Facile synthesis of core–shell carbon nanotubes@MnOOH nanocomposites with remarkable dielectric loss and electromagnetic shielding properties
Zhifan Zhanga,
Wenhui Danga,
Chengjun Dong*ab,
Gang Chenab,
Yude Wangb and
Hongtao Guan*ab
aSchool of Materials Science & Engineering, Yunnan University, 650091 Kunming, People's Republic of China. E-mail: dongchjun@hotmail.com; htguan06@ynu.edu.cn; Fax: +86 871 65035376; Tel: +86 871 65031124
bYunnan Province Key Lab of Micro-Nano Materials and Technology, Yunnan University, 650091 Kunming, People's Republic of China
First published on 6th September 2016
Abstract
Carbon nanotubes (CNTs)@MnOOH core–shell nanocomposites, in which the CNTs core is uniformly coated with a shell of MnOOH nanoflakes, were successfully synthesized using a facile hydrothermal method. The appearance of sodium dodecyl benzene sulfonate (SDBS) plays a key role in forming the unique CNTs@MnOOH core–shell. A high dielectric loss tangent value was observed for the samples with/without SDBS. Significantly, the CNTs@MnOOH core–shell nanocomposites exhibit an electromagnetic shielding effectiveness of 13–15 dB in the frequency range of 8–18 GHz, which can be attributed to the improved absorption loss, resulting from the increased electrical conductivity, and the improved impedance matching of the core–shell structure. Moreover, the increasing interfaces between the MnOOH and CNTs favor the electromagnetic attenuation performance. Our findings strongly confirm that the CNTs@MnOOH core–shell nanocomposites can be considered as a potential candidate for electromagnetic applications.
1. Introduction
Over recent years, due to the rise and popularity in the use of high operating frequency and bandwidth in electronic systems, there are more and more concerns on the deterioration of the microwave environment referred to as electromagnetic interference (EMI).1 EMI not only arouses malfunctions of precise electronic devices, but also has great harmful influences on human beings. Hence, in order to maintain the electromagnetic compatibility, lightweight EMI shielding materials are urgently needed. EMI shielding refers to the synergistic effects of reflection loss, absorption loss and multi-reflections of a shielding material against the penetration of EMI radiation through the material.1,2 The shielding effectiveness of a shield material correlates closely to its electric conductivity and electromagnetic parameters. In this sense, carbon materials, functional ceramics, metals and some metal oxides are usually applied as EMI shielding materials due to their good electrical and electromagnetic performance.2–5 Especially, the shielding properties of metal oxide materials are mainly dominated by their absorption loss, which can be controlled by the dielectric permittivity and magnetic permeability values.6In the large group of metal oxides, manganese oxides, in various forms, seem to be one of the most important engineering materials due to their considerable significance in several industrial applications, such as catalysts, molecular sieves and rechargeable batteries.7 Since its first report for microwave absorption use,8,9 manganese oxide has gained extensive and intensive studies for electromagnetic applications. To improve its electromagnetic performance, a series of attempts have been made to prepare manganese oxide with unique morphological structures10–13 and doped or combined with magnetic metal ions.14–16 However, it is still a big challenge to tailor manganese oxides with highly efficient EMI shielding effectiveness due to their relatively low electrical conductivity.
Due to their good electrical properties, carbon nanotubes (CNTs) are capable of dissipating electrostatic charges or shielding devices from electromagnetic radiation, which makes them one of the preferred and best characterized filler materials for military and civilian products.17,18 Eswaraiah et al. reported a maximum shielding effectiveness of about 18–20 dB in the frequency range of 8–12 GHz by a 7 wt% multi-wall carbon nanotubes (MWCNTs) loaded PVDF matrix. After loading 5 wt% MnO2 nanotubes (MNTs) and 1 wt% MWCNTs, the PVDF based composite delivered a shielding property of about 20 dB.1 Ting et al. have observed a microwave absorption of −23 dB at 7 GHz for epoxy composites filled with 1 wt% MWCNTs and 25 wt% MnO2 powder.19
Additionally, as a type of manganese oxide, MnOOH is of considerable importance in catalysts, batteries, molecular sieves and supercapacitor electrodes, due to its excellent chemical and physical properties. Meanwhile, MnOOH is an important precursor used for the synthesis of MnO2 based materials and compounds.20 However, MnOOH is rarely applied for electromagnetic use because of its weak electrical properties. If combined with CNTs, MnOOH is promised to have great performance in the electromagnetic absorption field. Nevertheless, CNTs tend to agglomerate into bundles when dispersed in either water or organic solvents due to the strong van der Waals interactions and thus, the use of surfactants seems to be quite necessary when dispersing CNTs in solution.21 Sodium dodecyl benzene sulfonate (SDBS), as a kind of anionic surfactant, has been shown to be able to stabilize CNTs dispersions.22 In the present work, the CNTs@MnOOH nanocomposites were synthesized with the aid of SDBS using a facile hydrothermal method. Then, their electromagnetic characteristics and EMI shielding effectiveness were investigated. The results show that with the assistance of SDBS, the CNTs were decorated with tiny MnOOH particulates to form a core–shell structure. The introduction of SDBS can improve the electromagnetic properties of the CNTs@MnOOH nanocomposite and thus, the EMI shielding effectiveness. To the utmost of our knowledge, this is the first report on the electromagnetic performance of a core–shell CNTs@MnOOH nanocomposite.
2. Materials and methods
2.1 Chemicals
Potassium permanganate (KMnO4), sodium dodecyl benzene sulfonate (SDBS), carbon nanotubes (CNTs) and other auxiliary reagents were purchased from Aladdin Reagent Co. Ltd. Deionized water was used throughout the experiments.2.2 Synthesis of the nanocomposites
The MnOOH combined CNT nanocomposites were synthesized using a simple hydrothermal method. The CNTs were first pretreated with 1 M hydrochloric acid (HCl) and then, rinsed with absolute ethanol and deionized water. Then, 0.1 g of CNTs was ultrasonically dispersed in 25 mL of deionized water for 2 h. Subsequently, 0.2 g of KMnO4 was added slowly into the CNTs suspension with magnetic stirring for another 12 h. The mixed solution was transferred to a Teflon-lined stainless autoclave and maintained at 150 °C for 6 h. After cooling down to room temperature, the black precipitate was centrifuged and rinsed several times with absolute ethanol and water, respectively. Finally, the black products were dried at 100 °C for 6 h and collected for further characterization. Regarding the composites with SDBS, 0.04 g SDBS was added to the mixture before being transferred to the autoclave. The subsequent procedure was similar to that described above. Hereafter, the nanocomposites with and without SDBS were labeled as CNTs@MnOOH and CNTs/MnOOH, respectively.2.3 Characterization
Powder X-ray diffraction (XRD) patterns were collected on a Rigaku TTR-III diffractometer with Cu Kα radiation (λ = 1.5418 Å) in the diffraction (2θ) range of 10–80°. The Raman spectra were recorded on a Renishaw inVia Raman microscope at room temperature with a 514 nm wavelength laser excitation and a power output of 3 mW. The particle morphologies and microstructures were observed on a JEM-2100 transmission electron microscope (TEM) and high-resolution transmission electron microscope (HRTEM) with an accelerating voltage of 200 kV. The samples for TEM analysis were prepared by dispersing the sample in absolute ethanol with sonication for a few seconds and then dropped on a copper grid with an amorphous carbon support film.23,242.4 Electromagnetic measurements
The electromagnetic properties of the nanocomposites were characterized by complex permittivity and permeability analysis. Firstly, the nanocomposites were homogeneously mixed with paraffin wax in a mass ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Afterwards, the mixture was pressed into toroidal-shaped samples (ϕout = 7.00 mm, ϕin = 3.04 mm) with a thickness of 2 mm. The complex permittivity and permeability of the nanocomposites were recorded on an Agilent N5230A vector network analyzer (VNA) in the frequency range of 8–18 GHz using a coaxial cable method.
1. Afterwards, the mixture was pressed into toroidal-shaped samples (ϕout = 7.00 mm, ϕin = 3.04 mm) with a thickness of 2 mm. The complex permittivity and permeability of the nanocomposites were recorded on an Agilent N5230A vector network analyzer (VNA) in the frequency range of 8–18 GHz using a coaxial cable method.
3. Results and discussion
3.1 Phase crystallinity
The XRD patterns of the products are shown in Fig. 1. It is found that all the products with/without SDBS are mainly indexed to monoclinic manganite-type manganese oxide (γ-MnOOH phases) (JCPDS no. 41-1379) with a space group P21/c(14). Without SDBS, a few peaks are observed for the tetragonal phase of Mn3O4 (JCPDS no. 24-0734) in CNTs/MnOOH (Fig. 1(c)). Besides, a weak peak was observed at about 11.6° for each sample because of the existence of the CNTs.25,26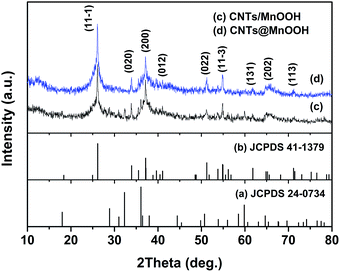 | ||
| Fig. 1 The XRD patterns of the as-synthesized CNTs/MnOOH (c) and CNTs@MnOOH (d), together with JCPDS 24-0374 (Mn3O4) (a) and JCPDS 41-1379 (MnOOH) (b). | ||
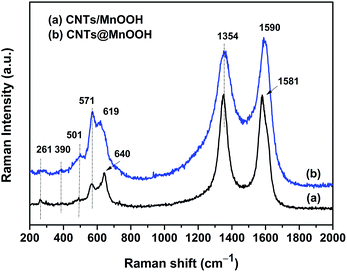
The local structure of the manganese oxides were effectively analyzed using Raman spectroscopy. As can be seen in Fig. 2, the existence of CNTs was clearly confirmed by the characteristic G band (1581 or 1590 cm−1) and D band (1352 cm−1).4,27 Moreover, the peaks at 261, 390, 501, 571 and 619 cm−1 were assigned to the Mn–O vibrational modes of MnOOH.28
An obvious peak at 640 cm−1 appears for CNTs/MnOOH, as shown in Fig. 2(a), which is in good accordance with the major vibrational features of Mn3O4,29 again indicating the presence of the Mn3O4 phase.
Hence, both the XRD and Raman analyses above illustrate that the introduction of SDBS was beneficial for the formation of pure MnOOH phases. It's worth noting that our outcomes are quite different from those of manganese oxide/carbon composites prepared using a similar process reported in the literature. For instance, Ma et al. reacted 1.0 g of acetylene black with 200 mL of 0.01 M KMnO4 solution at 70 °C to obtain birnessite-type MnO2 (JCPDS 42-1317).25 Xia et al. hydrothermally reduced 0.3 g of KMnO4 (dissolved in 25 mL of deionized water) with 0.1 g of CNTs at 150 °C for 6 h. The resulting products were found to be monoclinic potassium birnessite-type MnO2 (JCPDS 80-1098).30,31 Collectively, this demonstrates that not only the reaction temperature and time, but also the raw materials will comprehensively affect the process of the reduction reaction, thereby the final products.
3.2 Morphologies and microstructures
The evolution of the morphologies and microstructures with/without SDBS is compared in Fig. 3. Without SDBS, the MnOOH appears as assemblies of nanoparticles (10–20 nm in size) and nanowires (10 nm in diameter and several hundred nanometers in length) on the CNTs, as displayed in Fig. 3(a). The HRTEM image of the CNTs/MnOOH nanocomposites are shown in Fig. 3(c), in which the interplanar spacings were measured to be 0.34 and 0.52 nm, which corresponds to the diffraction of the CNTs and the (100) plane of the γ-MnOOH structure, respectively. In contrast, the CNTs@MnOOH nanocomposites synthesized with SDBS show totally different morphological features. As shown in Fig. 3(b), the MnOOH takes on structures of nanoflakes with lengths of about 30–50 nm. Apparently, most of the CNTs are uniformly decorated by the MnOOH nanoflakes, demonstrating the formation of the core–shell CNTs@MnOOH structure. The HRTEM image of the CNTs@MnOOH nanocomposites gives the same data to that found for the nanocomposite prepared without SDBS (Fig. 3(d)). | ||
| Fig. 3 TEM (a and b) and HRTEM (c and d) images of the CNTs/MnOOH (a and c) and CNTs@MnOOH (b and d) samples. | ||
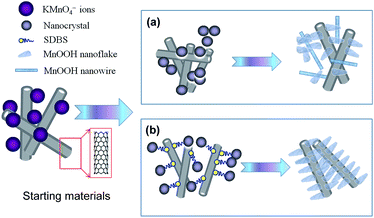
Up to now, a large series of reports are available on the preparation of MnO2/CNT composites based on the redox reaction between KMnO4 and CNTs.30–34 Instead of coupling MnOOH with CNTs,35 MnO2 was mainly obtained. Here, the formation of nanostructures between MnOOH and the CNTs was plausibly attributed to the hydrothermal treatment in our experiment. The possible formation mechanisms are proposed and are schematically illustrated in Fig. 4. At the very beginning of the reaction, MnOOH nanocrystals are formed on the surface of the CNTs after mixing the CNTs with KMnO4. Upon prolonging the reaction, the nanocrystalline MnOOH may further serve as nucleation sites for the crystals to grow. Due to the aggregation characteristics of CNTs, the CNTs tend to intertwine with each other and thus, the MnOOH nucleation sites are conglomerated (Fig. 4(a)) to form the morphological structures shown in Fig. 3(a). Upon the addition of SDBS, the reaction is totally different. The tail segments and most of the benzene rings in the SDBS structure are inclined to be positioned close to the CNTs surface while the sulfonate headgroups are exposed to water.22
Thus, great electrostatic repulsion between the CNTs causes the dispersion of the CNTs due to the negatively charged sulfonate groups. Under this circumstance, the MnOOH nanocrystallines formed on the surface of CNTs are grown freely, as shown in Fig. 4(b), and hence constitutes the uniform MnOOH coatings, as shown in Fig. 3(b).
3.3 Electromagnetic parameters
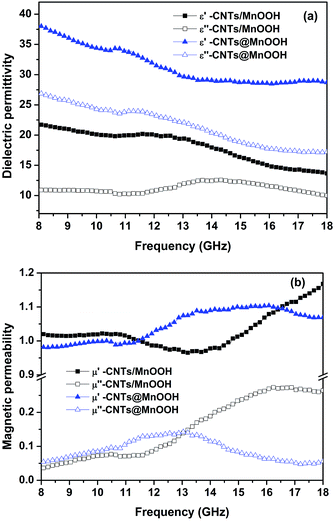
The electromagnetic properties, in terms of complex permittivity (εr = ε′r − jε′′r) and complex permeability (μr = μ′r − jμ′′r), of the CNTs/MnOOH and CNTs@MnOOH were examined using the coaxial cable method in the frequency range of 8–18 GHz, as shown in Fig. 5(a) and (b). The real permittivity (ε′r) for both samples all show decreasing trends upon increasing the frequency. From Fig. 5(a), the ε′r of CNTs/MnOOH and CNTs@MnOOH decrease from 21.8 and 38.1 to 13.7 and 28.7, respectively. It is also obvious that the CNTs@MnOOH nanocomposite exhibits much higher ε′r values than those of the CNTs/MnOOH nanocomposite. According to classical electromagnetic theory, the real part of permittivity means the polarization ability of materials under an alternating electromagnetic field. As revealed in Fig. 3(b), the shell of MnOOH nanoflakes decorated on the surfaces of CNTs causes additional charge carriers and interfaces in the CNTs@MnOOH nanocomposite, resulting in the much higher ε′r values.36 Furthermore, the residual bonds and clustered defects in the MnOOH and CNTs structures favor the orientation polarization, which further enhances the ε′r values.29,36 Upon increasing the external field, the polarization cannot follow the alternation of the field and thus, endows a reduction in the real permittivity. This phenomenon is the so-called frequency dispersive effect and has been proved to be beneficial for broadening the electromagnetic absorption bandwidth.37 In addition, the imaginary permittivity (ε′′r) values of the samples show much milder trends with increasing frequency. As displayed in Fig. 5(a), the CNTs@MnOOH nanocomposite has a high ε′′r value of 27.0 at 8 GHz and then decreases to 17.2 at 18 GHz. In contrast, the CNTs/MnOOH has much lower ε′′r values of 11.0 to 12.5 in the same frequency range.Fig. 5(b) presents the magnetic permeability values of both CNTs/MnOOH and CNTs@MnOOH. The real permeability (μ′r) for both samples demonstrates an initial decreasing tendency and then increases gradually with the frequency. The μ′r of CNTs/MnOOH gradually declines from 1.09 to 0.97 at 13.4 GHz and then increases with further increases in the frequency up to 18 GHz. The μ′r of the CNTs@MnOOH shows a similar variation with frequency except that its mutation point shift to a much lower frequency. On the other hand, the variation of μ′′r of the samples exhibits an inverse tendency to that of μ′r. As shown in Fig. 5(b), the μ′′r curves for both samples increase with frequency at first and then subsequently have a tendency to gradually decrease at 16.2 GHz and 13.2 GHz, respectively, for the CNTs/MnOOH and CNTs@MnOOH samples. Significantly, the μ′r values for both samples are larger than unity in most of the frequency ranges, illustrating their paramagnetic characteristics. For instance, the μ′r values of CNTs/MnOOH are all larger than 1.0 in the frequency range of 8–11.5 GHz and 14.6–18 GHz. In the case of CNTs@MnOOH, this phenomenon was observed when the frequency was higher than 11.5 GHz.
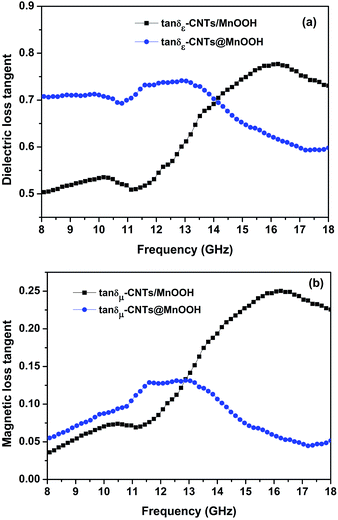
Loss tangents are often applied to indicate the loss properties of an electromagnetic absorbing material. The dielectric (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε = ε′′/ε′) and magnetic (tan
δε = ε′′/ε′) and magnetic (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ = μ′′/μ′) loss tangents of the samples were characterized. In Fig. 6(a), the tan
δμ = μ′′/μ′) loss tangents of the samples were characterized. In Fig. 6(a), the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε values for both samples gradually increase from 8 GHz and then, obtain a dip value at 11.2 GHz, which is coincident with the decreasing tendency of the dielectric permittivity. At the higher frequency bands over 11.2 GHz, there appears to be an obvious increase in tan
δε values for both samples gradually increase from 8 GHz and then, obtain a dip value at 11.2 GHz, which is coincident with the decreasing tendency of the dielectric permittivity. At the higher frequency bands over 11.2 GHz, there appears to be an obvious increase in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε that is followed by a subsequent decrease. The tan
δε that is followed by a subsequent decrease. The tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε of CNTs/MnOOH remains at 0.5–0.8 in the whole frequency range. After the addition of SDBS, the tan
δε of CNTs/MnOOH remains at 0.5–0.8 in the whole frequency range. After the addition of SDBS, the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε reaches as high as 0.7–0.75 in the frequency range below 13 GHz and then declines to about 0.6. Regardless the addition of SDBS, our results are much bigger than most other manganese oxide/carbon composites.4,38–40
δε reaches as high as 0.7–0.75 in the frequency range below 13 GHz and then declines to about 0.6. Regardless the addition of SDBS, our results are much bigger than most other manganese oxide/carbon composites.4,38–40
The great dielectric loss properties of CNTs/MnOOH and CNTs@MnOOH may be attributed to their excellent polarization performance, which was highly related to the chemical and physical properties of the materials, such as the crystal and morphological structures, surface areas and surface chemical states. As discussed in previous publications, the dielectric properties of manganese oxides mainly come from the Debye dipole polarization, space charge polarization and interfacial polarization, and their related relaxations.16 According to the free electron theory, the imaginary permittivity of a dielectric material can be given as follows:
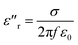 | (1) |
Based on eqn (1), the dynamic electrical conductivities as a function of frequency from 8–18 GHz were calculated for both CNTs/MnOOH and CNTs@MnOOH, as illustrated in Fig. 7. It is clearly shown that the electrical conductivity of the composites was enhanced upon the addition of SDBS, which is believed to the reason of uniform CNTs dispersion (Fig. 3(b)). Hence, CNTs@MnOOH shows a much higher ε′′r value than CNTs/MnOOH and then a higher dielectric loss.
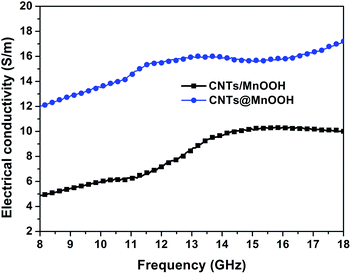 | ||
| Fig. 7 The dynamic electrical conductivity as a function of the frequency in the range from 8 to 18 GHz for both the CNTs/MnOOH and CNTs@MnOOH samples. | ||
Fig. 5(b) shows the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ curves obtained for our samples, suggesting a pronounced increase with frequency. After reaching a maximum value, tan
δμ curves obtained for our samples, suggesting a pronounced increase with frequency. After reaching a maximum value, tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ gives a sharp decrease. A decreasing shift of about 4 GHz was observed from CNTs/MnOOH (16 GHz) to CNTs@MnOOH (12 GHz), which also coincides with the magnetic permeability. The magnetic response of manganese oxides may be ascribed to the ferromagnetic coupling of Mn4+–O2−–Mn4+. When interacted with the electromagnetic field, there will be an induced eddy current loss, magnetic loss and residual loss in the composites.41
δμ gives a sharp decrease. A decreasing shift of about 4 GHz was observed from CNTs/MnOOH (16 GHz) to CNTs@MnOOH (12 GHz), which also coincides with the magnetic permeability. The magnetic response of manganese oxides may be ascribed to the ferromagnetic coupling of Mn4+–O2−–Mn4+. When interacted with the electromagnetic field, there will be an induced eddy current loss, magnetic loss and residual loss in the composites.41
To make a more clear explanation of the microwave response of the CNTs/MnOOH composites, the Debye dielectric relaxation Cole–Cole semicircle was performed, in which the ε′r and ε′′r of the dielectric permittivity are shown as follows:42,43
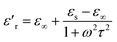 | (2) |
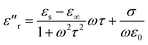 | (3) |
According to eqn (2) and (3), the relationship between ε′r and ε′′r is deduced as follows:
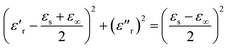 | (4) |
Thus, the plot of ε′r versus ε′′r is a single semicircle, corresponding to one Debye relaxation process.23,29,42 Fig. 8(a) and (b) show the Cole–Cole semicircle curves obtained for both samples. As shown, there is more than one circle, indicating several relaxation processes with a dissymmetrical distribution of the relaxation time.44 There are ordered micro-areas with nanometer scales in the unordered relaxation body of the manganese oxides, which play key roles in the dielectric relaxation of manganese oxide materials. When the discontinuous relaxation times are distributed around several special relaxation time τ0, the Cole–Cole semicircle is deflexed to several overlapped parts.44
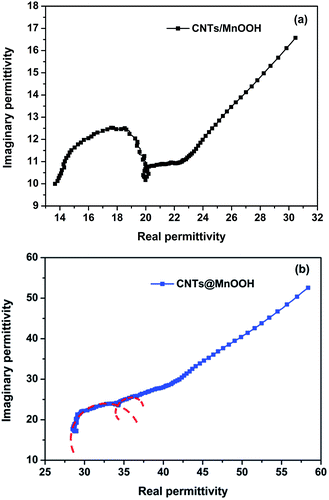 | ||
| Fig. 8 The Cole–Cole semicircles obtained for the CNTs/MnOOH (a) and CNTs@MnOOH samples (the red dashed lines indicate the fitting curve) (b). | ||
3.4 EMI shielding effectiveness
As a potential application field, the EMI shielding effectiveness (SE) of the CNTs/MnOOH and CNTs@MnOOH nanocomposites are carefully discussed upon the Schelkunoff formula.40 The EMI shielding for a shield material can be expressed using eqn (5).| SE = SEA + SER + SEM | (5) |
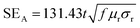 | (6) |
SER = 168.2 + 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lg(σr/(fμr)) lg(σr/(fμr))
| (7) |
SEM = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lg[1–2 × 10−0.1×SEA lg[1–2 × 10−0.1×SEA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos(0.23 × SEA) + 10−0.2×SEA] cos(0.23 × SEA) + 10−0.2×SEA]
| (8) |
Based on eqn (5)–(8), the EMI shielding effectiveness curves obtained for both the CNTs/MnOOH and CNTs@MnOOH samples are displayed in Fig. 9(a) and (b), which show clearly an increasing trend with frequency. The SER and SEM values obtained for both samples show similar values in the range of 1–4 dB and 0–1 dB, respectively. In addition, as shown in Fig. 9, the SEA values are much higher than the SER values, indicating that the primary shielding mechanism of the samples was absorption. Moreover, the CNTs@MnOOH sample exhibits a higher SEA value than CNTs/MnOOH, thus presenting a higher shielding effectiveness, especially in the frequency range below 13 GHz.
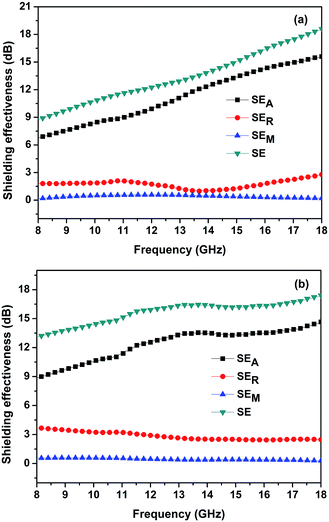 | ||
| Fig. 9 The SE of the CNTs/MnOOH (a) and CNTs@MnOOH (b) samples. SEA, SER and SEM represent the shielding caused by absorption loss, reflection loss and multi-reflection loss, respectively. | ||
The EMI shielding capacity of a shield material is associated closely with the microstructure of the fillers and the interfaces between the matrix and the fillers. In the present work, three main reasons may be used to explain the enhanced EMI shielding performance of CNTs@MnOOH. Firstly, as indicated in Fig. 3(b), the incorporation of SDBS improves the dispersion uniformity of the CNTs, thereby facilitating the improvement in the electrical conductivity. As investigated using eqn (1), a higher conductivity leads to higher ε′′r and thus, better energy conversion effectiveness.36 Secondly, the CNTs@MnOOH core–shell structure ameliorates the impedance matching between the input impedance of the composite (Zin) and the space impedance (Z0). When the EMI incident on the surface of the composite, the matching impedance will lead the incident wave transmitting into the composite and then, subsequently attenuated.46 Thirdly, the core–shell structure provides a large surface area and a large number of interfaces between the MnOOH and CNTs, producing space polarization and interfacial polarization, which plays a great role towards the EMI wave attenuation. Moreover, the charge carriers and both the defects and residual bonds on the surfaces also increase the absorption of the incident EMI waves. Therefore, the results indicate the present work shows that the as-synthesized CNTs@MnOOH nanocomposites can be considered as a potential candidate for EMI shielding.
4. Conclusions
CNTs@MnOOH nanocomposites were successfully synthesized via a facile hydrothermal method using SDBS as the surfactant. The addition of SDBS greatly improves the dispersion uniformity of the CNTs to form the CNTs@MnOOH core–shell structure and thus, its dielectric property is improved. An EMI shielding effectiveness of 13–15 dB in the frequency range of 8–18 GHz was observed for the CNTs@MnOOH nanocomposite. The enhanced shielding capacity can be ascribed to the absorption loss resulting from the improved electrical conductivity and the impedance matching of the core–shell structure.Acknowledgements
This work is financially supported by the National Nature Science Foundation of China (Grant No. 11564042), the Application Basic Research Fund of Yunnan Province (Grants No. 2013FB006 and 2014FB110), the Youth Talents Program of Yunnan University (Grant No. XT412003) and the Project of the Department of Education of Yunnan Province (2015Y008).Notes and references
- V. Eswaraiah, V. Sankaranarayanan and S. Ramaprabhu, Nanoscale Res. Lett., 2011, 6, 137 CrossRef PubMed.
- H. T. Guan, S. H. Liu, Y. P. Duan and C. Ji, Cem. Concr. Compos., 2006, 28, 468 CrossRef CAS.
- Z. H. Guo, S. Park, H. T. Hahn, S. Y. Wei, M. Moldovan, A. B. Karki and D. P. Young, J. Appl. Phys., 2007, 101, 09M511 Search PubMed.
- J. Kong, M. M. Kong, X. F. Zhang, L. X. Chen and L. N. An, ACS Appl. Mater. Interfaces, 2013, 5, 10367 CAS.
- C. J. Luo, W. Y. Duan, X. W. Yin and J. Kong, J. Phys. Chem. C, 2016, 120, 18721 CAS.
- C. G. Wei, C. J. Xu, B. H. Li, D. Nan, J. Ma and F. Y. Kang, Mater. Res. Bull., 2012, 47, 1740 CrossRef CAS.
- T. K. Gupta, B. P. Singh, V. N. Singh, S. Teotia, A. P. Singh, I. Elizabeth, S. R. Dhakate, S. K. Dhawan and R. B. Mathur, J. Mater. Chem. A, 2014, 2, 4256 CAS.
- H. T. Guan, S. H. Liu, Y. B. Zhao and Y. P. Duan, J. Electron. Mater., 2006, 35, 892 CrossRef CAS.
- H. T. Guan, Y. B. Zhao, S. H. Liu and S. P. Lv, Eur. Phys. J.: Appl. Phys., 2006, 36, 235 CrossRef CAS.
- M. Zhou, X. Zhang, L. Wang, M. J. Wei, L. Wang, K. W. Zhu and B. X. Feng, Mater. Chem. Phys., 2011, 130, 1191 CrossRef CAS.
- M. Zhou, X. Zhang, L. Wang, J. M. Wei, K. W. Zhu and B. X. Feng, J. Nanosci. Nanotechnol., 2013, 13, 904 CrossRef CAS PubMed.
- G. S. Wang, S. He, X. Luo, B. Wen, M. M. Lu, L. Guo and M. S. Cao, RSC Adv., 2013, 3, 18009 RSC.
- H. T. Guan, J. B. Xie, G. Chen and Y. D. Wang, Mater. Chem. Phys., 2014, 143, 1061 CrossRef CAS.
- Y. P. Duan, Z. Liu, H. Jing, Y. H. Zhang and S. Q. Li, J. Mater. Chem., 2012, 35, 18291 RSC.
- Y. P. Duan, J. L. Chen, Y. H. Zhang, M. Wen and T. M. Wang, J. Supercond. Novel Magn., 2014, 9, 2139 CrossRef.
- H. T. Guan, Y. Wang, C. J. Dong, G. Chen, X. C. Xiao and Y. D. Wang, Ceram. Int., 2015, 41, 5688 CrossRef CAS.
- T. H. Ting, Y. N. Jau and R. P. Yu, Appl. Surf. Sci., 2012, 7, 3184 CrossRef.
- Q. L. He, T. T. Yuan, X. Zhang, X. R. Yan, J. Guo, D. W. Ding, M. A. Khan, D. P. Young, A. Khasanov, Z. P. Luo, J. R. Liu, T. D. Shen, X. Y. Liu, S. Y. Wei and Z. H. Guo, J. Phys. Chem. C, 2014, 118, 24784 CAS.
- T. H. Ting, C. C. Chiang, P. C. Lin and C. H. Lin, J. Magn. Magn. Mater., 2013, 339, 100 CrossRef CAS.
- Z. C. Li, H. L. Bao, X. Y. Miao and X. H. Chen, J. Colloid Interface Sci., 2011, 357, 286 CrossRef CAS PubMed.
- L. A. Girifalco, M. Hodak and R. S. Lee, Phys. Rev. B: Condens. Matter Mater. Phys., 2000, 62, 13104 CrossRef CAS.
- M. Suttipong, N. R. Tummala, B. Kitiyanan and A. Striolo, J. Phys. Chem. C, 2011, 115, 17286 CAS.
- Y. Wang, H. T. Guan, S. F. Du and Y. D. Wang, RSC Adv., 2015, 8, 88979 RSC.
- J. Kong, T. Schmalz, G. Motz and A. H. E. Müller, J. Mater. Chem. C, 2013, 1, 1507 RSC.
- S. B. Ma, Y. H. Lee, K. Y. Ahn, C. M. Kim, K. H. Oh and K. B. Kim, J. Electrochem. Soc., 2006, 153, C27 CrossRef CAS.
- W. F. Zhao, Y. S. Tang, J. Xi and J. Kong, Appl. Surf. Sci., 2015, 326, 276 CrossRef CAS.
- J. Kong, M. J. Wang, J. H. Zou and L. N. An, ACS Appl. Mater. Interfaces, 2015, 7, 6733 CAS.
- C. M. Julien, M. Massot and C. Poinsignon, Spectrochim. Acta, Part A, 2004, 60, 689 CrossRef CAS.
- Y. Wang, H. T. Guan, C. J. Dong, X. C. Xiao, S. F. Du and Y. D. Wang, Ceram. Int., 2016, 1, 936 CrossRef.
- H. Xia, M. O. Lai and L. Lu, J. Mater. Chem., 2010, 20, 6896 RSC.
- H. Xia, Y. Wang, J. Y. Lin and L. Lu, Nanoscale Res. Lett., 2012, 7, 33 CrossRef PubMed.
- X. B. Jin, W. Z. Zhou, S. W. Zhang and G. Z. Chen, Small, 2007, 3, 1513 CrossRef CAS PubMed.
- J. L. Chen, Q. H. Chen and Q. Ma, J. Colloid Interface Sci., 2012, 370, 32 CrossRef CAS PubMed.
- X. L. Zhang, T. Wang, C. L. Jiang, F. Zhang, W. Y. Li and Y. B. Tang, Electrochim. Acta, 2016, 187, 465 CrossRef CAS.
- M. Zhang, Q. Xu, L. Sang, F. Ding and X. J. Liu, Chin. Sci. Bull., 2014, 59, 2973 CrossRef CAS.
- B. Wen, X. X. Wang, W. Q. Cao, H. L. Shi, M. M. Lu, G. Wang, H. B. Jin, W. Z. Wang, J. Yuan and M. S. Cao, Nanoscale, 2014, 6, 5754 RSC.
- S. H. Liu, J. M. Liu, X. L. Dong and Y. P. Duan, Electromagnetic shielding and absorbing materials, Chemical Industry Press, Beijing, 2014 Search PubMed.
- H. L. Lv, G. B. Ji, X. H. Liang, H. Q. Zhang and Y. W. Du, J. Mater. Chem. C, 2015, 3, 5056 RSC.
- L. Wang, Y. Huang, C. Li, J. J. Chen and X. Sun, Phys. Chem. Chem. Phys., 2015, 17, 5878 RSC.
- L. X. Yi, G. X. Hu and H. Li, Composites, Part A, 2015, 76, 233 CrossRef CAS.
- G. Blasse, J. Phys. Chem. Solids, 1965, 26, 1969 CrossRef CAS.
- K. S. Cole and R. H. Cole, J. Chem. Phys., 1941, 9, 341 CrossRef CAS.
- B. Wen, M. S. Cao, Z. L. Hou, W. L. Song, L. Zhang, M. M. Lu, H. B. Jin, X. Y. Fang, W. Z. Wang and J. Yuan, Carbon, 2013, 65, 124 CrossRef CAS.
- M. E. Lines and A. M. Glass, Principles and Applications of Ferroelectrics and Related Materials, Clarendon Press, Oxford, 2001 Search PubMed.
- M. H. Al-Saleh and U. Sundararaj, Carbon, 2009, 47, 1738 CrossRef CAS.
- J. H. Zhu, S. Y. Wei, N. Haldolaarachchige, D. P. Young and Z. H. Guo, J. Phys. Chem. C, 2011, 115, 15304 CAS.
| This journal is © The Royal Society of Chemistry 2016 |