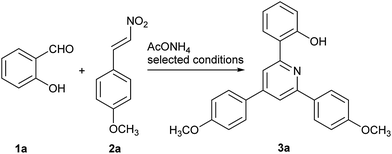
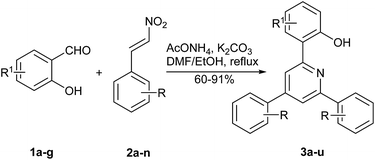
One-pot synthesis of 2,4,6-triarylpyridines from β-nitrostyrenes, substituted salicylic aldehydes and ammonium acetate†
Xushun Qing,
Ting Wang,
Feixiang Zhang and
Cunde Wang*
School of Chemistry and Chemical Engineering, Yangzhou University, 180 Siwangting Street, Yangzhou 225002, P. R. China. E-mail: wangcd@yzu.edu.cn; Fax: +86-514-8797-5244; Tel: +86-514-8797-5568
First published on 3rd October 2016
Abstract
A protocol for the synthesis of 2,4,6-trisubstituted pyridines from the β-nitrostyrenes, available substituted salicylic aldehydes and ammonium acetate was developed. The present strategy features high chemoselectivity and excellent tolerance for a broad range of functional groups, in which the β-nitrostyrenes generated from aldehydes and nitromethane, substituted salicylic aldehydes and ammonium acetate were respectively employed as simple and easily available substrates. This efficient method provides fast access to a variety of structurally diverse pyridine derivatives. The structures of two typical products were confirmed by X-ray crystallography.
Pyridines are privileged structural motifs and an important class of six-membered nitrogen containing heterocycles which are widely found in numerous natural products,1 pharmaceuticals,2 agrochemicals,3 electrochemicals,4 and functional organic materials.5 Among them, 2,4,6-triarylpyridines were widely investigated recently because of the broad spectrum of their biological and pharmaceutical properties (Fig. 1) such as anti-cancer, anti-malarial, vasodilator, anti-covulsant, anesthetic, anti-epileptic character and their use as pesticidial, fungicidal and herbicidal agrochemicals.6
Due to their π-stacking ability, 2,4,6-triarylpyridines were also used in supramolecular chemistry as building blocks7 or ligands in coordination chemistry and transition metal catalysis.8 Additionally, many synthetically symmetrical triarylthio-, triarylseleno- and triaryltelluoropyrylium salt compounds based on 2,4,6-triarylpyridine nucleus with the π-conjugated polyaryl motifs as the photosensitizers have been studied in photodynamic cell-specific cancer therapy.9 Given their immense pharmaceutical and material usefulness, the development of efficient syntheses of these functionalized 2,4,6-triarylpyridines has thus attracted many organic and medicinal chemists for decades. Traditionally, there were several methods for the preparation of the 2,4,6-triarylpyridine derivatives,10 including Chichibabin synthesis,11–13 and Kröhnke synthesis.14–16 Apart from ammonium acetate, various containing-nitrogen compounds were used as nitrogen sources in the annulation synthesis of 2,4,6-trisubstituted pyridines. For example, the intermolecular Michael addition reaction and an intramolecular condensation of 1-arylethylamines and ynones through the direct β-C(sp3)–H functionalization of enaminones under metal-free conditions directly constructed 2,4,6-trisubstituted pyridines.17 Particularly, ketones with amines as simple substrates have a structural advantage to provide, formally, the vinyl amine moiety in the assembly of 2,4,6-trisubstituted pyridines via HOTf-catalyzed and solvent-free system.18 Very recently, Wu and co-workers reported an iodine-triggered the decarboxylation–deamination of amino acids for the efficient preparation of 2,4,6-trisubstituted pyridines based on the catabolism and reconstruction behaviors of amino acids.19 Similarly, the combinational employment of iodine and triethylamine promoted effectively to trigger the oxime based synthesis of 2,4,6-trisubstituted pyridines.20 Guan and co-workers also described an iron-catalyzed cyclization of ketoxime carboxylates and N,N-dialkylanilines for the modular synthesis of diverse 2,4,6-trisubstituted pyridines.21 Additionally, the functionalization of existing pyridines was another prominent reaction, such as transition-metal catalyzed direct arylation of pyridines via C–H activation.22
Owing to large spectrum of fascinating applications on pharmaceuticals and materials, the development of methodologies for a direct access to highly substituted and specifically functionalized 2,4,6-triarylpyridines is a continuing challenge in modern synthetic organic chemistry.
Multicomponent reactions received increasing attention because of their efficiency and simplicity, appealing for synthetic and medicinal researchers interested in rapidly accessing new diversity and complexity of targeting moleculars.23 Recently a large number of reports have indicated that substituted β-nitrostyrenes are important synthetic intermediates and starting materials for the synthesis of a variety of useful building blocks, which widely were employed in the multicomponent reactions as an important component.24 Our previous results also showed that substituted β-nitrostyrenes as essential building blocks were efficiently used to construct heterocyclic cores via multicomponent reactions.25
In this context, nowadays, the multicomponent reactions of β-nitrostyrenes with 2-(iminomethyl)phenols generated from available substituted salicylic aldehydes and ammonium acetate largely leads the way in the synthesis of 2,4,6-triarylpyridines. Notably, this work represents a fascinating approach for the direct construction of two C–N bonds and two C–C bonds via cyclization in a single step to yield diversely asymmetric 2,4,6-triarylpyridines.
We commenced our investigation using 1a, 2a and ammonium acetate (AcONH4) as the model substrates (Table 1). Initially, in the presence of potassium carbonate (K2CO3) as the basic promoter, the reaction was carried out with various solvents, the results indicated that solvents such as EtOH, CH3CN, toluene and pyridine disfavored the desired transformation (Table 1, entries 1–4) and that polar solvent DMF was the best reaction media (Table 1, entry 5). However, a small improvement in the yield was observed with mixing solvent DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v, mL) (Table 1, entry 6). Further studies on the reaction without basic promoters indicated that the basic promoters should have to be used to stimulate the reaction (Table 1, entry 7). Encouraged by this result, we switched over to optimizing the reaction conditions, and different bases such as the common organic bases triethylamine, piperidine, 1,8-diazabicyclo [5.4.0]undec-7-ene (DBU) and strong inorganic base sodium hydroxide, but none of them was found to be better than K2CO3 (Table 1, entries 8–10) despite the complete time was shortened, the strong inorganic base such as sodium hydroxide (NaOH) failed to react under identical conditions (Table 1, entry 11), this maybe because that β-nitrostyrenes afforded the polymerization in the presence of a strong base such as NaOH. Then, we proceeded to investigate the optimum amount of K2CO3 required, and it was found that 2 equiv. was sufficient, as upon increasing the amount no change in the yield was observed (Table 1, entries 12 and 14). As shown in Table 1 (entry 6, entries 12–14), K2CO3 proved to be the best base among those tested to give the desired product in a maximum yield of 82%. Subsequently, ratio optimization of the mixed solvents showed that the ratio 4
1, v/v, mL) (Table 1, entry 6). Further studies on the reaction without basic promoters indicated that the basic promoters should have to be used to stimulate the reaction (Table 1, entry 7). Encouraged by this result, we switched over to optimizing the reaction conditions, and different bases such as the common organic bases triethylamine, piperidine, 1,8-diazabicyclo [5.4.0]undec-7-ene (DBU) and strong inorganic base sodium hydroxide, but none of them was found to be better than K2CO3 (Table 1, entries 8–10) despite the complete time was shortened, the strong inorganic base such as sodium hydroxide (NaOH) failed to react under identical conditions (Table 1, entry 11), this maybe because that β-nitrostyrenes afforded the polymerization in the presence of a strong base such as NaOH. Then, we proceeded to investigate the optimum amount of K2CO3 required, and it was found that 2 equiv. was sufficient, as upon increasing the amount no change in the yield was observed (Table 1, entries 12 and 14). As shown in Table 1 (entry 6, entries 12–14), K2CO3 proved to be the best base among those tested to give the desired product in a maximum yield of 82%. Subsequently, ratio optimization of the mixed solvents showed that the ratio 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (DMF/EtOH) was the optimum ratio (Table 1, entry 12, entries 15 and 18). The results indicated that the ratio of DMF/EtOH mixed solvents played an important part in the synthesis of 2,4,6-triarylpyridines via one-pot reaction due to that different ratio of DMF/EtOH mixed solvents should determine the temperature of the reaction system despite under refluxing. From the above results, the reaction conditions were affirmed: 1.0 equivalent of 1a, 2.05 equivalents of 2a, 1.1 equivalents of AcONH4, and 2.0 equivalents of K2CO3 in 5 mL mixed solvents DMF/EtOH (4
1 (DMF/EtOH) was the optimum ratio (Table 1, entry 12, entries 15 and 18). The results indicated that the ratio of DMF/EtOH mixed solvents played an important part in the synthesis of 2,4,6-triarylpyridines via one-pot reaction due to that different ratio of DMF/EtOH mixed solvents should determine the temperature of the reaction system despite under refluxing. From the above results, the reaction conditions were affirmed: 1.0 equivalent of 1a, 2.05 equivalents of 2a, 1.1 equivalents of AcONH4, and 2.0 equivalents of K2CO3 in 5 mL mixed solvents DMF/EtOH (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v, mL) at 122 °C (reaction system temperature) under refluxing for 5 h.
1, v/v, mL) at 122 °C (reaction system temperature) under refluxing for 5 h.
| Entry | Base | Solvent | T (°C) | t (h) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 1 mmol of 1a, 2.05 equiv. of 2a, 1.1 equiv. of AcONH4, appropriate basic reagent, 5 mL solvent, unless otherwise noted.b Isolated yield. | |||||
| 1 | K2CO3 (1.0) | EtOH | Reflux | 5 | 0 |
| 2 | K2CO3 (1.0) | MeCN | Reflux | 5 | Trace |
| 3 | K2CO3 (1.0) | PhMe | Reflux | 5 | Trace |
| 4 | K2CO3 (1.0) | Pyridine | Reflux | 5 | Trace |
| 5 | K2CO3 (1.0) | DMF | Reflux | 5 | 50 |
| 6 | K2CO3 (1.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 5 | 53 |
| 7 | (—) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 24 | 0 |
| 8 | Et3N (1.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 5 | 31 |
| 9 | Piperidine (1.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 4 | 33 |
| 10 | DBU (1.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 3 | 52 |
| 11 | NaOH (1.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 5 | Trace |
| 12 | K2CO3 (2.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 5 | 82 |
| 13 | K2CO3 (3.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 5 | 82 |
| 14 | K2CO3 (4.0) | DMF/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
118 | 5 | 81 |
| 15 | K2CO3 (2.0) | DMF/EtOH (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
98 | 8 | 78 |
| 16 | K2CO3 (2.0) | DMF/EtOH (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
122 | 5 | 91 |
| 17 | K2CO3 (2.0) | DMF/EtOH (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
124 | 5 | 90 |
| 18 | K2CO3 (2.0) | DMF/EtOH (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
131 | 5 | 85 |
The scope of this transformation was then investigated under the optimum conditions using different substituted salicylic aldehydes, and available substituted β-nitrostyrenes. The results are summarized in Table 2. Generally, aromatic β-nitroalkenes employed reacted smoothly to give the corresponding pyridines in good to excellent yields (Table 2, 3a–u, 60–91%), regardless of the functional groups at the para-, meta-, or ortho-position. A large range of functional groups on the benzene ring such as methyl, methoxyl, fluoro, chloro, bromo, iodo, nitro, and ethoxyl were all tolerated well. The electronic effects of the substituents on the yield showed the slight difference. Notably, β-nitrostyrenes bearing an electron-donating group on the aromatic ring (Table 2, entries 1–7) worked better than those bearing an electron-withdrawing group. The results also showed that the steric effects of the substituents are apparent, and the β-nitrostyrene with an iodo group afforded relatively low yield. Generally, the β-nitrostyrenes with para-position phenyl groups gave the products in higher yields than those with ortho-, or meta-position phenyl groups (Table 2, entries 8–10). Other β-nitrostyrenes such as 2-(2-nitrovinyl)thiophene, 2-(2-nitrovinyl)furan and 3-(2-nitrovinyl)thiophene also produced the furyl and thienyl products in good yields (Table 2, entries 17–19).
| Entry | R1 | R | Product | Yieldb (%) |
|---|---|---|---|---|
a Salicylic aldehydes 1a–g (1 mmol), β-nitrostyrenes 2a–n (2.05 mmol) and K2CO3 (2.0 equiv.), DMF/EtOH (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v, mL), reflux (122 °C), 9 h.b Isolated yield.c 2-(2-Nitrovinyl)furan.d 2-(2-Nitrovinyl)thiophene.e 3-(2-Nitrovinyl)thiophene. 1, v/v, mL), reflux (122 °C), 9 h.b Isolated yield.c 2-(2-Nitrovinyl)furan.d 2-(2-Nitrovinyl)thiophene.e 3-(2-Nitrovinyl)thiophene. |
||||
| 1 | H | p-OMe | 3a | 91 |
| 2 | 3-EtO | H | 3b | 80 |
| 3 | H | p-Me | 3c | 83 |
| 4 | H | H | 3d | 72 |
| 5 | 5-Cl | p-MeO | 3e | 70 |
| 6 | 5-Br | p-MeO | 3f | 72 |
| 7 | 3-Br-5-Cl | p-MeO | 3g | 62 |
| 8 | H | o-Cl | 3h | 61 |
| 9 | H | m-Cl | 3i | 67 |
| 10 | H | p-Cl | 3j | 86 |
| 11 | H | p-Br | 3k | 73 |
| 12 | H | p-I | 3l | 60 |
| 13 | 5-F | p-Cl | 3m | 69 |
| 14 | 5-Cl | p-Cl | 3n | 76 |
| 15 | 5-Br | p-Cl | 3o | 77 |
| 16 | 5-NO2 | p-Cl | 3p | 68 |
| 17c | H | Furan-2- | 3q | 78 |
| 18d | H | Thiophen-2- | 3r | 73 |
| 19e | H | Thiophen-3- | 3s | 71 |
| 20 | H | 3,4-OCH2CH2O | 3t | 64 |
| 21 | 3-Br-5-Cl | m-MeO | 3u | 66 |
Moreover, multisubstituted β-nitrostyrene also reacted under the conditions to afford the corresponding products in moderate yields (Table 2, entry 20). Similarly, substituted salicylic aldehydes bearing an electron-donating group on the aromatic ring were superior for the imine formation/addition reaction than those bearing an electron-withdrawing group (Table 2, entries 2, 5–7, 14–16). Unfortunately, when the salicylic aldehydes were replaced by the common aromatic aldehydes, the reactions were dramatically blocked and the desired products were not yielded. Because the solubility of compound 3p is very low in all organic solvents, just its 1H NMR was obtained, a clear 13C NMR was not gotten. Except compound 3p, other corresponding 2,4,6-trisubstituted pyridines were analyzed by their 1H NMR, 13C NMR and MS. Characteristic 1H chemical shift of Ar-OH, pyridine C(3)–H and C(5)–H at δ ca. 15.0 (s), 7.90 (s) and 7.70 (s), unequivocally indicated the exclusive chemical environment of hydroxyl and pyridine protons. Product 2,4,6-trisubstituted pyridines 3b and 3d were further characterized by single X-ray crystallography (Fig. 2).26
Moreover, for the products 3b and 3d, an intermolecular hydrogen bond was observed on the basis of its X-ray crystallographic data. See ESI† for the bond length of the intermolecular hydrogen bond of the products 3b and 3d. Additionally, the existence of the intermolecular hydrogen bond was further confirmed by the proton-NMR of the ArOH proton. In CDCl3 or DMSO-d6, the OH proton, hydrogen bonded with nitrogen of the pyridine moiety, resonates at ca. 15.0 ppm as a single peak, not as a common broad single peak, this was because the intramolecular hydrogen bond caused difficulties of exchanging the Ar-OH proton with deuterated solvents.
The mechanism proposed for the reaction is illustrated in Scheme 1. Firstly, in the presence of weak basic potassium carbonate, the reaction of salicylic aldehyde and ammonium acetate generated a stable intermediate 2-(iminomethyl)phenol [A], which has an intramolecular hydrogen bond. The formation of the intramolecular hydrogen bond not only stabilizes the structure of imine but also strengthens the basicity and nucleophilicity of imine. Nucleophilic addition of intermediate [A] to β-nitrostyrene afforded 1,4-dipole intermediate [B]. The intermolecular [4 + 2] cyclization of 1,4-dipole intermediate [B] with β-nitrostyrene gave the intermediate hexahydropyridine [C]. Finally, removal of two nitrous acids mediated by potassium carbonate and oxidative aromatization of the intermediate hexahydropyridine [C] produced the 2,4,6-triarylpyridine. Some control experiments were tested for our proposed mechanism. Under the optimized conditions, the model reaction of salicylic aldehyde, 1-chloro-4-(2-nitrovinyl)benzene and ammonium acetate was carried out in the optimized reaction conditions for 3 h aimed at yielding some key intermediates and supporting the mechanistic proposal, but the reaction system was very complicated after workup, the desired intermediates were not isolated by flash chromatography. After workup, the solid mixture was measured by high-resolution ESI mass spectra to afford a stronger ion peak: 392.0604 (m/z), which corresponded to the mass of the molecular ion (M + 1) for the final product 2-(4,6-bis(4-chlorophenyl)pyridin-2-yl)phenol (see ESI†). Additionally, the two mass of the molecular ion (M + 1): 484.4720 and 437.1934, should belong to 2-(4,6-bis(4-chlorophenyl)-3,5-dinitro-1,2-dihydropyridin-2-yl)phenol from the desired inter>mediate 2-(4,6-bis(4-chlorophenyl)-3,5-dinitropiperidin-2-yl)phenol and 2-(4,6-bis(4-chlorophenyl)-3-nitropyridin-2-yl)phenol from the desired intermediate 2-(4,6-bis(4-chlorophenyl)-3-nitro-1,2,3,6-tetrahydropyridin-2-yl)phenol. The results indicated there was a path that leads to the final product, the intermolecular cyclization gave the desired intermediate dinitropiperidines, next, HNO2 was removed by K2CO3 to give the desired intermediate nitrotetrahydropyridines, another HNO2 was removed by K2CO3 to give the desired intermediate dihydropyridines again, the final aromatization yielded the products by air.
Conclusions
In summary, we have developed a protocol for the synthesis of polysubstituted pyridines from the β-nitrostyrenes, available substituted salicylic aldehydes and ammonium acetate. The present strategy features high chemoselectivity and excellent tolerance for a broad range of functional groups, in which, the β-nitrostyrenes generated from aldehydes and nitromethane, substituted salicylic aldehydes and ammonium acetate were respectively employed as simple and easily available substrates. Moreover, the method also opens a new entry to β-nitrostyrene based N-heterocycle synthesis based on basic systems.Experimental section
All melting points were determined in a Yanaco melting point apparatus and are uncorrected. IR spectra were recorded in a Nicolet FT-IR 5DX spectrometer. The 1H NMR (400 or 600 MHz) and 13C NMR (100 or 150 MHz) spectra were recorded in a Bruker AV-400 or 600 spectrometer with TMS as internal reference in CDCl3 or DMSO-d6 solutions. The J values are given in hertz. Only discrete or characteristic signals for the 1H NMR are reported. High-resolution ESI mass spectra were obtained on a UHR-TOF maXis (ESI) mass spectrometer. X-ray crystallographic analysis was performed with a SMART APEX-II diffractometer. Flash chromatography was performed on silica gel (230–400 mesh) eluting with ethyl acetate–hexanes mixture. All reactions were monitored by thin layer chromatography (TLC). All reagents and solvents were purchased from commercial sources and purified commonly before used.The mixture of substituted salicylic aldehydes (1.0 mmol) and ammonium acetate (85 mg, 1.1 mmol) in the solution of DMF and EtOH (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 5 mL) was stirred at room temperature for 30 min. To the mixture was appropriate β-nitrostyrenes (2.05 mmol) and potassium carbonate (138.2 mg, 1.0 mmol) at room temperature. The resultant mixture was stirred at 140 °C for 5–8 h, and the completion of reaction was confirmed by TLC (hexanes/EtOAc, 10/1). Subsequently, the solvents were removed by vacuum, the residues was extracted with dichloromethane (15 mL × 2). The organic phase was washed with water (10 mL) and brine (15 mL), and dried over anhydrate sodium sulfate. After removal of dichloromethane, the crude product was purified by flash chromatography (silica gel, EtOAc/hexanes, 1/20) to give the desirable products 3a–u.
1, 5 mL) was stirred at room temperature for 30 min. To the mixture was appropriate β-nitrostyrenes (2.05 mmol) and potassium carbonate (138.2 mg, 1.0 mmol) at room temperature. The resultant mixture was stirred at 140 °C for 5–8 h, and the completion of reaction was confirmed by TLC (hexanes/EtOAc, 10/1). Subsequently, the solvents were removed by vacuum, the residues was extracted with dichloromethane (15 mL × 2). The organic phase was washed with water (10 mL) and brine (15 mL), and dried over anhydrate sodium sulfate. After removal of dichloromethane, the crude product was purified by flash chromatography (silica gel, EtOAc/hexanes, 1/20) to give the desirable products 3a–u.
2-(4,6-Bis(4-methoxyphenyl)pyridin-2-yl)phenol (3a)
Yellow solid; mp: 123.2–123.7 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm):14.94 (s, 1H), 7.89–7.86 (m, 4H), 7.67 (s, 1H), 7.63 (d, J = 8.4 Hz, 2H), 7.27 (dd, J = 7.8, 7.2 Hz, 1H), 6.99 (d, J = 8.4 Hz, 3H), 6.98 (d, J = 8.4 Hz, 2H), 6.88 (dd, J = 7.8, 7.2 Hz, 1H), 3.83 (s, 3H), 3.82 (s, 3H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 159.9, 159.8, 159.2, 156.8, 153.8, 149.6, 130.4, 129.9, 129.7, 127.4, 127.3, 125.3, 118.2, 117.7, 117.5, 115.0, 113.6, 113.5, 113.4, 54.4, 54.3; IR (KBr, cm−1) ν: 3435, 2931, 2836, 1748, 1604, 1514, 874, 823; HRMS (ESI) m/z [M + H]+ calcd for C25H22NO3: 384.1594; found: 384.1610.2-(4,6-Diphenylpyridin-2-yl)-6-ethoxyphenol (3b)
Yellow solid; mp: 158.3–158.9 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm):15.18 (s, 1H), 7.95 (s, 1H), 7.94 (d, J = 7.2 Hz, 2H), 7.75 (s, 1H), 7.64 (d, J = 6.6 Hz, 2H), 7.47–7.37 (m, 7H), 6.67 (d, J = 8.0 Hz, 1H), 6.78 (dd, J = 8.4, 7.8 Hz, 1H), 4.09 (q, J = 7.2 Hz, 3H), 1.45 (t, J = 7.2 Hz, 3H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 157.2, 145.1, 150.2, 149.7, 147.7, 137.5, 136.5, 128.6, 128.4, 128.2, 126.2, 126.0, 118.0, 125.4, 117.1, 116.9, 116.1, 115.0, 113.1, 63.3, 13.9; IR (KBr, cm−1) ν: 3447, 2880, 1750, 1606, 1541, 1492, 1428, 869, 817, 765; HRMS (ESI) m/z [M + H]+ calcd for C25H22NO2: 368.1645; found: 368.1658.2-(4,6-Di-p-tolylpyridin-2-yl)phenol (3c)
Yellow solid; mp: 160.1–160.8 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm): 14.85 (s, 1H), 7.90 (s, 1H), 7.83 (d, J = 7.8 Hz, 1H), 7.79 (d, J = 7.8 Hz, 2H), 7.69 (s, 1H), 7.54 (d, J = 7.8 Hz, 2H), 7.25–7.22 (m, 5H), 6.98 (d, J = 8.4 Hz, 1H), 6.85 (dd, J = 7.2, 7.8 Hz, 1H), 2.35 (s, 3H), 2.34 (s, 3H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 159.2, 156.9, 154.1, 150.0, 138.7, 138.5, 134.6, 134.3, 130.5, 128.9, 128.8, 126.0, 125.8, 125.3, 118.1, 117.7, 117.5, 115.6, 114.2, 20.3, 20.3; IR (KBr, cm−1) ν: 3444, 2926, 2838, 1750, 1604, 1540, 1509, 1434, 828, 748; HRMS (ESI) m/z [M + H]+ calcd for C25H22NO: 352.1696; found: 352.1710.2-(4,6-Diphenylpyridin-2-yl)phenol (3d)
Yellow solid; mp: 163.1–163.3 °C (PE/EA); 1H-NMR (CDCl3, 400 MHz) δ (ppm):14.71 (s, 1H), 7.98 (s, 1H), 7.93 (d, J = 8.0 Hz, 2H), 7.87 (d, J = 8.0 Hz, 1H), 7.77 (s, 1H), 7.68 (d, J = 8.0 Hz, 2H), 7.50–7.40 (m, 6H), 7.27 (dd, J = 7.2, 8.0 Hz, 1H), 7.00 (d, J = 8.0 Hz, 1H), 6.88 (dd, J = 7.6, 7.6 Hz, 1H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 159.1, 157.1, 154.3, 150.2, 137.5, 137.1, 130.6, 128.6, 128.4, 128.2, 128.1, 126.2, 125.9, 125.4, 118.0, 117.8, 117.6, 116.2, 114.7; IR (KBr, cm−1) ν: 3451, 1750, 1604, 1540, 1508, 1431, 864, 810, 748; HRMS (ESI) m/z [M + H]+ calcd for C23H18NO: 324.1383; found: 324.1396.2-(4,6-Bis(4-methoxyphenyl)pyridin-2-yl)-4-chlorophenol (3e)
Yellow solid; mp: 152.3–153.0 °C (PE/EA); 1H-NMR (DMSO-d6, 600 MHz) δ (ppm): 14.78 (s, 1H), 8.42 (d, J = 2.4 Hz, 1H), 8.41 (s, 1H), 8.16 (s, 1H), 8.12 (d, J = 8.4 Hz, 2H), 8.07 (d, J = 8.4 Hz, 2H), 7.37 (dd, J1 = 9.0 Hz, J2 = 3.0 Hz, 1H), 7.15 (d, J = 8.4 Hz, 2H), 7.13 (d, J = 8.4 Hz, 2H), 6.98 (d, J = 9.0 Hz, 1H), 3.87 (s, 3H), 3.86 (s, 3H); 13C-NMR (DMSO-d6, 100 MHz) δ (ppm): 160.1, 160.0, 157.7, 155.3, 153.8, 150.1, 130.2, 129.4, 129.2, 127.5, 127.3, 124.9, 122.5, 119.0, 119.0, 115.5, 113.7, 113.6, 113.5, 54.5, 54.4; IR (KBr, cm−1) ν: 3442, 2925, 2839, 1746, 1602, 1524, 1477, 1443, 875, 820; HRMS (ESI) m/z [M + H]+ calcd for C25H21ClNO3: 418.1204; found: 418.1209.2-(4,6-Bis(4-methoxyphenyl)pyridin-2-yl)-4-bromophenol (3f)
Yellow solid; mp: 168.2–168.6 °C (PE/EA); 1H-NMR (CDCl3, 400 MHz) δ (ppm): 14.99 (s, 1H), 7.94 (s, 1H), 7.85 (d, J = 8.4 Hz, 2H), 7.81 (s, 1H), 7.70 (s, 1H), 7.63 (d, J = 8.4 Hz, 2H), 7.33 (d, J = 8.8 Hz, 1H), 7.00 (d, J = 10.0 Hz, 2H), 6.98 (d, J = 9.6 Hz, 2H), 6.87 (d, J = 8.8 Hz, 1H), 3.83 (s, 3H), 3.82 (s, 3H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 160.0, 159.9, 158.3, 155.4, 153.9, 149.8, 132.9, 129.4, 129.4, 127.8, 127.4, 127.2, 119.8, 119.4, 115.4, 113.7, 113.5, 113.4, 109.4, 54.4, 54.4; IR (KBr, cm−1) ν: 3435, 2922, 2836, 1747, 1606, 1540, 1450, 1436, 820, 751; HRMS (ESI) m/z [M + H]+ calcd for C25H21BrNO3: 462.0699; found: 462.0705.2-(4,6-Bis(4-methoxyphenyl)pyridin-2-yl)-6-bromo-4-chlorophenol (3g)
Yellow solid; mp: 228.7–229.6 °C (PE/EA); 1H-NMR (DMSO-d6, 400 MHz) δ (ppm): 16.56 (s, 1H), 8.50 (s, 1H), 8.46 (s, 1H), 8.19 (s, 1H), 8.12 (d, J = 8.0 Hz, 2H), 8.04 (d, J = 8.0 Hz, 2H), 7.73 (s, 1H), 7.16 (d, J = 8.4 Hz, 2H), 3.84 (s, 6H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 160.1, 160.0, 154.9, 154.4, 153.7, 150.1, 132.8, 129.0, 128.6, 127.4, 127.3, 124.1, 122.3, 119.5, 116.8, 114.7, 113.6, 113.5, 111.9, 54.5, 54.4; IR (KBr, cm−1) ν: 3442, 2926, 2866, 1738, 1598, 1544, 1506, 1446, 875, 812, 753; HRMS (ESI) m/z [M + H]+ calcd for C25H20BrClNO3: 496.0310; found: 496.0303.2-(4,6-Bis(4-chlorophenyl)pyridin-2-yl)phenol (3h)
Yellow solid; mp: 187.1–187.6 °C (PE/EA); 1H-NMR (DMSO-d6, 400 MHz) δ (ppm): 13.90 (s, 1H), 8.42 (s, 1H), 8.26 (d, J = 7.6 Hz, 1H), 8.22 (s, 1H), 8.13 (d, J = 8.4 Hz, 2H), 8.11 (d, J = 8.4 Hz, 2H), 7.64 (d, J = 8.8 Hz, 2H), 7.62 (d, J = 8.8 Hz, 2H), 7.33 (dd, J = 7.2, 7.2 Hz, 1H), 6.97–6.74 (m, 2H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 159.0, 157.4, 153.3, 149.2, 135.7, 135.4, 135.0, 134.9, 130.9, 128.5, 128.4, 127.5, 127.2, 125.4, 118.0, 117.8, 117.6, 115.8, 114.8; IR (KBr, cm−1) ν: 3435, 1747, 1603, 1536, 1493, 1434, 819, 747; HRMS (ESI) m/z [M + H]+ calcd for C23H16Cl2NO: 392.0603; found: 392.0605.2-(4,6-Bis(2-chlorophenyl)pyridin-2-yl)phenol (3i)
Yellow solid; mp: 154.6–155.2 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm): 14.09 (s, 1H), 7.93 (s, 1H), 7.80 (d, J = 7.8 Hz, 1H), 7.61 (dd, J1 = 7.8, J2 = 2.4 Hz, 1H), 7.60 (s, 1H), 7.49–7.47 (m, 1H), 7.46 (dd, J1 = 7.8, J2 = 2.4 Hz, 1H), 7.39–7.38 (m, 1H), 7.35–7.30 (m, 4H), 7.25 (dd, J = 7.8, 7.8 Hz, 1H), 6.96 (d, J = 7.8 Hz, 1H), 6.86 (dd, J = 7.8, 7.8 Hz, 1H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 160.0, 157.6, 153.6, 148.9, 137.8, 137.7, 132.3, 132.3, 131.7, 131.3, 130.9, 130.5, 130.4, 130.2, 130.1, 127.3, 127.3, 126.5, 123.5, 119.0, 118.9, 118.7, 118.6; IR (KBr, cm−1) ν: 3444, 1747, 1602, 1544, 1490, 860, 784, 741; HRMS (ESI) m/z [M + H]+ calcd for C23H16Cl2NO: 392.0603; found: 392.0604.2-(4,6-Bis(3-chlorophenyl)pyridin-2-yl)phenol (3j)
Yellow solid; mp: 189.2–190.2 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm):14.23 (s, 1H), 7.94 (s, 1H), 7.85 (s, 1H), 7.84 (d, J = 8.4 Hz, 1H), 7.80 (d, J = 3.6 Hz, 1H), 7.67 (s, 1H), 7.63 (s, 1H), 7.54 (s, 1H), 7.41 (d, J = 4.2 Hz, 2H), 7.39 (d, J = 4.2 Hz, 2H), 7.28 (dd, J = 7.8, 7.2 Hz, 1H), 7.00 (d, J = 7.8 Hz, 1H), 6.89 (dd, J = 7.2, 7.8 Hz, 1H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 158.9, 157.5, 153.2, 149.0, 139.1, 138.8, 134.3, 134.2, 131.0, 129.5, 129.4, 128.8, 128.5, 126.4, 126.1, 125.5, 124.4, 124.2, 118.0, 117.7, 117.6, 116.2, 115.3; IR (KBr, cm−1) ν: 3434, 1748, 1605, 1519, 1448, 821, 726; HRMS (ESI) m/z [M + H]+ calcd for C23H16Cl2NO: 392.0603; found: 392.0604.2-(4,6-Bis(4-bromophenyl)pyridin-2-yl)phenol (3k)
Yellow solid; mp: 198.6–199.0 °C (PE/EA); 1H-NMR (CDCl3, 400 MHz) δ (ppm): 14.35 (s, 1H), 7.94 (s, 1H), 7.84 (d, J = 7.6 Hz, 3H), 7.68 (s, 1H), 7.60 (d, J = 8.0 Hz, 2H), 7.46 (d, J = 8.0 Hz, 2H), 7.44 (d, J = 8.4 Hz, 2H), 7.29 (dd, J = 7.2, 8.0 Hz, 1H), 7.00 (d, J = 8.4 Hz, 1H), 6.89 (dd, J = 7.2, 7.6 Hz, 1H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 159.0, 157.4, 153.4, 149.2, 135.8, 135.4, 135.0, 134.9, 130.9, 128.5, 128.4, 127.5, 127.2, 125.4, 118.0, 117.8, 117.6, 115.8, 114.8; IR (KBr, cm−1) ν: 3447, 1743, 1601, 1545, 1443, 875, 830, 767; HRMS (ESI) m/z [M + H]+ calcd for C23H16Br2NO: 479.9593; found: 479.9593.2-(4,6-Bis(4-iodophenyl)pyridin-2-yl)phenol (3l)
Yellow solid; mp: 239.1–239.4 °C (PE/EA); 1H-NMR (DMSO-d6, 600 MHz) δ (ppm):13.96 (s, 1H), 8.28 (d, J = 7.2 Hz, 1H), 8.22 (s, 1H), 7.99–7.90 (m, 8H), 7.62 (d, J = 8.8 Hz, 2H), 7.36 (dd, J = 7.8, 7.8 Hz, 1H), 6.99–6.97 (m, 2H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 158.9, 157.4, 153.5, 149.3, 137.4, 137.3, 136.7, 136.4, 130.9, 127.9, 127.6, 125.4, 118.0, 117.7, 117.6, 115.5, 114.8, 95.2, 94.8; IR (KBr, cm−1) ν: 3458, 1741, 1598, 1531, 1426, 819, 752; HRMS (ESI) m/z [M + H]+ calcd for C23H16I2NO: 575.9317; found: 575.9326.2-(4,6-Bis(4-chlorophenyl)pyridin-2-yl)-4-fluorophenol (3m)
Yellow solid; mp: 186.8–187.0 °C (PE/EA); 1H-NMR (DMSO-d6, 600 MHz) δ (ppm): 13.72 (s, 1H), 8.50 (s, 1H), 8.29 (s, 1H), 8.21 (dd, J1 = 10.8 Hz, J2 = 3.0 Hz, 1H), 8.18–8.16 (m, 4H), 7.68 (d, J = 8.4 Hz, 2H), 7.66 (d, J = 9.0 Hz, 2H), 7.22 (td, J1 = 9.0 Hz, J2 = 2.4 Hz, 1H), 6.99 (dd, J1 = 9.0 Hz, J2 = 4.8 Hz, 1H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 157.2 (d, J = 2.7 Hz), 155.9 (d, J = 1.5 Hz), 155.8 (d, J = 235 Hz), 154.4, 150.6, 136.3, 136.3, 136.1, 136.0, 129.6, 129.4, 128.5, 128.2, 119.6 (d, J = 30.8 Hz), 119.0, 118.7, 117.2, 115.9, 112.1 (d, J = 52.4 Hz); IR (KBr, cm−1) ν: 3441, 1749, 1606, 1543, 1486, 825, 736; HRMS (ESI) m/z [M + H]+ calcd for C23H15Cl2FNO: 410.0509; found: 410.0508.2-(4,6-Bis(4-chlorophenyl)pyridin-2-yl)-4-chlorophenol (3n)
Yellow solid; mp: 185.7–186.2 °C (PE/EA); 1H-NMR (DMSO-d6, 400 MHz) δ (ppm): 14.04 (s, 1H), 8.58 (s, 1H), 8.45 (s, 1H), 8.35 (s, 1H), 8.24 (d, J = 8.0 Hz, 2H), 8.22 (d, J = 8.0 Hz, 2H), 7.73 (d, J = 8.4 Hz, 2H), 7.71 (d, J = 8.4 Hz, 2H), 7.45 (d, J = 8.8 Hz, 1H), 7.07 (d, J = 8.8 Hz, 1H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 157.5, 155.9, 153.4, 149.5, 135.2, 135.2, 135.0, 134.9, 130.6, 128.6, 128.4, 127.5, 127.2, 124.9, 122.7, 119.0, 118.6, 116.3, 114.8; IR (KBr, cm−1) ν: 3436, 1746, 1601, 1540, 1487, 1434, 879, 823, 733; HRMS (ESI) m/z [M + H]+ calcd for C23H15Cl3NO: 426.0214; found: 426.0207.2-(4,6-Bis(4-chlorophenyl)pyridin-2-yl)-4-bromophenol (3o)
Yellow solid; mp: 195.3–195.7 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm): 14.43 (s, 1H), 7.90 (s, 1H), 7.84 (s, 1H), 7.75 (d, J = 8.4 Hz, 2H), 7.69 (s, 1H), 7.59 (d, J = 8.4 Hz, 2H), 7.46 (d, J = 8.4 Hz, 2H), 7.42 (d, J = 8.4 Hz, 2H), 7.34 (d, J = 8.8 Hz, 1H), 6.87 (d, J = 8.4 Hz, 1H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 158.1, 156.0, 153.5, 149.5, 135.4, 135.2, 135.1, 133.4, 128.6, 128.4, 127.9, 127.8, 127.5, 127.2, 119.5, 119.4, 116.3, 114.8, 109.8; IR (KBr, cm−1) ν: 3433, 1749, 1602, 1539, 1488, 1433, 873, 818, 730; HRMS (ESI) m/z [M + H]+ calcd for C23H15BrCl2NO: 469.9709; found: 469.9710.2-(4,6-Bis(4-chlorophenyl)pyridin-2-yl)-4-nitrophenol (3p)
Yellow solid; mp: 284.3–284.7 °C (PE/EA); 1H-NMR (DMSO-d6, 600 MHz) δ (ppm): 15.02 (s, 1H), 9.15 (s, 1H), 8.62 (s, 1H), 8.35 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 8.20 (d, J = 8.4 Hz, 2H), 8.18 (d, J = 7.8 Hz, 2H), 7.69 (d, J = 10.2 Hz, 2H), 7.68 (d, J = 8.4 Hz, 2H), 7.18 (d, J = 9.0 Hz, 1H); IR (KBr, cm−1) ν: 3441, 1747, 1603, 1540, 1484, 1430, 880, 848, 745; HRMS (ESI) m/z [M + H]+ calcd for C19H14NO3: 437.0454; found: 437.0441.3′,5′-Di(furan-2-yl)-[1,1′-biphenyl]-2-ol (3q)
Yellow solid; mp: 141.5–141.7 °C (PE/EA); 1H-NMR (CDCl3, 600 MHz) δ (ppm): 14.44 (s, 1H), 7.93 (s, 1H), 7.83 (d, J = 7.8 Hz, 1H), 7.75 (s, 1H), 7.53 (d, J = 12.6, 2H), 7.26 (dd, J = 8.4, 7.2 Hz, 1H), 7.02 (d, J = 3.6 Hz, 1H), 6.98 (d, J = 7.8 Hz, 1H), 6.95 (d, J = 3.6 Hz, 1H), 6.87 (dd, J = 7.8, 7.2 Hz, 1H), 6.51 (s, 2H); 13C-NMR (CDCl3, 150 MHz) δ (ppm): 159.1, 156.9, 151.0, 150.2, 145.3, 143.2, 142.9, 138.7, 130.6, 125.3, 117.8, 117.6, 111.3, 111.2, 110.1, 109.5, 108.6, 108.5; IR (KBr, cm−1) ν: 3437, 3117, 2689, 1669, 1588, 1447, 1295, 1194, 1081, 1011, 902, 833, 678; HRMS (ESI) m/z [M + H]+ calcd for C19H14NO3: 304.0968; found: 304.0976.3′,5′-Di(thiophen-2-yl)-[1,1′-biphenyl]-2-ol (3r)
Yellow solid; mp: 180.6–180.9 °C (PE/EA); 1H-NMR (DMSO-d6, 400 MHz) δ (ppm): 13.31 (s, 1H), 8.20 (s, 1H), 8.16 (d, J = 7.6 Hz, 1H), 8.12 (s, 1H), 8.10 (s, 1H), 8.06 (s, 1H), 7.82 (s, 1H), 7.75 (s, 1H), 7.33 (dd, J = 7.2, 7.6 Hz, 1H), 7.28–7.25 (m, 2H), 6.96–6.94 (m, 2H); 13C-NMR (DMSO-d6, 100 MHz) δ (ppm): 159.1, 157.6, 150.4, 144.2, 142.7, 140.4, 132.1, 129.7, 129.5, 129.4, 128.4, 128.3, 127.5, 119.6, 119.5, 118.2, 114.7, 113.4; IR (KBr, cm−1) ν: 3441, 1748, 1599, 1541, 831, 753; HRMS (ESI) m/z [M + H]+ calcd for C19H14NOS2: 336.0511; found: 336.0504.3′,5′-Di(thiophen-3-yl)-[1,1′-biphenyl]-2-ol (3s)
Yellow solid; mp: 176.2–176.8 °C (PE/EA); 1H-NMR (DMSO-d6, 400 MHz) δ (ppm): 14.54 (s, 1H), 8.53 (s, 1H), 8.37 (s, 1H), 8.29–8.21 (m, 3H), 7.99 (s, 1H), 7.76 (s, 3H), 7.32 (s, 1H), 6.94–6.93 (m, 2H); 13C-NMR (DMSO-d6, 100 MHz) δ (ppm): 159.8, 157.8, 150.9, 145.4, 140.6, 139.2, 131.9, 128.4, 128.1, 128.0, 127.1, 126.4, 126.2, 125.6, 119.6, 119.2, 118.3, 116.3, 115.0; IR (KBr, cm−1) ν: 3446, 1748, 1604, 1549, 1439, 851, 788, 754; HRMS (ESI) m/z [M + H]+ calcd for C19H14NOS2: 336.0511; found: 336.0507.2-(4,6-Bis(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)pyridin-2-yl)phenol (3t)
Yellow solid; mp: 143.8–144.2 °C (PE/EA); 1H-NMR (DMSO-d6, 400 MHz) δ (ppm): 14.57 (s, 1H), 8.23 (d, J = 6.0 Hz, 1H), 8.22 (s, 1H), 7.99 (s, 1H), 7.62 (s, 1H), 7.58 (s, 1H), 7.56 (d, J = 8.0 Hz, 1H), 7.53 (d, J = 8.4 Hz, 1H), 7.30 (dd, J = 7.2, 8.0 Hz, 2H), 7.00 (d, J = 9.6 Hz, 1H), 6.98 (d, J = 8.8 Hz, 1H), 6.92–6.90 (m, 2H), 4.29 (s, 8H); 13C-NMR (DMSO-d6, 100 MHz) δ (ppm): 159.7, 157.5, 154.2, 150.2, 145.4, 145.4, 144.3, 144.3, 131.8, 131.3, 130.6, 128.0, 121.0, 120.3, 119.5, 119.2, 118.2, 118.1, 118.0, 116.7, 116.2, 115.9, 115.2, 64.8, 64.2, 64.5; IR (KBr, cm−1) ν: 3442, 2926, 2866, 1738, 1598, 1544, 1506, 1448, 875, 812, 753; HRMS (ESI) m/z [M + H]+ calcd for C27H27NO5: 440.1492; found: 440.1495.2-(4,6-Bis(3-methoxyphenyl)pyridin-2-yl)-6-bromo-4-chlorophenol (3u)
Yellow solid; mp: 186.9–187.3 °C (PE/EA); 1H-NMR (DMSO-d6, 600 MHz) δ (ppm): 16.27 (s, 1H), 8.59 (s, 1H), 8.56 (s, 1H), 8.31 (s, 1H), 7.79 (s, 1H), 7.69 (d, J = 7.2 Hz, 1H), 7.67 (d, J = 7.2 Hz, 1H), 7.67 (s, 1H), 7.64 (s, 1H), 7.56 (dd, J = 7.8, 7.8 Hz, 1H), 7.51 (dd, J = 7.8, 8.4 Hz, 1H), 7.16 (d, J = 8.4 Hz, 1H), 7.14 (d, J = 7.8 Hz, 1H), 3.91 (s, 3H), 3.89 (s, 3H); 13C-NMR (CDCl3, 100 MHz) δ (ppm): 159.2, 159.1, 154.8, 154.7, 154.3, 150.8, 138.3, 137.8, 133.1, 129.4, 129.3, 124.2, 122.4, 119.4, 118.6, 118.5, 117.6, 115.1, 114.3, 113.7, 112.2, 111.9, 111.7, 54.5, 54.5; IR (KBr, cm−1) ν: 3433, 2925, 2841, 1750, 1593, 1538, 1455, 821, 724; HRMS (ESI) m/z [M + H]+ calcd for C25H20BrClNO3: 496.0310; found: 496.0311.Acknowledgements
Financial support of this research by the National Natural Science Foundation of China (NSFC 21173181) is gratefully acknowledged by authors. A Project was funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.Notes and references
- (a) J. P. Michael, Nat. Prod. Rep., 2005, 22, 627 RSC; (b) S. K. Srivastava, R. P. Tripathi and R. J. Ramachandran, Biol. Chem., 2005, 280, 30273 CrossRef CAS PubMed; (c) M. Abass, Heterocycles, 2005, 65, 901 CrossRef CAS; (d) V. C. Gibson, C. Redshaw and G. A. Solan, Chem. Rev., 2007, 107, 1745 CrossRef CAS PubMed.
- (a) D. F. Fisher and R. Sarpong, J. Am. Chem. Soc., 2010, 132, 5926 CrossRef PubMed; (b) C.-X. Yuan, C.-T. Chang, A. Axelrod and D. Siegel, J. Am. Chem. Soc., 2010, 132, 5924 CrossRef CAS PubMed; (c) S. D. Roughley and A. M. Jordan, J. Med. Chem., 2011, 54, 3451 CrossRef CAS PubMed; (d) T. P. Whitehead, C. Havel, C. Metayer, N. L. Benowitz and P. Jacob, Chem. Res. Toxicol., 2015, 28, 1007 CrossRef CAS PubMed.
- S. Kang, B. Song, J. Wu, M. He, D. Hu, L. Jin, S. Zeng, W. Xue and S. Yang, Eur. J. Med. Chem., 2013, 67, 14 CrossRef CAS PubMed.
- (a) Y. Shirota, J. Mater. Chem., 2005, 15, 75 RSC; (b) F. Ono, Y. Ishikura, Y. Tada, M. Endo and T. Sato, Synlett, 2008, 15, 2365 Search PubMed.
- (a) R. Makiura, S. Motoyama, Y. Umemura, H. Yamanaka, O. Sakata and H. Kitagawa, Nat. Mater., 2010, 9, 565 CrossRef CAS PubMed; (b) B. Wang, H. Yang, Y.-B. Xie, Y.-B. Dou, M.-J. Zhao and J.-R. Li, Chin. Chem. Lett., 2016, 27, 502 CrossRef CAS.
- (a) Z. A. E. Waller, P. S. Shirude, R. Rodriguez and S. Balasubramanian, Chem. Commun., 2008, 44, 1467 RSC; (b) A. Hernández-Campos, I. Velázquez-Martínez, R. Castillo, F. López-Vallejo, P. Jia, Y. Yu, M. A. Giulianotti and J. L. Medina-Franco, Chem. Biol. Drug Des., 2010, 76, 269 Search PubMed; (c) N. M. Smith, G. Labrunie, B. Corry, P. L. T. Tran, M. Norret, M. Djavaheri-Mergny, C. L. Raston and J. L. Mergny, Org. Biomol. Chem., 2011, 9, 6154 RSC; (d) B. Corry and N. M. Smith, Chem. Commun., 2012, 48, 8958 RSC; (e) R. Karki, P. Thapa, H. Y. Yoo, T. M. Kadayat, P. H. Park, Y. Na, E. Lee, K. H. Jeon, W. J. Cho, H. Choi, Y. Kown and E. S. Lee, Eur. J. Med. Chem., 2012, 49, 219 CrossRef CAS PubMed; (f) H.-B. Kwon, C. Park, K.-H. Jeon, E. Lee, S.-E. Park, K.-Y. Jun, T. M. Kadayat, P. Thapa, R. Karki, Y. Na, M. S. Park, S. Bae Rho, E.-S. Lee and Y. Kwon, J. Med. Chem., 2015, 58, 1100 CrossRef CAS PubMed.
- (a) G. W. V. Cave, M. J. Hardie, B. A. Roberts and C. L. Raston, Eur. J. Org. Chem., 2001, 3227 CrossRef CAS; (b) R. K. R. Jetti, A. Nagia, F. Xue and T. C. W. Mak, Chem. Commun., 2001, 37, 919 RSC; (c) U. S. Schubert and C. Eschbaumer, Angew. Chem., Int. Ed., 2002, 41, 2892 CrossRef CAS; (d) H. Hofmeier and U. S. Schubert, Chem. Commun., 2005, 41, 2423 RSC; (e) T. Smejkal and B. Breit, Angew. Chem., Int. Ed., 2008, 47, 311 CrossRef CAS PubMed; (f) D. Wu, L. Zhi, G. J. Bodwell, G. Cui, N. Tsao and K. Mllen, Angew. Chem., 2007, 119, 5513 CrossRef; (g) Y.-M. Zhang, M. Han, H.-Z. Chen, Y. Zhang and Y. Liu, Org. Lett., 2013, 15, 124 CrossRef CAS PubMed; (h) M. D. Wise, A. Ruggi, M. Pascu, R. Scopelliti and K. Severin, Chem. Sci., 2013, 4, 1658 RSC.
- (a) D. Bora, B. Deb, A. L. Fuller, A. M. Z. Slawin, J. DerekWoollins and D. K. Dutta, Inorg. Chim. Acta, 2010, 363, 1539 CrossRef CAS; (b) Y.-T. Chan, C. N. Moorefield, M. Soler and G. R. Newkome, Chem.–Eur. J., 2010, 16, 1768 CrossRef CAS PubMed; (c) E. Belhadj, A. El-Ghayoury, M. Mazari and M. Salle, Tetrahedron Lett., 2013, 54, 3051 CrossRef CAS; (d) S. Lin and X. Lu, Org. Lett., 2010, 12, 2536 CrossRef CAS PubMed; (e) P. Fang, M. R. Chaulagain and Z. D. Aron, Org. Lett., 2012, 14, 2130 CrossRef CAS PubMed.
- K. A. Leonard, M. I. Nelen, T. P. Simard, S. R. S. O. Darvies, A. R. Gollnickoseroff, S. L. Gibson, R. Hilf, L. B. Chen and M. R. Detty, J. Med. Chem., 1999, 42, 3593 Search PubMed.
- (a) R. L. Frank and R. P. Seven, J. Am. Chem. Soc., 1949, 71, 2629 CrossRef CAS; (b) S. Tu, T. Li, F. Shi, Q. Wang, J. Zhang, J. Xu, X. Zhu, X. Zhang, S. Zhu and D. Shi, Synthesis, 2005, 3045 CrossRef CAS; (c) M. M. Heravi, K. Bakhtiari, Z. Daroogheha and F. F. Bamoharram, Catal. Commun., 2007, 8, 1991 CrossRef CAS; (d) P. Wang, R. Hua and C.-J. Li, Curr. Org. Synth., 2013, 10, 655 CrossRef CAS.
- (a) N. Montazeri and S. Mahjoob, Chin. Chem. Lett., 2012, 23, 419 CrossRef CAS; (b) J. Safari, S. Gandomi-Ravandi and M. Borjeni, J. Chem. Sci., 2013, 125, 1063 CrossRef CAS; (c) M. A. Zolfigol, M. Safaiee, F. Afsharnadery, N. Bahrami-Nejad, S. Baghery, S. Salehzadeh and F. Maleki, RSC Adv., 2015, 5, 100546 RSC.
- (a) R. M. Martin, R. G. Bergman and J. A. Ellman, J. Org. Chem., 2012, 77, 2501 CrossRef CAS PubMed; (b) S. Yamamoto, K. Okamoto, M. Murakoso, Y. Kuninobu and K. Takai, Org. Lett., 2012, 14, 3182 CrossRef CAS PubMed; (c) J. Wu, W. Xu, Z.-X. Yu and J. Wang, J. Am. Chem. Soc., 2015, 137, 9489 CrossRef CAS PubMed.
- C. Chiu, Z. Tang and J. W. Ellingboe, J. Comb. Chem., 1999, 1, 73 CrossRef CAS.
- A. K. Verma, S. Koul, A. P. S. Pannu and T. K. Razdan, Tetrahedron, 2007, 63, 8715 CrossRef CAS.
- A. Kumar, S. Koul, T. K. Razdan and K. K. Kapoor, Tetrahedron Lett., 2006, 47, 837 CrossRef CAS.
- C. Zhu, B. Bi, Y. Ding, T. Zhang and Q.-Y. Chen, Tetrahedron, 2015, 71, 9251 CrossRef CAS.
- J. Shen, D. Cai, C. Kuai, Y. Liu, M. Wei, G. Cheng and X. Cui, J. Org. Chem., 2015, 80, 6584 CrossRef CAS PubMed.
- X. Zhang, Z. Wang, K. Xu, Y. Feng, W. Zhao, X. Xu, Y. Yana and W. Yi, Green Chem., 2016, 18, 2313 RSC.
- J.-C. Xiang, M. Wang, Y. Cheng and A.-X. Wu, Org. Lett., 2016, 18, 24 CrossRef CAS PubMed.
- H. Huang, J. Cai, L. Tang, Z. Wang, F. Li and G.-J. Deng, J. Org. Chem., 2016, 81, 1499 CrossRef CAS PubMed.
- M.-N. Zhao, Z.-H. Ren, L. Yu, Y.-Y. Wang and Z.-H. Guan, Org. Lett., 2016, 18, 1194 CrossRef CAS PubMed.
- (a) S. Reimann, P. Ehlers, A. Petrosyan, S. Kohse, A. Spannenberg, A. E. Surkus, T. V. Ghochikyan, A. S. Saghyan, S. Lochbrunner, O. Khn, R. Ludwig and P. Langer, Adv. Synth. Catal., 2014, 356, 1987 CrossRef CAS; (b) R. Khajuria, P. Kannaboina, K. K. Kapoor, A. Gupta, G. Raina, A. K. Jassal, L. K. Rana, M. S. Hundald and P. Das, Org. Biomol. Chem., 2015, 13, 5944 RSC; (c) J. J. Mousseau and A. B. Charette, Acc. Chem. Res., 2013, 46, 412 CrossRef CAS PubMed; (d) R. Rossi, F. Bellina, M. Lessi and C. Manzini, Adv. Synth. Catal., 2014, 356, 17 CrossRef CAS.
- (a) Multicomponent Reactions, ed. J. Zhu and H. Bienaymé, Wiley-VCH, Weinheim, Germany, 2005 Search PubMed; (b) M. Shiri, Chem. Rev., 2012, 112, 3508 CrossRef CAS PubMed; (c) A. Dömling, W. Wang and K. Wang, Chem. Rev., 2012, 112, 3083 CrossRef PubMed; (d) C. de Graaff, E. Ruijter and R. V. A. Orru, Chem. Soc. Rev., 2012, 41, 3969 RSC; (e) C. M. Marson, Chem. Soc. Rev., 2012, 41, 7712 RSC; (f) S. Brauch, S. S. Berkel and B. van Westermann, Chem. Soc. Rev., 2013, 42, 4948 RSC; (g) S. Kumar, A. A. Sawant, R. P. Chikhale, K. Karanjai and A. Thomas, J. Org. Chem., 2016, 81, 1645 CrossRef CAS PubMed; (h) Q. Wang, C. Wan, Y. Gu, J. Zhang, L. Gao and Z. Wang, Green Chem., 2011, 13, 578 RSC.
- (a) L. F. Tietze, S. Dietz, N. Boehnke, M. A. Duefert, I. Objartel and D. Stalke, Eur. J. Org. Chem., 2011, 6574 CrossRef CAS; (b) G. Xiong, M. Wei, Y. Zhou, Y. Li, F. Zhang and Y. Gong, Synthesis, 2011, 3439 CAS; (c) W. Song, W. Lu, J. Wang, P. Lu and Y. Wang, J. Org. Chem., 2010, 75, 3481 CrossRef CAS PubMed.
- (a) H. Liu, Z. Zhou, Q. Sun, Y. Li, Y. Li, J. Liu, P. Yan, D. Wang and C. Wang, ACS Comb. Sci., 2012, 14, 366 CrossRef CAS PubMed; (b) Z. Zhou, H. Liu, Q. Sun, Y. Li, J. Yang, J. Liu, P. Yan and C. Wang, Synlett, 2012, 23, 2255 CrossRef CAS; (c) Y. Li, Z. Xue, W. Ye, J. Liu, J. Yao and C. Wang, ACS Comb. Sci., 2014, 16, 113 CrossRef CAS PubMed; (d) J. Zhang, J. Yao, J. Liu, S. Xue, Y. Li and C. Wang, RSC Adv., 2015, 5, 48580 RSC; (e) W. Ye, Y. Li, L. Zhou, J. Liu and C. Wang, Green Chem., 2015, 17, 188 RSC; (f) S. Xue, J. Yao, J. Liu, L. Wang, X. Liua and C. Wang, RSC Adv., 2016, 6, 1700 RSC.
- Crystallographic data for 2,4,6-trisubstituted pyridines 3b and 3d have been deposited with the Cambridge Crystallographic Data Centre with the deposition number CCDC 1457745 and 1502300.†.
Footnote |
| † Electronic supplementary information (ESI) available: Reactions conditions and spectra. CCDC 1457745 and 1502300. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra18630k |
| This journal is © The Royal Society of Chemistry 2016 |