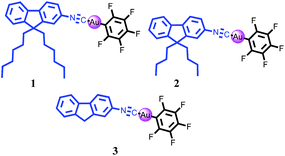
Fluorene-based mononuclear gold(I) complexes: the effect of alkyl chain, aggregation-induced emission (AIE) and mechanochromism characteristics†
Zhao Chen*a,
Yuting Nieb and
Sheng Hua Liu*b
aJiangxi Key Laboratory of Organic Chemistry, Jiangxi Science and Technology Normal University, Nanchang 330013, PR China. E-mail: chenzhao666@126.com
bKey Laboratory of Pesticide and Chemical Biology, Ministry of Education, College of Chemistry, Central China Normal University, Wuhan 430079, PR China. E-mail: chshliu@mail.ccnu.edu.cn
First published on 29th July 2016
Abstract
In this paper, three fluorene-based gold(I) complexes have been synthesized and characterized. All of these mononuclear gold(I) complexes exhibit obvious aggregation-induced emission (AIE) behavior. Furthermore, the luminogen 3 without containing alkyl chains also shows reversible mechanochromism property involving fluorescence color changes from green to faint yellow. The mechanism for AIE characteristics of these gold(I) complexes and mechanochromic behavior of luminogen 3 possibly involves a change in weak intermolecular π⋯π and C–H⋯F interactions and the generation of intermolecular gold–gold interactions.
Introduction
The development of highly emissive fluorescence materials has attracted much attention due to their potential applications in photoelectronic devices and display systems.1 However, the infamous aggregation caused quenching (ACQ) phenomenon leads to poor aggregate state emission efficiency, which has significantly limited the practical application of luminescent materials.2 Fortunately, in 2001, Tang et al. reported an aggregation-induced emission (AIE) phenomenon, which is opposite to the ACQ phenomenon.3 Luminogens with AIE property can emit efficiently in aggregated/solid states. Therefore, the discovery of AIE effect has contributed to the effective application of fluorescence materials. Mechanochromic materials, as a kind of “smart material”, have also attracted substantial attention on account of their promising applications in security papers and sensors.4 Bright solid state fluorescence and high color contrast are two extremely important factors for mechanochromism.5 Thus, it is very significative to synthesize mechanochromic luminogens possessing AIE property. So far, a number of AIE-active mechanochromic materials have been proven to be organic compounds.6 In contrast, AIE-active metal complexes exhibiting mechanochromic behavior are still rare. It is well known that gold(I) chemistry has attracted the widespread attention of scientists over the last two decades.7 To date, some mechanochromic gold(I) complexes have been reported.8 Nonetheless, gold(I) complexes with mechanochromism and AIE characteristics are still inadequate.9 On the other hand, fluorene-based derivatives are very valuable candidates in the domain of light emitting diodes.10 Unfortunately, the ACQ effect is one of the main bottlenecks that obstruct the efficient development of these fluorene-based materials. Hence, it is a meaningful research topic to develop AIE-active mechanochromic gold(I) complexes based on a fluorene skeleton. Herein, three fluorene-based mononuclear gold(I) complexes (Chart 1) have been designed and synthesized. The corresponding synthesis and characterization of these luminogens are depicted in detail. Furthermore, their photophysical and mechanochromic characteristics are explored as well. All of these gold(I) complexes exhibit excellent AIE characteristics. More interestingly, these gold(I) complexes show alkyl chain-dependent mechanofluorochromism. In addition, the crystalline state of solid sample 3 exhibits much stronger fluorescence than its amorphous state, which is very significative.11Result and discussion
Synthesis
The fluorene-based mononuclear gold(I) complexes 1–3 were prepared according to the reasonable routes described in Scheme 1. The intermediate products 1-c, 2-c and 3-b as starting materials were reacted with C6F5Au(tht) (tht = tetrahydrothiophene) to get the expected gold(I) complexes 1–3 in high yields, respectively.Aggregation-induced emission (AIE) properties of luminogens 1–3
In order to study the AIE properties of gold(I) complexes 1–3, their UV-Vis and photoluminescence (PL) spectra were systematically investigated. As shown in Fig. 1, the UV-Vis absorption spectra of luminogens 1–3 in DMF–H2O mixtures with high water fraction (fw) showed level-off tails in the visible region. Such tails can be attributed to the Mie scattering effect,12 which commonly indicates the presence of nano-aggregates.As presented in Fig. 2, dilute DMF solution (2.0 × 10−5 mol L−1) of complex 1 exhibited a single emission band with a maximum (λmax) at 350 nm, and the solution was practically nonluminescent under irradiation with UV light at 365 nm. However, when the fw value in the DMF solution was increased to 40%, a new yellow emission band with λmax at around 565 nm was observed. Moreover, when the water content was increased to 90%, the peak intensity of luminogen 1 at 565 nm was approximately 130-fold higher than the intensity in pure DMF, and the bright yellow fluorescence could be obtained under 365 nm UV light. This is because complex 1 is insoluble in water, and thus increasing the fw value in DMF–H2O mixtures resulted in the formation of aggregated particles. Actually, the aggregated particles obtained in DMF–H2O mixtures with high fw value have been confirmed by dynamic light-scattering (DLS) analysis (Fig. S1, ESI†). When a high percentage of water was added to the DMF solution containing small amounts of luminogen 1, nano-aggregates were formed, and the generation of intermolecular aurophilic interactions was possibly responsible for the strong yellow emission.13 Obviously, the yellow emission of luminogen 1 was associated with aggregate formation, and therefore 1 exhibited typical AIE property. As can be seen in Fig. 3, the aggregation-induced characteristics of complexes 2 and 3 in DMF–H2O mixtures with various fw value were similar to those of complex 1. As a consequence, 2 and 3 are also AIE-active luminogens.
Mechanochromism behavior of complex 3
The reversible mechanochromic property of complex 3 was investigated by PL spectroscopy. As evident from Fig. 4, the solid-state fluorescence spectrum of complex 3 exhibited two emission peaks with λmax at 467 nm and 498 nm, and the solid powder of 3 showed green luminescence upon UV irradiation at a wavelength of 365 nm. Intriguingly, two new emission bands were observed with λmax at 457 nm, 461 nm and 596 nm upon gentle grinding of solid powder 3, and the corresponding emission color was converted from green to faint yellow. Indeed, mechanofluorochromic phenomenon involving color changes from green to faint yellow is very rare, and it is different from our previous work.9d,9e Furthermore, the faint yellow luminescence reverted to its original unground green color after a 1 min treatment of the ground powder with dichloromethane vapor. In addition, as Fig. 5 shows, the repeatability of this reversible mechanochromic transformation is very superior. It was speculated that the observed mechanochromism behavior may be related to the change of molecular morphology. In order to better understand the possible mechanochromic mechanism, diverse solid states of luminogen 3 were tested by powder X-ray diffraction (XRD) measurements. As shown in Fig. 6, the XRD diffractogram of unground solid powder 3 exhibited many intense and sharp reflection peaks, which is indicative of its crystalline nature. However, the grinding gave rise to weakening and broadening of all the diffraction peaks, which suggested the use of mechanical force led to the transformation from crystalline state to amorphous state. | ||
| Fig. 5 Invertible grinding-fuming processes of the photoluminescence of complex 3 at 596 nm. Excitation wavelength: 365 nm. | ||
 | ||
| Fig. 6 XRD patterns of complex 3: unground, ground and after treatment with dichloromethane solvent vapor. | ||
Moreover, the initial clear and intense reflection peaks reappeared upon treatment of the ground sample with dichloromethane solvent vapor, which implies a restoration of its original crystalline phase state. Thus, the XRD experimental results proved that the mechanochromic property of 3 can be ascribed to the morphology conversion between stable crystalline state and metastable amorphous state. In contrast, as Fig. 7 shows, no mechanochromism phenomenon was observed for luminogens 1 and 2 containing alkyl chains of different lengths.
Experimentals
General
All manipulations were accomplished under an argon atmosphere by using standard Schlenk techniques, unless otherwise stated. All starting materials were obtained commercially as analytical-grade and used without further purification. Compounds 1-a,14 2-a14 and C6F5Au(tht) (tht = tetrahydrothiophene)15 were synthesized by procedures described in the corresponding literatures. 1H NMR (400 MHz) and 13C NMR (100.6 MHz) spectra were collected on American Varian Mercury Plus 400 spectrometer (400 MHz). 1H NMR spectra are reported as followed: chemical shift in ppm (δ) relative to the chemical shift of TMS at 0.00 ppm, integration, multiplicities (s = singlet, d = doublet, t = triplet, m = multiplet), and coupling constant (Hz). 13C NMR chemical shifts reported in ppm (δ) relative to the central line of triplet for CDCl3 at 77 ppm. 19F NMR chemical shifts are relative to C6F6 (δ = −163.00). EI-MS was measured using EI (TRACE MS2000). Elemental analyses (C, H, N) were performed by the Microanalytical Services, College of Chemistry, CCNU. UV-Vis spectra were recorded on U-3310 UV Spectrophotometer. Fluorescence spectra were obtained on a Fluoromax-P luminescence spectrometer (HORIBA JOBIN YVON INC.). Column chromatographic separations were carried out on silica gel (200–300 mesh). TLC was performed by using commercially prepared 100–400 mesh silica gel plates (GF254) and visualization was effected at 254 nm. The DMF/water mixtures with various water fractions were prepared by slowly adding ultra-pure water into the DMF solution containing small amounts of samples. XRD studies were recorded on a Shimadzu XRD-6000 diffractometer using Ni-filtered and graphite-monochromated Cu Kα radiation (λ = 1.54 Å, 40 kV, 30 mA). Dynamic light scattering (DLS) measurements were performed on the Zetasizer instrument ZEN3600 (Malvern, UK) with a 173° back scattering angle and He–Ne laser (λ = 633 nm).Synthetic procedures for 1-b and 2-b
A mixture of compound 1-a or 2-a (5 mmol) and formic acid (30 ml) was stirred for overnight at 110 °C. After completion of present reaction, formic acid was removed from reaction system by distillation, the residual mixture was extracted with dichloromethane (3 × 20 ml), the combined organic layers were washed with brine, dried (Na2SO4), and concentrated in vacuo. The residues were purified by column chromatography, affording the expected white solid product in a yield of 64%, 68%, respectively. 1-b: 1H NMR (400 MHz, CDCl3): δ (ppm) = 8.78–8.42 (m, 2H), 7.67–7.62 (m, 2H), 7.49–7.45 (m, 1H), 7.33–7.27 (m, 3H), 7.08–7.05 (m, 1H), 1.99–1.90 (m, 4H), 1.13–1.02 (m, 12H), 0.78–0.73 (m, 6H), 0.60 (t, J = 8 Hz, 4H). 13C NMR (100 MHz, CDCl3): δ (ppm) = 163.1, 159.0, 152.6, 151.9, 150.5, 150.3, 140.5, 140.1, 138.5, 137.7, 136.2, 135.7, 126.9, 126.8, 126.7, 122.7, 120.6, 119.9, 119.4, 119.3, 118.4, 117.7, 114.5, 113.3, 55.2, 40.3, 31.4, 29.6, 23.6, 22.5, 13.9. EI-MS: m/z = 377.25[M]+. Anal. calcd for C26H35NO: C, 82.71; H, 9.34; N, 3.71. Found: C, 82.77; H, 9.38; N, 3.66. 2-b: 1H NMR (400 MHz, CDCl3): δ (ppm) = 8.74–8.42 (m, 1H), 7.67–7.44 (m, 3H), 7.31 (s, 3H), 7.19–7.02 (m, 2H), 1.96 (t, J = 6 Hz, 4H), 1.10–1.04 (m, 4H), 0.69–0.57 (m, 10H). 13C NMR (100 MHz, CDCl3): δ (ppm) = 162.5, 158.8, 145.1, 144.4, 143.2, 142.9, 141.1, 140.8, 139.3, 138.6, 135.6, 135.3, 127.0, 126.8, 126.5, 125.0, 120.8, 120.2, 119.7, 119.6, 118.7, 118.1, 117.1, 116.0, 37.0. EI-MS: m/z = 321.26[M]+. Anal. calcd for C22H27NO: C, 82.20; H, 8.47; N, 4.36. Found: C, 82.26; H, 8.51; N, 4.31.Synthetic procedure for 3-a
A mixture of compound 2-aminofluorene (11 mmol) and formic acid (30 ml) was stirred for overnight at 110 °C. After completion of present reaction, collecting the white solid product by suction filtration. Yield = 58%. 1H NMR (400 MHz, CDCl3): δ (ppm) = 8.75–8.41 (m, 1H), 8.11–7.91 (m, 1H), 7.75–7.70 (m, 2H), 7.53 (t, J = 8 Hz, 1H), 7.40–7.28 (m, 3H), 7.10 (d, J = 4 Hz, 1H), 3.89 (d, J = 8 Hz, 2H). 13C NMR (100 MHz, CDCl3): δ (ppm) = 162.5, 158.8, 145.1, 144.4, 143.2, 142.9, 141.1, 140.8, 139.3, 138.6, 135.6, 135.3, 127.0, 126.8, 126.5, 125.0, 120.8, 120.2, 119.7, 119.6, 118.7, 118.1, 117.1, 116.0, 37.0. EI-MS: m/z = 209.02[M]+. Anal. calcd for C14H11NO: C, 80.36; H, 5.30; N, 6.69. Found: C, 80.41; H, 5.34; N, 6.63.Synthetic procedures for 1-c and 2-c
A CH2Cl2 suspension (15 ml) of 1-b or 2-b (2.5 mmol) and triethylamine (5 ml) was cooled to 0 °C. To the mixture was added dropwise a CH2Cl2 solution (10 ml) of triphosgene (2.8 mmol). The mixture was refluxed under an argon atmosphere for 3 h, then 10% aq. Na2CO3 (50 ml) was added dropwise at room temperature. The mixture was extracted with dichloromethane (3 × 20 ml). The combined organic layers were washed with brine, dried (Na2SO4), and concentrated in vacuo. The residues were purified by column chromatography, affording the expected light yellow liquid or green solid in a yield of 67%, 66%, respectively. 1-c: 1H NMR (400 MHz, CDCl3): δ (ppm) = 7.70–7.66 (m, 2H), 7.35–7.33 (m, 5H), 1.98–1.92 (m, 4H), 1.13–1.03 (m, 12H), 0.77 (t, J = 6 Hz, 6H), 0.60–0.52 (m, 4H). 13C NMR (100 MHz, CDCl3): δ (ppm) = 163.5, 152.0, 151.0, 142.2, 139.3, 128.1, 127.0, 125.3, 124.9, 122.9, 120.9, 120.2, 120.2, 55.3, 40.1, 31.4, 29.5, 23.6, 22.5, 13.9. EI-MS: m/z = 359.41[M]+. Anal. calcd for C26H33N: C, 86.85; H, 9.25; N, 3.90. Found: C, 86.89; H, 9.32; N, 3.84. 2-c: 1H NMR (400 MHz, CDCl3): δ (ppm) = 7.71–7.67 (m, 2H), 7.37–7.33 (m, 5H), 2.02–1.90 (m, 4H), 1.13–1.03 (m, 4H), 0.68 (t, J = 8 Hz, 6H), 0.59–0.51 (m, 4H). 13C NMR (100 MHz, CDCl3): δ (ppm) = 163.8, 152.0, 150.9, 142.2, 139.2, 128.1, 127.0, 125.3, 124.9, 122.9, 120.9, 120.2, 120.1, 55.3, 39.9, 25.8, 22.8, 13.6. EI-MS: m/z = 303.21[M]+. Anal. calcd for C22H25N: C, 87.08; H, 8.30; N, 4.62. Found: C, 87.14; H, 8.35; N, 4.57.Synthetic procedure for 3-b
A CH2Cl2 suspension (15 ml) of 3-a (3.8 mmol) and triethylamine (5 ml) was cooled to 0 °C. To the mixture was added dropwise a CH2Cl2 solution (10 ml) of triphosgene (4.2 mmol). The mixture was refluxed under an argon atmosphere for 3 h, then 10% aq. Na2CO3 (50 ml) was added dropwise at room temperature. The mixture was extracted with dichloromethane (3 × 20 ml). The combined organic layers were washed with brine, dried (Na2SO4), and concentrated in vacuo. The residues were purified by column chromatography, affording the expected light yellow solid in a yield of 80%. 1H NMR (400 MHz, CDCl3): δ (ppm) = 7.60 (d, J = 8 Hz, 1H), 7.55 (d, J = 4 Hz, 1H), 7.41 (d, J = 4 Hz, 1H), 7.33 (s, 1H), 7.27–7.21 (m, 3H), 3.69 (s, 2H). 13C NMR (100 MHz, CDCl3): δ (ppm) = 163.6, 144.0, 143.4, 142.6, 139.8, 127.7, 127.0, 125.1, 125.0, 122.9, 120.3, 120.1, 36.6. EI-MS: m/z = 191.16[M]+. Anal. calcd for C14H9N: C, 87.93; H, 4.74; N, 7.32. Found: C, 87.87; H, 4.70; N, 7.38.Synthetic procedures for 1 and 2
A mixture of C6F5Au(tht) (0.51 mmol) and 1-c or 2-c (0.5 mmol) was stirred in CH2Cl2 (20 ml) over night under an argon atmosphere at room temperature. After completion of present reaction, the solvent was evaporated. The residues were purified by column chromatography, affording the expected yellow sticky product or white solid product in a yield of 69%, 63%, respectively. 1: 1H NMR (400 MHz, CDCl3): δ (ppm) = 7.79–7.74 (m, 2H), 7.53 (d, J = 4 Hz, 1H), 7.50 (s, 1H), 7.42–7.37 (m, 3H), 2.05–1.92 (m, 4H), 1.14–1.01 (m, 12H), 0.77 (t, J = 8 Hz, 6H), 0.60–0.54 (m, 4H). 19F NMR (CDCl3): δ (ppm) = −116.86, −158.34, −163.21. Anal. calcd for C32H33AuF5N: C, 53.12; H, 4.60; N, 1.94. Found: C, 53.21; H, 4.68; N, 1.87. 2: 1H NMR (400 MHz, CDCl3): δ (ppm) = 7.79–7.74 (m, 2H), 7.55–7.47 (m, 2H), 7.44–7.37 (m, 3H), 2.06–1.93 (m, 4H), 1.14–1.05 (m, 4H), 0.69 (t, J = 8 Hz, 6H), 0.61–0.50 (m, 4H). 19F NMR (CDCl3): δ (ppm) = −116.54, −158.15, −163.02. Anal. calcd for C28H25AuF5N: C, 50.38; H, 3.78; N, 2.10. Found: C, 50.45; H, 3.70; N, 2.19.Synthetic procedure for 3
A mixture of C6F5Au(tht) (1.03 mmol) and 3-b (1 mmol) was stirred in CH2Cl2 (20 ml) over night under an argon atmosphere at room temperature. After completion of present reaction, the solvent was evaporated. A small amount of CH2Cl2 was added, and then a lot of n-hexane was added. Collecting the white solid product by suction filtration. Yield = 63%. 1H NMR (400 MHz, CDCl3): δ (ppm) = 7.89–7.83 (m, 2H), 7.71 (s, 1H), 7.63–7.56 (m, 2H), 7.48–7.41 (m, 2H), 3.99 (s, 2H). 19F NMR (CDCl3): δ (ppm) = −116.95, −158.43, −163.33. Anal. calcd for C20H9AuF5N: C, 43.26; H, 1.63; N, 2.52. Found: C, 43.34; H, 1.69; N, 2.45.Conclusions
In summary, three fluorene-based complexes containing mononuclear gold(I) units have been synthesized. Through study of their aggregation-induced properties it was revealed that all of these gold(I) complexes exhibited excellent AIE characteristics. Subsequent mechanochromic studies showed that complex 3 possessed reversible mechanochromic behavior, while complexes 1 and 2 containing alkyl chains of different lengths did not exhibit any mechanochromic behavior upon grinding, suggesting the effect of alkyl chain played a very important role in mechanochromism characteristics of complexes 1–3. The mechanism for AIE characteristics of 1–3 and mechanochromic behavior of 3 possibly involves a change in weak intermolecular π⋯π and C–H⋯F interactions and the generation of intermolecular gold–gold interactions. Undoubtedly, the results of this research should be helpful for the design of mechanical-stimuli-responsive gold(I) complexes with AIE property. Further studies on new gold(I) complexes possessing other fascinating characteristics are in progress.Acknowledgements
The authors acknowledge financial support from National Natural Science Foundation of China (No. 21272088, 21472059 and 21402057).Notes and references
- (a) Y. Sagara and T. Kato, Nat. Chem., 2009, 1, 605–610 CrossRef CAS PubMed; (b) M. Wang, G. Zhang, D. Zhang, D. Zhu and B. Z. Tang, J. Mater. Chem., 2010, 20, 1858–1867 RSC; (c) J. Wu, W. Liu, J. Ge, H. Zhang and P. Wang, Chem. Soc. Rev., 2011, 40, 3483–3495 RSC; (d) J. Mei, N. L. C. Leung, R. T. K. Kwok, J. W. Y. Lam and B. Z. Tang, Chem. Rev., 2015, 115, 11718–11940 CrossRef CAS PubMed; (e) H. Liu, L. Yao, B. Li, X. Chen, Y. Gao, S. Zhang, W. Li, P. Lu, B. Yang and Y. Ma, Chem. Commun., 2016, 52, 7356–7359 RSC; (f) Z. Chen, J. Liang, X. Han, J. Yin, G.-A. Yu and S. H. Liu, Dyes Pigm., 2015, 112, 59–66 CrossRef CAS; (g) Z. Chen, D. Wu, X. Han, Y. Nie, J. Yin, G.-A. Yu and S. H. Liu, RSC Adv., 2014, 4, 63985–63988 RSC.
- (a) R. T. K. Kwok, C. W. T. Leung, J. W. Y. Lam and B. Z. Tang, Chem. Soc. Rev., 2015, 44, 4228–4238 RSC; (b) J. Mei, Y. Hong, J. W. Y. Lam, A. Qin, Y. Tang and B. Z. Tang, Adv. Mater., 2014, 26, 5429–5479 CrossRef CAS PubMed.
- J. Luo, Z. Xie, J. W. Y. Lam, L. Cheng, H. Chen, C. Qiu, H. S. Kwok, X. Zhan, Y. Liu, D. Zhu and B. Z. Tang, Chem. Commun., 2001, 1740–1741 RSC.
- (a) Z. Chi, X. Zhang, B. Xu, X. Zhou, C. Ma, Y. Zhang, S. Liua and J. Xu, Chem. Soc. Rev., 2012, 41, 3878–3896 RSC; (b) Q. Qi, J. Qian, X. Tan, J. Zhang, L. Wang, B. Xu, B. Zou and W. Tian, Adv. Funct. Mater., 2015, 25, 4005–4010 CrossRef CAS; (c) B. Xu, Y. Mu, Z. Mao, Z. Xie, H. Wu, Y. Zhang, C. Jin, Z. Chi, S. Liu, J. Xu, Y.-C. Wu, P.-Y. Lu, A. Lien and M. R. Bryce, Chem. Sci., 2016, 7, 2201–2206 RSC; (d) D. A. Davis, A. Hamilton, J. L. Yang, L. D. Cremar, D. Van Gough, S. L. Potisek, M. T. Ong, P. V. Braun, T. J. Martinez, S. R. White, J. S. Moore and N. R. Sottos, Nature, 2009, 459, 68–72 CrossRef CAS PubMed.
- J. Wei, B. Liang, X. Cheng, Z. Zhang, H. Zhang and Y. Wang, RSC Adv., 2015, 5, 71903–71910 RSC.
- (a) T. Jadhav, B. Dhokale, Y. patil and R. Misra, RSC Adv., 2015, 5, 68187–68191 RSC; (b) Y. Cui, Y.-M. Yin, H.-T. Cao, M. Zhang, G.-G. Shan, H.-Z. Sun, Y. Wu, Z.-M. Su and W.-F. Xie, Dyes Pigm., 2015, 119, 62–69 CrossRef CAS; (c) Y. Zhan, P. Gong, P. Yang, Z. Jin, Y. Bao, Y. Li and Y. Xu, RSC Adv., 2016, 6, 32697–32704 RSC; (d) B. Xu, J. He, Y. Mu, Q. Zhu, S. Wu, Y. Wang, Y. Zhang, C. Jin, C. Lo, Z. Chi, A. Lien, S. Liu and J. Xu, Chem. Sci., 2015, 6, 3236–3241 RSC; (e) Y. Q. Dong, J. W. Y. Lam and B. Z. Tang, J. Phys. Chem. Lett., 2015, 6, 3429–3436 CrossRef CAS PubMed.
- (a) H. Schmidbaur, Gold Bull., 2000, 33, 3–10 CrossRef CAS; (b) J. Gil-Rubio, V. Cámara, D. Bautista and J. Vicente, Organometallics, 2012, 31, 5414–5426 CrossRef CAS; (c) W.-Y. Wong, K.-H. Choi, G.-L. Lu, J.-X. Shi, P.-Y. Lai and S.-M. Chan, Organometallics, 2001, 20, 5446–5454 CrossRef CAS; (d) W. J. Hunks, M. C. Jennings and R. J. Puddephatt, Inorg. Chem., 2000, 39, 2699–2702 CrossRef CAS PubMed; (e) W. Lu, W.-M. Kwok, C. Ma, C. T.-L. Chan, M.-X. Zhu and C.-M. Che, J. Am. Chem. Soc., 2011, 133, 14120–14135 CrossRef CAS PubMed; (f) H. Schmidbaur and A. Schier, Chem. Soc. Rev., 2012, 41, 370–412 RSC; (g) C. Jobbágy and A. Deák, Eur. J. Inorg. Chem., 2014, 4434–4449 CrossRef; (h) E. R. T. Tiekink, Coord. Chem. Rev., 2014, 275, 130–153 CrossRef CAS; (i) Z. Chen, D. Wu, X. Han, J. Liang, J. Yin, G.-A. Yu and S. H. Liu, Chem. Commun., 2014, 50, 11033–11035 RSC; (j) Z. Chen, X. Han, J. Zhang, D. Wu, G.-A. Yu, J. Yin and S. H. Liu, RSC Adv., 2015, 5, 15341–15349 RSC; (k) Z. Chen, J. Liang, Y. Nie, X. Xu, G.-A. Yu, J. Yin and S. H. Liu, Dalton Trans., 2015, 44, 17473–17477 RSC.
- (a) H. Ito, T. Saito, N. Oshima, N. Kitamura, S. Ishizaka, Y. Hinatsu, M. Wakeshima, M. Kato, K. Tsuge and M. Sawamura, J. Am. Chem. Soc., 2008, 130, 10044–10045 CrossRef CAS PubMed; (b) K. Kawaguchi, T. Seki, T. Karatsu, A. Kitamura, H. Ito and S. Yagai, Chem. Commun., 2013, 49, 11391–11393 RSC; (c) T. Seki, T. Ozaki, T. Okura, K. Asakura, A. Sakon, H. Uekusa and H. Ito, Chem. Sci., 2015, 6, 2187–2195 RSC; (d) S. Yagai, T. Seki, H. Aonuma, K. Kawaguchi, T. Karatsu, T. Okura, A. Sakon, H. Uekusa and H. Ito, Chem. Mater., 2016, 28, 234–241 CrossRef CAS; (e) H. Ito, M. Muromoto, S. Kurenuma, S. Ishizaka, N. Kitamura, H. Sato and T. Seki, Nat. Commun., 2013, 4, 2009–2013 Search PubMed; (f) T. Seki, K. Sakurada and H. Ito, Chem. Commun., 2015, 51, 13933–13936 RSC; (g) M. Jin, T. Seki and H. Ito, Chem. Commun., 2016, 52, 8083–8086 RSC; (h) J. Liang, F. Hu, X. Lv, Z. Chen, Z. Chen, J. Yin, G.-A. Yu and S. H. Liu, Dyes Pigm., 2012, 95, 485–490 CrossRef CAS; (i) T. Seki, Y. Takamatsu and H. Ito, J. Am. Chem. Soc., 2016, 138, 6252–6260 CrossRef CAS PubMed.
- (a) Z. Chen, P.-S. Huang, Z. Li, J. Yin, G.-A. Yu and S. H. Liu, Inorg. Chim. Acta, 2015, 432, 192–197 CrossRef CAS; (b) Z. Chen, J. Zhang, M. Song, J. Yin, G.-A. Yu and S. H. Liu, Chem. Commun., 2015, 51, 326–329 RSC; (c) Z. Chen, Z. Li, L. Yang, J. Liang, J. Yin, G.-A. Yu and S. H. Liu, Dyes Pigm., 2015, 121, 170–177 CrossRef CAS; (d) Z. Chen, Z. Li, F. Hu, G.-A. Yu, J. Yin and S. H. Liu, Dyes Pigm., 2016, 125, 169–178 CrossRef CAS; (e) Z. Chen, L. Yang, Y. Hu, D. Wu, J. Yin, G.-A. Yu and S. H. Liu, RSC Adv., 2015, 5, 93757–93764 RSC; (f) J. Liang, Z. Chen, L. Xu, J. Wang, J. Yin, G.-A. Yu, Z.-N. Chen and S. H. Liu, J. Mater. Chem. C, 2014, 2, 2243–2250 RSC.
- C.-C. Wu, Y.-T. Lin, K.-T. Wong, R.-T. Chen and Y.-Y. Chien, Adv. Mater., 2004, 16, 61–65 CrossRef CAS.
- Y. Dong, J. W. Y. Lam, A. Qin, Z. Li, J. Sun, H. H.-Y. Sung, I. D. Williams and B. Z. Tang, Chem. Commun., 2007, 40–42 RSC.
- C. W. T. Leung, Y. Hong, S. Chen, E. Zhao, J. W. Y. Lam and B. Z. Tang, J. Am. Chem. Soc., 2013, 135, 62–65 CrossRef CAS PubMed.
- J. Liang, Z. Chen, J. Yin, G.-A. Yu and S. H. Liu, Chem. Commun., 2013, 49, 3567–3569 RSC.
- S. P. Dudek, M. Pouderoijen, R. Abbel, A. P. H. J. Schenning and E. W. Meijer, J. Am. Chem. Soc., 2005, 127, 11763–11768 CrossRef CAS PubMed.
- R. Uson, A. Laguna, M. Laguna, D. A. Briggs, H. H. Murray and J. P. Fackler, Inorg. Synth., 1989, 26, 85–91 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: NMR spectra, mass spectra, size distribution curves of complexes 1–3 (2.0 × 10−5 mol L−1) in DMF–water mixtures with 90% volume fraction of water. See DOI: 10.1039/c6ra17806e |
| This journal is © The Royal Society of Chemistry 2016 |