Conjugated polymers with defined chemical structure as model carbon catalysts for nitro reduction†
Xiaoling Guoab,
Wei Qi *b,
Wei Liub,
Changhai Lianga,
Anmin Zhengc,
Xianfeng Yic and
Dangsheng Su*b
*b,
Wei Liub,
Changhai Lianga,
Anmin Zhengc,
Xianfeng Yic and
Dangsheng Su*b
aLaboratory of Advanced Materials and Catalytic Engineering, School of Chemical Engineering, Dalian University of Technology, Dalian, 116024, China
bShenyang National Laboratory for Materials Science, Institute of Metal Research, Chinese Academy of Sciences, Shenyang, 110016, China. E-mail: dssu@imr.ac.cn
cNational Center for Magnetic Resonance in Wuhan, Wuhan Institute of Physics and Mathematics, Chinese Academy of Sciences, Wuhan, 430071, China
First published on 4th October 2016
Abstract
A series of conjugated polymers (YPB-x) with designated chemical structure are synthesized as model carbon catalysts for mechanistic and kinetic studies of nitroaromatic reduction reactions. The reaction kinetics show that ketonic carbonyl groups are active sites for the nitrobenzene (NB) reduction. A catalytic reaction mechanism involving the activation of hydrazine with carbon catalysts and the subsequent reduction of NB is proposed to describe the catalytic process molecularly. The turnover frequency of heterogeneous carbon catalysts in the liquid phase is reported for the fair comparison of the intrinsic activity among different carbon catalysts.
1. Introduction
The development of novel and highly efficient catalysts meeting the requirements of green and sustainable chemistry is a hot topic in the fields of catalysis and materials science. Carbon, especially nanocarbon materials (such as carbon nanotubes (CNT), graphene, ultra-dispersed diamond (UDD), etc.), is considered as one of the most promising alternatives for conventional noble metal or metal oxide catalysts because of their adjustable chemical structure and composition, high stability and abundant resources.1–4 The enormous progress on the synthesis of novel nanocarbon materials provides sufficient options for various catalytic systems.5–8 Nevertheless, a critical challenge remains in the in-depth understanding and the structure–function relations of nanocarbon catalysis, since it is the foundation for the rational design of the catalysts.9 For example, carbon materials have been proven to show attractive catalytic activity in the reduction of nitroaromatics into corresponding aniline compounds for decades.10,11 The reaction is of significant importance in fine chemical synthesis, since the products (aniline) are important ingredients or intermediates for the production of resins, dyes or medicines.12 However, the detailed catalytic process by metal-free catalysts, such as the identity of the active sites and the reaction kinetics, is still unclear.13–15The difficulty in the mechanistic and kinetic study of carbon catalyzed process is not in measuring the reaction rates but in determining the identity and quantity of the active site. For example, several independent researches on carbon catalyzed nitro reduction reactions reached completely different conclusions on the identity of the active sites (either graphene defects or oxygen functionalities).16,17 The underlying problem is the complicated surface chemical compositions and structures (residual metal impurities, various functional groups, defects, etc.) on carbon materials, which can not be fully revealed through conventional characterization techniques (XPS or TPD, etc.), especially under reaction conditions.18–21
The utilization of model catalysts with well-defined chemical composition and structure is a feasible way to clarify the origin of the activity of carbon and reveal the catalytic process at molecular level. For example, a cyclotrimer of phenanthrenequinone (M = 618) with only ketonic carbonyl groups was reported as model carbon catalyst for the mechanistic and kinetic study in gas phase alkane oxidative dehydrogenation.22 However, the low thermal stability, relatively small surface area (5 m2 g−1) and limited conjugated size still make these small conjugated molecules obviously different from real nanocarbon catalysts.22 Larger conjugated and insoluble polymer with defined chemical structure and sufficient surface area is more appropriate to be used as model carbon catalyst.
Herein, we reported a series of conjugated polymers (Yamamoto derived phenanthrenequinone & benzene networks, denoted as YPB-x) with well-designed chemical structure and controllable content of ketonic carbonyl groups serving as heterogeneous model catalysts to reveal the identity of the active sites and the mechanism of carbon catalyzed nitrobenzene (NB) reduction reaction. The YPB-x polymers exhibit large conjugated systems, surface areas and abundant pores, which make them ideal analogue to nanocarbon catalysts. One of the obvious advantages of the obtained model catalysts is their spectroscopic transparency, and a plausible nitroaromatics reduction catalytic mechanism was proposed based on the Fourier transformed infrared spectroscopy (FT-IR) measurement on the model catalyst. The kinetic analysis results provide reliable chemical evidence that the ketonic carbonyl groups are the active sites for NB reduction reaction and the turnover frequency (TOF) of this carbon catalytic reaction system is given for the first time. The present research sheds light on the detailed mechanistic research based on model carbon catalysts with designated chemical structure.
2. Experimental
2.1 Catalyst preparation
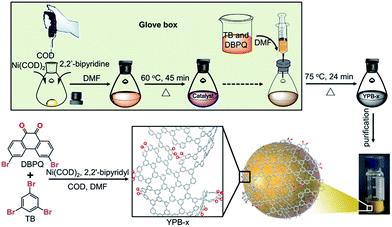
The model catalysts were synthesized through Yamamoto coupling reaction as illustrated in Scheme 1.23,24 The monomers, 1,3,5-tribromobenzene (TB) and 3,6-dibromo-phenanthrenequinone (DBPQ), were copolymerized in the presence of Ni catalyst, and the obtained polymers were named as YPB-x, where x is the mole ratio of TB and DBPQ. Two homopolymerization products of TB and DBPQ were also synthesized for comparison, and were named as YBN and YPQ, respectively.Taking YPB-6 as an example, the YPB series of polymers were synthesized through the following procedures. 0.660 g (2.4 mmol) Ni(COD)2, 0.376 g (2.4 mmol) 2,2′-bipyridine and 334 μL (2.4 mmol) 1,5-cyclooctadiene (COD) were dispersed in 15 mL of anhydrous DMF, and the mixture was stirred at 60 °C for 45 minutes under argon atmosphere to produce the catalysts for polymerization. Then, 15 mL of anhydrous DMF containing 0.189 g (0.6 mmol) TB and 0.036 g (0.1 mmol) DBPQ was dropwise added into above catalyst solution. After stirring at 75 °C for 24 h, the resulting mixture was washed with HCl(aq.), MeOH/H2O, hot acetone and tetrahydrofuran (THF) in sequence and was finally dried under vacuum. A yellow powder shaped product YPB-6 was obtained. The synthesis of other YPB-x copolymers is performed with similar strategies by changing the feed ratio of TB and DBPQ monomers. The homopolymerization product, YBN and YPQ compounds, are synthesized through a similar process with only one monomer added (TB and DBPQ, respectively).
The commercial carbon catalysts (graphene and CNT) were washed with concentrated HCl at room temperature (3 times, 12 h per time) to remove possible metal impurities. CNT was treated with concentrated HNO3 at 120 °C for 2 h to obtain the oxidized CNT (oCNT).14
2.2 Catalytic reaction
The catalytic reduction of NB was carried out using a reflux condenser system with hydrazine hydrate (HHA) and ethanol as reductant and solvent, respectively. In a typical procedure, 10.0 mg carbon catalysts (polymers or nanocarbon), 0.6 g NB and 2 mL N2H4·H2O (HHA) were dispersed in 1.5 mL of ethanol in a round bottom flask with stirring and refluxing at 80 °C. The reaction mixture was analysed and quantified by High Performance Liquid Chromatography (HPLC). For apparent catalytic activity comparison, NB conversion is controlled below 60% to make sure that the activity difference among catalysts could be clearly distinguished (reaction time ≤ 6 h). For kinetic analysis, a longer reaction time (typically 10–12 h) is needed, since it could provide enough data points and accuracy for a wide kinetic region. After one reaction run, the catalyst was filtered out and washed with solvent for more than 3 times, and then dried at 120 °C under vacuum for 24 h to give the recycled catalyst (named as r-YPB-6). The control experiment was performed as the same procedure as described above with only single reactant NB or HHA added, respectively. And the catalyst was recovered in the same way as r-YPB-6. The kinetic analysis was performed through sampling (100 μL each time) and analyzing the reaction mixture at given reaction time. The blank test for control experiment was performed as the same procedure as described above without addition of any catalysts.3. Results and discussion
3.1 Chemical structure of the model catalysts
The degree of polymerization and the chemical structure of YPB-x series are firstly examined with FT-IR measurements. As shown in Fig. 1a, the characteristic vibration bands belonging to monomers, DBPQ (ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) at 1677 cm−1) and TB (ν(C
O) at 1677 cm−1) and TB (ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Ar)) at 1590 cm−1, γ(
C(Ar)) at 1590 cm−1, γ(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H) bands below 1000 cm−1) could be clearly found in the FT-IR spectrum of YPB-6. The disappearance of the bands at about 1100 cm−1 (ν(C(Ar)–Br) for YPB-6 indicates the completed removal of –Br and the successful coupling reaction. The red shift of benzene skeleton deformational vibration (from 745 cm−1 of TB to 707 cm−1 of YPB-6) indicates the conjugation effect after polymerization.25 The normalized IR spectra of YPB-x series (Fig. S1†) shows the regularity of the increasing content of the ketonic carbonyl groups with the addition of monomer DBPQ, which is also consistent with the structure design of the polymer (as shown in Scheme 1).
C–H) bands below 1000 cm−1) could be clearly found in the FT-IR spectrum of YPB-6. The disappearance of the bands at about 1100 cm−1 (ν(C(Ar)–Br) for YPB-6 indicates the completed removal of –Br and the successful coupling reaction. The red shift of benzene skeleton deformational vibration (from 745 cm−1 of TB to 707 cm−1 of YPB-6) indicates the conjugation effect after polymerization.25 The normalized IR spectra of YPB-x series (Fig. S1†) shows the regularity of the increasing content of the ketonic carbonyl groups with the addition of monomer DBPQ, which is also consistent with the structure design of the polymer (as shown in Scheme 1).
Fig. 1b exhibits the solid-state 13C CP/MAS NMR spectra of YPB-6, YPQ and YBN, respectively. The peak at about 144 ppm could be assigned as the resonance of C–C bond that links two aromatic rings, providing evidence for the successful polymerization.25 The absence of the characteristic resonance signals belonging to ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–Br (123 ppm for TB)26 also indicates the completed polymerization. The signal at 178 ppm (–C
C–Br (123 ppm for TB)26 also indicates the completed polymerization. The signal at 178 ppm (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) indicates that the ketonic carbonyl groups are the only oxygen functionalities on the polymers.27,28 As shown in Table 1, the measured bulk and surface oxygen content (from the CHN/O elemental analysis and the SEM-EDS, respectively) exhibit similar trend as the calculated value, revealing the homogeneity of the chemical composition of the obtained polymers. The EA results also indicate that the oxygen content of the obtained YPB-x copolymers could be controlled through regulating the ratio of the two monomers as indicated by IR spectra (Fig. S1†). Moreover, the elemental analysis results suggest that there is no Ni and other metals in the synthesized polymers (Fig. S2–7†), which could avoid the argument of the catalytic activity of residual metals in carbon catalytic system.
O) indicates that the ketonic carbonyl groups are the only oxygen functionalities on the polymers.27,28 As shown in Table 1, the measured bulk and surface oxygen content (from the CHN/O elemental analysis and the SEM-EDS, respectively) exhibit similar trend as the calculated value, revealing the homogeneity of the chemical composition of the obtained polymers. The EA results also indicate that the oxygen content of the obtained YPB-x copolymers could be controlled through regulating the ratio of the two monomers as indicated by IR spectra (Fig. S1†). Moreover, the elemental analysis results suggest that there is no Ni and other metals in the synthesized polymers (Fig. S2–7†), which could avoid the argument of the catalytic activity of residual metals in carbon catalytic system.
| Catalyst | SBETa (m2 g−1) | Vtotb (cm3 g−1) | Smic/SDFTc (%) | Theoretical Od (wt%) | EA Oe (wt%) | EA Ce (wt%) | EDS Of (wt%) | EDS Cf (wt%) |
|---|---|---|---|---|---|---|---|---|
| a BET surface area.b Total pore volume calculated from the N2 adsorption/desorption measurements using NLDFT method.c Proportion of micropore in the whole surface area.d Theoretical oxygen content.e Weight content from CHN/O elemental analysis.f Surface element contents of polymers from EDS (average values of more than 30 spots for every samples). | ||||||||
| YBN | 1752 | 1.42 | 78.68 | 0 | 1.66 | 91.04 | 0.43 | 99.57 |
| YPB-40 | 1450 | 1.10 | 83.32 | 1.00 | 2.62 | 90.09 | 3.24 | 96.76 |
| YPB-15 | 1122 | 0.69 | 90.11 | 2.40 | 3.89 | 88.66 | 5.44 | 94.56 |
| YPB-6 | 817 | 0.48 | 92.04 | 4.58 | 6.31 | 87.32 | 6.13 | 93.87 |
| YPB-3 | 560 | 0.34 | 93.23 | 7.42 | 9.54 | 84.67 | 6.83 | 93.17 |
| YPQ | 8 | 0.01 | 0.00 | 15.53 | 18.27 | 76.36 | 19.58 | 80.42 |
| r-YPB-6 | 674 | 0.38 | 94.10 | 4.58 | 7.18 | 83.82 | — | — |
The morphology of the synthesized polymers is revealed by SEM and TEM. As shown in Fig. 2, the SEM images suggest that YPB-x and YBN are spherical with an average diameter of 450 ± 50 nm (Fig. 2a–e), while YPQ exhibit irregular shape and size (Fig. 2f). The formation of the spherical morphology could effectively minimize the surface energy during the polymerization process,29,30 and the spherical morphology of YPB-x and YBN polymers might be a result of the 3-dimensional polymerization from the three reactive ends (C–Br) of TB monomer. The TEM image of YPB-6 (Fig. 2g) reveals the amorphous structure of the spherical polymer since no obvious lattice fringe has been found, and the amorphous state is also confirmed by XRD analysis, as shown in Fig. S8.†
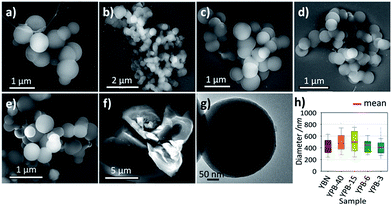 | ||
| Fig. 2 SEM images of (a) YBN, (b) YPB-40, (c) YPB-15, (d) YPB-6, (e) YPB-3, (f) YPQ; (g) TEM image of YPB-6, and (h) size distribution and the average diameter of YBN and YPB-x series. | ||
The porosity of the model catalysts is investigated by N2 adsorption–desorption analysis. YBN and YPB-x exhibit typical type-I isotherms,31 suggesting they are micro-porous materials (Fig. 3a, the micro-pores account for 80–95% of the total pore volume). The Brunauer–Emmett–Teller (BET) surface area and the total pore volume of YPB-x increase with the addition of TB from 560 m2 g−1/0.34 cm3 g−1 to 1450 m2 g−1/1.1 cm3 g−1, respectively, suggesting a promotion in the porosity of the polymers due to the cross-linking effect of monomer TB (Table 1). In addition, the BET surface area and pore volume of YPQ (8 m2 g−1/0.01 cm3 g−1, Fig. 3a) are much lower than that of YPB-x species. Apparently, the cross-linker TB plays an important role in both regulating the content of the ketonic carbonyl groups and determining the morphology and microstructure of the obtained model catalysts.
 | ||
| Fig. 3 (a) Nitrogen adsorption/desorption isotherms of YPB-x and YBN; (b) TGA curves of YPB-x, YBN and YPQ (10% O2 and Ar as balance gas, 50 mL min−1, 10 °C min−1). | ||
The thermal and solvent stability of the model catalysts are tested with TGA under oxygenous atmosphere and the dispersion–separation recycle experiments in various solvents, respectively. No apparent weight loss of YPB-x is observed below 330 °C in the presence of oxygen (Fig. 3b), suggesting their relatively high thermal stability and resistance to oxidation. The residual weights of the YPB compounds are less than 0.5 wt% after TGA tests indicating the absence of metal, which is consistent with the elemental analysis results, EDS and XRD analysis (Table S1 and Fig. S14†). The morphology and size of the YPB-x model catalysts also show negligible changes after dispersion in non-polar or polar solvents for more than 24 h (Fig. S10†), suggesting that there is no dissolving or swelling for them in solution. The relatively high stability benefits the applications of YPB-x as model heterogeneous catalysts in both gas and liquid phases.
3.2 Nitrobenzene reduction under the catalysis of model catalysts
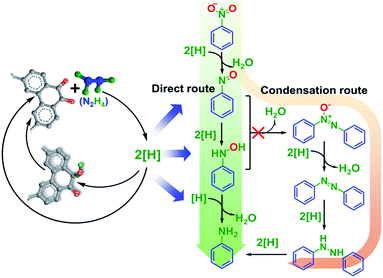
NB reduction is selected as a model reaction to examine the activity of the model catalysts and explore the mechanism of carbon catalyzed nitro reduction reactions. The NB reduction reactions are performed under a relatively mild condition (T = 80 °C) to make sure that the chemical structure of the model catalysts are well maintained. The NB conversion is controlled below 60% with an aniline selectivity >90% to ensure that the activity differences among various model catalysts could be clearly distinguished. YPB-6 is chosen as a representative catalyst for further studies because all of the synthesized polymers share similar morphology and chemical composition, and YPB-6 exhibits moderate surface area and oxygen content among them.The catalytic reaction pathways are firstly investigated by FT-IR measurements. As shown in Fig. 4, YPB-6-NB and YPB-6-HHA represent the catalysts that recovered from the control experiments with only single type of reactant added (NB or HHA, respectively), and the reactions or interactions between the catalysts and the reactants (NB and HHA) could be clearly observed in the IR spectra of these recovered catalysts. The IR signal of YPB-6-NB (the catalyst contacted with only NB in the reaction system) is almost identical with fresh YPB-6, indicating that the interactions between NB and the polymer catalysts may be weak. On the contrary, the stretching vibration band of the ketonic carbonyl groups (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O at 1680 cm−1) in the spectrum of YPB-6-HHA (the catalyst contacted with HHA only) decreased and broadened significantly comparing with fresh YPB-6, suggesting the partial reduction of YPB-6 in the presence of HHA. The slight increase of the signal at ∼1050 cm−1 (ν(N–H)) may also indicate the interaction between YPB-x catalysts and HHA. The catalyst under reduced state (denoted as [H] in Scheme 2) get re-oxidized easily and goes back to its initial state after exposing to NB as shown in Fig. 4 (r-YPB-6, recovered YPB-6 catalyst after interacting with both HHA and NB), indicating that the formation of the reducing species is a critical step in carbon catalyzed NB reduction process.
O at 1680 cm−1) in the spectrum of YPB-6-HHA (the catalyst contacted with HHA only) decreased and broadened significantly comparing with fresh YPB-6, suggesting the partial reduction of YPB-6 in the presence of HHA. The slight increase of the signal at ∼1050 cm−1 (ν(N–H)) may also indicate the interaction between YPB-x catalysts and HHA. The catalyst under reduced state (denoted as [H] in Scheme 2) get re-oxidized easily and goes back to its initial state after exposing to NB as shown in Fig. 4 (r-YPB-6, recovered YPB-6 catalyst after interacting with both HHA and NB), indicating that the formation of the reducing species is a critical step in carbon catalyzed NB reduction process.
The morphology and the major chemical structure of the YPB catalysts (the carbon framework and the ketonic carbonyl functionalities) are well maintained after one catalytic cycle as indicated by SEM and FT-IR spectra (Fig. S11† and 4). The slight differences between the IR spectra of the recovered and fresh YPB-6 (bands at 2850–2950 cm−1) could be attributed to the physical adsorption of the two reactants and solvent as proved by the adsorption test and the EA of r-YPB-6 (N account for 2.06%). The surface area of YPB-6 decreases correspondingly (from 817 m2 g−1 to 674 m2 g−1) after catalyzing the NB reduction due to the adsorption of the reactants. The immediate problem arising from the decreases of the surface area is the loss of the activity. The reuse experiment suggests that around 20% of the catalytic activity decreases (Fig. S17†) due to the blocking of the active sites.
NB conversion increases with the reaction temperature and the dosage of HHA (as shown in Fig. S16†). The NB conversion rates are independent on the concentration of HHA when the molar ratio between HHA and NB is above 8 (HHA/NB > 8), and it could be observed that the reduction of NB catalysed by YPB-6 follows pseudo first-order kinetics under such conditions where HHA is in large excessive as shown in Fig. 5a.13 In addition, the catalytic activity of YPB-x polymers, tested under the same reaction condition, shows a linear positive dependence on the content of the ketonic carbonyl groups (Fig. 5b). This result provides direct chemical evidence that ketonic carbonyl groups are the active sites for the activation of HHA and the subsequent NB reduction as discussed above. The linear dependence also suggests that the intrinsic activities of the ketonic carbonyl groups are identical for YPB-x series, and the surface area (in a certain range) has little influence on the activity (turnover frequency, TOF) of carbon catalysts. It should be noted that the quantity of the active oxygen species shown in Fig. 5b is actually the one taking off the contribution of the micropores smaller than the kinetic diameter of NB (0.59 nm), which is based on a simple assumption that the ketonic carbonyl groups are homogeneously dispersed on YPB-x.
 | ||
Fig. 5 (a) Kinetics of NB reduction reaction under the catalysis of YPB-6 (x, conversion of NB), (b) plot of NB conversion rate as a function of active oxygen amount ( YPB-x, YPB-x,  YBN, YBN,  Blank). Blank). | ||
Two reaction pathways, namely the direct and condensation routes (as shown in Scheme 2), are normally proposed for the reduction of nitro compounds.32 There is only less than 1% by-products (azobenzene and azoxybenzene) detected in our model catalyst catalyzed NB reduction process, indicating that the direct route may dominate the reaction pathway. The hypothesis is consistent with the kinetic analysis (Fig. 5a) that the reactions are in first order, and the first step in the direct route (forming nitrosobenzene) is considered as the rate-determining step.33 Combining the IR measurements on the model catalysts, product distribution and kinetic analysis of the catalytic reaction, we proposed a plausible mechanism for carbon catalyzed aromatic nitro compounds reduction as shown in Scheme 2. The ketonic carbonyl groups at the surface of carbon serve as active sites for the reaction, and they could activate and interact with hydrazine forming active reducing species ([H]), which could further reduce NB into the target product aniline. The reduction of NB follows the direct route through a sequential hydrogenation process with aniline and H2O as final products.
Finally, a simple comparison of the catalytic performance of YPB-6 and other types of carbon catalysts is shown in Table 2. The apparent catalytic activity are expressed and compared as the reaction rates (mmol g−1 h−1) at given NB conversion (15% in Table 2). YPB-6 exhibits similar apparent catalytic activity as nanocarbon catalysts, although it has lower oxygen content than nanocarbon (3.17% vs. 10.31% and 5.70% for grapheme oxide and o-CNT). Homogeneous phenanthraquinone exhibits higher apparent catalytic activity, but its intrinsic activity (TOF) is lower than YPB-6 (23.6 h−1 vs. 6.08 h−1).33 To the best of our knowledge, it is the first report on the TOF of carbon catalysts in liquid phase heterogeneous systems. It is difficult to evaluate the TOF for nanocarbon catalysts (graphene oxide and o-CNT), since the surface concentration of the accessible active sites can not be accurately measured.
| Catalysta | Oxygenb (at%) | SBET (m2 g−1) | Reaction rated (mmol g−1 h−1) | Selectivity (%) | TOF (h−1) |
|---|---|---|---|---|---|
| a Reaction conditions: 10 mg of catalyst, 0.6 g of NB, 8.0 equivalent of HHA, 1.5 mL ethanol, 80 °C, 6 h.b Atomic percentage (at%).c Not determined.d Reaction rate calculated at NB conversion at 15%. | |||||
| YPB-6 | 3.17 | 817 | 50.2 | 96.4 | 23.6 |
| Graphene oxide | 10.31 | 539 | 59.5 | 77.0 | n.d.c |
| o-CNT | 5.70 | 231.5 | 48.8 | 97.8 | n.d.c |
| Phenanthra-quinone | 8.33 | n.d.c | 88.3 | 74.1 | 6.08 |
4. Conclusions
In conclusion, a series of conjugated polymers with single type of oxygen functionalities, the ketonic carbonyl groups, are synthesized using the bottom-up strategy, and which are considered as proper candidate for model carbon catalysts for kinetic and mechanistic studies. Nitrobenzene reduction is selected as a model reaction to show the catalytic activity of the model catalysts. The mechanistic analysis reveals that the ketonic carbonyl groups on carbon catalysts could activate hydrazine forming activated reducing species, which subsequently react with nitrobenzene through typical direct route yielding the target product of aniline. The ketonic carbonyl groups are identified as the active sites for carbon catalyzed nitrobenzene reduction reactions, and the intrinsic activity (TOF) for carbon materials in liquid phase is obtained from the model catalysts. The present research indicates a possible strategy for the fair comparison of the intrinsic catalytic activity of carbon materials in liquid phase heterogeneous reactions, and it also sheds light on the detailed mechanistic and kinetic research on model carbon catalysts with designated chemical structure.Acknowledgements
The authors acknowledge financial support from the NSFC of China (21303226, 21133010, 21573256, 51221264 and 21261160487), the "Strategic Priority Research Program" of the Chinese Academy of Sciences (Grant No. XDA09030103) and the Youth Innovation Promotion Association of CAS. The authors thank Prof. Dr Robert Schlögl from Fritz Haber Institute (Berlin, Germany) for fruitful discussion.Notes and references
- D. Su, S. Perathoner and G. Centi, Chem. Rev., 2013, 113, 5782–5816 CrossRef CAS PubMed.
- D. Su, J. Zhang, B. Frank, A. Thomas, X. C. Wang, J. Paraknowitsch and R. Schlögl, ChemSusChem, 2010, 3, 169–180 CrossRef CAS PubMed.
- X. Sun, R. Wang and D. Su, Chin. J. Catal., 2013, 34, 508–523 CrossRef CAS.
- D. Chen, A. Holmen, Z. Sui and X. Zhou, Chin. J. Catal., 2014, 35, 824–841 CrossRef CAS.
- Y. Yan, J. Miao, Z. Yang, F. X. Xiao, H. B. Yang, B. Liu and Y. Yang, Chem. Soc. Rev., 2015, 44, 3295–3346 RSC.
- D. Haag and H. H. Kung, Top. Catal., 2014, 57, 762–773 CrossRef CAS.
- D. Su, N. I. Maksimova, G. Mestl, V. L. Kuznetsov, V. Keller, R. Schlögl and N. Keller, Carbon, 2007, 45, 2145–2151 CrossRef CAS.
- R. Wang, X. Sun, B. Zhang, X. Sun and D. Su, Chem.–Eur. J., 2014, 20, 6324–6331 CrossRef CAS PubMed.
- W. Qi and D. Su, ACS Catal., 2014, 4, 3212–3218 CrossRef CAS.
- B. H. Han, D. H. Shin and S. Y. Cho, Tetrahedron Lett., 1985, 26, 6233–6234 CrossRef CAS.
- M. Spiro, Catal. Today, 1990, 7, 167–178 CrossRef CAS.
- C. H. Campos, E. Rosenberg, J. L. G. Fierro, B. F. Urbano, B. L. Rivas, C. C. Torres and P. Reyes, Appl. Catal., A, 2015, 489, 280–291 CrossRef CAS.
- J. W. Larsen, M. Freund, K. Y. Kim, M. Sidovar and J. L. Stuart, Carbon, 2000, 38, 655–661 CrossRef CAS.
- S. Wu, G. Wen, B. Zhong, B. Zhang, X. Gu, N. Wang and D. Su, Chin. J. Catal., 2014, 35, 914–921 CrossRef CAS.
- V. Höller, D. Wegricht, I. Yuranov, L. Kiwi-Minsker and A. Renken, Chem. Eng. Technol., 2000, 23, 251–255 CrossRef.
- Y. Gao, D. Ma, C. Wang, J. Guan and X. Bao, Chem. Commun., 2011, 47, 2432–2434 RSC.
- S. Wu, G. Wen, J. Wang, J. Rong, B. Zong, R. Schlögl and D. S. Su, Catal. Sci. Technol., 2014, 4, 4183–4187 CAS.
- J. L. Figueiredo, M. F. R. Pereira, M. M. A. Freitas and J. J. M. Órfão, Carbon, 1999, 37, 1379–1389 CrossRef CAS.
- M. F. R. Pereira, J. J. M. Órfão and J. L. Figueiredo, Appl. Catal., A, 1999, 184, 153–160 CrossRef CAS.
- J. Zhang, X. Liu, R. Blume, A. Zhang, R. Schlögl and D. S. Su, Science, 2008, 322, 73–77 CrossRef CAS PubMed.
- W. Qi, W. Liu, X. L. Guo, R. Schlögl and D. S. Su, Angew. Chem., Int. Ed., 2015, 54, 13682–13685 CrossRef CAS PubMed.
- J. Zhang, X. Wang, Q. Su, L. Zhi, A. Thomas, X. Feng, D. Su, R. Schlögl and K. Müllen, J. Am. Chem. Soc., 2009, 131, 11296–11297 CrossRef CAS PubMed.
- T. Yamamoto, A. Morita, Y. Miyazaki, T. Maruyama, H. Wakayama, Z. Zhou, Y. Nakamura, T. Kanbara, S. Sasaki and K. Kubota, Macromolecules, 1992, 25, 1214–1223 CrossRef CAS.
- J. Schmidt, M. Werner and A. Thomas, Macromolecules, 2009, 42, 4426–4429 CrossRef CAS.
- K. Endo, T. Ida, S. Shimada, J. V. Ortiz, K. Deguchi, T. Shimizu and K. Yamada, J. Mol. Struct., 2012, 1027, 20–30 CrossRef CAS.
- National Institute of Advanced Industrial Science and Technology (AIST), Japan, Spectral Database for Organic Compounds, SDBS, 2015, http://www.sdbs.db.aist.go.jp.
- B. M. Alam, T. Aouak, N. M. Alandis and M. M. Alam, Int. J. Polym. Mater. Polym. Biomater., 2014, 64, 229–241 CrossRef.
- Q. Sun, X. Meng, X. Liu, X. Zhang, Y. Yang, Q. Yang and F. S. Xiao, Chem. Commun., 2012, 48, 10505–10507 RSC.
- C. Scherdel, T. Scherb and G. Reichenauer, Carbon, 2009, 47, 2244–2252 CrossRef CAS.
- G. H. Zheng and C. Y. Pan, Macromolecules, 2006, 39, 95–102 CrossRef CAS.
- K. S. W. Sing, D. H. Everett, R. A. W. Haul, L. Moscou, R. A. Pierotti, J. Rouquerol and T. Siemieniewska, Pure Appl. Chem., 1985, 57, 603–619 CrossRef CAS.
- V. Kratky, M. Kralik, M. Mecarova, M. Stolcova, L. Zalibera and M. Hronec, Appl. Catal., A, 2002, 235, 225–231 CrossRef CAS.
- S. Wu, G. Wen, X. Liu, B. Zhong and D. Su, ChemCatChem, 2014, 6, 1558–1561 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra18201a |
| This journal is © The Royal Society of Chemistry 2016 |



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: