High fluorescence intensity poly(aryl ether ketone)s containing tetraphenylethylene moieties: preparation, characterization and fluorescent properties
Feng Li,
Jiashuang Luan,
Shuling Zhang,
Yuchao Luo,
Yinlong Du,
Cong Gao and
Guibin Wang*
Alan G. MacDiarmid Institute, Department of Chemistry, Jilin University, No. 2699, Qianjin street, Changchun, P. R. China. E-mail: wgb@jlu.edu.cn; Fax: +86 431 85168889; Tel: +86 431 85168889
First published on 25th August 2016
Abstract
A novel monomer containing tetraphenylethylene-(2,6-difluorophenyl)(4-(3-(4-1,2,2-triphenyl vinyl)phenoxy)propyl)phenyl)methanone (2F-TPE) was designed and synthesized by reaction of (4-(3-bromopropyl)phenyl)(2,6-difluorophenyl)methanone and 1-(4-hydroxyphenyl)-1,2,2-triphenylethene. A series of high fluorescence intensity poly(aryl ether ketone) copolymers containing tetraphenylethylene (TPE–PAEK) were prepared, which is the first report of preparing PAEK featuring tetraphenylethylene groups by polycondensation. The fluorescence and thermal properties of TPE–PAEK were characterized using fluorescence spectrophotometer, differential scanning calorimetry and thermogravimetric analysis respectively. The results indicated that tetraphenylethylene-based copolymers possessed high fluorescence intensity at a low concentration both in solids and in water/THF mixtures. TPE–PAEK still presented strong fluorescence intensity at elevated temperature up to 90 °C. In addition, TPE–PAEK possess excellent thermal stability.
Introduction
Photoluminescent materials with great fundamental and technological implications represent a rapidly developing field and have received widespread attention in recent years, such as great potential in rollable displays, fluorescent chemo/biosensors, bio-imaging, organic light-emitting diodes (OLEDs), as well as other optical devices.1–6High-performance polymers have attracted considerable attention over the past decade owing to an increasing demand for their usage to replace metals or ceramics. Poly(aryl ether ketone)s (PAEKs) are one of important high-performance aromatic polymers with excellent mechanical properties, good electrical characteristics and superior thermal stability, which possess some potential applications in aerospace, automobile, microelectronic devices, light-emitting diodes and other high technology fields.7–10 Because of these superior properties, high-performance polymers such as PAEK are the promising polymeric matrix for the photoluminescent materials, but the research of PAEK-based photoluminescent materials is rarely reported. The introduction of luminophore into materials is the most efficient strategy to prepare photoluminescent materials, such as inorganic nanoparticles and organic nanoparticles. Yang's group firstly added CdSe/ZnS quantum dot into trifluoromethylphenyl-substituted fluorinated poly(ether ether ketone) (FPEEK) to prepare flexible photoluminescent, transparent films.11 The emission intensity of quantum dots (QDs) in the composite films is rather sensitive to the change of environmental temperature. However, when the temperature was up to 90 °C, the films showed a weak emission intensity. In addition, quantum dots generally contained heavy metal such as Cd, Se and Td, which is unfriendly to the environment, and quantum dots can hardly homodispersed in the polymer, which will lead to aggregation-caused quenching (ACQ). The well-designed conjugated polymers containing polyfluorene, polythiophene and polycarbazole have many notable advantages, such as photo- or electro-luminescence, easy fabrication as uniform films, mechanical flexibility, and so on.12
However, these polymer-based materials also have some intrinsic disadvantages, such as poor color purity, low quantum yield, low luminescence intensity, short lifetime,13 and especially the extremely detrimental ACQ,14 which limits their practical applications for light-emitting diodes and solid-state lighting. In 2001, Tang et al. first described a novel phenomenon opposite to the ACQ effect: propeller-shaped molecules such as hexaphenylsilole (HPS) and tetraphenylethylene (TPE) emit strongly in the solid state but are weakly emissive in solution.15 Because of the aggregation-induced emission (AIE) effect, it seems possible to design efficient solid emitter. The AIE molecules also give us a new way to overcome the defect of ACQ.
Therefore, a novel difluoride monomer containing tetraphenylethylene-((2,6-difluorophenyl)(4-(3-(4-1,2,2-triphenyl vinyl)phenoxy)propyl)phenyl)methanone (2F-TPE) was synthesized. After that, a series of novel tetraphenylethylene-based poly(arylether ketone)s (TPE–PAEK) were prepared through the condensation of 2F-TPE monomer, 2,2-bis(4-hydroxyphenyl)hexafluoropropane (bisphenol AF) and difluorobenzophenone. The fluorescent behaviors of TPE–PAEK in THF and water/THF mixtures were investigated. Furthermore, the fluorescence responses of TPE–PAEK in water/THF to temperature and the correspondingly thermal properties of the copolymers were discussed in detail.
Experimental
Materials
Anhydrous aluminium chloride was supplied by Beijing Chemical Reagent. 2,2-Bis(4-hydroxyphenyl)hexafluoropropane (bisphenol AF) was supplied by Aladdin Reagent (Shanghai) Co. Ltd. 2,6-Difluorobenzoyl chloride, 3-phenylpropyl bromide were purchased from Alfa-Aesar Ltd. 1-(4-Hydroxyphenyl)-1,2,2-triphenylethene was synthesized according to a published method.16 (4-(3-Bromopropyl)phenyl)(2,6-difluorophenyl)methanone (BDFM) was synthesized in our laboratory according to a published method.17 Difluorobenzophenone (DFB) was purchased from Changzhou Huashan Chemical Co. Ltd, China. Tetramethylenesulfone (TMS) was obtained from Jinzhou Oil Refinery and purified by vacuum distillation before used. Other commercially available materials and solvents were used without further purification.Synthesis of ((2,6-difluorophenyl)(4-(3-(4-1,2,2-triphenyl vinyl)phenoxy)propyl)phenyl)methanone (2F-TPE)
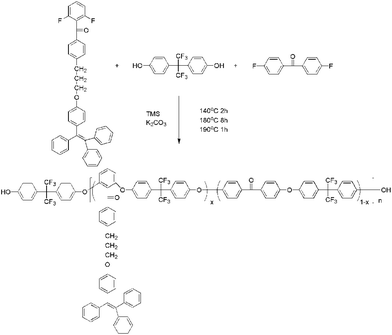
2F-TPE was synthesized as shown in Scheme 1. First, a 25 ml three-neck flask was equipped with an addition funnel, mechanical stirrer and nitrogen inlet was charged with 1-(4-hydroxyphenyl)-1,2,2-triphenylethene (0.696 g, 2 mmol), potassium hydroxide (0.1143 g, 2.04 mmol), DMF (12 ml). The solution was heated to 80 °C and stirred for 30 min. 0.678 g (2 mmol) BDFM was added and reacted for 4 h. After heated to 90 °C for 4 h, it was cooled down to room temperature and poured into 500 ml 10% wt cold hydrochloric acid aqueous, and the faint yellow precipitate was filtered and dissolved in toluene, the solution was dried by anhydrous magnesium sulfate. The crude product was purified on a silica-gel column using toluene as eluent. After recrystallized from ether and dried in a vacuum oven at 50 °C for 24 h, 0.59 g of light green solid was obtained. Yield: 50%.Preparation of side-chain-type tetraphenylethylene polyaryletherketone (TPE–PAEK)
A typical synthesis procedure of 10%-TPE–PAEK, where 10% refers to the feed percent of 2F-TPE, was as follows. Firstly, a 50 ml three-neck round-bottomed flask was equipped with a dropping funnel, mechanical stirrer and nitrogen inlet was charged with 2F-TPE, 4,4′-difluorodiphenyl methanone, 2,2-(4-hydroxyphenyl)hexafluoropropane, and K2CO3 were added. Then, toluene (9 ml) and sulfolane (17.2 ml) were added into the flask. The mixture was kept at the room temperature for a few minutes. The nitrogen was sent into the flask to remove the air and then heated to 140 °C and kept for 2 h. After that, the mixture was heated to 180 °C to keep for 8 h and then heated to 190 °C for 1 h in nitrogen atmosphere under magnetic stirring. Then, the solution was poured into 1000 ml 10% wt cold hydrochloric acid aqueous. The resulting fiber-like copolymer was filtered and washed with boiled deionized water and cool ethanol under nitrogen atmosphere for 5 times. The copolymer was dried under vacuum at 50 °C for 12 h to give pure side-chain-type poly(aryl ether ketone) containing pendant group of tetraphenylethylene (10%-TPE–PAEK).10%-TPE–PAEK. The yield was 60%. GPC: Mn: 2696 g mol−1; Mw: 4628 g mol−1; polydispersity index (PDI): 1.72.
30%-TPE–PAEK. The yield was 61%. GPC: Mn: 3379 g mol−1; Mw: 6139 g mol−1; polydispersit– index (PDI): 1.82.
50%-TPE–PAEK. The yield was 60%. GPC: Mn: 3006 g mol−1; Mw: 4638 g mol−1; polydispersity index (PDI): 1.54.
Measurements
FT-IR spectra were measured by a Fourier-transform infrared spectrometer. 1H-NMR experiments were carried out on a Bruker 510 spectrometer (1H, 500 MHz) using DMSO-d6 as solvents. TOF-MS were measured by a SHIMADZU Axima-CFR Time of Flight Mass Spectrometer using acetone as solvent. Thermogravimetric analysis (TGA) on a Perkin Elemer Pyris 1 thermal analyzer system was employed to assess thermal stability of the copolymer. Before the analysis, the copolymers were dried and kept in the TGA furnace at 100 °C under nitrogen for 30 min in order to remove water. The samples were evaluated in the range of 100–800 °C at a heating rate of 10 °C min−1 in nitrogen. Differential scanning calorimeter (DSC) measurements were performed on a TA DSC Q2000 instrument at a heating rate of 10 °C min−1 from 50 °C to 300 °C under nitrogen. The glass transition temperature (Tg) of the copolymer were reported as the midpoint of the step transition in the second heating run. Fluorescence (FL) spectra of the copolymer were recorded on a Shimadzu spectrometer. The FL lifetime of the copolymer were performed on an Edinburgh Instrument FLS920.Results and discussion
Synthesis of monomer
A new difluoride monomer containing tetraphenylethylene was designed and synthesized as shown in Scheme 1. The structure of 2F-TPE was confirmed by FT-IR, TOF-MS and 1H-NMR spectroscopy. The structure of the monomer 3 (2F-TPE) was proven using 1H-NMR spectroscopy (Fig. 1) and FT-IR (Fig. 3a). The signals at 6.60, 6.95, 7.10 were assigned to the electron-rich phenyl groups on the tetraphenylethylene parts. The signals at 7.68, 7.29, 7.71, 7.43 were assigned to the protons in the benzophenone structure of monomer and the signals at 2.0, 2.83, 3.88 were the protons of the CH2 parts. The absorption bands at 3054 cm−1 were characterization of benzene ring in the tetraphenylethylene parts. Bands at 2924 cm−1 and 2854 cm−1 were assigned to CH2 parts. Bands at 1762 cm−1 and 1273 cm−1 were characterization of carbonyl groups in aromatic ketone and ether bonds respectively. Bands at 1596 cm−1 were assigned to the benzene ring in the segment of benzophenone. Therefore, 2F-TPE was synthesized successfully.Synthesis of poly(aryl ether ketone) copolymers
A series of poly(aryl ether ketone) copolymers were successfully synthesized through a standard aromatic nucleophilic substitution reaction with 2F-TPE, 4,4-difluorobenzophenone and 2,2-(4-hydroxyphenyl)hexafluoropropane, and were named as x-TPE–PAEK (as shown in Table 1 and Scheme 2, x is the ratio of 2F-TPE in the copolymer). The fluorescence intensity of the copolymers was controlled by the content of 2F-TPE in the copolymerization procedure. All the copolymers (x-TPE–PAEKs) were soluble in THF, chloroform and polar aprotic solvents. The structure of the copolymers (x-TPE–PAEKs) were confirmed by 1H-NMR spectroscopy and FT-IR. The 1H-NMR spectra of the copolymers were presented in Fig. 2. Signals from 1.5–4 were assigned to the protons in the monomer 2F-TPE, the benzene protons in the 2F-TPE, 4,4-difluorobenzophenone and 2,2-(4-hydroxyphenyl)hexafluoropropane were overlapped from 6 to 8.| Copolymers | Tg (°C) | Td5% (°C) | Td10% (°C) |
|---|---|---|---|
| 10%-TPE–PAEK | 151.1 | 463.3 | 518.8 |
| 30%-TPE–PAEK | 147.3 | 453.2 | 487.8 |
| 50%-TPE–PAEK | 144.2 | 437.7 | 458.5 |
Copolymer structure was confirmed. Fig. 3b confirmed the chemical structure of TPE–PAEK-x: bands at 3440 cm−1 were assigned to hydroxyl groups, bands at 1494–1474 cm−1 and 1594 cm−1 were characterizatic of benzene in the main chain. 1652 cm−1 was assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of aromatic ketone in the main chain. Bands at 2921 cm−1 and 2859 cm−1 were characterization of methylene groups. And the characterization of –CF3 group and ether bonds band were at 1162 cm−1 and 1252 cm−1 respectively.
O bond of aromatic ketone in the main chain. Bands at 2921 cm−1 and 2859 cm−1 were characterization of methylene groups. And the characterization of –CF3 group and ether bonds band were at 1162 cm−1 and 1252 cm−1 respectively.
Because of the specific structure with need for further research and better interaction between the copolymers and water for the stable existence of the copolymers in the mixture, we designed the structure of the copolymer with phenolic hydroxyl groups on each side of copolymers' main chain.
Thermal properties of x-TPE–PAEKs
The thermal stabilities of x-TPE–PAEKs were evaluated using a TGA instrument under a nitrogen atmosphere. The 5% weight loss temperatures (Td5%) of x-TPE–PAEKs were above 430 °C, indicating that the copolymers possessed excellent thermal stabilities. The glass transition temperature (Tg) of x-TPE–PAEKs is in the range of 144–151 °C and decreases with increasing the molar content of 2F-TPE monomers. The phenomenon should be owed to the introduction of the side chains with CH2 groups, which leads to the decrease of the rigidity of the copolymer chains, following by the enhancement of the movement of segments.Fluorescence behavior of the copolymers
It is well known that TPE is an AIE active molecule. In this work, we investigated the fluorescence properties of copolymers containing TPE groups (TPE–PAEKs) in THF and THF/water mixtures (Fig. 4). The fluorescence of the TPE–PAEKs is too strong so that the fluorescence intensity of the TPE–PAEKs in 0.2 mg ml−1 is above the detection range of the instrument. Hence, we need to dilute the concentration of the solution to satisfy the requirement of the measurement. As shown in Fig. 5, the copolymer of 10%-TPE–PAEK exhibited nearly none fluorescence emission at 432 nm in THF, which should be owe to the free intramolecular rotation of the TPE moieties in the copolymer side chain backbone. While 95% water as a poor solvent was added to the THF solution, an emission peak at 471 nm appeared and could reach a tremendously higher value than that in THF solution as shown in Fig. 5a. Similar to the copolymer of 10%-TPE–PAEK, the copolymer of 30%-TPE–PAEK was almost non-emissive in THF solution. When 95% water added to the THF solution, the conjugated polymer chains aggregated and the free rotation of the polymer was restricted. Therefore, the non-radiative decay channels were blocked, leading to the significant fluorescence enhancement. Compared with 10%-TPE–PAEK, the emission intensity rapidly enhanced and the fluorescence intensity of 30%-TPE–PAEK in THF/water mixtures almost reached a higher value than that in THF solution (Fig. 5b). As shown in Fig. 5c, with the increasing content of TPE monomer from 30% to 50%, the fluorescence intensity of the copolymers has an obvious enhancement. In Fig. 6 and 7, the fluorescence color images of their solutions and solids exhibited the AIE feature of x-TPE–PAEKs. However, as shown in Fig. 6b, the copolymer could emit much stronger fluorescence in solid than in water/THF solution, which should be ascribed that the intramolecular rotation of the TPE groups were restricted by a tighter accumulation of molecular chains. In addition, the fluorescence quantum yields of all samples were shown in Table 2. | ||
| Fig. 5 Emission spectra of (a) 10%-TPE–PAEK, (b) 30%-TPE–PAEK and (c) 50%-TPE–PAEK in THF or THF/water (5/95 v/v) mixtures (λex = 355 nm, CTPE–PAEK = 0.1 mg ml−1). | ||
Generally, we discussed the aggregation-induced emission of polymer based on the influence of the aggregation structure controlled by temperature and pH on the behavior of fluorescence emission. In this work, the effect of the temperature on the fluorescence intensity of TPE–PAEKs was investigated, as seen in Fig. 7. The emission intensity of TPE–PAEKs decreases with the increase of temperature in the range of 20–90 °C. When the temperature is below 30 °C, the emission intensity of TPE–PAEKs is nearly no decrease because of the restriction of the copolymer chains and the TPE groups. And the change keeps a linear relationship within 30 °C to 90 °C with a sensitivity as high as 3% per degree as shown in Fig. 8. The fluorescence intensity of the solution decreased with the increase of temperature. The relationship between the increase of temperature and decrease of fluorescence intensity within a certain range is linear. The main reason is that the increase of temperature reduces the viscosity of the solution. As a result, solvent and solute molecules kinetic energy increases, followed by the increase of the probability of collision between the fluorescent molecule and other molecules. Excited fluorescent molecules will transfer their energy by intermolecular and intramolecular collisions and return to the ground state in the form of a non-fluorescence emission, which resulted in the fluorescence quenched.18 Compared with other polymers systems with similar properties such as tetraphenylethylene carbazole molecules and PNIPAM polymers containing tetraphenylethylene structure,19,20 TPE–PAEKs still maintain a relatively high fluorescence intensity at 50 °C and reveal fluorescence at 90 °C owing to the PAEKs' characteristic rigid conjugated structure.21
Conclusions
A difluoride monomer containing tetraphenylethylene group was designed and successfully synthesized. Based on this monomer, the poly(aryl ether ketone) copolymers containing tetraphenylethylene group were firstly prepared by polycondensation. The resultant copolymers showed distinct AIE behavior in both solvent/poor solvent mixtures and the solid state, indicating that the presence of the TPE groups could provide the luminogen with AIE feature and suppress the ACQ effect. Additionally, the copolymers also showed excellent thermal stabilities and could also have a strong fluorescence intensity in solvent/poor solvent mixtures at 90 °C. These novel copolymers with great AIE feature is expected to applicate for rollable displays, fluorescent chemo/biosensors, as well as other optical devices, which will fit some harsh environment for temperature determination in many fields such as health care and aerodynamics studies.Acknowledgements
This work is financially supported by the Ph.D. Program Foundation of the Ministry of Education of China (No. 20120061110017), Jilin Science and Technology Development Plan Program of China (20150204001GX) and Jilin Provincial Economic Structure Strategic Adjustment Guide Fund Special Project (High Technology Industry) (2015Y045).Notes and references
- O. A. Efremova, K. A. Brylev, Y. A. Vorotnikov, L. Vejsadova, M. A. Shestopalov, G. F. Chimonides, P. Mikes, P. D. Topham, S.-J. Kim, N. Kitamura and A. J. Sutherland, J. Mater. Chem. C, 2016, 4, 497–503 RSC.
- P. Tao, W.-L. Li, J. Zhang, S. Guo, Q. Zhao, H. Wang, B. Wei, S.-J. Liu, X.-H. Zhou, Q. Yu, B.-S. Xu and W. Huang, Adv. Funct. Mater., 2016, 26, 881–894 CrossRef CAS.
- B. Liu, H. Nie, X. Zhou, S. Hu, D. Luo, D. Gao, J. Zou, M. Xu, L. Wang and Z. Zhao, Adv. Funct. Mater., 2015, 26(5), 776–783 CrossRef.
- E. Manna, F. Fungura, R. Biswas, J. Shinar and R. Shinar, Adv. Funct. Mater., 2015, 25, 1226–1232 CrossRef CAS.
- W. Z. Yuan, H. Zhao, X. Y. Shen, F. Mahtab, J. W. Y. Lam, J. Z. Sun and B. Z. Tang, Macromolecules, 2009, 42, 9400–9411 CrossRef CAS.
- E. Zhao, H. Li, J. Ling, H. Wu, J. Wang, S. Zhang, J. W. Y. Lam, J. Z. Sun, A. Qin and B. Z. Tang, Polym. Chem., 2014, 5, 2301–2308 RSC.
- J. Pang, K. Shen, D. Ren, S. Feng, Y. Wang and Z. Jiang, J. Mater. Chem. A, 2012, 1, 1465–1474 RSC.
- A. L. Mong and D. Kim, J. Power Sources, 2016, 304, 301–310 CrossRef.
- V. L. Rao, J. Macromol. Sci., Polym. Rev., 1995, 661–712 CrossRef CAS.
- B. Zoetebier, S. Tas, G. J. Vancso, K. Nijmeijer and M. A. Hempenius, J. Polym. Sci., Part A: Polym. Chem., 2015, 53, 2786–2793 CrossRef CAS.
- L. Zhu, P. Huo, Q. Wang, F. Liang, C. Zhang, J. Li, X. Qu, G. Wang and Z. Yang, Chem. Commun., 2013, 49, 5283–5285 RSC.
- T. Fulghum, S. M. Abdul Karim, A. Baba, P. Taranekar, T. Nakai, T. Masuda and R. C. Advincula, Macromolecules, 2006, 39, 1467–1473 CrossRef CAS.
- R. Resmi, S. R. Amrutha and M. Jayakannan, J. Polym. Sci., Part A: Polym. Chem., 2009, 47, 2631–2646 CrossRef CAS.
- C. T. Chen, Chem. Mater., 2004, 16, 4389–4400 CrossRef CAS.
- J. Luo, Z. Xie, J. W. Y. Lam, L. Cheng, H. Chen, C. Qiu, H. S. Kwok, X. Zhan, Y. Liu, D. Zhu and B. Z. Tang, Chem. Commun., 2001, 1740–1741, 10.1039/B105159H.
- C. Yu, Y. Wu, F. Zeng, X. Li, J. Shi and S. Wu, Biomacromolecules, 2013, 14, 4507–4514 CrossRef CAS PubMed.
- J. Pang, H. Zhang, X. Li and Z. Jiang, Macromolecules, 2007, 40, 9435–9442 CrossRef CAS.
- Y. Hong, J. W. Y. Lam and B. Z. Tang, Chem. Commun., 2009, 45, 4332–4353 RSC.
- Z. Yang, Z. Chi, T. Yu, X. Zhang, M. Chen, B. Xu, S. Liu, Y. Zhang and J. Xu, J. Mater. Chem., 2009, 19, 5541–5546 RSC.
- Y. Jiang, X. Yang, C. Ma, C. Wang, Y. Chen, F. Dong, B. Yang, K. Yu and Q. Lin, ACS Appl. Mater. Interfaces, 2014, 6, 4650–4657 CAS.
- S. Bao, Polym. Chem., 2015, 6, 3537–3542 RSC.
| This journal is © The Royal Society of Chemistry 2016 |