Brönsted acid-mediated annulations of 1-cyanocyclopropane-1-carboxylates with arylhydrazines: efficient strategy for the synthesis of 1,3,5-trisubstituted pyrazoles†
Abstract
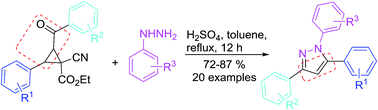
1-Cyanocyclopropane-1-carboxylates are reacted with arylhydrazines to afford 1,3,5-trisubstituted pyrazoles under the influence of a Brönsted acid. Formally, this transformation can be regarded as an annulation of three-membered rings with α-carbonyl and hydrazines. This newly efficient method provides access to a variety of structurally diverse pyrazole derivatives. The structures of five typical products were confirmed by X-ray crystallography.

 Please wait while we load your content...
Please wait while we load your content...