Mixed calcium and zinc salts of N-(3-amino-benzoic acid)terpene-maleamic acid: preparation and its application as novel thermal stabilizer for poly(vinyl chloride)
Abstract
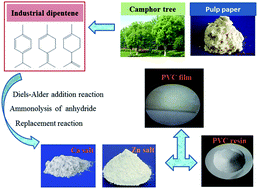
Dipentene-maleic anhydride (DPMA) was prepared by the Diels–Alder addition of dipentene with maleic anhydride. DPMA was converted via ammonolysis with para-aminobenzoic acid (PABA) to form N-(3-amino-benzoic acid)terpene-maleamic acid (ABDPMA), which was then converted to zinc soap (ABDPMA-Zn) or calcium soap (ABDPMA-Ca). Their chemical structures were confirmed by FT-IR and ICP-AES. Thermal stabilizing effects of ABDPMA-Ca/ABDPMA-Zn were compared with ABTMA-Ca/ABTMA-Zn (ABTMA: N-(3-amino-benzoic acid)tung-maleamic acid), C36DA-Ca/C36DA-Zn (C36DA: dimer fatty acid), ZnMA/ZnO (MA: maleic acid), EFC/ZnSt2/ESBO (EFC: calcium salt of epoxidised fatty acid; ESBO: epoxidised soybean oil, and CaSt2/ZnSt2 (St: stearic acid). Thermal stabilities of poly(vinyl chloride) (PVC) compounds were determined using Congo Red test, discoloration test, ultraviolet-visible spectroscopy analysis and thermogravimetric analysis. Dynamic mechanical and tensile properties of the PVC compounds were also studied. Besides better plasticization performance, ABDPMA-Ca/ABDPMA-Zn improved the long thermal stability of PVC compared with ABTMA-Ca/ABTMA-Zn, C36DA-Ca/C36DA-Zn, and CaSt2/ZnSt2.

 Please wait while we load your content...
Please wait while we load your content...