UV-assisted reduction of in situ electrospun antibacterial chitosan-based nanofibres for removal of bacteria from water
Lebea N. Nthunyaac,
Monaheng L. Masheaneac,
Soraya P. Malingaa,
Tobias G. Barnardb,
Edward N. Nxumaloc,
Bhekie B. Mambac and
Sabelo D. Mhlanga *c
*c
aDepartment of Applied Chemistry and the DST/Mintek Nanotechnology Innovation Centre-Water Research Node, University of Johannesburg, P. O. Box 17011, Doornfontein, 2028, Johannesburg, South Africa
bWater and Health Research Centre, University of Johannesburg, P. O. Box 17011, Doornfontein, 2028, Johannesburg, South Africa
cNanotechnology and Water Sustainability Research Unit, College of Science, Engineering and Technology, University of South Africa, Johannesburg, Florida 1709, South Africa. E-mail: mhlansd@unisa.ac.za; Tel: +27 11 471 2104
First published on 4th October 2016
Abstract
A greener synthesis of low-swelling uniformly-sized chitosan (CTS)-based nanofibres decorated with silver (Ag) and silver/iron (Ag/Fe) nanoparticles is reported. The synthesis was achieved by electrospinning a solution of CTS blended with varying amounts of polyacrylamide (PAA), polyethylene glycol (PEG) and Ag+ or Ag+/Fe3+ ions. These nanofibres were subjected to UV irradiation under ionised water vapour at low temperature (70 °C). The effect of UV irradiation time on the reduction of the NPs was confirmed using UV-Vis spectroscopy. The microstructure and chemical composition of the Ag and Ag/Fe modified nanofibres was studied using transmission electron microscopy (TEM), energy dispersive X-ray spectroscopy (EDS), X-ray diffraction (XRD) and ultraviolet-visible spectroscopy (UV-Vis). TEM revealed that the average diameter of the CTS-based nanofibres, AgNPs, and Ag/Fe NPs supported on the CTS-based nanofibres were 471 ± 89 nm, 18 ± 2.5 and 32 ± 8.7 nm respectively. XRD and EDS analysis confirmed the presence of Ag and Fe in the nanofibers. The biocidal effect of the Ag and Ag/Fe NPs supported on the CTS-based nanofibres was investigated using Gram positive (Bacillus cereus, Enterococcus faecalis) and Gram negative (Escherichia coli, Klebsiella pneumoniae, Klebsiella oxytoca, Pseudomonas aeruginosa, Proteus mirabilis, Shigella boydii, Shigella sonnei, Enterobacter cloacae) bacterial strains. The nanofibres exhibited a strong biocidal effect on the bacteria suggesting that they can be used as efficient antimicrobial materials in water systems that are contaminated by bacteria.
Introduction
The quality of drinking water in most rural communities in developing countries remains very poor. A classic example is a recent study conducted by Mpenyana-Monyatsi and co-workers that has shown the presence of high levels of bacterial contaminants in different drinking water sources (mainly bore-wells) in the Mpumalanga province, South Africa.1 Bacterial contamination of drinking water sources is known to be the major source of water-borne diseases and other health related problems which include diarrhea, nausea and gastroenteritis.2 Approximately one billion of the world's population has no access to pre-treated water and about 43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 deaths in South Africa that occur annually, are related to consumption of untreated water which contains pathogens.3,4
000 deaths in South Africa that occur annually, are related to consumption of untreated water which contains pathogens.3,4
In an attempt to remove bacterial contaminants from water, different treatment processes have been employed. These include application of antibiotics, chlorination, and membrane technologies.5–7 However, these processes have drawbacks associated with them. For example bacteria has been shown to become tolerant to antibiotics and hence resist their mode of antibacterial activity.7 Chlorination results in chlorinated organic by-products which are known to be toxic to humans.6 Membrane technologies are energy-driven and susceptible to biofouling, hence require high operation and maintenance costs.5
Recent studies have reported on the application of nanoparticles (NPs) supported on polymer materials in water treatment.8 However, the efficient removal of waterborne pathogens from drinking water following environmentally benign technologies remains a challenge. Methods that avoid problem shifting or the use of harmful or toxic chemicals are highly desired, especially in the synthesis of NPs and their application in water purification. NPs possess unique physical or chemical properties different from those of their counterpart macroscopic materials.9 The level of toxicity and median lethal dose (LD50) of NPs towards bacteria depends on their sizes and shapes. NPs with smaller sizes are able to penetrate through the membranes of different cells of microscopic organisms including bacteria, thereby inhibiting their growth and killing them.10 Extensive research has been done on Ag NPs due to the high antibacterial properties inherent to these NPs.11
Polymeric materials with different geometries such as nanofibres have been used as support materials for NPs used for different applications such as wound dressing and disinfection.12 These materials act as an anchor and provide dispersion of the NPs with controlled sizes, shapes and distribution.13 Nanofibrous materials also provide a porous structure and high surface area that can be used to disperse the NPs. Most polymers are chosen based on their biodegradability, biocompatibility, non-toxicity, ease of availability and their low cost for the synthesis of these NP supporting materials.14–17 An example of those polymers is chitosan (CTS). Electrospun CTS embedded with Ag NPs has been used in different antibacterial applications including water treatment, with high efficiencies.18
Electrospinning is a common technique that has been used for the synthesis of nanofibres with desired nanoscale sizes. In order to prepare nanofibres containing antibacterial NPs, the NPs are deposited on the nanofibres either ex situ or in situ. In the ex situ procedure, the nanofibres are electrospun with the electrospinning polymer solution containing the already prepared NPs (i.e. prepared in a separate experiment). In the in situ procedure, the polymer solution is mixed with the metal ion precursor and electrospun. Thereafter, the metal ions nanofibres are reduced to their respective active NPs. Different methods which include photo-reduction,19 photochemical reduction,20 argon plasma18 the use of solvents11,21 have been used to reduce the metal ions dispersed on the nanofibres. To the best of our knowledge, the synthesis of Ag and Ag/Fe supported on CTS-based nanofibres (a natural biodegradable polymer) using a comparatively greener UV-assisted ionized water vapour reduction process has not been reported. The polymer backbone of the nanofibres was obtained by carefully selecting a recipe of compatible polymers that were added to the CTS solution (in optimized amounts) to make it electrospinnable.
Experimental
Materials
CTS (MW = 150![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, 75 degree of deacetylation), PAA (MW = 700
000 g mol−1, 75 degree of deacetylation), PAA (MW = 700![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1), PEG (MW = 6600 g mol−1), iron chloride hexahydrate (FeCl3·6H2O), silver nitrate (AgNO3), Mueller Hinton broth and agar, p-iodonitrotetrazolium chloride and glacial acetic acid (CH3COOH) were purchased from Sigma Aldrich, Germany. B. cereus (ATCC no. = 10876), E. faecalis (ATCC no. = 7080), E. coli (ATCC no. = 11775 and 25922), K. pneumoniae (ATCC no. = 13882 and 31488), K. oxytoca (ATCC no. = 8724), P. aeruginosa (ATCC no. = 27853), P. mirabilis (ATCC no. = 12453), S. boydii (ATCC no. = 9207), S. sonnei (ATCC no. = 25931) and E. cloacae (ATCC no. = 13047) strains were purchased from American Type Culture Collection (ATCC). De-ionised water was prepared in our laboratory using direct-Q® (Millipore) system supplied by Merck Millipore. All reagents used in this study were used as received.
000 g mol−1), PEG (MW = 6600 g mol−1), iron chloride hexahydrate (FeCl3·6H2O), silver nitrate (AgNO3), Mueller Hinton broth and agar, p-iodonitrotetrazolium chloride and glacial acetic acid (CH3COOH) were purchased from Sigma Aldrich, Germany. B. cereus (ATCC no. = 10876), E. faecalis (ATCC no. = 7080), E. coli (ATCC no. = 11775 and 25922), K. pneumoniae (ATCC no. = 13882 and 31488), K. oxytoca (ATCC no. = 8724), P. aeruginosa (ATCC no. = 27853), P. mirabilis (ATCC no. = 12453), S. boydii (ATCC no. = 9207), S. sonnei (ATCC no. = 25931) and E. cloacae (ATCC no. = 13047) strains were purchased from American Type Culture Collection (ATCC). De-ionised water was prepared in our laboratory using direct-Q® (Millipore) system supplied by Merck Millipore. All reagents used in this study were used as received.
Electrospinning of CS-based nanofibers
To synthesize Ag and Ag/Fe NPs supported on CTS-based nanofibres, the following procedure was used: AgNO3 was dissolved in a solution of CTS/PAA/PEG which was prepared using 50% CH3COOH in a beaker for the deposition of Ag NPs on the CTS-based nanofibres. In another beaker, AgNO3 and FeCl3 were dissolved in a similar solution of CTS/PAA/PEG for the deposition of Ag/Fe NPs on the CTS-based nanofibres. The amount of metal ion salts was calculated to give 4% of Ag NPs and 4% of Ag/Fe NPs (i.e., 2% Ag NPs and 2% Fe NPs) relative to the CTS/PAA/PEG polymer powders. A 3% CTS/PAA polymer concentration (ratio of 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23) was used as the electrospinning solution. A 5 wt% of PEG relative to CTS was added to reduce the surface tension of CTS. The prepared solutions were transferred to a 10 mL plastic syringe fitted with a needle of 0.8 mm internal diameter. The syringe was placed on a NE-4000 double syringe pump. A high voltage generator (EV11M, TEL Atomic) was used to induce an electric field between the collecting plate and the tip of the needle. The positive terminal of the DC generator was connected to the tip of the syringe needle and the negative terminal connected to the aluminum foil (collecting plate). The earth terminals were connected to the syringe pump. The nanofibres were synthesized at the following optimized electrospinning conditions: syringe injection flow rate of 0.7 mL h−1, a distance of 21 cm between the aluminum foil and the tip of the needle, and a voltage of 24 kV at room temperature.
23) was used as the electrospinning solution. A 5 wt% of PEG relative to CTS was added to reduce the surface tension of CTS. The prepared solutions were transferred to a 10 mL plastic syringe fitted with a needle of 0.8 mm internal diameter. The syringe was placed on a NE-4000 double syringe pump. A high voltage generator (EV11M, TEL Atomic) was used to induce an electric field between the collecting plate and the tip of the needle. The positive terminal of the DC generator was connected to the tip of the syringe needle and the negative terminal connected to the aluminum foil (collecting plate). The earth terminals were connected to the syringe pump. The nanofibres were synthesized at the following optimized electrospinning conditions: syringe injection flow rate of 0.7 mL h−1, a distance of 21 cm between the aluminum foil and the tip of the needle, and a voltage of 24 kV at room temperature.
UV-assisted reduction
The electrospun CTS-based nanofibres containing Ag+ and Ag+/Fe3+ ions were irradiated with UV light at λmax = 249 nm using a UV lamp (model no. 3040, Photochemical Reactors Ltd) fitted to a horizontal furnace (Fig. 1). The reduction of the metal ions into NPs was achieved at 70 °C. The nanofibres were irradiated at different times from 30–210 min in order to determine the optimum time for the reduction of the metal ions into NPs. | ||
| Fig. 1 An experimental set-up for the UV-assisted reduction of Ag+ and Ag+/Fe3+ NPs supported on CTS-based nanofibres. | ||
Characterization of nanofibres
The antibacterial CTS-based nanofibres were characterized using UV-Vis spectroscopy (Perkin Elmer Shimadzu 2450 spectrophotometer) for the determination of the effect of UV irradiation time on the intensity of NPs formed on the surface of the nanofibres. Transmission electron microscopy (TEM, JEOL JEM-2010) was used to study the size and dispersion of the NPs on the surface of nanofibres. TEM coupled with energy dispersive X-ray spectroscopy (TEM-EDS) was used to confirm the presence of elemental Ag and Fe on the nanofibres. X-ray diffraction (XRD) patterns of the nanofibres were acquired with a Rigaku Ultimate IV diffractometer equipped with a Cu Kα radiation source, a scintillation counter detector and a K-β filter. The XRD patterns were analyzed qualitatively using “PDXL” software, provided with JCPDS-PDF2 database and the card number was ICDD.Antibacterial activity of the nanofibres
The antimicrobial activity of the Ag and Ag/Fe NPs supported on the CTS-based nanofibres was tested against B. cereus, E. faecalis, E. coli, K. pneumoniae, K. oxytoca, P. aeruginosa, P. mirabilis, S. boydii, S. sonnei and E. cloacae. The bacterial strains were grown and maintained on a Mueller-Hinton agar during experiments. The plates were incubated at 37 °C for 24 h. The strains were grown in a liquid culture by inoculating Mueller-Hinton broth with the bacterial colony of interest. All strains were grown at 37 °C with constant mild shaking until an optical density of 0.6 at 600 nm (OD600) was reached. The basic disc diffusion test was used to test the antibacterial activity of the CTS-based nanofibre mats embedded with Ag and Ag/Fe NPs. The method was used as described by the Clinical and Laboratory Standards Institute.22 Briefly; bacterial lawns were created from bacterial suspension with a turbidity equivalent to 0.5 MacFarland standard on Mueller-Hinton agar plate. The nanofibres (cut to similar shapes of similar sizes) were added and incubated at 37 °C for 16 hours and the bacterial inhibition noted.The minimum inhibitory concentration of Ag and Ag/Fe NPs towards bacteria required in the CTS-based nanofibres was determined using a 96 well plate method reported by Eloff.23 Suspensions of the nanofibres were prepared by cutting, grinding and suspending nanofibres in water. The initial concentrations of the CTS-based nanofibre suspensions (12.5 mg mL−1) were serial diluted at different dilution factors, i.e. 2×, 4×, 8×, 16×, 32× and 64×. The bacterial cultures (50 μL) with similar OD600 were added to each well containing the test samples. The micro titre plates were closed and incubated at 37 °C for 24 h. Bacterial viability was illustrated with the addition p-iodonitrotetrazolium chloride (50 μL) to each well and incubated for 40 min using a method by Eloff.23 The wells that turned purple indicated the presence of viable bacteria and thus no growth inhibition.
Results and discussion
UV-reduction of Ag+ and Fe3+ ions on CTS-based nanofibres
The Ag or Ag/Fe NPs were formed on electrospun CTS-based nanofibres by subjecting the nanofibres bearing the metals ions to UV irradiation in the presence of ionized water vapour. When the nanofibres were irradiated with UV light at 249 nm under the constant flow of ionized water vapour at 70 °C, the nanofibres containing Ag+ ions changed colour from a white to a yellowish brown colour. The nanofibres containing Ag+/Fe3+ ions changed colour from a white to light yellowish colour. These colour changes gave an indication that the Ag+ and Fe3+ ions on the CTS-based nanofibres were reduced to Ag0 and Fe0 respectively. No colour change was observed when nanofibres containing no metal ions (blank run) were subjected to UV-irradiation. During UV irradiation, water vapour ionizes to form the H2O+ and releases the electrons which are accepted by the positively charged Ag+ and Fe3+ ions. When metal ions are exposed to these electrons, they are reduced to Ag0 and Fe0 as shown by eqn (1)–(3).24 The reduction of the metals ions was however confirmed by the use of relevant techniques discussed in the forthcoming sections.| H2Ovapour + hv → H2O+ + e− | (1) |
| Ag+ + e− → Ag0 | (2) |
| Fe3+ + e− → Fe0 | (3) |
XRD analysis
The presence of Ag and Fe NPs on the CTS-based nanofibres was confirmed by XRD analysis and the results are presented in Fig. 2. The JCPDS card values for the following planes (003) and (101) at 2-theta = 25.89° and 43.33° are characteristics diffractions of carbon from the CTS backbone (Fig. 2a). The JCPDS card values for the following planes (111), (200), (220), (311), (222) at 2-theta = 38.26°, 44.47°, 64.71° and 77.75°, 81.92° are characteristic diffractions of Ag NPs (Fig. 2b). The JCPDS card values for the following planes (110), (200), (211) at 2-theta = 44.49°, 64.73° and 81.91° are characteristics diffractions of Fe NPs (Fig. 2c). The diffraction patterns of Ag NPs on Fig. 2c were observed at the similar planes with no shift of diffraction angles. This suggested that there was no change of chemical surrounding on the Ag NPs in the presence of Fe NPs.The spectral peaks of Ag and Fe NPs were observed to be superimposed on one another (Fig. 2i(c)). The NPs were however identified by their mirror planes. Since, distinct peaks of both NPs could not be identified, the peaks on spectra (c) were deconvoluted using origin software and distinct peaks of Ag and Fe NPs were observed as shown in Fig. 2ii.
UV-Vis of reduced Ag and Ag/Fe NPs on CTS-based nanofibres
The UV-Vis absorption spectra and plots of maximum absorbance vs. time of Ag and Ag/Fe NPs on the CTS-based nanofibres are shown in Fig. 3 and 4. The maximum absorption peaks of Ag NPs were observed at the wavelength range of 382–403 nm which correspond to the surface plasmon band of Ag NPs (Fig. 3i).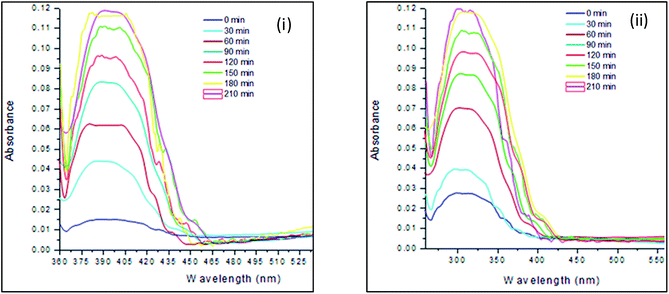 | ||
| Fig. 3 UV-Vis absorption spectra of Ag and Ag/Fe NPs on CTS-based nanofibres reduced at different irradiation times: (i) Ag NPs and (ii) Ag/Fe NPs. | ||
The surface plasmon resonance increased as the holding time of UV irradiation was increased (Fig. 4). The increase in the intensity of the surface plasmon resonance indicated that the number of Ag NPs produced increased with an increase in irradiation time. However, the reduction capacity became slower at longer reduction times (between 180 min and 210 min) (Fig. 3 and 4). This was due to depletion of reducible metal ions with time. Initially, the absorption peaks were broad from the beginning of UV irradiation time. This suggested that the Ag NP size distribution was changed as the time of UV irradiation was increased.
The maximum absorption peaks of Ag/Fe NPs were observed at the maximum wavelength of 301 nm at lower reduction times (Fig. 3ii). The maximum absorption shifted from lower wavelengths to higher wavelengths (301–320 nm) as the UV irradiation time was increased from 30 min to 210 min. This wavelength shift resulted to the narrow and symmetric absorption peaks relative to UV-assisted reduction of Ag+ ions which implied the non-changing distribution of the Ag/Fe NPs on the CTS-based nanofibres.
The surface plasmon resonance absorption of Ag/Fe NPs also increased with an increase in irradiation time (Fig. 3i and 6). The increase in surface plasmon confirmed the increase in density of Ag/Fe NPs produced. At shorter irradiation time, a high amount of metal ions were not reduced. At longer UV irradiation time, most of the Ag+ and Fe3+ ions were reduced to Ag and Fe NPs, hence, less Ag+ and Fe3+ ions were left for reduction to NPs.
TEM and EDS analysis
The presence of elemental Ag and Fe in the CTS-based nanofibres was also confirmed by the EDS analysis (Ag: 2.6–3.0 kV and Fe: 6.5 kV) (Fig. 5). The intense signals observed for C, O, N, Cr and Cu were as a result of C, O and N atoms present in the CTS-based nanofibres whilst Cr and Cu are components of TEM instrument where Cr forms part of the Cr alloys in the instrument and Cu grid was used as a sample holder. The intense peaks of Cr and Cu mask the peaks of Ag and Fe NPs since Ag and Fe ions were added in small quantities during the synthesis of the antibacterial CTS-based nanofibres.The Ag and Ag/Fe NPs on the CTS-based nanofibres formed as the UV irradiation time was varied are shown by the TEM images in Fig. 6 and 7. The number of microscopically visible Ag and Ag/Fe NPs increased with an increase in UV irradiation time. These results correspond to UV-Vis analysis results shown in Fig. 3 and 4. The Fe NPs were observed to improve the dispersion of the Ag NPs on the surface of the nanofibres (Fig. 7).
 | ||
| Fig. 6 TEM images of Ag NPs (black spots) on CTS-based nanofibres reduced at different UV irradiation times. (a) 30 min, (b) 60 min, (c) 90 min, (d) 120 min and (e) 210 min. | ||
 | ||
| Fig. 7 TEM images of Ag/Fe NPs (black spots) on CTS-based nanofibres reduced at different UV irradiation times. (a) 30 min, (b) 60 min, (c) 90 min, (d) 120 min and (e) 210 min. | ||
Fig. 8 shows the effect of UV irradiation time on the average size of NPs. The average sizes of Ag NPs were found to be 39 ± 7 nm, 30 ± 9 nm, 12 ± 5 nm, 45 ± 4 nm and 23 ± 6 nm at 30, 60, 90, 120 and 210 min respectively (Fig. 8). The average sizes of Ag/Fe NPs were found to be 36 ± 5 nm, 53 ± 6 nm, 36 ± 8 nm, 56 ± 5 nm and 48 ± 9 nm at 30, 60, 90, 120, 210 min respectively (Fig. 8). There was no observable trend on the size of NPs when the UV irradiation time was increased. However, the magnitude of errors associated to the average sizes of NPs showed variation of sizes of Ag NPs and Ag/Fe NPs. The large deviation of sizes of Ag NPs showed uneven size distribution of Ag NPs and the smaller the deviation of sizes of Ag/Fe showed a better even size distribution of Ag/Fe NPs. These results further confirmed the distribution of NPs on the hosting materials as explained by the UV-Vis results. The NP sizes were calculated from scale bars of the TEM images (at least 50 for each) and the ImageJ software.
 | ||
| Fig. 8 The effect of UV irradiation time on the size of Ag (left bars) and Ag/Fe NPs (right bars) supported on the on CTS-based nanofibres. | ||
Antimicrobial tests using CTS-based nanofibres
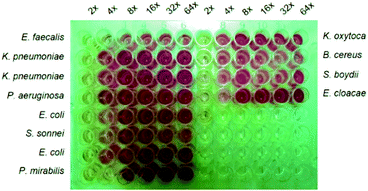
No direct measurable zone of bacterial inhibition could be associated with the NPs containing mats since the nanofibres bound agents could not diffuse into the agar as per the normal antibacterial test. Clear zones were more likely due to the sufficient contact of the NPs with the bacterial strains. Alternative factors such as uneven distribution of the Ag NPs on the surface of the nanofibres and too low concentrations of the NPs on the mats could further have contributed to the failure of the observation of the zone of bacterial inhibition growth. The fact that there was no diffusion of the NPs into the media to produce a measurable inhibition zone shows that the antibacterial agents did not leach of the nanofibres.
 | ||
| Fig. 10 Minimum Inhibition Concentration (MIC) of antibacterial CS based nanofibres decorated with Ag NPs on 12 strains of bacteria. | ||
 | ||
| Fig. 11 Minimum Inhibition Concentration (MIC) of antibacterial CS based nanofibres decorated with Ag/Fe NPs on 12 strains of bacteria. | ||
The differences in the required concentration of antibacterial agents needed to produce a noticeable antibacterial activity on different strains of bacteria could be due to response and resistance of these strains linked to bacterial structure, bacterial cell wall, cell membrane, and thickness of their peptidoglycan layer, which respond differently to different bacteria hence help them resist antibacterial agents differently.
The MIC was analysed for a two way student t-test at a 0.05 significant level using IBM SPSS statistics 24 software. The mean values of the minimum inhibition concentration of both Ag and Ag/Fe NPs supported on CTS-based nanofibres were not significantly different as evidenced by the p values ≤ 0.05 (Table 1). As such, the MIC can be comfortably reported as CTS-based nanofibres (6.71 mg mL−1) that contain Ag (0.27 mg mL−1) and Ag/Fe (0.27 mg mL−1) combined.
| Biocides | Test against | Sig. (2-tailed) | Mean difference | Std deviation | Std error mean | 95% confidence interval of the difference | |
|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||
| Ag supported on CTS-based nanofibres | E. faecalis | 0.000 | 2.34100 | 0.34314 | 0.10851 | 2.0955 | 2.5865 |
| K. pneumoniae | 0.000 | 6.25900 | 0.02923 | 0.00924 | 6.2381 | 6.2799 | |
| K. pneumoniae | 0.000 | 6.40500 | 0.37319 | 0.11801 | 6.1380 | 6.6720 | |
| P. aeruginosa | 0.000 | 4.50500 | 0.11346 | 0.03588 | 4.4238 | 4.5862 | |
| E. coli | 0.000 | 4.76600 | 0.12747 | 0.04031 | 4.6748 | 4.8572 | |
| P. mirabilis | 0.000 | 3.57700 | 0.07804 | 0.02468 | 3.5212 | 3.6328 | |
| K. oxytoca | 0.000 | 6.50800 | 0.06052 | 0.01914 | 6.4647 | 6.5513 | |
| B. cereus | 0.000 | 6.59500 | 0.04649 | 0.01470 | 6.5617 | 6.6283 | |
| S. boydii | 0.000 | 6.65900 | 0.36828 | 0.11646 | 6.3955 | 6.9225 | |
| E. cloacae | 0.000 | 6.61500 | 0.08330 | 0.02634 | 6.5554 | 6.6746 | |
| S. sonnei | 0.000 | 6.63500 | 0.09336 | 0.02952 | 6.5682 | 6.7018 | |
| E. coli | 0.000 | 4.29800 | 0.03910 | 0.01236 | 4.2700 | 4.3260 | |
| Ag/Fe supported on CTS-based nanofibres | E. faecalis | 0.000 | 3.82800 | 0.12497 | 0.03952 | 3.7386 | 3.9174 |
| K. pneumoniae | 0.000 | 6.18400 | 0.05542 | 0.01752 | 6.1444 | 6.2236 | |
| K. pneumoniae | 0.000 | 6.46000 | 0.07272 | 0.02300 | 6.4080 | 6.5120 | |
| P. aeruginosa | 0.000 | 6.70600 | 0.08356 | 0.02642 | 6.6462 | 6.7658 | |
| E. coli | 0.000 | 4.47000 | 0.06289 | 0.01989 | 4.4250 | 4.5150 | |
| P. mirabilis | 0.000 | 3.64270 | 0.04046 | 0.01280 | 3.6138 | 3.6716 | |
| K. oxytoca | 0.000 | 6.43000 | 0.10477 | 0.03313 | 6.3550 | 6.5050 | |
| B. cereus | 0.000 | 6.67800 | 0.03645 | 0.01153 | 6.6519 | 6.7041 | |
| S. boydii | 0.000 | 6.82500 | 0.05297 | 0.01675 | 6.7871 | 6.8629 | |
| E. cloacae | 0.000 | 4.27100 | 0.04358 | 0.01378 | 4.2398 | 4.3022 | |
| S. sonnei | 0.000 | 4.59500 | 0.09652 | 0.03052 | 4.5260 | 4.6640 | |
| E. coli | 0.000 | 6.63600 | 0.06204 | 0.01962 | 6.5916 | 6.6804 | |
Conclusions
CTS-based nanofibres embedded with Ag and Ag/Fe NPs were successfully synthesized using UV-ionized water vapour assisted photochemical reduction at low temperature. This process is safe since the reagents used are environmentally friendly. The intensity of the NPs dispersed on the surface of the nanofibres was found to increase with an increase in irradiation time. The nanofibres exhibited strong biocidal effect on all bacterial strains tested. The addition of Fe NPs on the nanofibres containing Ag NPs showed the similar antibacterial activity as the nanofibres containing Ag NPs only. Since Fe NPs were added in the equivalent quantities of Ag to make up the total of 4% of NPs supported on the nanofibres, it meant that the amount of Ag was reduced compared to the nanofibres containing Ag without Fe NPs. The addition of Fe NPs can subsequently reduce the cost of antibacterial CTS-based nanofibres since Fe salts are cheaper than Ag salts. The Ag and Ag/Fe NPs supported on CTS-based nanofibres exhibited the bactericidal effects on all strains tested, although some strains showed more resistance. This showed that the bacterial strains exhibited different tolerances towards biocidal activity of the antibacterial agents (Ag and Ag/Fe NPs). However, the NPs exhibited strong antibacterial activity on all bacterial strains suggesting that the CTS-based nanofibres can be used for antibacterial purification of water.Acknowledgements
This work was done partly at the University of Johannesburg and some characterizations were performed at the University of South Africa and the University of Witwatersrand. It was funded by National Research Foundation Nanotechnology Flagship grant number 97823 and the DST/Mintek Nanotechnology Innovation Centre – Water Research Node. Support from these organizations is highly appreciated.References
- L. Mpenyana-Monyatsi, M. S. Onyango and M. N. B. Momba, Pol. J. Environ. Stud., 2012, 21, 1349–1358 CAS.
- B. Ye, L. Yang, Y. Li, W. Wang and H. Li, Int. J. Environ. Res. Public Health, 2013, 10, 879–891 CrossRef CAS PubMed.
- L. C. Simoes and M. Simoes, RSC Adv., 2013, 3, 2520–2533 RSC.
- G. C. Pegram, N. Rollins and Q. Espey, Water SA, 1998, 24, 11–20 Search PubMed.
- A. G. Fane, R. Wang and M. X. Hu, Angew. Chem., Int. Ed., 2015, 54, 3368–3386 CrossRef CAS PubMed.
- M. Deborde and U. von Gunten, Water Res., 2008, 42, 13–51 CrossRef CAS PubMed.
- J. L. Armstrong, D. S. Shigeno, J. J. Calomiris and R. J. Seidler, Appl. Environ. Microbiol., 1981, 42, 277–283 CAS.
- K. Simeonidis, S. Mourdikoudis, E. Kaprara, M. Mitrakas and L. Polavarapu, Environ. Sci.: Water Res. Technol., 2015, 2, 43–70 Search PubMed.
- T. Pradeep and Anshup, Thin Solid Films, 2009, 517, 6441–6478 CrossRef CAS.
- K. Schwirn, L. Tietjen and I. Beer, Environ. Sci. Eur., 2014, 26, 4 CrossRef.
- M. Rai, A. Yadav and A. Gade, Biotechnol. Adv., 2009, 27, 76–83 CrossRef CAS PubMed.
- A. M. Abdelgawad, S. M. Hudson and O. J. Rojas, Carbohydr. Polym., 2014, 100, 166–178 CrossRef CAS PubMed.
- D. Li and R. B. Kaner, J. Am. Chem. Soc., 2006, 128, 968–975 CrossRef CAS PubMed.
- Z. Anwar, M. Gulfraz and M. Irshad, J. Radiat. Res. Appl. Sci., 2014, 7, 163–173 CrossRef CAS.
- H. Y. Zhou, D. J. Zhou, W. F. Zhang, L. J. Jiang and J. B. Li, Front. Mater. Sci., 2011, 5, 367–378 CrossRef.
- J. Puls, S. A. Wilson and D. Holter, J. Polym. Environ., 2010, 19, 152–165 CrossRef.
- P. K. Sahoo, T. Biswal and R. Samal, J. Nanotechnol., 2011, 2011, 1–11 CrossRef.
- D. Annur, Z.-K. Wang, J.-D. Liao and C. Kuo, Biomacromolecules, 2015, 16, 3248–3255 CrossRef CAS PubMed.
- W. K. Son, J. H. Youk and W. H. Park, Carbohydr. Polym., 2006, 65, 430–434 CrossRef CAS.
- H.-H. Park, X. Zhang, Y.-J. Choi, H.-H. Park and R. H. Hill, J. Nanomater., 2011, 2011, 1–7 Search PubMed.
- M. Ghaffari-Moghaddam and H. Eslahi, Arabian J. Chem., 2014, 7, 846–855 CrossRef CAS.
- Clinical Laboratory Standard Institude, CLSI document M02-A11, CLSI, 2012, vol. 32, pp. 1–76 Search PubMed.
- J. N. Ellof, Planta Med., 1998, 64, 711–713 CrossRef PubMed.
- S. L. Guberman, Dissociative Recombination of Molecular Ions with Electrons, Springer Science + Business Media LLC, New York, 2003 Search PubMed.
- J. M. Andrews, J. Antimicrob. Chemother., 2001, 48, 5–16 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |




