Controllable wetting state high adhesion hydrophobic surface†
Abstract
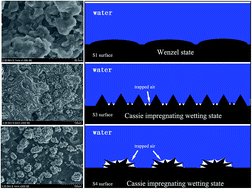
High adhesion hydrophobic surfaces with high apparent contact angles were achieved by a convenient approach. The morphology of the high adhesion surface both at the micrometre scale and at the nanometre scale can be changed by variation in the amount of carboxyl functionalized multi-wall carbon nanotubes. The contact angle values for the as-prepared surfaces are different, increasing from about 122.8° to 151.4° by increasing the amount of MWCNT–COOH. The as-prepared surface with a tunable adhesion can be achieved and the maximum volume of the water droplet adhered on the surface can be adjusted from about 5 μL to 35 μL. The variation in the contact angle and the maximum volume of the water droplet adhered on the surface are both attributed to the different morphologies of the surfaces. To analyze the correlation between the surface morphology and water droplet wetting, we suggest that the wetting state of the water droplet on the surface is changed, which results from the appearance of the micro/nano scale structure. Furthermore, a process of water droplet no-loss transport was achieved by the difference in adhesion of the surface.

 Please wait while we load your content...
Please wait while we load your content...