Highly thermally conductive UHMWPE/graphite composites with segregated structures†
Abstract
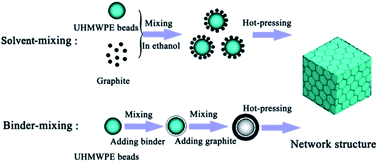
Polymer composites with segregated structures based on ultrahigh molecular weight polyethylene (UHMWPE) and graphite flakes were fabricated by a novel binder-mixing method and the traditional solvent-mixing method. Compared with the solvent-mixing method, the thermal conductivity of composites fabricated by the binder-mixing method improved on average by 26.27 percent at the volume fraction of graphite flakes from 2.22–18.83 vol%. Optical and SEM images showed that the binder-mixing method results in the formation of a more continuous and more homogeneous conductive network and wider thermally conductive paths, leading to the greatly improved thermal conductivity.

 Please wait while we load your content...
Please wait while we load your content...