Hepatoprotective effect of orally applied water-soluble pristine C60 fullerene against CCl4-induced acute liver injury in rats
Abstract
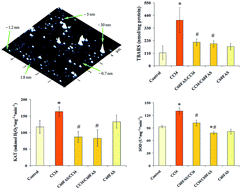
The development of novel more efficient antioxidants is one of the most perspective approaches for the treatment of the harmful effects of toxins and their metabolites. Due to its remarkable antioxidant properties, pristine C60 fullerene has recently been proposed as a promising candidate for many biomedical applications. The present study aimed to determine whether single oral administration of water-soluble pristine C60 fullerene in small dose (1.5 mg kg−1 bwt) could prevent acute liver injury caused by single intraperitoneal injection of carbon tetrachloride (CCl4; 1.0 mL kg−1 bwt) in rats. We performed structural examination of the liver and monitored the serum markers relevant to hepatocyte integrity as well as liver functionality. The antioxidative potential of pristine C60 fullerene was assessed in vivo by measuring lipid peroxidation. We also measured the activity of catalase and superoxide dismutase in order to evaluate the antioxidative defense system. Biochemical and pathological results obtained in this study indicate that water-soluble pristine C60 fullerene in a single dose of 1.5 mg kg−1 has a hepatoprotective effect against CCl4-induced toxicity via its antioxidant properties. According to histopathological examinations and some biological tests, water-soluble pristine C60 fullerene is more effective against liver injury when administered before a toxicant than after one. Moreover, our results show that the low dose of C60 fullerene used has no acute toxicity in rodents. We concluded that water-soluble pristine C60 fullerene can be considered as a powerful liver-protective agent.


 Please wait while we load your content...
Please wait while we load your content...