Fluorescent tracer of dopamine enables selective labelling and interrogation of dopaminergic amacrine cells in the retina of living zebrafish†
Abstract
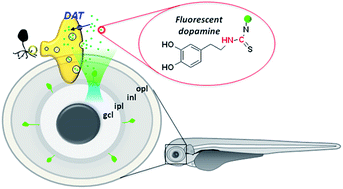
The retinal dopaminergic amacrine cell plays multiple roles in vision; it optimizes the encoding of visual stimuli, serves as an output of the circadian rhythm, and influences trophic processes. Specific identification of such cells in vivo remains challenging. A fluorescent tracer that mimics natural dopamine would help elucidate the distribution and the functional roles of dopaminergic neurons. Here we report a fluorescent tracer of dopamine for the selective labeling and study of retinal dopaminergic amacrine cells in vivo. This tracer has low toxicity and effective selectivity, and enables fluorescent determination of the evoked release of dopamine and the femtosecond laser ablation of single dopaminergic amacrine cells in living zebrafish. Our approach is readily adaptable by other research groups, and is expected to accelerate biomedical research in domains related to the pathophysiology of retinal dopaminergic amacrine cells.

 Please wait while we load your content...
Please wait while we load your content...