanide trications and different ligands is being presently carried out in our laboratories.
Acknowledgements
The authors appreciate the financial support of the following Brazilian Agencies: FACEPE (Pronex), and CNPq. The authors also thank Centro de Tecnologias Estratégicas do Nordeste, CETENE, and Dr Júlia F. Campos for the MALDI-TOF mass spectrometry measurements.References
- Z. Wang, H. Yang, P. He, Y. He, J. Zhao and H. Tang, Dalton Trans., 2016, 45, 2839–2844 RSC.
- D. Wu, X. Xin, X. Pang, M. Pietraszkiewicz, R. Hozyst, X. Sun and Q. Wei, ACS Appl. Mater. Interfaces, 2015, 7, 12663–12670 CAS.
- H. Terraschke, L. Ruiz Arana, P. Lindenberg and W. Bensch, Analyst, 2016, 141, 2588–2594 RSC.
- J. Liu, J.-S. Miao and H.-B. Wu, Luminescence, 2015, 30, 393–396 CrossRef CAS PubMed.
- F. Cao, Z. Yuan, J. Liu and J. Ling, RSC Adv., 2015, 5, 102535–102541 RSC.
- R. J. Meier, J. M. B. Simbürger, T. Soukka and M. Schäferling, Chem. Commun., 2015, 51, 6145–6148 RSC.
- Z. Deng, X. Hu, Y. Wang, Y. Yin, B. Peng and Z. Xu, J. Biomater. Sci., Polym. Ed., 2015, 26, 16–31 CrossRef CAS PubMed.
- J. Ye, J. Wang, Q. Li, X. Dong, W. Ge, Y. Chen, X. Jiang, H. Liu, H. Jiang and X. Wang, Biomater. Sci., 2016, 4, 652–660 RSC.
- L. Jia, T. Zhou, J. Xu, Z. Xu, M. Zhang, Y. Wang, Z. Li and T. Zhu, J. Mater. Sci., 2016, 51, 1324–1332 CrossRef CAS.
- H. Li, R. Lan, C.-F. Chan, L. Jiang, L. Dai, D. W. J. Kwong, M. H.-W. Lam and K.-L. Wong, Chem. Commun. (Camb)., 2015, 51, 14022–14025 RSC.
- L. Zhou, Y. Jiang, R. Cui, Y. Li, X. Zhao, R. Deng and H. Zhang, J. Lumin., 2016, 170, 692–696 CrossRef CAS.
- J. P. Martins, P. Martín-Ramos, C. Coya, M. R. Silva, M. E. S. Eusebio, A. De Andrés, Á. L. Álvarez and J. Martín-Gil, J. Lumin., 2015, 159, 17–25 CrossRef CAS.
- J. Sun, B. Song, Z. Ye and J. Yuan, Inorg. Chem., 2015, 54, 11660–11668 CrossRef CAS PubMed.
- P. K. Shahi, A. K. Singh, S. B. Rai and B. Ullrich, Sens. Actuators, A, 2015, 222, 255–261 CrossRef CAS.
- Q. Xu, Z. Li and H. Li, Chem.–Eur. J., 2016, 22, 3037–3043 CrossRef CAS PubMed.
- N. B. D. Lima, A. I. S. Silva, S. M. C. Gonçalves and A. M. Simas, J. Lumin., 2016, 170, 505–512 CrossRef CAS.
- J. D. L. Dutra, T. D. Bispo and R. O. Freire, J. Comput. Chem., 2014, 35, 772–775 CrossRef CAS PubMed.
- M. a. M. Filho, J. D. L. Dutra, G. B. Rocha, R. O. Freire and A. M. Simas, RSC Adv., 2013, 3, 16747 RSC.
- J. J. P. Stewart, MOPAC2016, http://openmopac.net/MOPAC2016.html, accessed September 2016.
- N. B. D. Lima, J. D. L. Dutra, S. M. C. Gonçalves, R. O. Freire and A. M. Simas, Sci. Rep., 2016, 6, 21204 CrossRef CAS PubMed.
- E. Niyama, H. F. Brito, M. Cremona, E. E. S. Teotonio, R. Reyes, G. E. S. Brito and M. C. F. C. Felinto, Spectrochim. Acta, Part A, 2005, 61, 2643–2649 CrossRef CAS PubMed.
- A. Beeby, I. M. Clarkson, R. S. Dickins, S. Faulkner, D. Parker, L. Royle, A. S. de Sousa, J. A. G. Williams and M. Woods, J. Chem. Soc., Perkin Trans. 2, 1999, 493–504 RSC.
- N. B. D. Lima, S. M. C. Gonçalves, S. a. Júnior and A. M. Simas, Sci. Rep., 2013, 3, 2395 Search PubMed.
- T. C. W. M. J. H. Li Yigen, Y. Yansheng and L. Yuanying, Chin. J. Struct. Chem., 1988, 7, 165 Search PubMed.
- A. T. H. L. L. Van Meervelt, A. Froyen, W. D'Olieslager, C. Walrand-Gorller, I. Drisque, G. S. D. King and S. Maes, Bull. Soc. Chim. Belg., 1996, 105, 377 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: excitation spectra; emission spectra; lifetime curves; infrared spectra; and 1H, 19F, and 31P NMR spectra. See DOI: 10.1039/c6ra20609c |
| This journal is © The Royal Society of Chemistry 2016 |