Room temperature copper-catalyzed oxidative amidation of terminal alkynes for the synthesis of α-ketoamides using O-benzoyl hydroxylamines as aminating reagent and oxidant†
Guodong Shen*,
Lingyu zhao,
Yichen Wang and
Tongxin Zhang*
School of Chemistry and Chemical Engineering, School of Pharmacy, Liaocheng University, Liaocheng 252000, Shandong, PR China. E-mail: shenguodong33@163.com; xintongzhang123@163.com; Fax: +86-635-8239680; Tel: +86-635-8239680
First published on 11th August 2016
Abstract
A novel and convenient copper-catalyzed oxidative amidation for the synthesis of α-ketoamides has been successfully developed, which uses easily available O-benzoyl hydroxylamines as aminating reagent and oxidant. The reaction proceeds smoothly at room temperature and is compatible with a range of substrates to give the desired products in moderate to good yields.
α-Ketoamides are important functional scaffolds in many natural products, biological compounds, pharmaceuticals, and synthetic intermediates.1 Significant efforts have been made toward the development of efficient methods for their preparation. Traditional approaches for accessing α-ketoamides mainly involve the condensation of their corresponding α-keto acids or α-keto acyl halides with amines.2 Some alternative methods, such as double carbonylation reactions,3 oxidative reactions4 and other methods5 have also been developed. Recently, the copper-catalyzed oxidative reaction for the synthesis of α-ketoamides has attracted much attention,4a–d especially using commercially available terminal alkynes as coupling partners.6 For example, Jiao et al. and Shah et al. have successfully developed copper-catalyzed methods for the preparation of α-ketoamides involving terminal alkynes as coupling partners with amines (Scheme 1, eqn (1) and (2)).6a,b While generally effective, the methods require external oxidants, additives and heating conditions. Therefore, developing new methods for the synthesis of α-ketoamides using easily available starting materials under mild conditions is still of promising interest. In the past decade, O-benzoyl hydroxylamines (BzO-NR1R2) have received great attention because of easy handling and preparation and high reactivity in amination reactions as a convenient nitrogen source under mild conditions.7 Herein, we present a novel and convenient copper-catalyzed oxidative amidation of terminal alkynes for the synthesis of α-ketoamides employing easily available O-benzoyl hydroxylamines (BzO-NR1R2) as dually a reactive aminating reagent and oxidant at room temperature (Scheme 1, eqn (3)).
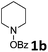
Initially, the synthesis of α-ketoamides with phenylacetylene 1a and N-(benzoyloxy)piperidine 1b was investigated in the presence of copper catalyst to identify and optimize the reaction parameters (Table 1). To our delight, when the reaction was carried out with CuI (10 mol%) and DBU (2 eq.) in THF (3.0 mL) at ambient temperature under nitrogen, the desired product could be detected (Table 1, entry 1). It was found that the copper catalysts such as CuCl2, CuI, CuBr, Cu(OTf)2 affected the reaction yield significantly (Table 1, entries 1–4). The best result was obtained in the presence of 10 mol% of Cu(OTf)2 (0.05 mmol, 0.018 g), phenylacetylene (2.0 mmol, 0.204 g) and N-(benzoyloxy)piperidine (0.5 mmol, 0.103 g), DBU (1.0 mmol, 0.152 g) in THF (3.0 mL) at room temperature for 12 h, and 84% yield was got (Table 1, entry 4), while increasing the catalyst loading did not improve the product yield (Table 1, entry 5). No reaction occurred in the absence of copper catalyst (Table 1, entry 6). Notably, the copper-catalyzed oxidative amidation reaction did not have external oxygen source (Table 1, entries 1–5). Meanwhile, addition of other oxidants such as benzoyl peroxide (BPO, 1 eq.) or oxygen into the catalytic system decreased the yield obviously (Table 1, entries 7 and 8). Therefore, we surmised that N-(benzoyloxy)piperidine could serve as dually aminating reagent and oxidant. During the study of bases, we noticed that the addition of DBU to the reaction showed the best efficiency (Table 1, entries 9–13). Then, the reaction was investigated in other solvents and THF was found to be the best choice (Table 1, entries 14–16). The reaction temperature was also screened and the reaction yields decreased obviously at lower or higher reaction temperatures (Table 1, entries 17 and 18). Thus, the optimized reaction conditions for the oxidative amidation of terminal alkynes involved using O-benzoyl hydroxylamines as dually aminating reagent and oxidant, Cu(OTf)2 as the catalyst, DBU as the base, THF as the solvent and conducting the reaction at room temperature under nitrogen.
| Entry | Catalyst | Base | Solvent | T (°C) | 1cb (%) |
|---|---|---|---|---|---|
| a General reaction conditions: copper catalyst (0.05 mmol, 10 mol%), phenylacetylene (2.0 mmol, 0.204 g, 4.0 equiv.) and N-(benzoyloxy)piperidine (0.5 mmol, 0.103 g, 1.0 equiv.), base (1.0 mmol, 2.0 equiv.), solvent (3.0 mL), N2, 12 h.b Isolated yield based on 1b.c Cu(OTf)2 (0.1 mmol) was added as the catalyst.d No copper catalyst was used, n.r. = no reaction.e Benzoyl peroxide (BPO, 0.5 mmol) was added.f The reaction was performed under oxygen balloon.g No base was used. | |||||
| 1 | CuI | DBU | THF | RT | 52 |
| 2 | CuBr | DBU | THF | RT | 68 |
| 3 | CuCl2 | DBU | THF | RT | 62 |
| 4 | Cu(OTf)2 | DBU | THF | RT | 84 |
| 5 | Cu(OTf)2 | DBU | THF | RT | 82c |
| 6 | — | DBU | THF | RT | n.r.d |
| 7 | Cu(OTf)2/BPO | DBU | THF | RT | 38e |
| 8 | Cu(OTf)2/O2 | DBU | THF | RT | 46f |
| 9 | Cu(OTf)2 | Na2CO3 | THF | RT | n.r. |
| 10 | Cu(OTf)2 | K2CO3 | THF | RT | n.r. |
| 11 | Cu(OTf)2 | t-BuOK | THF | RT | Trace |
| 12 | Cu(OTf)2 | Et3N | THF | RT | n.r. |
| 13 | Cu(OTf)2 | — | THF | RT | n.r.g |
| 14 | Cu(OTf)2 | DBU | Toluene | RT | 70 |
| 15 | Cu(OTf)2 | DBU | Dioxane | RT | Trace |
| 16 | Cu(OTf)2 | DBU | H2O | RT | n.r. |
| 17 | Cu(OTf)2 | DBU | THF | 35 | 72 |
| 18 | Cu(OTf)2 | DBU | THF | 15 | 24 |
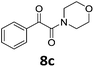
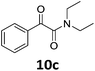
With the optimized reaction conditions in hand, we evaluated the scope of the copper-catalyzed oxidative amidation for the synthesis of α-ketoamides. As shown in Table 2, the reaction yields were slightly influenced by the electronic effect of substituents of terminal alkynes. Terminal alkynes with electron-donating methyl, tertiary butyl and pentyl groups (Table 2, entries 2–4) and electron-withdrawing fluoro and chloro groups (Table 2, entries 5–6) generated the desired products in moderate to good yields. Various O-benzoyl hydroxylamines with acyclic and cyclic N-alkyl substituents were explored by reacting with phenylacetylene (1a) under the optimum reaction conditions (Table 2, entries 7–12). The five-membered cyclic amines 2b bearing a pyrrolidine moiety, the six-membered cyclic amines 1b and 3b bearing a piperidine and morpholine moiety, and the seven-membered cyclic amines 4b bearing a azepane moiety are compatible to the reaction conditions (Table 2, entries 7–9). O-benzoyl hydroxylamines which were derived from N,N-diethyl 5b, N,N-di-n-butyl 6b, and N-n-butyl-N-ethyl 7b amines were smoothly converted to α-ketoamides 10c–14c in 60–76% yields (Table 2, entries 10–14). An O-benzoyl-N-phenylhydroxylamine 8b, which is prepared from primary phenylamine, could not be utilized in this protocol to furnish the desired compound 15c, presumably because of the weak oxidative ability of 8b (Table 2, entry 15). Aliphatic alkynes 1-hexyne 7a also could not reacted with N-(benzoyloxy)piperidine 1b under the optimized reaction condition (Table 2, entry 16).
| Entry | a | b | c | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: copper(II) trifluoromethanesulfonate (0.05 mmol, 0.018 g, 10 mol%), terminal alkyne a (2.0 mmol, 4.0 equiv.) and O-benzoyl hydroxylamine b (0.5 mmol, 1.0 equiv.), DBU (1.0 mmol, 0.152 g, 2.0 equiv.), THF (3.0 mL), N2, RT, 12 h.b Isolated yield based on b. | ||||
| 1 | 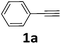 |
 |
 |
84 |
| 2 |  |
1b |  |
82 |
| 3 |  |
1b |  |
64 |
| 4 |  |
1b |  |
72 |
| 5 | 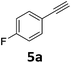 |
1b | 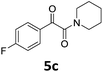 |
80 |
| 6 | 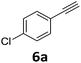 |
1b |  |
78 |
| 7 | 1a |  |
 |
80 |
| 8 | 1a | 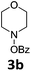 |
 |
72 |
| 9 | 1a |  |
 |
62 |
| 10 | 1a |  |
 |
68 |
| 11 | 1a | 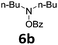 |
 |
76 |
| 12 | 1a | 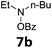 |
 |
60 |
| 13 | 2a | 5b |  |
65 |
| 14 | 6a | 5b |  |
61 |
| 15 | 1a |  |
 |
0 |
| 16 |  |
1b |  |
0 |
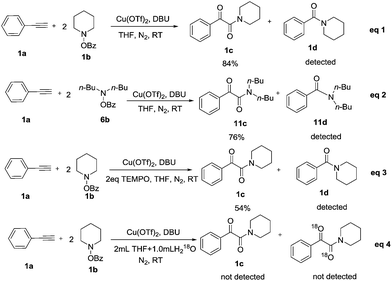
To gain some understanding of the mechanism for this reaction, some control experiments were tentatively examined under the optimized reaction conditions (for mechanism details, see ESI†). We have concluded that oxygen source of the product 1c come from N-(benzoyloxy)piperidine 1b (Table 1, entries 1–8), and the detected phenyl(piperidin-1-yl)methanone 1d also confirmed our speculation (Scheme 2, eqn (1)). We used O-benzoyl-N,N-dibutylhydroxylamine 6b to repeat the reaction, N,N-dibutylbenzamide 11d was also detected from the reaction (Scheme 2, eqn (2)). When the reaction was carried out with two equivalents of 2,2,6,6-tetramethylpiperidine 1-oxyl (TEMPO), it could proceed smoothly and the reaction may not undergo a radical mechanism (Scheme 2, eqn (3)). When 2.0 mL THF and 1.0 mL H218O used as the solvent to repeat the reaction, no product was detected, and the oxygen of the products did not come from the H2O in THF (Scheme 2, eqn (4)).
In summary, we have developed a novel and convenient copper-catalyzed oxidative amidation of terminal alkynes for synthesis of α-ketoamides. This protocol displays attractive features including using easily available O-benzoyl hydroxylamines (BzO-NR1R2) as aminating reagent and oxidant, and conducting the reaction at room temperature. The reaction also exhibits some functional group tolerance and allows for the preparation of a number of α-ketoamides in moderate to good yields. The importance of the α-ketoamides scaffold would render this protocol attractive for both synthetic and medicinal chemistry.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 21402079) and the Shandong Provincial Natural Science Foundation of China (ZR2015PB004).Notes and references
- (a) J. L. Jesuraj and J. Sivaguru, Chem. Commun., 2010, 46, 4791 RSC; (b) Z. Zhang, Q. Zhang, Z. Ni and Q. Liu, Chem. Commun., 2010, 46, 1269 RSC; (c) Y. H. Chen, Y. H. Zhang, H. J. Zhang, D. Z. Liu, M. Gu, J. Y. Li, F. Wu, X. Z. Zhu, J. Li and F. J. Nan, J. Med. Chem., 2006, 49, 1613 CrossRef CAS PubMed; (d) S. Alvarez, R. Alvarez, H. Khanwalkar, P. Germain, G. Lemaire, F. Rodriguez-Barrios, H. Gronemeyer and A. R. de Lera, Bioorg. Med. Chem., 2009, 17, 4345 CrossRef CAS PubMed; (e) L. Yang, D. X. Wang, Z. T. Huang and M. X. Wang, J. Am. Chem. Soc., 2009, 131, 10390 CrossRef CAS PubMed; (f) G. G. Xu and F. A. Etzkorn, Org. Lett., 2010, 12, 696 CrossRef CAS PubMed; (g) D. Tomita, K. Yamatsugu, M. Kanai and M. Shibasaki, J. Am. Chem. Soc., 2009, 131, 6946 CrossRef CAS PubMed; (h) H. Xu and C. Wolf, Angew. Chem., Int. Ed., 2011, 50, 12249 CrossRef CAS PubMed.
- (a) R. P. Singh and J. M. Shreeve, J. Org. Chem., 2003, 68, 6063 CrossRef CAS PubMed; (b) G. M. Dubowchik, V. M. Vrudhula, B. Dasgupta, J. Ditta, T. Chen, S. Sheriff, K. Sipman, M. Witmer, J. Tredup, D. M. Vyas, T. A. Verdoorn, S. Bollini and A. Vinitsky, Org. Lett., 2001, 3, 3987 CrossRef CAS PubMed; (c) A. Chiou, T. Markidis, V. C. Kokotou, R. Verger and G. Kokotos, Org. Lett., 2000, 2, 347 CrossRef CAS PubMed; (d) G. M. Dubowchik, J. L. Ditta, J. J. Herbst, S. Bollini and A. Vinitsky, Bioorg. Med. Chem. Lett., 2000, 10, 559 CrossRef CAS PubMed; (e) J. Chen and R. F. Cunico, J. Org. Chem., 2004, 69, 5509 CrossRef CAS PubMed; (f) R. Hua, H. Takeda, Y. Abe and M. Tanaka, J. Org. Chem., 2004, 69, 974 CrossRef CAS PubMed.
- (a) J. Liu, R. Zhang, S. Wang, W. Sun and C. Xia, Org. Lett., 2009, 11, 1321 CrossRef CAS PubMed; (b) E. R. Murphy, J. R. Martinelli, N. Zaborenko, S. L. Buchwald and K. F. Jensen, Angew. Chem., Int. Ed., 2007, 46, 1734 CrossRef CAS PubMed; (c) M. Iizuka and Y. Kondo, Chem. Commun., 2006, 1739 RSC; (d) T. Fukuyama, S. Nishitani, T. Inouye, K. Morimoto and I. Ryu, Org. Lett., 2006, 8, 1383 CrossRef CAS PubMed.
- (a) C. Zhang, Z. J. Xu, L. R. Zhangand and N. Jiao, Angew. Chem., Int. Ed., 2011, 50, 11088 CrossRef CAS PubMed; (b) F. T. Du and J. X. Ji, Chem. Sci., 2012, 3, 460 RSC; (c) C. Zhang, X. Zong, L. Zhang and N. Jiao, Org. Lett., 2012, 14, 3280 CrossRef CAS PubMed; (d) J. Zhang, Y. Wei, S. Lin, F. Liang and P. Liu, Org. Biomol. Chem., 2012, 10, 9237 RSC; (e) J. M. Grassot, G. Masson and J. Zhu, Angew. Chem., Int. Ed., 2008, 47, 947 CAS; (f) M. Bouma, G. Masson and J. Zhu, J. Org. Chem., 2010, 75, 2748 CrossRef CAS PubMed; (g) W. Wei, Y. Shao, H. Hu, F. Zhang, C. Zhang, Y. Xu and X. Wan, J. Org. Chem., 2012, 77, 7157 CrossRef CAS PubMed; (h) X. Zhang and L. Wang, Green Chem., 2012, 14, 2141 RSC; (i) W. P. Mai, H. H. Wang, Z. C. Li, J. W. Yuan, Y. M. Xiao, L. R. Yang, P. Mao and L. B. Qu, Chem. Commun., 2012, 48, 10117 RSC.
- (a) R. Mossetti, T. Pirali, G. C. Tron and J. Zhu, Org. Lett., 2010, 12, 820 CrossRef CAS PubMed; (b) D. Coffinier, L. E. Kaim and L. Grimaud, Org. Lett., 2009, 11, 1825 CrossRef CAS PubMed; (c) Q. Liu, S. Perreault and T. Rovis, J. Am. Chem. Soc., 2008, 130, 14066 CrossRef CAS PubMed; (d) Z. F. Al-Rashid, W. L. Johnson, R. P. Hsung, Y. Wei, P. Y. Yao, R. Liu and K. Zhao, J. Org. Chem., 2008, 73, 8780 CrossRef CAS PubMed.
- (a) C. Zhang and N. Jiao, J. Am. Chem. Soc., 2010, 132, 28 CrossRef CAS PubMed; (b) M. Kumar, S. Devari, A. Kumar, S. Sultan, Q. N. Ahmed, M. Rizvi and B. A. Shah, Asian J. Org. Chem., 2015, 4, 438 CrossRef CAS.
- (a) N. Matsuda, K. Hirano, T. Satoh and M. Miura, Angew. Chem., Int. Ed., 2012, 51, 3642 CrossRef CAS PubMed; (b) R. P. Rucker, A. M. Whittaker, H. Dang and G. Lalic, J. Am. Chem. Soc., 2012, 134, 6571 CrossRef CAS PubMed; (c) N. Matsuda, K. Hirano, T. Satoh and M. Miura, J. Am. Chem. Soc., 2013, 135, 4934 CrossRef CAS PubMed; (d) Q. Xiao, L. M. Tian, R. C. Tan, Y. Xia, D. Qiu, Y. Zhang and J. B. Wang, Org. Lett., 2012, 14, 4230 CrossRef CAS PubMed; (e) X. Y. Yan, C. Chen, Y. Q. Zhou and C. J. Xi, Org. Lett., 2012, 14, 4750 CrossRef CAS PubMed; (f) N. Matsuda, K. Hirano, T. Satoh and M. Miura, Org. Lett., 2011, 13, 2860 CrossRef CAS PubMed; (g) E. J. Yoo, S. Ma, T. S. Mei, K. S. L. Chan and J. Q. Yu, J. Am. Chem. Soc., 2011, 133, 7652 CrossRef CAS PubMed; (h) C. Grohmann, H. Wang and F. Glorius, Org. Lett., 2013, 15, 3014 CrossRef CAS PubMed; (i) Z. Dong and G. Dong, J. Am. Chem. Soc., 2013, 135, 18350 CrossRef CAS PubMed; (j) J. He, T. Shigenari and J. Q. Yu, Angew. Chem., Int. Ed., 2015, 54, 6545 CrossRef CAS PubMed; (k) M. Shang, S. H. Zeng, S. Z. Sun, H. X. Dai and J. Q. Yu, Org. Lett., 2013, 15, 5286 CrossRef CAS PubMed; (l) K. Wu, Z. L. Fan, Y. Xue, Q. Z. Yao and A. Zhang, Org. Lett., 2014, 16, 42 CrossRef CAS PubMed; (m) T. Matsubara, S. Asako, L. Ilies and E. Nakamura, J. Am. Chem. Soc., 2014, 136, 646 CrossRef CAS PubMed; (n) Q. Gou, G. Liu, Z. N. Liu and J. Qin, Chem.–Eur. J., 2015, 21, 15491 CrossRef CAS PubMed; (o) J. He, T. Shigenari and J. Q. Yu, Angew. Chem., 2015, 127, 6645 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra15219h |
| This journal is © The Royal Society of Chemistry 2016 |