Limited Ce doped Ni–Co as a highly efficient catalyst for H2-SCR of NO with good resistance to SO2 and H2O at low temperature
Xuanxuan Cai,
Wei Sun,
Waqas Qamar Zaman,
Limei Cao and
Ji Yang*
School of Resources and Environmental Engineering, State Environmental Protection Key Laboratory of Environmental Risk Assessment and Control on Chemical Process, East China University of Science and Technology, Shanghai 200237, PR China. E-mail: yangji@ecust.edu.cn; Fax: +86-21-64251668; Tel: +86-21-64251668
First published on 20th September 2016
Abstract
The Ni1−xCexCo1.95Pd0.05O4 catalysts doped with limited Ce were fabricated using a sol–gel method and employed to investigate the SO2-resistance in the selective catalytic reduction of NO by hydrogen (H2-SCR). The results indicated that the inclusion of a limited amount of cerium not only effectively enhanced the catalytic activity of Ni1−xCexCo1.95Pd0.05O4 catalysts but also improved their resistance to SO2. Notably, when the Ce doping was 0.07 at%, the catalyst showed high sulfur resistance and exhibited a broader temperature window of 140–350 °C with a maximum NOX conversion of 98% at 190 °C. Also, addition of 5% H2O into the feed gas slightly decreased the deNOX performance of all catalysts, the NOX conversion nearly recovered to the initial level with H2O withdrawal, and the resistance to H2O also increased gradually in correspondence to the increase in Ce doping. Meanwhile, an increase of GHSV from 4800 to 9600 mL h−1 g−1, the NO/H2 ratios from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 to 1
10 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and the O2 content from 0% to 6%, gradually decreased the removal efficiency of NO. The XPD and XRS analysis depicted fine cubic spinel-type structures and Ce bearing two chemical valence states doped in the spinels. In the H2-TPR and NH3-TPD analysis, the Ce doping resulted in the temperature shift of reductive peaks and weak acid sites to lower temperature and the increase of strong acid sites. These results enhanced the redox property and led to a positive effect on SCR reaction, which were consistent with the SCR activity data.
1 and the O2 content from 0% to 6%, gradually decreased the removal efficiency of NO. The XPD and XRS analysis depicted fine cubic spinel-type structures and Ce bearing two chemical valence states doped in the spinels. In the H2-TPR and NH3-TPD analysis, the Ce doping resulted in the temperature shift of reductive peaks and weak acid sites to lower temperature and the increase of strong acid sites. These results enhanced the redox property and led to a positive effect on SCR reaction, which were consistent with the SCR activity data.
1. Introduction
Nitrogen oxides (NOX) from fossil fuel combustion1 are major air pollutants, which have serious environmental implications,2 such as causing photochemical smog, acid rain, and greenhouse effects.3 The current efficient processing technology for NOX emission control in industry is NH3-SCR, however, many problems persist in NH3-SCR, namely catalyst deterioration, NH3-slip, ash odor and so on.4 To solve this issue, selective catalytic reduction by hydrogen (H2-SCR) could greatly replace the NH3-SCR, which is due to the efficient removal of NOX without the emergence of any secondary pollutants at the lowest possible temperature.5,6Moreover, the noble metal catalysts as well as the precious metal doped catalysts in the process of H2-SCR exhibit good catalytic activity for NOX removal at relatively low temperatures.7–10 For example, higher than 95% NO conversion with 78–92% N2 selectivity at 100–400 °C was studied at Pt/MgO–CeO2 catalyst,8 and G. L. Chiarello et al.10 researched the Pd/SiO2 catalyst with 90% NOX conversion at 170–300 °C. Nevertheless, noble metals catalysts have poor resistance to sulfur dioxide, and that lead to the formation of formed the sulfates and sulfites on the active sites of catalysts. Eventually such species results in the deactivation of SCR activity and reduces the NO removal at low temperature.11,12 Such as, the activities of Pt/SiO2 and Pd/SiO2 were greatly influenced by 20 ppm SO2 in the simulated gas stream.13 Thus, improving sulfur tolerance of the catalyst should be salient for practical usage.
For this problem, recently, we have used the limited cerium doping into Ni–Co spinel catalyst to improve the SO2-resistance, the results exhibits that a high removal efficiency of NOX along with excellent SO2 resistance is achieved through the H2-SCR in the presence of oxygen at relatively lower temperature. The good performance is due to the cerium particular properties, revealing two stable oxidation states of Ce4+ and Ce3+, that results in the reserve and release of oxygen into cerium by shifting between Ce4+ and Ce3+ under oxidizing and reducing conditions.14,15 In our study, we also focus on the effects of GHSV, NO/H2 ratios and O2 content on the SCR activity. Moreover, the durability and tolerance of catalysts to H2O is also investigated for practical consideration.
2. Experimental
2.1. Catalyst preparation
The catalysts were prepared using sol–gel method with inorganic salts, where the molar ratio of citric acid![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni(NO3)3·6H2O
Ni(NO3)3·6H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce(NO3)3·6H2O
Ce(NO3)3·6H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co(NO3)3·6H2O
Co(NO3)3·6H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PdCl2 was 3
PdCl2 was 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 − x
1 − x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x
x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.95
1.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05. Then these compounds were dissolved into deionized water, further addition of the ammonia solution was to adjust the pH of the mixture solution at 5–6, during this process, the polyethylene glycol (PEG) 400 was added to make the high specific surface area and uniform particle size.16 Then the mixture was stirred for 3 h at room temperature, and evaporated redundant water at 80 °C to form a wet gel, later on the gel was dried at 130 °C. Finally, after grinding the obtained material into fine powder, the powder was calcinated in muffle furnace at 400 °C for 4 h to obtain crystallized Ni1−xCexCo1.95Pd0.05O4.
0.05. Then these compounds were dissolved into deionized water, further addition of the ammonia solution was to adjust the pH of the mixture solution at 5–6, during this process, the polyethylene glycol (PEG) 400 was added to make the high specific surface area and uniform particle size.16 Then the mixture was stirred for 3 h at room temperature, and evaporated redundant water at 80 °C to form a wet gel, later on the gel was dried at 130 °C. Finally, after grinding the obtained material into fine powder, the powder was calcinated in muffle furnace at 400 °C for 4 h to obtain crystallized Ni1−xCexCo1.95Pd0.05O4.
2.2. Catalyst characterization
The samples X-ray diffraction (XRD) measurements have been conducted by using a Rigaku D/MAX 2550 diffractometer (Japan) with Cu Kα radiation (λ = 1.54056 Å) operating at 40 kV and 100 mA. The elemental composition of the samples was characterized by energy dispersive spectrometer (EDS) using a Falion 60s spectrometer. The morphological features of samples were studied by Scanning Electron Microscopy (SEM) using Nova NanoSEM 450.The Brunauer–Emmett–Teller (BET) surface area of the catalysts were measured with a nitrogen adsorption instrument (Micromeritics, Tristar II 3020) by using N2 adsorption/desorption at liquid nitrogen temperature. Prior to BET analysis, the samples were degassed at 300 °C for 3 h and cooled to room temperature.
The H2 Temperature Programmed Reduction (H2-TPR) analysis were conducted on Auto Chem II 2920 equipped with a TCD detector. In a typical TPR experiment, about 50 mg sample was pretreated in a He stream at 300 °C for 1 h and cooled to room temperature before the H2-TPR analysis. Then, the H2-TPR runs were performed from room temperature to 800 °C at a heating rate of 10 °C min−1 in a 10% H2/Ar mixture with a flow rate of 40 mL min−1.
Temperature Programmed Desorption of ammonia (NH3-TPD) was performed on Auto Chem II 2920 analyzer. For NH3-TPD experiments, 50 mg of the oven-dried sample was treated under helium flow at 300 °C for 60 min to remove the adsorbed H2O and cooled to room temperature by other gases. Then, the saturation of NH3 adsorption was achieved with 10% NH3/He mixture at 50 °C for 60 min with a total flow rate of 40 mL min−1. After NH3 adsorption, the samples were purged by He (40 mL min−1) for another 60 min. Finally, the NH3-TPD was performed from room temperature to 800 °C at a heating rate of 10 °C min−1 under a constant flow of He (50 mL min−1). The NH3 desorption was monitored by a Thermal Conductivity Detector (TCD).
X-ray Photoelectron Spectroscopy (XPS) were performed in a Quantum 2000 Scanning ESCA Microprobe (Physical Electronics) using a focused monochromatic Al Kα X-ray (1486.7 eV) source to analyze the surface atomic concentration and chemical states of the catalysts, the X-ray source operated at 100 W and 100 μm-diameter beam. The binding energy scale was calibrated by the carbon C 1s at 284.6 eV as known standards. The de-convolution of XPS peak was carried out using Casa XPS program.
The Raman spectroscopy experiments were conducted using an Iuvia microscope instrument (Iuvia Reflerx) with a wavelength of 514.5 nm.
2.3. Catalytic activity test
The activity measurements of the catalysts were carried out in a fixed-bed flow micro-reactor at 50–350 °C with powder catalyst of 1 g, the thermocouple controlled the catalyst bed temperature through interpolating into the fixed-bed reactor, and the fixed-bed reactor was heated with an artificial intelligence temperature controller of type Al-518/518P. The reactant gas typically consisted of 1071 ppm NO, 1071–10710 ppm H2, 0–6% O2, 5% H2O (when used), 500 ppm SO2 (when used), and while N2 of high purity (99%) was used as a carrier gas. While the water vapor was obtained by passing N2 through a heated gas-wash bottle including deionized water. Moreover, the total gas flow rate of the inlet gas was always maintained at 155 mL min−1 with a GHSV of 9600 mL h−1 g−1, except the changes of GHSV. The gas effluent stream from the reactor was monitored by a NO–NO2–NOX Analyzer (Thermo Scientific, Model 42i), and the nitric oxide conversion was defined by using the following equation,
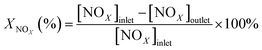 | (1) |
3. Results and discussion
3.1. Catalysts characterization
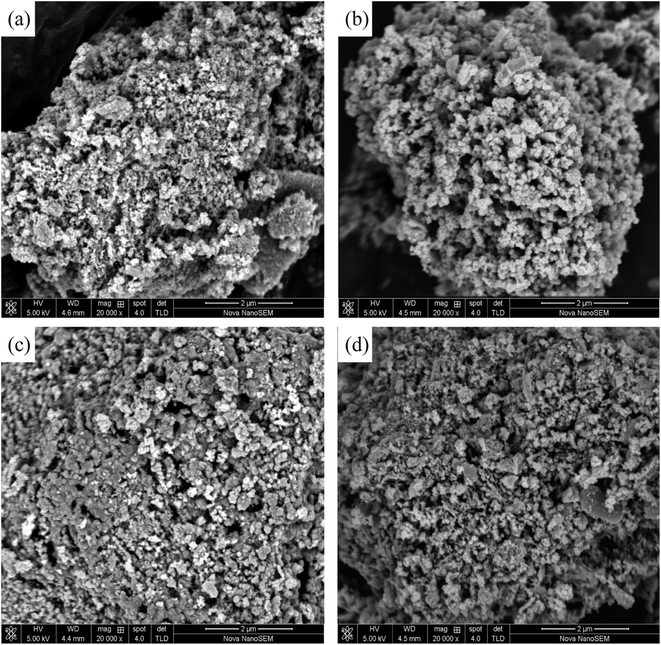 | ||
| Fig. 2 SEM patterns of Ni1−xCexCo1.95Pd0.05O4 used in this study; (a) 0.03 at% Ce; (b) 0.07 at% Ce; (c) 0.1 at% Ce; (d) 0.2 at% Ce. | ||
| Catalysts | SBET (m2 g−1) | Elemental composition (at%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Ni | Co | Pd | Ce | Pd/Ni | Co/Ni | Ce/Ni | ||
| NiCo1.95Pd0.05O4 | 12.1 | 20.29 | 38.97 | 1.00 | 0.00 | 0.049 | 1.92 | 0.000 |
| Ni0.97Ce0.03Co1.95Pd0.05O4 | 6.7 | 21.73 | 39.90 | 1.43 | 0.94 | 0.066 | 1.84 | 0.043 |
| Ni0.95Ce0.05Co1.95Pd0.05O4 | 7.4 | 20.93 | 41.03 | 1.41 | 1.86 | 0.067 | 1.96 | 0.089 |
| Ni0.93Ce0.07Co1.95Pd0.05O4 | 16.1 | 19.71 | 39.85 | 1.53 | 2.13 | 0.078 | 2.02 | 0.108 |
| Ni0.9Ce0.1Co1.95Pd0.05O4 | 12.7 | 19.18 | 38.56 | 1.04 | 2.43 | 0.054 | 2.01 | 0.126 |
| Ni0.8Ce0.2Co1.95Pd0.05O4 | 21.1 | 15.63 | 36.02 | 1.14 | 4.35 | 0.079 | 2.31 | 0.278 |
3.2. Catalysts performance
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 ppm H2, 2% O2 and N2 as balance gas. As shown in Fig. 4a and b, initially the NOX conversion over the Ni1−xCexCo1.95Pd0.05O4 catalysts increased to maximum and later decreased as the temperature increased from 50 to 350 °C. While the NOX conversion over NiCo1.95Pd0.05O4 catalyst was over 80% with the temperature window of 170–260 °C, and the maximum conversion obtained was found to be 93% at 230 °C. The previous research21 indicated the Ce doping could effectively improve the SCR performance. Hence, in the present study, the effect of different concentrations of doped Ce on the conversion for H2-SCR over the Ni1−xCexCo1.95Pd0.05O4 catalysts was examined. The results shown in Fig. 4a, exhibit distinctly increased SCR activities in correspondence to the increase of amount of doped Ce. When the Ce doping was 0.03 at%, the maximum NOX conversion was approximately 95% at 240 °C, while the temperature window over 80% was broadened in the range of 170–310 °C, which is illustrated in Fig. 4b. As the concentration of doped cerium increases, the temperature window of NOX conversion became more and more wide, and the temperature of the highest NO conversion shifted to a lower temperature, except the Ce doping of 0.03 at% and 0.05 at%. When the Ce doping was 0.07 at%, the temperature window reached to the maximum of 140–350 °C, and the maximum NOX conversion of 98% was achieved at 190 °C. Then the temperature window decreased but was still larger than the original value as upon the further increase in the amount of doped Ce. These results indicated that the Ce doping effectively enhanced the SCR activity of Ni1−xCexCo1.95Pd0.05O4 catalysts, which was due to the contributive effect of cerium.21 The sequence of NO conversions for Ni1−xCexCo1.95Pd0.05O4 with different amount of doped cerium decreased as follow:
700 ppm H2, 2% O2 and N2 as balance gas. As shown in Fig. 4a and b, initially the NOX conversion over the Ni1−xCexCo1.95Pd0.05O4 catalysts increased to maximum and later decreased as the temperature increased from 50 to 350 °C. While the NOX conversion over NiCo1.95Pd0.05O4 catalyst was over 80% with the temperature window of 170–260 °C, and the maximum conversion obtained was found to be 93% at 230 °C. The previous research21 indicated the Ce doping could effectively improve the SCR performance. Hence, in the present study, the effect of different concentrations of doped Ce on the conversion for H2-SCR over the Ni1−xCexCo1.95Pd0.05O4 catalysts was examined. The results shown in Fig. 4a, exhibit distinctly increased SCR activities in correspondence to the increase of amount of doped Ce. When the Ce doping was 0.03 at%, the maximum NOX conversion was approximately 95% at 240 °C, while the temperature window over 80% was broadened in the range of 170–310 °C, which is illustrated in Fig. 4b. As the concentration of doped cerium increases, the temperature window of NOX conversion became more and more wide, and the temperature of the highest NO conversion shifted to a lower temperature, except the Ce doping of 0.03 at% and 0.05 at%. When the Ce doping was 0.07 at%, the temperature window reached to the maximum of 140–350 °C, and the maximum NOX conversion of 98% was achieved at 190 °C. Then the temperature window decreased but was still larger than the original value as upon the further increase in the amount of doped Ce. These results indicated that the Ce doping effectively enhanced the SCR activity of Ni1−xCexCo1.95Pd0.05O4 catalysts, which was due to the contributive effect of cerium.21 The sequence of NO conversions for Ni1−xCexCo1.95Pd0.05O4 with different amount of doped cerium decreased as follow:
Ni0.93Ce0.07Co1.95Pd0.05O4 > Ni0.9Ce0.1Co1.95Pd0.05O4 > Ni0.8Ce0.2Co1.95Pd0.05O4 > Ni0.95Ce0.05Co1.95Pd0.05O4 > Ni0.97Ce0.03Co1.95Pd0.05O4 > NiCo1.95Pd0.05O4.
Among them, the Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst exhibited high deNOX performance with the maximum NOX conversion of 98% at a relatively low temperature and wide temperature window of 140–350 °C.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 and O2 content from 0% to 6% were investigated at 50–350 °C range, and the results were shown in Fig. 4c–e.
10 and O2 content from 0% to 6% were investigated at 50–350 °C range, and the results were shown in Fig. 4c–e.As illustrated in Fig. 4c, the NOX conversion at a low GHSV was higher than that at a high GHSV. The NOX conversion first increased and remained over 90%, then finally declined as the temperature increased further. In the temperature range of 150–270 °C, Ni0.93Ce0.07Co1.95Pd0.05O4 exhibited high NOX conversion at GHSVs of 4800–9300 mL h−1 g−1, and the NOX conversion faintly decreased upon the increase of GHSV. While, a high GHSV means requirement of the less catalyst that may subsequently reduce the cost of the SCR system. Hence, a high GHSV of 9300 mL h−1 g−1 was used in our work.
However, H2 is a strong reducing agent and its concentration could significantly influence the SCR activity, so the effect of NO/H2 ratios on Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst was studied, which was shown in Fig. 4d. The NOX conversion first clung to the maximum around 200 °C, and then decreased as the temperature increased further at different NO/H2 ratios. As the NO/H2 ratios descended from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, the H2 concentration increased, resulting in the improvement of deNOX performance, which showed that the maximum NOX conversion enhanced from 60% to 96%. While the Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst exhibited high catalytic activity at low NO/H2 ratio, and in industry, the lower NO/H2 ratio is usually used to obtain a relatively higher NOX conversion. Therefore, the Ce doping significantly improved the catalytic activity and economic performance.
10, the H2 concentration increased, resulting in the improvement of deNOX performance, which showed that the maximum NOX conversion enhanced from 60% to 96%. While the Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst exhibited high catalytic activity at low NO/H2 ratio, and in industry, the lower NO/H2 ratio is usually used to obtain a relatively higher NOX conversion. Therefore, the Ce doping significantly improved the catalytic activity and economic performance.
Previous study has showed that the content of O2 was a crucial parameter for the H2-SCR reaction.23,24 Generally, the oxygen content is 4–6% in the flue gas from circulating fluidized bed boiler.25 Hence, in the present study the oxygen content was max 6%. Fig. 4e showed the O2 influence on Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst, in the absence of O2, the NOX conversion ascended to the maximum of 100% and then remained stable at the high efficiency within a temperature range of 150–350 °C. When the O2 content increased from 0% to 6%, the removal efficiency of NOX gradually decreased from 100% to 75% at 200 °C, which was due to the consumption of the hydrogen reductant by oxygen. That was in consistence to the results of varying NO/H2 ratios effects. When the O2 content was 6%, the NOX conversion still maintained higher removal level of over 75% at 170–250 °C range, which indicated that the addition of cerium efficiently enhanced the oxygen resistance of catalyst.
Previous studies21,28 have demonstrated that the doped cerium could improve the Mn–Ce/TiO2 catalytic activity and resistance to SO2. In the experiments performed in the study at hand, the main task was to adjust the amount of doped cerium to enhance the catalyst performance. As shown in Fig. 5a, the doped Ce Ni1−xCexCo1.95Pd0.05O4 catalysts with doped Ce exhibited high NOX conversion (92–97%) in 60 min, after addition of 500 ppm SO2 to the gas feed stream, although there was obvious decrease in the deNOX performance, yet the NOX conversion showed comparatively a higher value of 61–72% than of 50% on NiCo1.95Pd0.05O4 catalyst, which indicated the doped Ce catalysts exhibited less sensitivity to SO2. In addition, as the Ce doping increased, the SO2-resistance of Ni1−xCexCo1.95Pd0.05O4 catalysts also increased, which was confirmed by the increase of NOX conversion from 60.8% to 71.6% at 150 min in the presence of 500 ppm SO2 with the doped Ce increased from 0.03 to 0.2. After the removal of SO2, the NO conversion of Ni1−xCexCo1.95Pd0.05O4 catalysts was greatly recovered, and it was also contributed to the successive increase in the amount of doped Ce, especially when the doped Ce increased to 0.07, 0.1 and 0.2, the NOX conversion nearly recovered to initial value. Therefore, the addition of cerium weak sulfation on the catalyst surface, it was probable that the SO2 preferentially reacted with cerium instead of active component, which inhibited the formation of sulfate on active sites of catalysts, it was consistent with the literature.29 Thus these factors greatly improve the sulfur-resistance of Ni1−xCexCo1.95Pd0.05O4 catalysts. Moreover, the Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst could quickly recover to the initial value whilst exhibiting high NOX conversion of 95%.
The effect of H2O on the selective catalytic reduction of NOX with hydrogen over the Ni1−xCexCo1.95Pd0.05O4 catalysts is demonstrated in Fig. 5b. It was found that the initial NOX conversions of the catalysts were about 91–95% and stabilized for 60 min in absence of H2O. After the addition of 5% H2O in the gas feed stream, the deNOX performance of all catalysts showed minimal decrease for 200 min, which was due to H2O competitive adsorption with NO as well as with H2,30,31 resulting in the desorption of NO originally adsorbed on the catalyst.32 The NOX conversion of the NiCo1.95Pd0.05O4 couldn't recover to the initial level, that was attributed to the reduction in the number of the actives sites which was available for the adsorption and reaction of NO and H2.33 However, the catalyst with the addition of cerium showed better H2O-resistance, the NOX conversion rate continuously increased to almost the original level. As the Ce doped, the hydrogen adsorption of catalysts was promoted, which resulted in the decrease of H2O adsorption, and increased the resistance to H2O.
Therefore, the results manifested that the Ce doping could efficiently enhance the resistance to H2O and SO2, and among all, the Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst displayed good characteristics against the resistive of H2O and SO2 in the low-temperature SCR reaction.
3.3. Catalysts activity analysis
In order to study the reason for the improvement of catalytic activity, the H2-TPR, NH3-TPD analysis and XPS analysis were conducted, and the results were shown as follows.The redox properties and acidic properties of catalysts are an indicator to determine the catalytic activity, thus the Ni1−xCexCo1.95Pd0.05O4 catalysts were demonstrated using H2-TPR and NH3-TPD analysis, respectively, and the results are exhibited in Fig. 6. As shown in Fig. 6a, the profile of NiCo1.95Pd0.05O4 catalyst had a sharp reduction peak at 128 and a broad peak at 352 °C. According to the literature,34,35 the reduction peak at 128 °C was assigned to the reduction of both Pd2+ and Co3+, which was the form of Pd2+ to Pd0 and Co3+ to Co2+, respectively.36,37 While the broad peak appeared at 352 °C, corresponding to the co-reduction of Co2+ to Co0 and Ni2+ to Ni0.36,37 However, when the Ce doped in catalyst, the temperature of the reduction peaks shifted to low temperature, which was due to the interaction between Pd2+ and Ce4+.38 The peak around 100 °C corresponded to the co-reduction of Pd2+, Co3+, Ce4+ and Ce3+, while the peak around 252 °C was assigned to the reduction of both Co2+ and Ni2+. Upon the increase of Ce doping content, the temperature of peaks shifted greatly to low temperature, which was advantageous to the hydrogen adsorption, resulted in the enhancement of the redox property of Ni1−xCexCo1.95Pd0.05O4 catalysts, hence improving the SCR activity.
 | ||
| Fig. 6 H2-TPR and NH3-TPD profiles of Ni1−xCexCo1.95Pd0.05O4 catalysts used in this study. (a) H2-TPR; (b) NH3-TPD. | ||
Temperature programmed desorption of ammonia (NH3-TPD) has been employed to investigate the acidic properties of all catalysts, and the results obtained are shown in Fig. 6b. All catalysts displayed one broad NH3 desorption peak spanned in the temperature range of 100–350 °C, and the desorption of NH3 at different temperature regions was determined by the strength of acid sites and the amount of acid, while the peak was corresponded to the weak and medium strength of acid sites.15,39 For the NiCo1.95Pd0.05O4 catalyst, a shoulder over 600 °C was observed, and no peak formation may be in result of the temperature of NH3-TPD not exceeding 700 °C. However, when the Ce was doped in the catalyst, the temperature of weak acid sites shifted to low temperature, which eventually further weakened the strength of weak acid, and the result was facilitating to NO stripping and beneficial to SCR reaction. Meanwhile, one broad peak from 550 °C to 700 °C was observed, that was assigned to the chemisorbed NH3 molecules adsorbed by strong acid sites.40 As the increased of Ce doping, the intensity and area of the peak centred at 650 °C was enlarged, which resulted in a positive effect on SCR reaction. Therefore, the Ce doping could improve the SCR activity and thus widened the NOX conversion temperature window,27 which was in good agreement with the SCR activity of the Ni1−xCexCo1.95Pd0.05O4 catalysts (Fig. 4).
In order to further analyze the Ni1−xCexCo1.95Pd0.05O4 catalysts, in this study, the Ce content of 0 at%, 0.03 at%, 0.07 at% and 0.1 at% were selected to investigate the surface characteristics. Fig. 7 shows the surface chemical sate of Ni, Co, Pd and Ce in the Ni1−xCexCo1.95Pd0.05O4 catalysts, which was conducted by XPS analysis. The Ni 2p spectra is given in Fig. 7a, all samples showed two spin–orbit doublets characteristic of Ni2+ and Ni3+ with two shakeup satellites. The Ni2+ peaks could be found at the binding energy of 854.4 eV and 871.6 eV, while the fitting peaks with an energy of 856.43 eV and 873.8 eV corresponded to Ni3+, respectively.18,41
 | ||
| Fig. 7 XPS spectra of Ni1−xCexCo1.95Pd0.05O4 used in this study. (a) Ni 2p, (b) Co 2p, (c) Pd 3d, (d) Ce 3d. | ||
Fig. 7b demonstrated the Co 2p spectra with two types of species of 2p3/2 and 2p1/2, the peaks at 779.4 eV and 794.8 eV were from Co3+, while the binding energy at 781.4 eV and 796.7 eV were associated with Co2+, respectively,42–44 which indicated that the low spin Co3+ species were dominantly positioned in the octahedral sites than the Co2+ species.45,46
The Pd 3d XPS pattern indicated the fitting peaks at the binding energy of 337.1 eV and 342.5 eV assigned to 3d5/2 and 3d3/2, respectively.47 The Pd5/2 binding energy of 337.1 eV was in between metallic Pd and PdO, which demonstrated the Pd doping stably dissolve into the spinel crystal lattice.48
The Ce 3d XPS spectra was illustrated in Fig. 7d, where v and u represented the spin–orbit coupling of 3d5/2 and 3d3/2, respectively. The binding energy peaks of v, v′′, v′′′, u, u′′ and u′′′, locating at 881.9 eV, 888.7 eV, 897.7 eV, 900.32 eV, 907.6 eV and 916.5 eV, respectively, corresponded to Ce4+. And the peaks v0, v′ and u′ at the binding energy of 879.3 eV, 884.8 eV and 900.32 eV were assigned to Ce3+.49 While the Ce4+ peaks were more over that of Ce3+, it indicated that the Ce co-existed as Ce4+ and Ce3+ and the Ce4+ was dominant in the Ni1−xCexCo1.95Pd0.05O4 catalysts.50 Meanwhile, the low-temperature deNOX performance was efficiently enhanced, which was due to the possibility of oxygen storage and release by the coexistence of Ce4+ and Ce3+ in the catalysts, and that was consistent with the results of SCR activity test of Ni1−xCexCo1.95Pd0.05O4 catalysts. A shift was observed in Ni, Co and Pd XPS spectra with an addition of doping Ce, which was due to the big electronegativity of cerium, it resulted in a shift to lower bingding energy of Ni 2p, Co 2p and Pd 3d.
4. Conclusions
The nickel cobaltite spinels doped with cerium were investigated for the SO2-resistence in the process of NO selective reduction by hydrogen. It is confirmed that the addition of a small amount (0.03–0.07 at%) of cerium not only effectively enhances the catalytic activity of Ni1−xCexCo1.95Pd0.05O4 catalysts but also improves their resistance towards SO2. On introducing 500 ppm SO2 to the flue gas, the deNOX performance results in a significant decrease but still shows a higher value of 61–72% than that of NiCo1.95Pd0.05O4 catalyst. Further rising Ce inclusion in the catalyst increased the SO2-resistance and in varying dopant amount also exhibited a linear relation towards SO2-resistance. When the SO2 was removed, the NOX conversion of Ni1−xCexCo1.95Pd0.05O4 catalysts greatly recovered, especially when the Ce doping was 0.07 at%, the NOX conversion quickly recovered to the initial value, and exhibited a broad temperature window of 140–350 °C with a maximum NOX conversion of 98% at 190 °C. Meanwhile, addition of 5% H2O to inlet gas, slightly decreases the deNOX performance of all catalysts for 200 min. The NOX conversion nearly recovered to the initial level upon the withdrawal of H2O, among of all, the Ni0.93Ce0.07Co1.95Pd0.05O4 catalyst showed better H2O-resistance.The effect of GHSV, NO/H2 ratios and O2 content on the NOX conversion over the Ni0.93Ce0.07Co1.95Pd0.05O4 catalysts were also investigated. As the increase of GHSV from 4800 to 9600 mL h−1 g−1, the NOX conversion faintly decreased from 98% to 96% in the temperature range of 150–270 °C. A decrease of NO/H2 ratios from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, resulted in the increase of deNOX performance, and showed the maximum NOX conversion increase from 60% to 96%. As the O2 content increased from 0% to 6%, the removal efficiency of NOX gradually decreased from 100% to 75% at 200 °C, although the O2 content reached up to 6%, the NOX conversion still maintained high removal level of over 75% at 170–250 °C range, these results indicated that the addition of cerium efficiently enhanced oxygen resistance of the catalyst.
10, resulted in the increase of deNOX performance, and showed the maximum NOX conversion increase from 60% to 96%. As the O2 content increased from 0% to 6%, the removal efficiency of NOX gradually decreased from 100% to 75% at 200 °C, although the O2 content reached up to 6%, the NOX conversion still maintained high removal level of over 75% at 170–250 °C range, these results indicated that the addition of cerium efficiently enhanced oxygen resistance of the catalyst.
The XPD and XRS analysis showed the good cubic spinel-type structures, and the Ce doped in the spinels with two chemical valence states of Ce3+ and Ce4+, that was conducive to oxygen store and release, enhancing the SCR activity. In the H2-TPR and NH3-TPD analysis, when the Ce was doped in catalyst, resulted in the shift of reductive temperature to low temperature, that enhanced the redox property; weakened the strength of weak acid, which was facilitating to NO stripping and beneficial to SCR reaction; enhancing the strong acid sites, added positive effect on SCR reaction. These results were consistent with the SCR activity data.
Acknowledgements
This research is based on work supported by the National Natural Science Foundation of China (21277045). Thanks to Zhenhua Zhou for providing solutions to the experimental process and writing assistance.References
- C. N. Costa and A. M. Efstathiou, J. Phys. Chem. C, 2007, 111, 3010–3020 CAS.
- A. Fritz and V. Pitchon, Appl. Catal., B, 1997, 13, 1–25 CrossRef CAS.
- G. Qi, R. T. Yang and F. C. Rinaldi, J. Catal., 2006, 237, 381–392 CAS.
- G. G. Olympiou and A. M. Efstathiou, Chem. Eng. J., 2011, 170, 424–432 CrossRef CAS.
- M. Machida, S. Ikeda, D. Kurogi and T. Kijima, Appl. Catal., B, 2001, 35, 107–116 CAS.
- S. S. Kim and S. C. Hong, J. Ind. Eng. Chem., 2010, 16, 992–996 CrossRef CAS.
- F. J. P. Schott, P. Balle, J. Adler and S. Kureti, Appl. Catal., B, 2009, 87, 18–29 CAS.
- C. N. Costa and A. M. Efstathiou, Appl. Catal., B, 2007, 72, 240–252 CAS.
- Y. U. Qing, F. Kong, L. I. Landong and W. U. Guangjun, Chin. J. Catal., 2010, 31, 261–263 Search PubMed.
- G. L. Chiarello, D. Ferri, J. D. Grunwaldt, L. Forni and A. Baiker, J. Catal., 2007, 252, 137–147 CAS.
- B. Q. Jiang, Z. B. Wu, Y. Liu, S. C. Lee and W. K. Ho, J. Phys. Chem. C, 2010, 114, 4961–4965 CAS.
- H. Chang, X. Chen, J. Li, L. Ma, C. Wang, C. Liu, J. W. Schwank and J. Hao, Environ. Sci. Technol., 2013, 47, 5294–5301 CrossRef CAS PubMed.
- T. Yoshinari, K. Sato, M. Haneda, Y. Kintaichi and H. Hamada, Appl. Catal., B, 2003, 41, 157–169 CAS.
- X. Li, S. Zhang, Y. Jia, X. Liu and Q. Zhong, J. Nat. Gas Chem., 2012, 21, 17–24 CAS.
- Z. Wu, R. Jin, Y. Liu and H. Wang, Catal. Commun., 2008, 9, 2217–2220 CrossRef CAS.
- J. Fan and W. Huang, Chin. J. Catal., 2011, 32, 139–143 CAS.
- H. Kavas, A. Baykal, M. S. Toprak, Y. Köseoğlu, M. Sertkol and B. Aktaş, J. Alloys Compd., 2009, 479, 49–55 CrossRef CAS.
- Z. Yu, H. Li, X. Zhang, N. Liu, W. Tan, X. Zhang and L. Zhang, Biosens. Bioelectron., 2016, 75, 161–165 CrossRef CAS PubMed.
- Z. Q. Liu, K. Xiao, Q. Z. Xu, N. Li, Y. Z. Su, H. J. Wang and S. Chen, RSC Adv., 2013, 3, 4372–4380 RSC.
- M. N. Iliev, P. Silwal, B. Loukya, R. Datta, D. H. Kim, N. D. Todorov, N. Pachauri and A. Gupta, J. Appl. Phys., 2013, 114, 033514–033515 Search PubMed.
- S. Y. Jiang and R. X. Zhou, Fuel Process. Technol., 2015, 133, 220–226 CrossRef CAS.
- W. U. Peng, Y. Liu, F. Zhang, W. U. Guangjun, L. Baocheng and N. Guan, Chin. J. Catal., 2008, 29, 191–196 Search PubMed.
- L. Yuan, X. Zheng, K. Duan, H. Hu, J. Wang, S. I. Woo and Z. Liu, Front. Environ. Sci. Eng., 2013, 7, 457–463 CrossRef CAS.
- S. Yang, X. Wang, W. Chu, Z. Song and S. Zhao, Appl. Catal., B, 2011, 3, 380–385 Search PubMed.
- L. Huilin, Z. Guangbo, B. Rushan, C. Yongjin and D. Gidaspow, Fuel, 2000, 79, 165–172 CAS.
- H. Chang, J. Li, J. Yuan, L. Chen, Y. Dai, H. Arandiyan, J. Xu and J. Hao, Catal. Today, 2013, 201, 139–144 CrossRef CAS.
- K. J. Lee, M. S. Maqbool, P. A. Kumar, K. H. Song and H. P. Ha, Catal. Lett., 2013, 143, 988–995 CAS.
- S. Yun, T. Aikebaier, Q. Xie, S. Chen and H. Yu, Appl. Catal., B, 2014, 150–151, 630–635 Search PubMed.
- R. Jin, L. Yue, W. Yan, W. Cen, Z. Wu, H. Wang and X. Weng, Appl. Catal., B, 2014, 148–149, 582–588 CrossRef CAS.
- R. Burch and M. D. Coleman, Appl. Catal., B, 1999, 23, 115–121 CAS.
- M. Leicht, F. J. P. Schott, M. Bruns and S. Kureti, Appl. Catal., B, 2012, 117–118, 275–282 CAS.
- X. Lu, C. Song, S. Jia, Z. Tong, X. Tang and Y. Teng, Chem. Eng. J., 2015, 260, 776–784 CAS.
- M. D. Amiridis, I. E. Wachs, G. Deo and J.-M. Jehng, J. Catal., 1996, 161, 247–253 CAS.
- F. Bustamante, F. Córdoba, M. Yates and C. M. D. Correa, Appl. Catal., A, 2002, 234, 127–136 CAS.
- J. M. Giraudon, A. Elhachimi, F. Wyrwalski, S. Siffert, A. Aboukaïs, J. F. Lamonier and G. Leclercq, Appl. Catal., B, 2007, 75, 157–166 CrossRef CAS.
- T. H. Lim, S. J. Cho, H. S. Yang, M. H. Engelhard and D. H. Kim, Appl. Catal., A, 2015, 505, 62–69 CAS.
- Y. Gou, X. Liang and B. Chen, J. Alloys Compd., 2013, 574, 181–187 CAS.
- H. Zhu, Z. Qin, W. Shan, W. Shen and J. Wang, J. Catal., 2004, 225, 267–277 CAS.
- M. Imran, D. H. Kim, W. A. Al-Masry, A. Mahmood, A. Hassan, S. Haider and S. M. Ramay, Polym. Degrad. Stab., 2013, 98, 904–915 CAS.
- Z. Huang, Z. Zhu, Z. Liu and Q. Liu, J. Catal., 2003, 214, 213–219 CAS.
- J. H. Li, C. C. Wang, C. J. Huang, Y. F. Sun, W. Z. Weng and H. L. Wan, Appl. Catal., A, 2010, 382, 99–105 CAS.
- Z.-Q. Liu, K. Xiao, Q.-Z. Xu, N. Li, Y.-Z. Su, H.-J. Wang and S. Chen, RSC Adv., 2013, 3, 4372 RSC.
- J. Yang, M. Cho and Y. Lee, Biosens. Bioelectron., 2016, 75, 15–22 CAS.
- X. Wang, W. Wen, J. Mi, X. Li and R. Wang, Appl. Catal., B, 2015, 176–177, 454–463 CAS.
- J. G. Kim, D. L. Pugmire, D. Battaglia and M. A. Langell, Appl. Surf. Sci., 2000, 165, 70–84 CAS.
- G. A. Babu, G. Ravi and Y. Hayakawa, International Journal of Science and Engineering Applications, 2014, 1, 17–20 Search PubMed.
- J. M. Giraudon, A. Elhachimi, F. Wyrwalski, S. Siffert, A. Aboukaïs, J. F. Lamonier and G. Leclercq, Appl. Catal., B, 2007, 75, 157–166 CrossRef CAS.
- G. C. M. Rodríguez and B. Saruhan, Appl. Catal., B, 2010, 93, 304–313 CrossRef.
- S. Watanabe, X. Ma and C. Song, J. Phys. Chem. C, 2009, 113, 14249–14257 CAS.
- B. Shen, Y. Wang, F. Wang and T. Liu, Chem. Eng. J., 2014, 236, 171–180 CAS.
| This journal is © The Royal Society of Chemistry 2016 |




