Efficient capture of low partial pressure H2S by tetraethyl ammonium amino acid ionic liquids with absorption-promoted solvents
Bing Wanga,
Kai Zhanga,
Shuhang Ren*a,
Yucui Houb and
Weize Wu*a
aState Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing, 100029, China. E-mail: rensh@mail.buct.edu.cn; wzwu@mail.buct.edu.cn
bDepartment of Chemistry, Taiyuan Normal University, Taiyuan, 030031, China
First published on 17th October 2016
Abstract
To capture H2S from gas mixtures, ionic liquids (ILs), especially functional ILs, are regarded as novel absorbents and can absorb a large amount of H2S at high pressures. However, the solubility of dilute H2S in functional ILs is still very low and the viscosities of H2S-absorbed ILs are very high resulting in long absorption equilibrium times. To enhance the solubility and reduce the equilibrium time, we designed mixtures of functional ILs, tetraethyl ammonium amino acids, and ethylene glycol (EG) to capture dilute H2S of 0.005 MPa. It has been found that these mixtures can absorb a large amount of H2S from mixed gas, and EG content, temperature, H2S mole fraction and type of ILs have significant influence on the absorption. At 40 °C, the solubility of H2S in the mixture can reach up to 2.36 mol H2S per kg mixture. EG in the ILs mixtures not only decreases the viscosity of ILs before and after the absorption, but also enhance the absorption capacity of H2S in ILs. Low temperatures, high H2S partial pressures and low pKb of –NH2 (or –NH) in amino acids formed the anion of ILs have positive effects on H2S absorption in the mixtures. The mechanism of H2S absorption by functional IL and EG mixtures has also been proposed.
Introduction
Hydrogen sulfide (H2S), which is not only toxic to human beings, but also harmful to many catalytic reactions in industry,1 mainly exists in syngas and natural gas. Many researchers focus on the capture of H2S, and many kinds of traditional solvents,2–5 which can be divided into physical and chemical absorbents, are used to absorb H2S from gas mixtures. Up to now, aqueous solution of alkanolamines is one of the most effective absorbents to capture of H2S from syngas and natural gas. The alkanolamines, including monoethanolamine (MEA), diethanolamine (DEA) and N-methyldiethanolamine (MDEA), have been adopted widely in commercial units. However, there exist some disadvantages for these absorbents. For example, the high vapor pressure of alkanolamines can cause their volatile loss, which may result in pollution to the absorbed gas.Ionic liquids (ILs), which have extremely low vapor pressure, tunable structure, high thermal and chemical stability, and excellent solvent power, are regarded as a kind of novel and environmentally benign materials for gas capture. Until now, many types of ILs have been synthesized and used for gas capture, especially for CO2 (ref. 6–12) and SO2,13–17 but only a few researches mention to the capture of H2S. IL 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim][PF6]) was firstly used for the capture of H2S, and the solubility of H2S in the IL was about 18.5 mol H2S per kg IL at a H2S partial pressure of 2 MPa and a temperature of 20 °C.18 After that, several kinds of imidazolium-based ILs,19–31 such as 1-butyl-3-methylimidazolium bis-(trifluoromethyl)sulfonylimide ([Bmim][Tf2N]), 1-hexyl-3-methylimidazolium hexafluorophosphate ([Hmim][PF6]), and 1-ethyl-3-methylimidazolium ethylsulfate ([Emim][EtSO4]), were used to absorb H2S. Moreover, some calculations with different models, such as a modified perturbed hard sphere chain equation of state, were carried out based on the solubility of H2S in these ILs.32–41 Quaternary ammonium-based ILs were also used for H2S capture and showed high absorption capacities at high partial pressures.42,43 Duan et al.42 synthesized a series of caprolactam tetrabutyl ammonium bromide ILs to capture H2S, and the solubility of H2S could reach 1.67 mol H2S per kg IL (5.40 wt%) at 30 °C and 0.1 MPa of H2S. Although all of these ILs can absorb H2S, only physical interaction was found between these ILs and H2S. As we know, the partial pressures of H2S in normal gas mixtures are very low, such as 0.005 MPa in a natural gas.44 When the partial pressure of H2S is very low, these ILs can hardly absorb H2S.18 Hence, Wu et al.45 synthesized a group of functional ILs, which contain free tertiary amine groups in anion to chemically interact with H2S, and used the functional ILs to absorb H2S. The absorption capacity of H2S in the ILs at 0.0044 MPa of H2S and 60 °C could reach up to about 1.5 mol per kg IL, which is much higher than that in previous normal ILs at the same conditions.
The above results indicate that functional groups are important for functional ILs to absorb H2S at low partial pressures due to chemical interaction. As we know, tertiary amine groups are less alkalescent than amino functional group –NH2 (or –NH), which may have stronger interaction between IL and H2S. Hence, it is possible that functional ILs with –NH2 (or –NH) group can enhance the absorption capacity of H2S with low partial pressures. In this work, inspired by Wu's work,45 we synthesized tetraethyl ammonium amino acid functional ILs and found that they could capture H2S with high absorption capacity of H2S. On the other hand, during the capture of H2S, the viscosity of functional ILs become very high, which is unfavorable for their applications. It was reported that ethylene glycol (EG) had a very low vapor pressure (0.017 kPa at 25 °C) and a low viscosity,46 which make EG an ideal solvent for the absorption. Hence, we added EG as a solvent into the functional ILs to form absorbent mixtures for capturing H2S. It was found that these IL and EG mixtures could capture large amounts of H2S at low partial pressures with a low viscosity of the IL and EG mixtures. Importantly, we found that EG could enhance the absorption capacity of H2S by the functional ILs, and there was a synergetic effect between the functional ILs and EG. The results indicated that at 0.005 MPa of H2S and 40 °C, the absorption capacity of H2S in the IL and EG mixture could reach up to 2.36 mol H2S per kg absorbent, which is much higher than that in previous solvents at the same conditions.
Experimental section
Materials
H2S (5%, N2 as make-up gas) and N2 (99.99%) were supplied from Beijing Haipu Gases Co., Ltd., Beijing, China. Tetraethyl ammonium hydroxide (aqueous solution of 25 wt%), L-alanine (99%), β-alanine (98.5%), L-proline (99%), glycine (99%), ethylene glycol (99%) and N-methyl-2-pyrrolidone (99.5%) were purchased from Shanghai Jingchun Chemicals Co., Ltd, Shanghai, China. All reagents and solvents were analytical reagents. All of the ILs, shown in Fig. 1, were synthesized following the procedure reported by Jiang et al.9 All of the ILs were dried using a sweeping method,47 until their water contents were less than 0.1% in weight.Apparatus and procedures
The absorption experiment consisted mainly of a cylinder containing H2S/N2 gas mixture, a test tube with an inner diameter of 12 mm and a length of 200 mm, a rotameter (Beijing Forth Automation Meter Factory, China), and a constant temperature water bath. The setup of the experiment is similar to our previous work used for SO2 capture.48The absorption experiment was carried out at an ambient pressure. In a typical experiment, H2S/N2 gas mixture with a desired H2S partial pressure was bubbled through a tested mixture in the test tube. The tested mixture contained about 1 g IL and certain weight of solvent. The vent gas was bubbled through a glass bottle containing NaOH aqueous solution for absorbing the remaining H2S. The flow rate (50 mL min−1) was monitored by the rotameter and calibrated by a soap film fluid meter. The test tube was most partly immersed into the water bath, the temperature of which was maintained within ±0.1 °C by using a temperature controller (Model A2, Beijing Changliu Co., Ltd., China). After a given time for absorption, the weight of the test tube was measured by an electrical balance (ME204E, Mettler-Toledo International Inc.) with an accuracy of 0.1 mg, and the content of absorbed H2S in the mixture was calculated by the mass deference before and after absorption. The reproducibility of the measurements was better than ±2.5%, and it was estimated that the data of H2S absorption capacity were accurate to ±5%.
The regeneration experiment was carried out at an ambient pressure. After reaching saturated absorption, the test tube, containing IL, EG and H2S mixture, was partly immersed into the oil bath with a temperature of 100 °C. Pure N2 gas was bubbled through the mixture for 4.5 h, and the vent gas was bubbled through a glass bottle containing NaOH aqueous solution for absorbing the desorbed H2S. The flow rate (100 mL min−1) was monitored by the rotameter and calibrated by a soap film fluid meter. After 4.5 h, the content of H2S in the sampled mixture was measured following the standard iodometry (GB11060.1-89, a standard method of natural gas determination of hydrogen sulfide content iodometric titration in China). Briefly, the sample was dissolved in an aqueous solution, and iodine solution and hydrochloric acid solution was added. Then the aqueous mixture was analyzed by titration of iodine aqueous solution, the concentration of which was determined by sodium thiosulfate, and a small amount of fresh starch solution was added as an indicator.
Results and discussion
Selection of solvents for the absorption of H2S
Before solvents was added into the ILs, the absorption of H2S by pure [N2222][L-Ala] was measured, and the result is shown in Fig. 2. The absorption rate is very fast in the first 10 min, and the solubility of H2S in the IL reaches 0.60 mol H2S per kg absorbent. After that, the absorption rate decreases sharply and the absorption reaches equilibrium even in about 1400 min with a capacity of 1.88 mol H2S per kg absorbent. At the end of the absorption, the IL became almost solid and the viscosity was too high to be measured. This makes the IL cannot be used in industry application.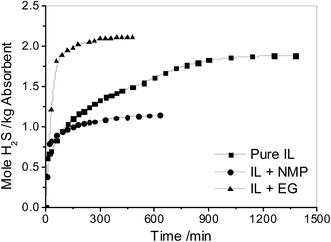 | ||
Fig. 2 Effect of solvents on the absorption of H2S by [N2222][L-Ala] and solvent mixtures (the mass ratio of IL to solvent is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C with a H2S partial pressure of 0.005 MPa in gas. 1) at 40 °C with a H2S partial pressure of 0.005 MPa in gas. | ||
It was reported that the addition of solvents in ILs could decrease the viscosity of the ILs during the absorption of acid gases.48 Therefore, two kinds of solvents with low vapor pressures were chosen to reduce the viscosity of ILs.49 As shown in Table 1, N-methyl-2-pyrrolidone (NMP) and EG were separately added into [N2222][L-Ala] with equal weight, and both of them could reduce the viscosity of mixtures before and after the absorption. However, the solubility of H2S in these mixtures is quite different. When NMP was added into the IL, the solubility of H2S is 1.14 mol H2S per kg absorbent. But when EG was added into the IL, the solubility of H2S reach 2.10 mol H2S per kg absorbent, even higher than that in pure IL. This means that EG can enhance the absorption of H2S in IL. In addition, the vapor pressure of EG is extremely low, just 0.017 kPa at 25 °C.46 As a result, EG was used as a solvent for H2S capture in the next work.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) before and after absorption of H2S at 40 °C with a H2S partial pressure of 0.005 MPa in gas
1) before and after absorption of H2S at 40 °C with a H2S partial pressure of 0.005 MPa in gas
| Absorbent | η (before absorption)/mPa s | η (after absorption)/mPa s |
|---|---|---|
| [N2222][L-Ala] | 71 | Almost solid |
| [N2222][L-Ala] + EG | 31 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 156 156 |
| [N2222][L-Ala] + NMP | 8 | Glass |
Effect of EG on the absorption of H2S
Fig. 3 shows the effect of mass ratio of EG to [N2222][L-Ala] on the H2S solubility in [N2222][L-Ala] and EG mixtures at 40 °C with a H2S partial pressure of 0.005 MPa in gas. When the mass ratio of IL to EG is 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, the absorption of H2S by the mixture needs a long time of 870 min to reach equilibrium. The solubility of H2S in the mixture is 1.76 mol H2S per kg absorbent. When the mass ratio of IL to EG is 1
3, the absorption of H2S by the mixture needs a long time of 870 min to reach equilibrium. The solubility of H2S in the mixture is 1.76 mol H2S per kg absorbent. When the mass ratio of IL to EG is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the absorption can reach equilibrium in 480 min, and the solubility in the mixture is increased to 2.10 mol H2S per kg absorbent. This means that the addition of EG can enhance the H2S solubility in the mixture, and also raise the H2S absorption rate.
1, the absorption can reach equilibrium in 480 min, and the solubility in the mixture is increased to 2.10 mol H2S per kg absorbent. This means that the addition of EG can enhance the H2S solubility in the mixture, and also raise the H2S absorption rate.
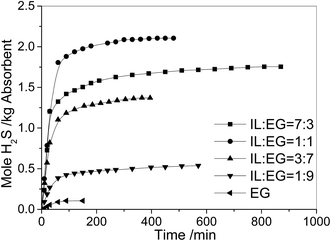 | ||
| Fig. 3 Absorption of H2S as a function of time in [N2222][L-Ala] and EG mixtures with different mass ratios at 40 °C with a H2S partial pressure of 0.005 MPa in gas. | ||
However, due to the low solubility of H2S in pure EG, the solubility of H2S in the mixtures decreases with the further increase of EG content. For example, when the mass ratio of IL to EG reaches 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, the solubility of H2S in the mixture decreases to 1.37 mol H2S per kg absorbent.
7, the solubility of H2S in the mixture decreases to 1.37 mol H2S per kg absorbent.
Effect of temperature on the absorption of H2S
Temperature can influence on the solubility of gases in liquid solutions. In this work, the effect of temperature on H2S absorption in [N2222][L-Ala] and EG mixture was investigated, and the results are shown in Fig. 4. It can be seen that the increase of temperature can decrease the solubility of H2S in the IL and EG mixture with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mass ratio of IL to EG. For example, at 30.0 °C, the solubility of H2S in the mixture is 2.22 mol H2S per kg absorbent, and at 50.0 °C, it decreases to 2.05 mol H2S per kg absorbent. Therefore, a decrease of temperature is favorable for H2S absorption, while an increase of temperature is favorable for H2S desorption, which suggests the absorbent can be regenerated by heating treatment.
1 mass ratio of IL to EG. For example, at 30.0 °C, the solubility of H2S in the mixture is 2.22 mol H2S per kg absorbent, and at 50.0 °C, it decreases to 2.05 mol H2S per kg absorbent. Therefore, a decrease of temperature is favorable for H2S absorption, while an increase of temperature is favorable for H2S desorption, which suggests the absorbent can be regenerated by heating treatment.
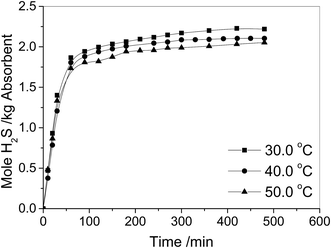 | ||
Fig. 4 Absorption of H2S as a function of time in [N2222][L-Ala] and EG mixtures (the mass ratio of IL to EG is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at different temperatures with a H2S partial pressure of 0.005 MPa in gas. 1) at different temperatures with a H2S partial pressure of 0.005 MPa in gas. | ||
Effect of H2S partial pressure on the absorption of H2S
The partial pressure of H2S in gas also shows influence on the capture of H2S from gas. It can be seen from Fig. 5 that when H2S partial pressure decreases, the absorption capacity of H2S in the mixture decreases. For example, when the partial pressure of H2S is 0.005 MPa, the solubility of H2S in the mixture is 2.10 mol H2S per kg absorbent. When the partial pressure of H2S decreases to 0.0005 MPa, the solubility of H2S in the mixture is just 1.25 mol H2S per kg absorbent. However, when the partial pressure decreases from 0.005 MPa to 0.002 MPa, the solubility of H2S in the mixture do not changed obviously, just from 2.10 to 2.07 mol H2S per kg absorbent. | ||
Fig. 5 Absorption of H2S as a function of time in [N2222][L-Ala] and EG mixtures (the mass ratio of IL to EG is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C with different partial pressures of H2S in gas. 1) at 40 °C with different partial pressures of H2S in gas. | ||
Reuse of [N2222][L-Ala] and EG mixture
The reuse of [N2222][L-Ala] and EG mixture was also tested in this work. As shown in Fig. 6, no obvious change was observed during the reuse of the mixture at 40 °C with a H2S partial pressure of 0.005 MPa in gas, indicating that the [N2222][L-Ala] and EG mixture can be reused. But there still remain a little amount of H2S in the mixture after desorption. However, the remaining H2S in the mixture is almost a constant value, about 0.38 mol H2S per kg absorbent, and it doesn't influence the solubility of H2S in the next cycle. As discussed in the mechanism section, there is a weak chemical interaction between the –NH2 group on [N2222][L-Ala] and H2S, which may cause the high H2S residue after the desorption. | ||
Fig. 6 The reuse of [N2222][L-Ala] and EG mixture (the mass ratio of IL to EG is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C with a H2S partial pressure of 0.005 MPa in gas. 1) at 40 °C with a H2S partial pressure of 0.005 MPa in gas. | ||
Effect of ILs on the capture of H2S
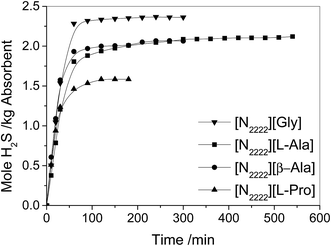
The absorption of H2S in different IL and EG mixtures as a function of time was investigated and the results are shown in Fig. 7. The effects of ILs on the solubility of H2S are shown in Table 2. The solubilities of H2S in the four IL and EG mixtures are varied from 2.36 to 1.59 mol H2S per kg absorbent. Interestingly, the solubility of H2S in the different tetraethyl ammonium amino acid IL and EG mixtures is enhanced with the decrease of pKb of –NH2 (or –NH) in amino acids, which are formed the anion of ILs. For instance, the pKb of –NH2 in β-alanine is 10.08, and the solubility of H2S in the mixture is 2.06 mol H2S per kg absorbent. But the pKb of –NH2 in glycine is 9.58, and the solubility of H2S in the mixture is 2.36 mol H2S per kg absorbent. As far as we know, this is the highest solubility absorbent reported in the literature up to now, and the solubility of H2S is even higher than that in aqueous solution of alkanolamines at the same condition.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C with a H2S partial pressure of 0.005 MPa in gas, and the pKb of –NH2 (or –NH) in amino acids formed the anion of ILs
1) at 40 °C with a H2S partial pressure of 0.005 MPa in gas, and the pKb of –NH2 (or –NH) in amino acids formed the anion of ILs
| Solubility of H2S in absorbents | |||
|---|---|---|---|
| IL | Mole H2S per kg absorbent | Mole ratio of H2S to ILa | pKb of –NH2 or –NH in amino acidb |
| a These data is calculated from the absorption capacity of H2S in the IL and EG mixtures.b These data was measured at 25 °C.51 | |||
| [N2222][Gly] | 2.36 | 0.94 | 9.58 |
| [N2222][L-Ala] | 2.12 | 0.90 | 9.71 |
| [N2222][β-Ala] | 2.06 | 0.89 | 10.08 |
| [N2222][L-Pro] | 1.59 | 0.75 | 10.47 |
The mechanism of H2S absorption by tetraethyl ammonium amino acid ILs with the absence and presence of EG
Wu et al.30 reported that both physical and chemical interactions existed between H2S and carboxylate-based ILs. They proposed a reaction mechanism that two IL molecules interact with one H2S molecule. As the partial pressure of H2S is extremely low in this work, just 0.005 MPa, the physical solubility can almost be ignored. Chemical interaction between IL and H2S is main force for the absorption.Table 3 shows the solubility of H2S in [N2222][L-Ala] with different mass ratios of IL to EG, subtracting the solubility of H2S in pure EG. The H2S solubility in [N2222][L-Ala] is 0.90 mol H2S per mol IL, when the mass ratio of IL to EG is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. With the increase of EG in the absorbents, the solubility of H2S in the ILs increases. However, in the absence of EG in the mixture, the solubility of H2S in [N2222][L-Ala] is just 0.41 mol H2S per mol IL. This means that EG may not only enhance the solubility of H2S in IL, but also change the mechanism of H2S absorption.
1. With the increase of EG in the absorbents, the solubility of H2S in the ILs increases. However, in the absence of EG in the mixture, the solubility of H2S in [N2222][L-Ala] is just 0.41 mol H2S per mol IL. This means that EG may not only enhance the solubility of H2S in IL, but also change the mechanism of H2S absorption.
As we know, amino acids contain –COOH and –NH2 (or –NH) groups on their molecules, as shown in Fig. 1. When mixed with [N2222]OH, the –COOH groups in the amino acids react with [N2222]OH and the ILs are formed. The –NH2 (or –NH) group on the amino acids became an alkaline group in the ILs, which can chemically react with H2S. As shown in Table 2, the pKb of –NH2 (or –NH) group plays an important role on the absorption of H2S. The lower the pKb of –NH2 or –NH group in amino acids is, the higher the solubility of H2S in the ILs is. This indicates that the –NH2 or –NH group may lead to the absorption of H2S in ILs. The result also shows a way to enhance the solubility of H2S in ILs.
The absorption mechanism of H2S by IL was investigated with 1H NMR and FT-IR. The 1H NMR and FT-IR spectra are shown in Fig. 8 and 9, respectively. From the 1H NMR spectra in Fig. 8, it can be seen that before and after the absorption of H2S without EG in the IL, the resonances of the methyl protons (δ = 1.12 ppm) and methine protons (δ = 3.21 ppm) on the [L-Ala] anion shift downfield to δ = 1.15 and 3.26 ppm after H2S absorption. The FT-IR results in Fig. 9 also show the change of IL before and after the absorption. On comparing the FT-IR spectra before and after H2S absorption, the weak absorption band at 1616 cm−1 shift to a strong absorption band at 1621 cm−1, which was attributed to the new N–H deformation vibration.13 This means that chemical interaction exists between the –NH2 group and H2S, and is not very strong.
 | ||
| Fig. 8 1H NMR spectra of absorbents before and after H2S absorption: (a) [N2222][L-Ala]; (b) [N2222][L-Ala] + H2S; (c) [N2222][L-Ala] + EG; (d) [N2222][L-Ala] + EG + H2S. | ||
 | ||
| Fig. 9 FT-IR spectra of absorbents before and after H2S absorption: (a) [N2222][L-Ala]; (b) [N2222][L-Ala] + H2S; (c) [N2222][L-Ala] + EG; (d) [N2222][L-Ala] + EG + H2S. | ||
However, when EG was added into the IL, the resonances of the methyl protons (δ = 1.12 ppm) and methine protons (δ = 3.21 ppm) on the [L-Ala] anion shift upfield to δ = 1.08 and 3.17 ppm, respectively. There may be a hydrogen bond formed between –NH2 group in [L-Ala] anion and EG. After H2S absorption in the mixture of EG and [N2222][L-Ala], the resonances of the methyl protons (δ = 1.08 ppm) and methine protons (δ = 3.17 ppm) on the [L-Ala] anion shift downfield to δ = 1.33 and 3.62 ppm, respectively. It can be seen that there is also a chemical interaction between the –NH2 group and H2S with EG in the IL, and the interaction is stronger than that without EG in the IL. This means that EG can enhance the capacity of H2S in tetraethyl ammonium amino acid ILs. Moreover, on comparing the FT-IR spectra, shown in Fig. 9, of IL with EG before and after H2S absorption, the weak absorption band at 1616 cm−1 also shifts to a strong absorption band at 1621 cm−1, which was assigned to the new N–H deformation vibration.13
Based on the results above, we proposed a possible mechanism of H2S absorption by tetraethyl ammonium amino acid ILs in the absence and presence of EG in Fig. 10. To confirm the mechanism proposed, an equal weight of glycerol was added into the IL, and it was found that the solubility of H2S in the IL was 0.81 mol H2S per mol IL at 40 °C with 5% of H2S in nitrogen, which indicates that the solubility of H2S in the IL can be enhanced by glycerol.
 | ||
| Fig. 10 The proposed mechanism for the absorption of H2S by [N2222][L-Ala] in the absence and presence of EG. | ||
According to the proposed mechanism shown in Fig. 10, in the absence of EG, there is an absorption equilibrium between IL and H2S, and stoichiometrically, the maximum solubility of H2S in IL is 0.5 mol H2S per mol IL, which is very close to the measured solubility of H2S in pure IL or mixed with NMP. However, when EG is added into IL, there is new absorption equilibrium among IL, H2S and EG. The solubility of H2S in IL can stoichiometrically reach 1.0 mol H2S per mol IL. At absorption equilibrium, there exists a chemical equilibrium constant (K), which is a function of temperature, type of ILs and concentration of EG. The increase of temperature can decrease the K value and then the solubility of H2S, which is demonstrated in Fig. 4. The change of ILs can also influence the K value. With the decrease of the pKb of –NH2 (or –NH) in anion, the basicity of the ILs is raised and then increases the K value, resulting in increasing the solubility of H2S. EG can not only react with H2S, but also enhance the basicity of aliphatic amine by the solvation effect.50 Hence, the increase of EG concentration can also increase the K value. Due to that the H2S absorption follows the absorption equilibrium, when the mass ratio of EG to [N2222][L-Ala] is increased much higher than that the stoichiometric reaction needs as shown in Table 3, the solubilities of H2S in IL are around 1.0 mol H2S per mol IL of stoichiometric value. Although the partial pressure does not change the K value, the increase of H2S partial pressure can shift the equilibrium to the absorption direction, increasing the solubilities of H2S in IL. During regenerating the absorbents, the increase of temperature decreases the K value and bubbling with N2 decreases the H2S partial pressure, both are favorable to the desorption of H2S in IL.
Conclusions
In summary, a series of tetraethyl ammonium amino acid ILs were synthesized and mixed with EG to absorb dilute H2S. The effects of solvent type, mass ratio of EG to IL, temperature, and H2S partial pressure on the absorption of H2S in the ILs were studied. It has been found that these IL and EG mixtures can capture large amounts of H2S under low H2S partial pressures. The solubility of H2S in the mixture of [N2222][Gly] and EG with a mass ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is 2.36 mol H2S per kg absorbent, which is the highest capacity of H2S in solvents up to now at 40 °C and 0.005 MPa. EG in the ILs not only decreases the viscosity of the absorbent, but also enhance the solubility of H2S in the ILs. The temperature has a negative effect on the absorption, while the H2S partial pressure has a positive one. The pKb of –NH2 (or –NH) in amino acids formed the anion of ILs also influence the capacity of the mixture, and the decrease of pKb can enhance the capacity. Finally, the possible mechanism of the absorption by tetraethyl ammonium amino acid ILs with the absence and presence of EG has been proposed. This work provides a way to design functional ILs, enhance the absorption capacity of H2S in the ILs, and decrease the viscosity of the ILs during absorption.
1 is 2.36 mol H2S per kg absorbent, which is the highest capacity of H2S in solvents up to now at 40 °C and 0.005 MPa. EG in the ILs not only decreases the viscosity of the absorbent, but also enhance the solubility of H2S in the ILs. The temperature has a negative effect on the absorption, while the H2S partial pressure has a positive one. The pKb of –NH2 (or –NH) in amino acids formed the anion of ILs also influence the capacity of the mixture, and the decrease of pKb can enhance the capacity. Finally, the possible mechanism of the absorption by tetraethyl ammonium amino acid ILs with the absence and presence of EG has been proposed. This work provides a way to design functional ILs, enhance the absorption capacity of H2S in the ILs, and decrease the viscosity of the ILs during absorption.
Acknowledgements
The authors thank Professors Zhenyu Liu and Qingya Liu for their help and the financial support of the National Natural Science Foundation of China (No. 21306007), the Research Fund for the Doctoral Program of Higher Education of China (20130010120005) and the long-term subsidy mechanism from the Ministry of Finance and the Ministry of Education of PRC (BUCT).References
- B. J. Wood, W. E. Isakson and H. Wise, Ind. Eng. Chem. Prod. Res. Dev., 1980, 19, 197–204 CrossRef CAS.
- F. Murrieta-Guevara and A. T. Rodriguez, J. Chem. Eng. Data, 1984, 29, 456–460 CrossRef CAS.
- B. Munder, H. Lidal and O. C. Sandall, J. Chem. Eng. Data, 2000, 45, 1201–1204 CrossRef CAS.
- P. J. G. Huttenhuis, N. J. Agrawal, J. A. Hogendoorn and G. F. Versteeg, J. Pet. Sci. Eng., 2007, 55, 122–134 CrossRef CAS.
- M. Shokouhi, H. Farahani, M. Hosseini-Jenab and A. H. Jalili, J. Chem. Eng. Data, 2015, 60, 499–508 CrossRef CAS.
- L. A. Blanchard, D. Hancu, E. J. Beckman and J. F. Brennecke, Nature, 1999, 399, 28–29 CrossRef.
- L. A. Blanchard, Z. Y. Gu and J. F. Brennecke, J. Phys. Chem. B, 2001, 105, 2437–2444 CrossRef CAS.
- E. D. Bates, R. D. Mayton, L. Ntai and J. H. Davis, J. Am. Chem. Soc., 2002, 124, 926–927 CrossRef CAS PubMed.
- Y. Y. Jiang, G. N. Wang, Z. Zhou, Y. T. Wu, J. Geng and Z. B. Zhang, Chem. Commun., 2008, 505–507 RSC.
- Y. Q. Zhang, S. J. Zhang, X. M. Lu, Q. Zhou, W. Fan and X. P. Zhang, Chem.–Eur. J., 2009, 15, 3003–3011 CrossRef CAS PubMed.
- B. E. Gurkan, J. C. Fuente, E. M. Mindrup, L. E. Ficke, B. F. Goodrich, E. A. Price, W. F. Schneider and J. F. Brennecke, J. Am. Chem. Soc., 2010, 132, 2116–2117 CrossRef CAS PubMed.
- C. M. Wang, H. M. Lou, D. E. Jiang, H. R. Li and S. Dai, Angew. Chem., 2010, 122, 6114–6117 CrossRef.
- W. Z. Wu, B. X. Han, H. X. Gao, Z. M. Liu, T. Jiang and J. Huang, Angew. Chem., Int. Ed., 2004, 43, 2415–2417 CrossRef CAS PubMed.
- J. Huang, A. Riisager, P. Wasserscheid and R. Fehrmann, Chem. Commun., 2006, 4027–4029 RSC.
- X. L. Yuan, S. J. Zhang and X. M. Lu, J. Chem. Eng. Data, 2007, 52, 596–599 CrossRef CAS.
- C. M. Wang, G. K. Cui, X. Y. Lou, Y. J. Xu, H. R. Li and S. Dai, J. Am. Chem. Soc., 2011, 133, 11916–11919 CrossRef CAS PubMed.
- G. K. Cui, J. J. Zheng, X. Y. Luo, W. J. Lin, F. Ding, H. R. Li and C. M. Wang, Angew. Chem., Int. Ed., 2013, 125, 10814–10818 CrossRef.
- F. Y. Jou and A. E. Mather, Int. J. Thermophys., 2007, 28, 490–495 CrossRef CAS.
- C. S. Pomelli, C. Chiappe, A. Vidis, G. Laurenczy and P. J. Dyson, J. Phys. Chem. B, 2007, 111, 13014–13019 CrossRef CAS PubMed.
- M. Rahmati-Rostami, C. Ghotbi, M. Hosseini-Jenab, A. N. Ahmadi and A. H. Jalili, J. Chem. Thermodyn., 2009, 41, 1052–1055 CrossRef CAS.
- M. Shokouhi, M. Adibi, A. H. Jalili, M. Hosseini-Jenab and A. Mehdizadeh, J. Chem. Eng. Data, 2010, 55, 1663–1668 CrossRef CAS.
- A. H. Jalili, A. Mehdizadeh, M. Shokouhi, A. N. Ahmadi, M. Hosseini-Jenab and F. Fateminassab, J. Chem. Thermodyn., 2010, 42, 1298–1303 CrossRef CAS.
- M. B. Shiflett, A. M. S. Niehaus and A. Yokozeki, J. Chem. Eng. Data, 2010, 55, 4785–4793 CrossRef CAS.
- H. Sakhaeinia, V. Taghikhani, A. H. Jalili, A. Mehdizadeh and A. A. Safekordi, Fluid Phase Equilib., 2010, 298, 303–309 CrossRef CAS.
- H. Sakhaeinia, A. H. Jalili, V. Taghikhani and A. A. Safekordi, J. Chem. Eng. Data, 2010, 55, 5839–5845 CrossRef CAS.
- A. H. Jalili, M. Safavi, C. Ghotbi, A. Mehdizadeh, M. Hosseini-Jenab and V. Taghikhani, J. Phys. Chem. B, 2012, 116, 2758–2774 CrossRef CAS PubMed.
- A. H. Jalili, M. Shokouhi, G. Maurer and M. Hosseini-Jenab, J. Chem. Thermodyn., 2013, 67, 55–62 CrossRef CAS.
- M. Safavi, C. Ghotbi, V. Taghikhani, A. H. Jalili and A. Mehdizadeh, J. Chem. Thermodyn., 2013, 65, 220–232 CrossRef CAS.
- M. B. Shiflett and A. Yokozeki, Fluid Phase Equilib., 2010, 294, 105–113 CrossRef CAS.
- K. Huang, D. N. Cai, Y. L. Chen, Y. T. Wu, X. B. Hu and Z. B. Zhang, AIChE J., 2013, 59, 2227–2235 CrossRef CAS.
- A. Haghtalab and A. Kheiri, J. Chem. Thermodyn., 2015, 89, 41–50 CrossRef CAS.
- F. Llovell, R. M. Marcos, N. MacDowell and L. F. Vega, J. Phys. Chem. B, 2012, 116, 7709–7718 CrossRef CAS PubMed.
- S. Mortazavi-Manesh, M. A. Satyro and R. A. Marriott, AIChE J., 2013, 59, 2993–3005 CrossRef CAS.
- J. J. Chen, W. W. Li, H. Q. Yu and X. L. Li, AIChE J., 2013, 59, 3824–3833 CrossRef CAS.
- R. Shahriari, M. R. Dehghani and B. Behzadi, Fluid Phase Equilib., 2012, 313, 60–72 CrossRef.
- X. Y. Ji, C. Held and G. Sadowski, Fluid Phase Equilib., 2014, 363, 59–65 CrossRef CAS.
- H. J. Liu, S. Dai and D. E. Jiang, J. Phys. Chem. B, 2014, 118, 2719–2725 CrossRef CAS PubMed.
- M. A. Ahmadi, R. Haghbakhsh, R. Soleimani and M. B. Bajestani, J. Supercrit. Fluids, 2014, 92, 60–69 CrossRef CAS.
- C. A. Faundez, J. F. Diaz-Valdes and J. O. Valderrama, Fluid Phase Equilib., 2014, 375, 152–160 CrossRef CAS.
- J. Sanchez-Badillo, M. Gallo, S. Alvarado and D. Glossman-Mitnik, J. Phys. Chem. B, 2015, 119, 10727–10737 CrossRef CAS PubMed.
- M. Ramdin, S. P. Balaji, J. M. Vicent-Luna, J. J. Gutierrez-Sevillano, S. Calero, T. W. Loos and T. J. H. Vlugt, J. Phys. Chem. B, 2014, 118, 23599–23604 CAS.
- B. Guo, E. H. Duan, Y. F. Zhong, L. Gao, X. S. Zhang and D. S. Zhao, Energy Fuels, 2011, 25, 159–161 CrossRef CAS.
- K. Huang, X. M. Zhang, Y. Xu, Y. T. Wu and X. B. Hu, AIChE J., 2014, 60, 4232–4240 CrossRef CAS.
- W. Y. Xu and C. A. Jiang, The Handbook of Natural Gas Utilization, China Petrochemical Press, Beijing, China, 2nd edn, 2006 Search PubMed.
- K. Huang, D. N. Cai, Y. L. Chen, Y. T. Wu, X. B. Hu and Z. B. Zhang, ChemPlusChem, 2014, 79, 241–249 CrossRef CAS.
- I. Short, A. Sahgal and W. Hayduk, J. Chem. Eng. Data, 1983, 28, 63–66 CrossRef CAS.
- S. H. Ren, Y. C. Hou, W. Z. Wu and W. N. Liu, J. Chem. Eng. Data, 2010, 55, 5074–5077 CrossRef CAS.
- S. H. Ren, Y. C. Hou, W. Z. Wu, X. T. Chen, J. L. Fan and J. W. Zhang, Ind. Eng. Chem. Res., 2009, 48, 4928–4932 CrossRef CAS.
- J. Y. Lenoir, P. Renault and H. Renon, J. Chem. Eng. Data, 1971, 16, 340–342 CrossRef.
- H. J. Xu, C. F. Zhang and Z. S. Zheng, Ind. Eng. Chem. Res., 2002, 41, 6175–6180 CrossRef CAS.
- CRC Handbook of Chemistry and Physics, ed. D. P. Lide, CRC Press, Boca Raton, FL, 85th edn, 2005 Search PubMed.
| This journal is © The Royal Society of Chemistry 2016 |