Glucosamine-induced growth of highly distributed TiO2 nanoparticles on graphene nanosheets as high-performance photocatalysts†
Lanbing Gua,
Haijiao Zhang *a,
Zheng Jiaoa,
Minjie Lic,
Minghong Wua and
Yong Lei
*a,
Zheng Jiaoa,
Minjie Lic,
Minghong Wua and
Yong Lei *b
*b
aInstitute of Nanochemistry and Nanobiology, School of Environmental and Chemical Engineering, Shanghai University, Shanghai 200444, P. R. China. E-mail: hjzhang128@shu.edu.cn
bInstitute of Physics & IMN MacroNano, Ilmenau University of Technology, Ilmenau 98693, Germany. E-mail: yong.lei@tu-ilmenau.de
cDepartment of Chemistry, College of Science, Shanghai University, Shanghai 200444, P. R. China
First published on 11th July 2016
Abstract
Here, a composite composed of ultrasmall TiO2 nanoparticles and reduced graphene nanosheets (RGO) has been intelligently designed by a facile glucosamine-assisted hydrothermal strategy in alkaline conditions. The experimental results indicate that the induction of glucosamine plays a vital role in the formation of the desirable TiO2@graphene products, which can effectively regulate the growth and dispersity of TiO2 nanoparticles on the graphene backbone. Consequently, highly dispersed TiO2 nanoparticles with an average diameter of about 13 nm are firmly anchored on graphene. Importantly, the achieved products possess a large surface area of 238.0 m2 g−1, good thermal stability, and high crystallinity along with the increase of calcination temperatures. In particular, the TiO2@graphene obtained through thermal treatment at 700 °C exhibits better photocatalytic activity for photodegradation of methyl orange (MO) than those produced at other calcination temperatures, and it also has a good photocatalytic ability for degrading rhodamine B (RhB) and methyl blue (MB).
1. Introduction
Currently, due to a global concern of environmental pollution, photocatalysis using semiconductor oxides as catalysts has emerged as a green pollution treatment technology.1–4 Among the existing available photocatalysts, TiO2 nanomaterials are playing a more and more vital role by virtue of their fascinating advantages including non-toxicity, low-cost, strong redox ability, and good chemical and thermal stability.5–8 Therefore, TiO2 has served as an efficient photocatalyst in many practical applications such as water disinfection, purification of polluted water and air, hydrogen production and so on.9–12 Nevertheless, the relatively poor charge transport ability and wide band gap (∼3.2 eV) of TiO2 restrict light absorption and accelerate recombination between electrons and hole pairs, thus hindering their further industrial applications.13–15To overcome these difficulties, many approaches have been developed including non-metal doping, noble metal deposition, and constructing composites.16,17 Of all, the combination of TiO2 nanostructures and two dimensional (2D) graphene nanosheets has been deemed as one of the most promising strategies for fabricating high-performance TiO2-based photocatalysts. The reason is mainly that graphene as the support possesses outstanding properties regarding excellent electrical conductivity and large surface area, as well as good chemical stability.18,19 For example, Hu et al. have adopted a one-step approach to synthesize the TiO2/graphene hybrids, which exhibit an enhanced photocatalytic activity for methylene orange (MO) degradation under visible light irradiation.20 Cho et al. also have reported that the TiO2/graphene composites synthesized by a surfactant-assisted method display an improved visible-light photocatalytic activity.21
Although significant progress has been made for the fabrication of TiO2/graphene hybrids, there still exist a few problems restricting the enhancement of their photocatalytic activity. Firstly, during the synthetic process, TiO2 oligomers generated by the rapid hydrolysis of titanium source tend to aggregate into larger TiO2 nanoparticles,22,23 which will greatly decrease the surface–volume ratio of TiO2 and increase the diffusion length of photo-generated electrons/holes, thus resulting in a dissatisfied photocatalytic performance. On the other hand, it is difficult to grow highly-distributed TiO2 nanoparticles on the matrix due to an insufficient use of active sites in graphene,24,25 thus affecting the synergistic effect between TiO2 nanoparticles and graphene. Additionally, a number of expensive surfactants and organic solvents are also been exploited for the generation of more TiO2 nanoparticles on graphene.26,27 Those are environmentally harmful and high costly. Consequently, it is urgently desirable to develop a facile, easy-to-operate, and cost-effective route for preparing TiO2/graphene hybrids with ideal architectures and superior photocatalytic performance.
Herein, we demonstrate an innovative, scalable and facile glucosamine-induced hydrothermal route for synthesis of ultrasmall TiO2 nanoparticles loaded on graphene sheets uniformly in an alkaline medium. In the whole synthetic process, no any organic solvent or surfactant is utilized, meaning a cost-effective and environmentally friendly protocol. The microstructure and the morphological evolution of the products are thoroughly investigated by different techniques. The photocatalytic performances of the products are further evaluated for degradation of three kinds of organic dyes including methyl orange (MO), rhodamine B (RhB) and methylene blue (MB) under UV light irradiation. Especially, the effects of glucosamine on the materials synthesis and the thermal treatment temperature on the photocatalytic properties of composites are also discussed in detail.
2. Experimental section
2.1 Materials
Tetrabutyl orthotitanate (TBOT), tetrapropylammonium hydroxide (TPAOH, 25 wt%), glucosamine, natural graphite powder, phosphorus-pentoxide (P2O5), potassium hypermanganate (KMnO4), hydrogen-peroxide (H2O2), potassium persulfate (K2S2O8), sulfuric acid (H2SO4), methyl orange (MO), rhodamine B (RhB), methyl blue (MB), commercial TiO2 (Degussa P25), the reagents mentioned are all purchased from Sinopharm Chemical Reagent Co., Ltd. All reagents were employed as received without further purification. Distilled water (DI) was used in all experiments.2.2 Synthesis of TiO2@graphene composites
Graphene oxide (GO) nanosheet was firstly prepared from natural graphite powder by a modified Hummers' method.28 Briefly, the reduced graphene oxide (RGO) was further obtained by a thermal reduction of the as-synthesized GO at 500 °C for 2 h in a tube furnace under nitrogen atmosphere.29In a typical procedure, 30 mg of RGO was dispersed in 40 mL of deionized water by sonication for 2 h. Then, 120 mg of glucosamine was added into the RGO suspension solution under stirring for 30 min. Next, 265 μL of TBOT was added into above mixture drop by drop and stirred at room temperature for 1 h to form a homogeneous solution. Subsequently, 350 μL of TPAOH (25 wt%) was added to the mixture solution. Afterwards, the solution was subjected to the flask with magnetic stirring at 80 °C for 6 h. After reaction, the solution was transferred into a Teflon-lined stainless steel autoclave and kept at 180 °C for 24 h. The resulting hybrid was collected by centrifugation, rinsed by DI water and ethanol for three times and dried at 60 °C for 12 h. Finally, the resulting powder was annealed in the temperature range of 400–800 °C for 2 h in a N2-flowing tube furnace. The products obtained were denoted as the TiO2@graphene-T (T represents the calcination temperature). Additionally, the reference products were also prepared with a similar experimental process but without the addition of glucosamine or TPAOH.
2.3 Materials characterizations
The samples were characterized by scanning electron microscopy (SEM, JEOL JSM-6700F), transmission electron microscopy (TEM, JEOL 200CX), high resolution transmission electron microscopy (HRTEM, JSM-2010F). Elemental qualitative analysis was conducted by the energy-dispersive X-ray spectroscopy (EDX, OXFORD INCA) which was mounted in the JSM-6700F. X-ray powder diffraction (XRD) patterns were recorded on the Japan Rigaku D/max-2550 instrument using CuKα radiation (λ = 0.154 nm). Raman spectra were collected using a Renishawin Via system with an excitation wavelength of 514 nm. X-ray photoelectron spectra (XPS) were detected with a Kratos Analytical Axis UltraDLD spectrometer with MgKα radiation. N2 adsorption–desorption isotherms were measured on a QUADRASORB SI surface area & pore size analyzer at 77 K, after the product was annealed at 150 °C for 6 h. The Brunauer–Emmett–Teller (BET) specific surface area was calculated by using the desorption data. Thermal analysis (TG, STA409, Netzsch, Germany) was performed at a heating rate of 10 °C min−1 from room temperature to 800 °C in air atmosphere. UV-Vis absorption spectra were tested on a HITACHI U-3010 UV-Vis spectrophotometer. The photoluminescence (PL) spectra were obtained on a Hitachi F-7000 fluorescence spectrophotometer.2.4 Photodegradation tests
The photocatalytic measurements were carried out in a SGY-IB photochemical reactor under UV light irradiation (λ < 400 nm) with a 300 W Hg lamp as light source. To exhibit the effectiveness of the products in photocatalysis, the photodegradation behaviors of different organic dyes including methyl orange (MO), rhodamine B (RhB), and methyl blue (MB) in aqueous solution were investigated. Here, taking MO as the example, typically, 10 mg of TiO2@graphene catalyst was added to 50 mL of aqueous solution containing 10 mg L−1 MO in a 100 mL beaker. After ultrasonic treatment for 20 min, the mixture solution was stirring in the dark at ambient temperature for 60 min to achieve adsorption–desorption equilibrium. Then, 3–5 mL of the suspension was extracted to detect the initial concentration of MO solution, which was recorded as the initial concentration C0. During the following 20 min reaction process, vigorous agitation was performed to ensure the uniform irradiation of the TiO2@graphene photocatalyst suspension. Every 5 min 3–5 mL of the suspension was extracted and next centrifuged immediately to separate catalysts. In the end, the solution was measured using a Hitachi U-3010 UV-Vis spectrophotometer to determine the concentration of remaining dye over time, which was recorded as Ct.3. Results and discussion
A schematic illustration for the growth process of TiO2@graphene composites through a facile glucosamine-induced hydrothermal route is presented in Scheme 1. In brief, when glucosamine possessing amino groups is added to the RGO solution, it is likely to preferentially adsorb onto the graphene sheets with negative charges owing to powerful electrostatic interaction between RGO and the glucosamine with positive charges.30,31 Thus, the amino groups enable glucosamine to serve as the linker. Simultaneously, the glucosamine also plays a role of inhibitor for slowing the growth of TiO2 nanocrystals and ensures an uniform deposition of amorphous TiO2 precursor (titanium oligomers) with the hydrolysis of TBOT.32 Afterwards, the Ti4+ ions begin to slowly hydrolyze at the room temperature and generate some titanium species. At the aging stage, partial amorphous TiO2 precursor is converted to anatase phase under basic condition of TPAOH and a number of small TiO2 nanoparticles are successfully decorated onto the surface of graphene. All these make TiO2 nanocrystals to more easily grow on the graphene. Through further hydrothermal and thermal treatments, anatase TiO2 nanoparticles with high crystallinity and good thermal stability will highly disperse onto the grapheme matrix.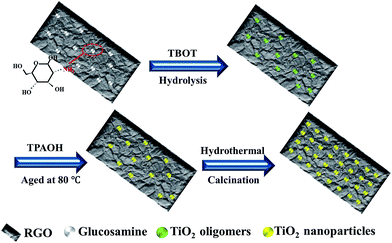 | ||
| Scheme 1 Schematic illustration for the growth process of highly distributed TiO2@graphene composites. | ||
3.1 Structure and morphology
SEM and TEM techniques are used to examine the microstructure and morphology of the products. As shown in Fig. 1a and b, the typical SEM images at different magnification of the as-synthesized product after annealing at 700 °C indicate that a lot of small TiO2 nanoparticles uniformly disperse on the surface of grapheme nanosheets. Fig. 1c and d are the corresponding TEM images of the product. From the images, the composite is composed of a large amount of TiO2 nanoparticles and wrinkle grapheme nanosheets, demonstrating a well-defined morphology and good monodispersity. Moreover, no dissociative TiO2 nanoparticles or unloaded graphene sheets can be obviously seen. In addition, the graphene nanosheets are very thin and nearly transparent, which seems like single layer nanosheet of high quality. The RGO nanosheets as the support also prevent the aggregation of TiO2 nanoparticles to a certain extent. The result is in agreement with the SEM analysis, confirming a successful assembly of TiO2@graphene composite. Seen from the EDX analysis (Fig. 1e), the dominant elements including Ti, C and O are detected, indicating a high chemical purity of the products, while the Cu element is derived from the supporting grid for SEM analysis. And the size of TiO2 nanoparticles is calculated according to the particle size distribution in Fig. 1f, and most of them have a small diameter of about 13 nm. | ||
| Fig. 1 (a, b) SEM images, (c, d) TEM images at different magnification, (e) the corresponding EDX pattern, and (f) particle size distribution of the typical TiO2@graphene product annealed at 700 °C. | ||
Fig. 2a and b show the typical HRTEM images of the TiO2@graphene product. It is clearly distinguished from TiO2 nanoparticles and graphene, indicating TiO2 well-grown on the graphene matrix, which can further strengthen synergistic interactions between the TiO2 nanoparticles and graphene.33 The image notes distinctly a high crystalline nature with a lattice spacing of 0.352 nm, corresponding to the (101) planes of TiO2.34 For further investigating the composition and distribution of the sample, the STEM image and the corresponding elemental mapping analysis are also conducted, as depicted in Fig. 2c–f. The obtained TiO2@graphene is mainly composed of Ti, C and O elements, which are homogeneously distributed throughout the sample. That reveals the TiO2 nanoparticles are highly dispersed in the graphene, in accordance with the EDX result.
 | ||
| Fig. 2 (a, b) HRTEM images, (c) STEM image of TiO2@graphene-700 product, and (d–f) the corresponding elemental mapping images. | ||
To insight into the roles of glucosamine and TPAOH, the four controlled experiments are designed while keeping other conditions the same. The morphological evolution of four products is investigated by TEM, as displayed in Fig. S1.† At the absence of glucosamine, only a small amount of TiO2 nanoparticles are loaded on the graphene and most of them are free (Fig. S1a†). When glucose is added substituting for glucosamine, we can observe from Fig. S1b† that the morphology is similar to the typical product we synthesized except for some agglomeration. The results suggest that glucosamine can serve as the linker agent due to the specific amino groups in molecular structure relative to that of glucose, resulting in a powerful electrostatic attraction between glucosamine and graphene. Moreover, many hydroxyl groups on the glucosamine surface can make TiO2 grow on the graphene more easily. On the other hand, glucosamine also acts as an inhibitor for preventing the aggregation of TiO2 nanocrystals.35–37 Without TPAOH, bulk TiO2 particles seriously get together on the graphene, generating an ill-defined morphology (Fig. S1c†). However, when using equal molar NaOH instead of TPAOH (Fig. S1d†), the TiO2 formed on the graphene is small nanoparticles although the uniformity is no good than that one obtained in TPAOH system. From the results, we can draw a conclusion that an alkaline system is benefit for the formation of ultrasmall TiO2 particles and the ideal morphology. So the structure of the products can be adjusted by both TPAOH and glucosamine.
Previous studies have proved that the calcination temperature has a major impact on the structure and performance of the nanomaterials.38–40 To this end, a series of TiO2@graphene composites are obtained at different annealing temperature from 400 to 800 °C (Fig. S2†). It can be seen from Fig. S2a–c† that the morphologies of the products annealed below 800 °C have no big changes and apparent sintering phenomenon doesn't occur during the thermal treatment, suggesting a good thermal stability of the products. Upon the temperature is increased to 800 °C, TiO2 nanoparticles begin to sinter seriously and 2D structure of graphene is also destroyed, which may affect the photocatalytic properties.
Based on the TEM observations, the effect of the calcination temperature on the crystallinity of TiO2 samples is further investigated. XRD technique is employed to monitor the microscopic changes in terms of the crystal growth or possible phase transition. As noted in Fig. 3a, the characteristic diffraction peaks for all TiO2@graphene samples are at 25.3, 37.8, 48.0, 53.9, 55.1, 62.7, 68.8, 70.3, and 75.0° corresponding to the planes of (101), (004), (200), (105), (211), (204), (116), (220), and (215) of anatase TiO2 (JCPDS card no. 21-1272).41 No typical diffraction peaks belonging to the separate grapheme is observed in the composites. The reason can be deduced that the main characteristic peak of RGO is close to the main peak of anatase TiO2 at 25.3°.42 Notably, the anatase phase of TiO2 materials is kept well in the range of calcination temperatures from 400 to 800 °C, in which no phase transition to rutile happens and no impurities are found even at the temperature as high as 800 °C. It is generally acknowledged that the anatase phase of TiO2 has a better photocatalytic activity than rutile, because it includes a higher reduction potential and a lower recombination rate of photogenerated carriers in comparison to the rutile products.43 Beyond that, the intensity of anatase peaks gradually increases as the temperature increases, suggesting an enhancement of crystallization. That is in favor of the photocatalytic process, since good crystallinity is helpful for the charge transfer of the excited electron and electron hole pair, thus decreasing the recombination rate.44
 | ||
| Fig. 3 (a) XRD patterns of various TiO2@graphene composites calcined at different temperature, and (b) Raman spectra of various products. | ||
Fig. 3b shows the Raman spectra of TiO2@graphene, bare TiO2 nanoparticles and GO. Clearly, five typical peaks of anatase TiO2 at 149, 199, 397, 513 and 639 cm−1 are observed for the TiO2@graphene composite and bare TiO2 nanoparticles. At the same time, two characteristic bands of carbon appear at 1352 cm−1 (D band) and at 1598 cm−1 (G band) for both TiO2@graphene and bare GO. The D band presents the edges defects and disordered carbon atoms, whereas the G band is ascribed to the zone centre E2g mode, corresponding to the ordered sp2-bonded carbon atoms. The intensity change of between D band and G band, namely ID/IG, is used to monitor the structural change of the grapheme nanosheets. The ID/IG of TiO2@graphene is 1.1, higher than that of bare GO (0.87). That indicates the defects are reduced after the removal of the oxygenous groups on the GO after thermal treatment, suggesting that the GO has reverted to graphene and the electronic interactions between ultrasmall TiO2 nanoparticles and graphene substrate, which is desirable for photoelectron transport between TiO2 semiconductor and graphene.
N2 adsorption–desorption isotherm of TiO2/graphene is collected, as illustrated in Fig. 4a. The product annealed at 700 °C shows a distinct hysteresis loop at the relatively pressure p/p0 = 0.4–1.0, corresponding to a type IV isotherm, revealing its mesoporous structure. The specific surface area of TiO2@graphene-700 is calculated to be about 238.0 m2 g−1. The high specific surface area of the composite can provide more active sites and effectively facilitate the adsorption and subsequent photocatalytic process. The corresponding pore size distribution plot from Fig. 4b shows that the sample has a small mesoporous mainly peaked at 3.83 nm, which may be mainly attributed to the aggregation among the TiO2 nanoparticles, in accordance with the previous study.32 The thermal decomposition behaviors of TiO2@graphene products are measured by TG analysis. As shown in Fig. 4c, the product synthesized without glucosamine has a weight loss of 61.62% between 360 and 480 °C (predominantly DTA peak at around 462.43 °C). The weight loss at first is due to the absorbed water in the product and the rest loss is mainly attributed to the removal of the carbon. Besides that, no notable weight loss is observed at above 500 °C. Therefore, the residuals and other organic components can be completely removed after annealing at 700 °C. In addition, the typical product we synthesized possesses a weight loss of 43.24%, as shown in Fig. 4d, in which the DTA peak is at around 560.68 °C, and the weight loss is between 500 and 600 °C. By contrast, the TiO2 contents in the composite synthesized without glucosamine (only 38.38%) is much little than 56.76% of the typical product, meaning glucosamine plays a crucial role for the effective growth of TiO2 nanoparticles on the graphene.
The typical XPS spectra of as-prepared TiO2@graphene sample are displayed in Fig. 5. Seen from Fig. 5a, the full scale XPS survey spectrum shows the C, O, and Ti photoelectron lines, consistent with the EDX pattern. Ti2p XPS spectrum in Fig. 5b exhibits two characteristic peaks at 459.2 and 464.79 eV, corresponding to the Ti2p3/2 and Ti2p1/2 spin–orbit peaks of TiO2.45–47 In the spectrum of O1s, the broad peak can be fit into two symmetric peaks at 530.2 and 533.3 eV, which are contributed to the Ti–O–Ti and Ti–O–C bond, respectively, presenting a strong covalent bond connection between graphene and TiO2. For investigating the carbon states in the composite, spectrum of C1s is measured, as shown in Fig. 5d. Deconvolution of the C1s peak in the XPS spectrum performs four types of carbon bonds, namely, C–C (284.51 eV), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (284.84 eV), –COOH (288.6 eV) and C–O (286.5 eV).48 The small peak corresponding to the oxygenate groups suggests a transformation from GO to RGO. It is believed that a small amount of residual oxygenate groups on RGO are favorable for maintaining a good dispersion of the nanoparticles.49,50
C (284.84 eV), –COOH (288.6 eV) and C–O (286.5 eV).48 The small peak corresponding to the oxygenate groups suggests a transformation from GO to RGO. It is believed that a small amount of residual oxygenate groups on RGO are favorable for maintaining a good dispersion of the nanoparticles.49,50
 | ||
| Fig. 5 XPS spectra of TiO2@graphene-700: (a) survey scan spectrum, (b) Ti2p, (c) O1s, and (d) C1s spectra. | ||
3.2 Photocatalytic properties
Since the morphology, particle size, crystal phase, and crystallinity are responsible for the photocatalytic performance of materials. Thereby, the photocatalytic activities of a series of TiO2@graphene samples annealed at different temperatures are tested by photodegradation of methyl orange (MO). Fig. 6a shows the degradation results of MO under UV light over various photocatalysts. Compared with uncalcined sample (namely, precursor), the calcined TiO2@graphene samples demonstrate better catalytic performances owing to their enhanced crystallinity. Nontheless, the product calcined at 800 °C is an exception, its photocatalytic performance is the worst among them because of seriously sintered TiO2 nanoparticles and destroyed grapheme structure in the composite, which is consistent with the above observation from TEM image (Fig. S2d†). It is obvious that TiO2@graphene-700 catalyst shows the best photocatalytic performance in all samples and 96.23% of MO can be degradated within 15 min, which is comparable to that of Degussa P25 (about 98.97% of MO can be degradated within 15 min). The current photocatalytic degradation rate may be sequenced as follows: TiO2@graphene-700 > TiO2@graphene-600 > TiO2@graphene-500 > TiO2@graphene-400 > precursor > TiO2@graphene-800. Fig. 6b records the spectral changes of MO solution induced by the TiO2@graphene-700 photocatalyst, where the biggest absorbance peak is at 464 nm. The color change is a good indicator for tracking the reaction process. As can be seen from the inset digital photos, the resultant solutions change from orange to almost colorless. Significantly, the TiO2@graphene-700 photocatalyst also exhibits a good cycling stability, the degradation rates are scarcely changed after 5 cycles, as observed in Fig. 6c. At the last cycle, near all MO in the solution can still be degraded within 20 min.To confirm the universal superiority of our materials, other model dyes such as MB and RhB are also degraded by using TiO2@graphene-700 as the photocatalyst, as shown in Fig. 6d. In two cases, nearly all MB and RhB can be completely degraded within 20 min, and about 97.13% and 95.61% of MB and RhB can be removed from the solution. Fig. 6e and f show the temporal evolution of the spectral changes of MB and RhB solutions, corresponding to the characteristic peaks at 664 and 552 nm, respectively. Similarly, the color changes from bright color to nearly colorless record the reaction process according to the digital images.
From the above photocatalytic results, we deduce the superior photocatalytic performance of TiO2@graphene-700 can be explained as follows: (1) ultrasmall TiO2 nanoparticles loading uniformly on the surface of graphene lead to the larger specific surface areas (238.0 m2 g−1), thus offering more active sites for adsorption of organic dyes; (2) the mesostructure constructed by a variety of small TiO2 nanoparticles is benefit to the diffusion and transport of organic dyes during the photocatalytic process; (3) the successful assembly of TiO2@graphene composite can improve the photon utilization efficiency due to well photo absorbed ability and excellent electrical conductivity of graphene. In the photocatalytic reaction, when photo-generated e−/h+ pairs appear in ultrasmall TiO2 nanoparticles, the electrons on the TiO2 surface transfer quickly to the graphene, which can effectively slow down the recombination of e−/h+ pairs (Scheme 2 and Fig. S3†),20,40 thus enhancing the photocatalytic performance.51,52
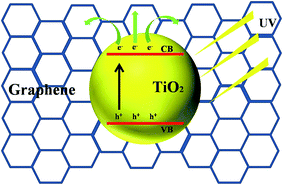 | ||
| Scheme 2 Schematic diagram for the photodegradation towards organic dyes over TiO2@graphene composites under UV irradiation. | ||
4. Conclusions
In the study, we provide an effective one-pot hydrothermal method for preparing TiO2@graphene nanohybrids by using glucosamine as a morphology-controller agent and auxiliary dispersant. Such a protocol can achieve the TiO2@graphene of high quality, in which ultrasmall TiO2 nanoparticles with good monodispersity are homogeneously attached onto the graphene surface, resulting in a large surface area of 238.0 m2 g−1. Meanwhile, the structures and photocatalytic performances of TiO2@graphene composites are greatly affected by the calcination temperature. In particular, the product annealed at 700 °C exhibits the best photocatalytic ability for the decomposition of MO in comparison to commercial P25. Additionally, the product also shows good photocatalytic effects for MB and RhB. Consequently, the notable enhancement of photocatalytic performance for TiO2@graphene composites mainly comes from their large surface area, high crystallinity, good dispersity, and the synergistic effects between the two components. The promising results suggest that the glucosamine-induced growth approach for ultrasmall TiO2 nanoparticles anchored onto the graphene nanosheets can be utilized for achieving other graphene-based hybrid materials.Acknowledgements
The work is supported by the National Natural Science Foundation of China (NSFC No, 11275121, 21471096, and 21371116), and Program for Innovative Research Team in University (IRT13078).Notes and references
- H. Tong, S. Ouyang, Y. Bi, N. Umezawa, M. Oshikiri and J. Ye, Nano-photocatalytic materials: possibilities and challenges, Adv. Mater., 2012, 24, 229–251 CrossRef CAS PubMed.
- Y. Miao, H. J. Zhang, S. Yuan, Z. Jiao and X. D. Zhu, Preparation of flower-like ZnO architectures assembled with nanosheets for enhanced photocatalytic activity, J. Colloid Interface Sci., 2016, 462, 9–18 CrossRef CAS PubMed.
- Y. Mi, L. Y. Wen, Z. J. Wang, D. W. Cao, Y. G. Fang and Y. Lei, Building of anti-restack 3D BiOCl hierarchitecture by ultrathin nanosheets towards enhanced photocatalytic activity, Appl. Catal., B, 2015, 176, 331–337 CrossRef.
- H. J. Zhang, M. X. Ying, R. M. Gao, L. Hu, Z. Jiao and X. D. Zhu, Carbon-mediated fabrication of core-shell structured SnO2@TiO2 nanocomposite with excellent photocatalytic performance, RSC Adv., 2015, 5, 58439–58448 RSC.
- Q. Zhang, J. B. Joo, Z. Lu, M. Dahl, D. Q. L. Oliveira, M. Ye and Y. Yin, Self-assembly and photocatalysis of mesoporous TiO2 nanocrystal clusters, Nano Res., 2010, 4, 103–114 CrossRef.
- Y. Lin, S. Zhou, X. Liu, S. Sheehan and D. Wang, TiO2/TiSi2 heterostructures for high-efficiency photoelectrochemical H2O splitting, J. Am. Chem. Soc., 2009, 131, 2772–2773 CrossRef CAS PubMed.
- T. Salim, Z. Yin, S. Sun, X. Huang, H. Zhang and Y. M. Lam, Solution-processed nanocrystalline TiO2 buffer layer used for improving the performance of organic photovoltaics, ACS Appl. Mater. Interfaces, 2011, 3, 1063–1067 CAS.
- F. P. Cai, Y. B. Tang, H. Shen, C. Wang, A. Ren, L. Xiao, S. Gu and W. W. D. Shi, Graphene oxide-assisted synthesis and photocatalytic hydrogen production of mix-phase titanium dioxide (TiO2) nanosheets, CrystEngComm, 2015, 17, 1086–1091 RSC.
- O. K. Varghese, M. Paulose and C. A. Grimes, Long vertically aligned titania nanotubes on transparent conducting oxide for highly efficient solar cells, Nat. Nanotechnol., 2009, 4, 592–597 CrossRef CAS PubMed.
- K. Wang, M. Wei, M. A. Morris, H. Zhou and J. D. Holmes, Mesoporoustitania nanotubes: their preparation and application as electrode materials for rechargeable lithium batteries, Adv. Mater., 2007, 19, 3016–3020 CrossRef CAS.
- D. Chen, F. Huang, Y. B. Cheng and R. A. Caruso, Mesoporous anatase TiO2 beads with high surface areas and controllable pore sizes: a superior candidate for high-performance dye-sensitized solar cells, Adv. Mater., 2009, 21, 2206–2210 CrossRef CAS.
- E. L. Ratcliff, B. Zacher and N. R. Armstrong, Selective interlayers and contacts in organic photovoltaic cells, J. Phys. Chem. Lett., 2011, 2, 1337–1350 CrossRef CAS PubMed.
- D. Chen and R. A. Caruso, Recent progress in the synthesis of spherical titania nanostructures and their applications, Adv. Funct. Mater., 2013, 23, 1356–1374 CrossRef CAS.
- H. J. Zhang, G. D. Du, W. Q. Lu, L. L. Cheng, X. D. Zhu and Z. Jiao, Porous TiO2 hollow nanospheres: synthesis, characterization and enhanced photocatalytic properties, CrystEngComm, 2012, 14, 3793–3801 RSC.
- H. Li, B. Liu, Y. H. Wang, S. Yin, X. L. Ma, X. C. Wang, Q. S. Wu, R. F. Shen and H. Chen, A facile one-step hydrothermal synthesis of a B-doped graphene/rod-shaped TiO2 nanocomposite, RSC Adv., 2014, 4, 37992–37997 RSC.
- S. Liu, E. Guo and L. Yin, Tailored visible-light driven anatase TiO2 photocatalysts based on controllable metal ion doping and ordered mesoporous structure, J. Mater. Chem., 2012, 22, 5031–5041 RSC.
- L. F. He, N. Du, C. D. Wang, X. F. Chen and W. J. Zhang, A facile synthesis of graphene-supported mesoporous TiO2 hybrid sheets with uniform coverage and controllable pore diameters, Microporous Mesoporous Mater., 2015, 206, 95–101 CrossRef CAS.
- R. Kumar, R. K. Singh, P. K. Dubey, D. P. Singh, R. M. Yadav and R. S. Tiwari, Hydrothermal synthesis of a uniformly dispersed hybrid graphene–TiO2 nanostructure for optical and enhanced electrochemical applications, RSC Adv., 2015, 5, 7112–7120 RSC.
- S. Jiang, R. W. Wang, M. J. Pang, H. B. Wang, S. J. Zeng, X. Z. Yue, L. Ni, Y. R. Yu, J. Y. Dai, S. L. Qiu and Z. T. Zhang, Assembling porous carbon-coated TiO2(B)/anatase nanosheets on reduced graphene oxide for high performance lithium-ion batteries, Electrochim. Acta, 2015, 182, 406–415 CrossRef CAS.
- C. Y. Hu, F. Chen, T. W. Lu, C. J. Lian, S. Z. Zheng, Q. H. Hu, S. W. Duo and R. B. Zhang, Water-phase strategy for synthesis of TiO2–graphene composites with tunable structure for high performance photocatalysts, Appl. Surf. Sci., 2014, 317, 648–656 CrossRef CAS.
- K. M. Cho, K. H. Kim, H. O Choi and H. T. Jung, A highly photoactive, visible-light-driven graphene/2D mesoporous TiO2 photocatalyst, Green Chem., 2015, 17, 3972–3978 RSC.
- W. Li, F. Wang, Y. P. Liu, J. X. Wang, J. P. Yang, L. J. Zhang, A. A. Elzatahry, D. A. Dahyan, Y. Y. Xia and D. Y. Zhao, General strategy to synthesize uniform mesoporous TiO2/graphene/mesoporous TiO2 sandwich-like nanosheets for highly reversible lithium storage, Nano Lett., 2015, 15, 2186–2193 CrossRef CAS PubMed.
- M. M. Zhen, M. Q. Sun, G. D. Gao, L. Liu and Z. Zhou, Synthesis of mesoporous wall-structured TiO2 on reduced graphene oxide nanosheets with high rate performance for lithium-ion batteries, Chem.– Eur. J., 2015, 21, 5317–5322 CrossRef CAS PubMed.
- W. J. Liu, J. Y. Cai, Z. X. Ding and Z. H. Li, TiO2/RGO composite aerogels with controllable and continuously tunable surface wettability for varied aqueous photocatalysis, Appl. Catal., B, 2015, 174, 421–426 CrossRef.
- M. C. Long, Y. L. Qin, C. Chen, X. Y. Guo, B. H. Tan and W. M. Cai, Origin of visible light photoactivity of reduced graphene oxide/TiO2 by in situ hydrothermal growth of undergrown TiO2 with graphene oxide, J. Phys. Chem. C, 2013, 117, 16734–16741 CAS.
- X. Xin, X. F. Zhou, J. H. Wu, X. Y. Yao and Z. P. Liu, Scalable synthesis of TiO2/grapheme nanostructured composite with high-rate performance for lithium ion batteries, ACS Nano, 2012, 6, 11035–11043 CrossRef CAS PubMed.
- C. Z. Zhu, S. J. Guo, P. Wang, L. Xing, Y. X. Fang, Y. M. Zhai and S. J. Dong, One-pot, water-phase approach to high-quality graphene/TiO2 composite nanosheets, Chem. Commun., 2010, 46, 7148–7150 RSC.
- Y. Xu, H. Bai, G. Lu, C. Li and G. Shi, Flexible graphene films via the filtration of water-soluble noncovalent functionalized graphene sheets, J. Am. Chem. Soc., 2008, 130, 5856–5857 CrossRef CAS PubMed.
- H. J. Zhang, P. P. Xu, G. D. Du, Z. W. Chen, K. Oh, D. Y. Pan and Z. Jiao, A facile one-step synthesis of TiO2/graphene composites for photodegradation of methyl orange, Nano Res., 2011, 4, 274–283 CrossRef CAS.
- W. J. Liu, J. G. Cai, Z. X. Ding and Z. H. Li, TiO2/RGO composite aerogels with controllable and continuously tunable surface wettability for varied aqueous photocatalysis, Appl. Catal., B, 2015, 174, 421–426 CrossRef.
- G. H. Qin, H. J. Zhang and C. Y. Wang, Ultrasmall TiO2 nanoparticles embedded in nitrogen doped porous graphene for high rate and long life lithium ion batteries, J. Power Sources, 2014, 272, 491–500 CrossRef CAS.
- J. X. Qiu, C. Lai, Y. Z. Wang, S. Li and S. Q. Zhang, Resilient mesoporous TiO2/graphene nanocomposite for high rate performance lithium-ion batteries, Chem. Eng. J., 2014, 256, 247–254 CrossRef CAS.
- L. L. Tan, W. J. Ong, S. P. Chai and A. R. Mohamed, Noble metal modified reduced graphene oxide/TiO2 ternary nanostructures for efficient visible-light-driven photoreduction of carbon dioxide into methane, Appl. Catal., B, 2015, 166, 251–259 CrossRef.
- A. J. Wang, W. Yu, Y. Fang, Y. L. Song, D. Jia, L. L. Long, M. P. Cifuentes, M. G. Humphrey and C. Zhang, Facile hydrothermal synthesis and optical limiting properties of TiO2-reduced graphene oxide nanocomposites, Carbon, 2015, 89, 130–141 CrossRef CAS.
- B. C. Qiu, M. Y. Xing and J. L. Zhang, Mesoporous TiO2 nanocrystals grown in situ on graphene aerogels for high photocatalysis and lithium-ion batteries, J. Am. Chem. Soc., 2014, 136, 5852–5855 CrossRef CAS PubMed.
- P. H. Shao, J. Y. Tian, W. X. Shi, S. S. Gao and F. Y. Cui, Eco-friendly one-pot synthesis of ultradispersed TiO2 nanocrystals/graphene nanocomposites with high photocatalytic activity for dye degradation, J. Mater. Chem. A, 2015, 3, 19913–19919 CAS.
- P. Liang, H. J. Zhang, Y. J. Bai, Y. Y. Feng and Y. Wang, A designed TiO2/carbon nanocomposite as a high-efficiency lithium-ion battery anode and photocatalyst, Nanotechnology, 2015, 10, 871–878 Search PubMed.
- Z. S. Wu, G. M. Zhou, L. C. Yin, W. Ren, F. Li and H. M. Cheng, Graphene/metal oxide composite electrode materials for energy storage, Nano Energy, 2012, 1, 107–131 CrossRef CAS.
- S. L. Yang, C. Y. Cao, P. P. Huang, L. Peng, Y. B. Sun, F. Wei and W. G. Song, Sandwich-like porous TiO2/reduced graphene oxide (rGO) for high-performance lithium-ion batteries, J. Mater. Chem. A, 2015, 3, 8701–8705 CAS.
- N. Ma, Y. W. Qiu, Y. C. Zhang, H. Y. Liu, Y. N. Yang, J. W. Wang, X. Y Li and C. Cui, Reduced graphene oxide enwrapped pinecone-liked Ag3PO4/TiO2 composites with enhanced photocatalytic activity and stability under visible light, J. Alloys Compd., 2015, 648, 818–825 CrossRef CAS.
- M. G. Wang, J. Han, H. X. Xiong, R. Guo and Y. D. Yin, Nanostructured hybrid shells of r-GO/AuNP/m-TiO2 as highly active photocatalysts, ACS Appl. Mater. Interfaces, 2015, 7, 6909–6918 CAS.
- J. Zhang, Z. P. Zhu, Y. P. Tang and X. L. Feng, Graphene encapsulated hollow TiO2 nanospheres: efficient synthesis and enhanced photocatalytic activity, J. Mater. Chem. A, 2013, 1, 3752–3756 CAS.
- R. R. Liu, W. J. Guo, B. Sun, J. L. Pang, M. S. Pei and G. W. Zhou, Composites of rutile TiO2 nanorods loaded on graphene oxide nanosheet with enhanced electrochemical performance, Electrochim. Acta, 2015, 156, 274–282 CrossRef CAS.
- D. H. Wang, H. J. Zhang, J. X. Guo, H. J. Xu, X. D. Zhu and Z. Jiao, Template-free fabrication of rattle-type TiO2 hollow microspheres with superior photocatalytic performance, RSC Adv., 2014, 4, 37311–37319 RSC.
- D. L. Shieh, Y. S. Lin, J. H. Yeh, S. C. Chen, B. C. Lin and J. L. Lin, N-doped, porous TiO2 with rutile phase and visible light sensitive photocatalytic activity, Chem. Commun., 2012, 48, 2528–2530 RSC.
- G. H. Qin, X. Y. Zhang and C. Y. Wang, Design of nitrogen doped graphene grafted TiO2 hollow nanostructures with enhanced sodium storage performance, J. Mater. Chem. A, 2014, 2, 12449–12458 CAS.
- L. Peng, H. J. Zhang, Y. J. Bai, Y. Y. Feng and Y. Wang, A designed TiO2/carbon nanocomposite as a high-efficiency lithium-ion battery anode and photocatalyst, Chem.– Eur. J., 2015, 21, 14871–14878 CrossRef CAS PubMed.
- M. M. Zhen, S. Q. Guo, G. D. Gao, Z. Zhou and L. Liu, TiO2-B nanorods on reduced graphene oxide as anode materials for Li-ion batteries, Chem. Commun., 2015, 51, 507–510 RSC.
- J. S. Lee, K. H. You and C. B. Park, Highly photoactive, low bandgap TiO2 nanoparticles wrapped by graphene, Adv. Mater., 2012, 24, 1084–1088 CrossRef CAS PubMed.
- G. F. Gu, J. L. Cheng, X. D. Li, W. Ni, Q. Guan, G. X. Qu and B. Wang, Facile synthesis of graphene supported ultralong TiO2 nanofibers from the commercial titania for high performance lithium-ion batteries, J. Mater. Chem. A, 2015, 3, 6642–6648 CAS.
- Y. H. Tang, G. Zhang, C. B. Liu, S. L. Luo, X. L. Xu, L. Chen and B. G. Wang, Magnetic TiO2–graphene composite as a high-performance and recyclable platform for efficient photocatalytic removal of herbicides from water, J. Hazard. Mater., 2013, 252–253, 115–122 CrossRef CAS PubMed.
- Y. Y. Liang, H. L. Wang, H. S. Casalongue, Z. Chen and H. J. Dai, TiO2 nanocrystals grown on graphene as advanced photocatalytic hybrid materials, Nano Res., 2010, 3, 701–705 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra15028d |
| This journal is © The Royal Society of Chemistry 2016 |


