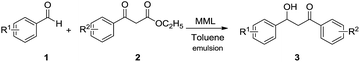
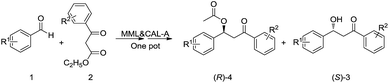
One-pot bienzymatic cascade combining decarboxylative aldol reaction and kinetic resolution to synthesize chiral β-hydroxy ketone derivatives†
Fan Xu‡
,
Jian Xu‡,
Yujing Hu,
Xianfu Lin and
Qi Wu*
Department of Chemistry, Zhejiang University, Hangzhou 310027, Zhejiang, PR China. E-mail: llc123@zju.edu.cn; Web: http://mypage.zju.edu.cn/en/0003155 Fax: +86 571 87952618; Tel: +86 571 87951588
First published on 8th August 2016
Abstract
A bienzymatic one-pot sequential cascade for the synthesis of (S)-β-hydroxy ketones and acylated (R)-β-hydroxy ketone derivatives was successfully developed. An immobilized lipase from Mucor miehei (MML) catalysed promiscuous decarboxylative aldol reaction and a lipase A or B from Candida antarctica (CAL-A or CAL-B) catalysed kinetic resolution of racemic β-hydroxy ketone were combined in this one-pot protocol, reducing the purification step between the two reactions. Twelve chiral β-hydroxy ketones and the same number of corresponding acylated derivatives were obtained with excellent ee values and high yields through this method, and the scaling up experiment also worked without apparent loss of reaction rate and stereoselectivity.
Introduction
Chiral β-hydroxy ketones are important building blocks for the synthesis of biologically active compounds, including macrolides antibiotics, cancer drugs and many natural products.1 They are also crucial components in the building of complex polyol architectures.2 Chiral β-hydroxy ketones are usually synthesized through asymmetric aldol reaction, which could be catalyzed by numerous catalysts, comprising chiral proline derivatives,3a–c chiral amines,3d,e chiral metal complexes,3f etc. Hydrolases have also been widely reported as being capable of promiscuously catalysing asymmetric aldol reactions for gaining access to a series of β-hydroxy ketone products.4 Comparing to the organic catalysts, hydrolases usually displayed comparable or improved catalytic efficiency & selectivity, higher environmental affinity & economic effect, and greater availability. Besides, after being immobilized, hydrolases could be recycled for several times without major loss of catalytic activity, which could enhance their practical value and reduce their cost. However, most direct enzymatic aldol reactions require cyclohexanone as the ketone substrate, while acyclic ketones usually display very low reactivity.Recently, a lipase-catalysed decarboxylative aldol reaction was successfully developed to produce linear β-hydroxy ketones. Yu et al. found that lipase B from Candida antarctica (CAL-B) could efficiently catalyse the decarboxylative aldol reaction between aldehyde and a series of different β-ketoesters under mild reaction condition.5 Further efforts were made to gain insight of this enzymatic reaction.5b,6 Basic additive like cyclen, triethylamine or imidazole had to be applied to improve the reaction results, and only limited stereoselectivity could be witnessed for all of these catalysts. Thus, in order to obtain optical pure β-hydroxy ketones or its derivatives, an additional kinetic resolution reaction step (KR), which is usually catalysed by enzyme as well, should be performed for the racemic aldol adduct. KR of β-hydroxy ketones were reported in several articles,5b,7 but in most cases these reaction results were far from ideal.
Multistep one-pot reactions, or cascades, showed great potential for manufacturing fine chemicals and pharmaceutical intermediates.8 In order to develop an efficient and green method to synthesize chiral β-hydroxy ketones, a one-pot enzymatic cascade combining decarboxylative aldol reaction and kinetic resolution would be a wise option. An efficient one-pot cascade usually requires the compatibility of the individual reaction steps with one another. Zhang et al. once used lipase CAL-B to catalyse decarboxylative aldol reaction in water containing acetonitrile with cyclen as the additive, then used CRL to catalyse KR of β-hydroxy ketones in toluene.5b Unfortunately, the two reactions could not be combined in a one-pot system, so Zhang had to choose a stepwise approach. Edin et al. also tried to combine (S)-proline and PCL (lipase form Pseudomonas cepacia) to synthesize chiral (S)-β-hydroxy ketones with high ee values,9 but the substrate scope was quite limited.
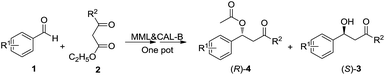
Our group have made attempts to combine the enzymatic promiscuous reaction and KR in a one-pot or stepwise manner for the synthesis of chiral molecules.10 In this research, we found that immobilized lipase from Mucor miehei (MML) could catalyse decarboxylative aldol reaction quite well with no additives other than water. Meanwhile, lipase A and lipase B from Candida antarctica (CAL-A and CAL-B) were found to be capable of catalysing KR of different β-hydroxy ketones, although several articles failed to apply CAL-B for this KR reaction.5b,7c To our delight, we observed that MML was compatible with CAL-B or CAL-A in a one-pot reaction system (Scheme 1), thus reduced the purification step between the two reactions. A series of different β-hydroxy ketone derivatives were successfully synthesized through this one-pot cascade with high yields and excellent ee values.
Results and discussion
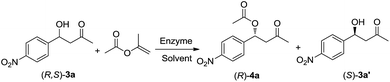
As KR step is the core step to attach stereoselectivity in this one-pot cascade attempt, we started our research by optimizing the reaction condition of the KR of β-hydroxy ketones. Racemic aldol adduct 3a was chosen as the model substrate, and isopropenyl acetate was applied as the acyl donor. Several lipases were found to be capable of catalysing KR of 3a. CAL-A was most active towards this reaction, but its stereoselectivity was not exactly strict (entry 4, Table 1). CAL-B gave the best performance of all the tested lipases, a yield of 49% and ee of 99% for (R)-4a could be obtained (entry 6, Table 1) when the reaction media TBME was replaced by diisopropyl ether (DIPE). The influence of other solvents was displayed in Table S1 (see in ESI).† A study on the influence of ratio of acyl donor/β-hydroxy ketone 3a (Fig. S1†) suggested that 4 eq. of acyl donor was most suitable.| Entry | Enzyme source | Solvent | ee of 3a′ b/% | Yield of 3a′ b/% | ee of 4a b/% | Yield of 4a b/% |
|---|---|---|---|---|---|---|
| a Reaction conditions: 0.4 mmol racemic β-hydroxy ketone 3a, 40 mg enzyme, 2 mmol isopropenyl acetate, 2 mL TBME, 37 °C, 72 h.b Determined by chiral HPLC analysis using AD-H column. | ||||||
| 1 | MML | TBME | 2 | 98 | — | 1 |
| 2 | PS-IM | TBME | 64 | 60 | 97 | 38 |
| 3 | TL-IM | TBME | 37 | 72 | 98 | 27 |
| 4 | CAL-A | TBME | 99 | 34 | 49 | 64 |
| 5 | CAL-B | TBME | 85 | 54 | 99 | 45 |
| 6 | CAL-B | DIPE | 99 | 50 | 99 | 49 |
| 7 | CAL-B | Toluene | 46 | 68 | 99 | 30 |
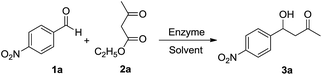
Previous studies on enzymatic decarboxylative aldol reaction all reported that high polar solvents were the optimized reaction media.5,6 As effective solvents for KR reaction were all ones with medium polarity, the investigation of decarboxylative aldol reaction was initiated by enzyme screening in DIPE. Of all the enzymes tested, MML provided the most promising result (entry 9, Table 2), and β-hydroxy ketone 3a could be obtained with a yield of 41%. Since water was one of the substrates, we added 4.0 μL water into the reaction system, and MML's catalytic activity was greatly boosted with the yield of 3a about 94% (entry 10, Table 2). CAL-B, the optimized catalyst for KR step, could not provide an acceptable result (entry 6, 7, Table 2) even after an optimization of adding water amount (Fig. S2†). Although basic additives could further enhance CAL-B's performance,5 they would also lead to a remarkable elimination side-reaction on the acyl product 4a in the subsequent KR step. So MML was considered as the optimized catalyst for this reaction. In TBME and DIPE, the additional water could be fully dissolved, and MML showed similar catalytic activity (entry 9, 11, Table 2). In toluene, however, due to its low water content and water solubility, in order to improve the yield of 3a to 90% (entry 13 Table 2), more extra water had to be provided, and the biphasic reaction mixture had to be emulsified by ultrasonication before adding the enzyme. Evitt et al. once claimed that lipase (CAL-B) did not catalyse decarboxylative aldol reaction but the hydrolysis of β-ketoester, and the subsequent decarboxylative aldol reaction was carried out spontaneously.6a By utilizing Zhang's method,5b a control reaction (same reaction condition as described in entry 9, Table 2) without 4-nitrobenzaldehyde (1a) was carried out for 24 h, then MML was removed by filtration and the decarboxylative aldol reaction was induced by the addition of 1a. No aldol product could be detected in 24 h. After adding MML into the reaction mixture again, remarkable formation of 3a could be observed. It indicates that MML is involved in the entire reaction process.
| Entry | Enzyme source | Solvent | H2O/μL | Yieldb/% |
|---|---|---|---|---|
| a Reaction condition: 0.4 mmol 4-nitrobenzaldehyde, 0.8 mmol ethyl acetoacetate, 40 mg enzyme, 2 mL solvent, 37 °C, 3 d.b Determined by HPLC.c n.d. means not detected.d The reaction mixture was emulsified by ultrasonication. | ||||
| 1 | No cat. | DIPE | — | n.d.c |
| 2 | BSA | DIPE | — | n.d.c |
| 3 | PS-IM | DIPE | — | 13 |
| 4 | TL-IM | DIPE | — | 26 |
| 5 | CAL-A | DIPE | — | 21 |
| 6 | CAL-B | DIPE | — | 29 |
| 7 | CAL-B | DIPE | 4.0 | 63 |
| 9 | MML | DIPE | — | 41 |
| 10 | MML | DIPE | 4.0 | 94 |
| 11 | MML | TBME | 4.0 | 95 |
| 12d | MML | Toluene | 4.0 | 83 |
| 13d | MML | Toluene | 6.0 | 90 |
Water affected not only the catalytic activity of enzyme but also the reaction equilibrium. As all the solvent, reagent and enzyme were in the same batch during the investigation, the total water content could be calculated by adding the residual water amount (0.19%, v/v, determined by Karl Fischer titration) and the additional water amount. As shown in Fig. S3,† when the total water content was 0.39–1.19% (1.1–3.3 eq.), 3a could be obtained with more than 90% yield. On the other hand, water was harmful for the subsequent KR reaction (Fig. S4†), so a minimum content of 0.39% (1.1 eq.) was chosen for further investigation (4.0 μL additional water in this work). As most of water would be consumed in the decarboxylative aldol reaction, the subsequent KR step in a one-pot cascade would not be affected as long as the first step was fully carried out, so this one-pot cascade had to be performed in a sequential mode.
The effect of molar ratio of β-ketoester was studied as well (Fig. S5†), and a ratio of 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was applied for the further research. With optimized reaction conditions, the substrate scope of this decarboxylative aldol reaction was investigated. Excellent conversion could be observed for substituted benzaldehydes with strong electron-withdrawing group such as nitro, trifluoromethyl or cyano group (entry 1–4, Table 3), while halogen substituted or non-substituted benzaldehydes displayed dramatically decreased reactivity (entry 5–8, Table 3). The slightly lower yields of β-hydroxy ketone 3b and 3d were caused by the side elimination reaction. Altering the structure of β-ketoester could further extend the product scope (entry 9–11, Table 3).
1 was applied for the further research. With optimized reaction conditions, the substrate scope of this decarboxylative aldol reaction was investigated. Excellent conversion could be observed for substituted benzaldehydes with strong electron-withdrawing group such as nitro, trifluoromethyl or cyano group (entry 1–4, Table 3), while halogen substituted or non-substituted benzaldehydes displayed dramatically decreased reactivity (entry 5–8, Table 3). The slightly lower yields of β-hydroxy ketone 3b and 3d were caused by the side elimination reaction. Altering the structure of β-ketoester could further extend the product scope (entry 9–11, Table 3).
| Entry | 3 | R1 | R2 | Conv.b/% | Yieldb,c/% |
|---|---|---|---|---|---|
| a Reaction condition: 0.4 mmol aldehyde, 0.6 mmol β-ketoester, 4.0 μL H2O, 40 mg MML, 2 mL DIPE, 37 °C, 72 h.b Determined by HPLC.c The values in parentheses are isolated yields. | |||||
| 1 | 3a | p-NO2 | CH3 | 98 | 95 (92) |
| 2 | 3b | m-NO2 | CH3 | 97 | 80 (73) |
| 3 | 3c | p-CF3 | CH3 | 98 | 97 (92) |
| 4 | 3d | p-CN | CH3 | 94 | 85 (80) |
| 5 | — | p-Br | CH3 | 19 | 18 |
| 6 | — | p-F | CH3 | 4 | 3 |
| 7 | — | p-Cl | CH3 | 5 | 5 |
| 8 | — | H | CH3 | 2 | 1 |
| 9 | 3e | p-NO2 | n-C3H7 | 97 | 95 (91) |
| 10 | 3f | p-NO2 | c-C3H5 | 95 | 92 (89) |
| 11 | 3g | p-NO2 | C6H5 | 96 | 94 (88) |
Due to the similarity of the reaction conditions, we tried to combine the two reaction steps in a one-pot reaction system. At first, a series of one-pot cascade attempts was made with different water contents (Table S2†). A water content of 0.39% (1.1 eq., 4.0 μL extra water) was found to be most effective, and both of the chiral products could be obtained with more than 98% ee and 47% yields. Motivated by the excellent result, we broadened the substrate scope, and a series of (S)-β-hydroxy ketones and acylated (R)-β-hydroxy ketone derivatives were synthesized through this one-pot cascade. Most of them were obtained with 95–99% ee values and 42–49% yields. R1 group showed no influence on the stereoselectivity on the KR step (entry 1–4, Table 4), but β-hydroxy ketone derivatives bearing m-nitro or p-CN group were more easily to be eliminated so their yields were slightly lower (entry 2, 4, Table 4). Enlarging R2 from methyl to n-propyl (3e) or phenyl group (3g) greatly decreased the efficiency of the KR step in the one-pot protocol. For β-hydroxy ketone 3e, the stereoselectivity of CAL-B was still excellent, so prolonging the reaction time of KR to 7 d would lead to an acceptable result (entry 5, Table 4). For 1,3-biphenyl β-hydroxy ketone 3g, however, CAL-B's stereoselectivity was remarkably decreased (entry 7, Table 4). In order to promote the reaction result for this kind of β-hydroxy ketone, CAL-B might have to be replaced by another enzyme. Enlarging R2 from methyl to c-propyl group had no negative effect on either the rate or stereoselectivity of KR reaction (entry 6, Table 4). The reaction results of a single KR reaction were listed in Table S3.† The two reactions gave similar final results no matter combined in a one-pot system or carried out separately, but a one-pot procedure could reduce the purification step between the two reactions thus simplify the handling of the whole synthesis procedure.
| Entry | 3 | 4 | R1 | R2 | ee of 3 b/% | Yield of 3 b/% | ee of 4 b/% | Yield of 4 b/% |
|---|---|---|---|---|---|---|---|---|
| a Reaction conditions: 0.4 mmol aldehyde, 0.6 mmol β-ketoester, 4.0 μL H2O, 40 mg CAL-B, 40 mg MML, 2 mL diisopropyl ether, 37 °C, 3 d; after completion of decarboxylative aldol reaction, adding 1.6 mmol isopropenyl acetate into the reaction mixture, 3 d.b Determined by chiral HPLC analysis. The values in parentheses are isolated yields.c The reaction time of KR step was prolonged to 7 d. | ||||||||
| 1 | 3a | 4a | p-NO2 | CH3 | 99 | 48 (46) | 99 | 47 (46) |
| 2 | 3b | 4b | m-NO2 | CH3 | 99 | 42 (39) | 98 | 42 (38) |
| 3 | 3c | 4c | p-CF3 | CH3 | 98 | 49 (48) | 99 | 48 (47) |
| 4 | 3d | 4d | p-CN | CH3 | 94 | 45 (42) | 95 | 42 (39) |
| 5c | 3e | 4e | p-NO2 | n-C3H7 | 90 | 53 (51) | 98 | 42 (37) |
| 6 | 3f | 4f | p-NO2 | c-C3H5 | 98 | 49 (46) | 96 | 48 (46) |
| 7c | 3g | 4g | p-NO2 | C6H5 | 57 | 57 | 80 | 32 |
Because CAL-B was not the best option for catalysing KR of 1,3-biphenyl β-hydroxy ketones, a brief optimization was carried out for KR of substrate 3g. Although CAL-A's stereoselectivity was still not as strict as CAL-B, its catalytic activity was much higher, and the ee value of unreacted (S)-3g could reach 99% in 3 d (entry 1, Table 5). We tested CAL-A's performance in different organic solvents and found that toluene could greatly enhance the stereoselectivity of the KR reaction (entry 5, Table 5). Decreasing the reaction time from 3 d to 2 d could further increase the ee of 4g to 90% with ee of 3g′ unchanged (entry 7, Table 5). The study on the influence of molar ratio of acyl donor (Fig. S6†) suggested that 4 eq. of acyl donor was optimized for the overall result.
| Entry | Enzyme | Solvent | ee of 3g′ b/% | Yield of 3g′ b/% | ee of 4g b/% | Yield of 4g b/% |
|---|---|---|---|---|---|---|
| a Reaction condition: 0.4 mmol racemic β-hydroxy ketone 3g, 40 mg enzyme, 2 mmol isopropenyl acetate, 2 mL TBME, 37 °C, 3 d.b Determined by chiral HPLC analysis using AD-H column.c n.d. means not detected.d The reaction time was prolonged to 7 d.e The reaction time was decreased to 2 d. | ||||||
| 1 | CAL-A | Diisopropyl ether | 99 | 39 | 60 | 58 |
| 2 | CAL-Bd | Diisopropyl ether | 57 | 61 | 80 | 34 |
| 3 | PS-IM | Diisopropyl ether | <1 | >99 | — | n.d.c |
| 4 | TL-IM | Diisopropyl ether | <1 | >99 | — | n.d.c |
| 5 | CAL-A | Toluene | 99 | 51 | 87 | 49 |
| 6 | CAL-A | TBME | 99 | 41 | 67 | 57 |
| 7 | CAL-Ae | Toluene | 99 | 49 | 90 | 48 |
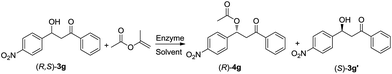
As the KR of 1,3-biphenyl β-hydroxy ketone required toluene as the solvent, and the decarboxylative aldol reaction could also be catalysed by MML in emulsified toluene (entry 13, Table 2), we first tested the substrate scope of decarboxylative aldol reaction with this alternative reaction condition. A series of 1,3-biphenyl β-hydroxy ketones were synthesized with 86–93% yields. Changing the solvent from diisopropyl ether to toluene showed minor difference on the yield of 3g (entry 11, Table 3; entry 1, Table 6). The benzaldehyde substrates also had to be substituted with strong electron-withdrawing group to achieve excellent conversions, while the substituent group on the β-ketoesters displayed minor influence on the reaction results. These β-hydroxy ketones tend to be eliminated, so the yields were much lower than the conversions. Then we investigated the one-pot cascade catalysed by MML & CAL-A. This cascade was found highly efficient for synthesizing (S)-1,3-biphenyl β-hydroxy ketones, with 98–99% ee values and 42–45% yields. Corresponding acylated (R)-1,3-biphenyl β-hydroxy ketone derivatives could be obtained with 90–95% ee values and 42–46% yields. para-Substituted R2 group showed minor influence on the KR result; meta-substituted R2 group, however, greatly decreased the reaction rate so the reaction time had to be prolonged to 7 d for an acceptable result (entry 6, Table 7). The reaction results of a single KR reaction were listed in Table S4.†
| Entry | 3 | R1 | R2 | Conv.b/% | Yieldb,c/% |
|---|---|---|---|---|---|
| a Reaction condition: 0.4 mmol aldehyde, 0.6 mmol β-ketoester, 6.0 μL H2O, 40 mg MML, 2 mL toluene, 37 °C, 72 h.b Determined by HPLC.c The values in parentheses are isolated yields. | |||||
| 1 | 3g | p-NO2 | H | 95 | 93 |
| 2 | 3h | p-CF3 | H | 90 | 86 (83) |
| 3 | 3i | p-CF3 | p-Cl | 96 | 90 (86) |
| 4 | 3j | p-CF3 | p-CH3 | 94 | 89 (83) |
| 5 | 3k | p-CF3 | p-OCH3 | 91 | 86 (81) |
| 6 | 3l | p-CF3 | m-OCH3 | 94 | 88 (84) |
| Entry | 3 | 4 | R1 | R2 | ee of 3 b/% | Yield of 3 b/% | ee of 4 b/% | Yield of 4 b/% |
|---|---|---|---|---|---|---|---|---|
| a Reaction conditions: 0.4 mmol aldehyde, 0.8 mmol β-ketoester, 6.0 μL H2O, 40 mg CAL-A, 40 mg MML, 2 mL toluene, 37 °C, 3 d; after completion of decarboxylative aldol reaction, adding 1.6 mmol isopropenyl acetate into the reaction mixture, 2 d.b Determined by chiral HPLC analysis. The values in parentheses are isolated yields.c The reaction time of KR step was prolonged to 7 d. | ||||||||
| 1 | 3g | 4g | p-NO2 | H | 99 | 44 (43) | 90 | 42 (40) |
| 2 | 3h | 4h | p-CF3 | H | 99 | 44 (41) | 95 | 45 (41) |
| 3 | 3i | 4i | p-CF3 | p-Cl | 99 | 45 (43) | 92 | 43 (39) |
| 4 | 3j | 4j | p-CF3 | p-CH3 | 99 | 44 (42) | 94 | 42 (42) |
| 5 | 3k | 4k | p-CF3 | p-OCH3 | 99 | 43 (41) | 94 | 46 (44) |
| 6c | 3l | 4l | p-CF3 | m-OCH3 | 98 | 42 (43) | 90 | 43 (38) |
So far, this one-pot cascade had only been carried out under analytical level. In order to test that if this method is capable of preparing chiral β-hydroxy ketones of larger scale, we increased the amount of substrates, catalysts and solvent by 50 times for the synthesis of (S)-3a and (R)-4a. The reaction progress was traced by chiral LC. As indicated by Fig. 1, the reaction progress was not hampered by scaling the reaction up to a gram level. After a total reaction time of 6 days, (S)-3a could be isolated with 47% yield and 99% ee while the acyl product (R)-4a could also be isolated with 45% yield and 99% ee. Other chiral β-hydroxy ketones or acylated derivatives could also be obtained through a scaling up one-pot cascade. The enzymatic hydrolysis of acylated (R)-β-hydroxy ketone derivatives had been demonstrated by Zhang et al.5b and through this method (R)-β-hydroxy ketones could also be obtained efficiently. The catalysts could be washed with diisopropyl ether or toluene several times and then recycled for at least three times without affecting stereoselectivity or reaction rate.
CAL-B's excellent performance on the KR of β-hydroxy ketones in this work was quite surprising, as in other works CAL-B showed no catalytic activity towards the KR of 3a.5b,7c To confirm this result, a molecular docking and molecular dynamics (MD) for 10 ns were performed with the acyl products (R) or (S)-4a separately using the modeling suite YASARA.11a The acyl products (R) or (S)-4a were introduced as tetrahedral intermediates (oxyanions) covalently bound to catalytically active Ser105. From Fig. S7,† we can see the RMSD of the CA backbone of CAL-B remained constant and the structure tended to be stable after 10 ns simulation. The final balanced structure of both CAL-B-(R)-4a and CAL-B-(S)-4a were extracted for further analysis. In the case of the preferential product (R)-4a, the large-sized phenyl group was oriented toward the active site entrance and the medium-sized acetonyl group was positioned in the stereospecificity pocket surrounded by residues Trp104, Ser47, Thr40, and Ala281. Meanwhile, the acyl moiety of the product (R)-4a was accommodated in the acid-binding pocket surrounded by residues Val189, Thr138, Leu140, and Gln157, whereas the carbonyl oxygen atom was efficiently stabilized by the oxyanion hole defined, inter alia, by residues Gln106 and Thr40. All the hydrogen bonds required for catalysis were spatially arranged (Fig. 2a). Two important distance (d1: distance between NE2 of H224 and OG of S105; d2: distance between NE2 of H224 and the ester oxygen atom of the product) were short enough for efficient nucleophilic attack even for the whole 10 ns simulation process (Fig. 2c). In the case of the non-preferential product (S)-4a, the MD experiment revealed that although the large-sized phenyl group and the stereospecificity pocket, meanwhile the carbonyl oxygen atom could be stabilized by residues Gln106 and Thr40 through hydrogen bonds (Fig. 2b), the critical distance (d2) was too long for effective nucleophilic attack to occur. During the 10 ns simulation process, most d2 values were larger than 3.5 Å (Fig. 2d). These results explained the high enantioselectivity of CAL-B in the esterification of β-hydroxy ketones.
Experimental
Materials and methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate = 6
ethyl acetate = 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1–3
1–3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate = 10
ethyl acetate = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1–6
1–6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate = 3
ethyl acetate = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.Characterization data
Conclusions
In summary, we have successfully demonstrated a one-pot bienzymatic cascade in organic media to synthesize chiral β-hydroxy ketones for the first time. After detailed optimizations of a series of reaction factors, twelve different (S)-β-hydroxy ketones and a same number of acylated (R)-β-hydroxy ketones could be obtained under mild reaction condition with easy handworks. The ee values of most chiral compounds were in a range of 94–99%, while the total yields of both chiral products were all above 85%. This enzymatic one-pot cascade could still be very efficient no matter being scaled up to a gram level or the catalysts being recycled for three times. Additionally, CAL-B and CAL-A catalysed KR of β-hydroxy ketones were successfully demonstrated for the first time, while so far there was very few efficient way to prepare chiral 1,3-biphenyl β-hydroxy ketones with high optical purity under mild condition. This one-pot bienzymatic cascade was highly practical as the product chiral β-hydroxy ketones could serve as synthetic intermediates for many biologically active compounds.Acknowledgements
We acknowledge the financial support received from the National Natural Science Foundation of China (NSFC) (No. 21472169), the Zhejiang Provincial Natural Science Foundation (No. LY14B020006), and the Fundamental Research Funds for the Central Universities (No. 2016QNA3012).Notes and references
- (a) B. M. Trost, I. Fleming and C. H. Heathcock, Comprehensive Organic Synthesis, Pergamon: Oxford, 1991, vol. 2 Search PubMed; (b) T. Mukaiyama, Tetrahedron, 1999, 55, 8609 CrossRef CAS; (c) K. C. Nicolaou, D. Vourloumis, N. Winssinger and P. S. Baran, Angew. Chem., Int. Ed., 2000, 39, 44 CrossRef CAS; (d) D. A. Evans, A. M. Ratz, B. E. Huff and G. S. Sheppard, J. Am. Chem. Soc., 1995, 117, 3448 CrossRef CAS.
- (a) B. Schetter and R. Mahrwald, Angew. Chem., Int. Ed., 2006, 45, 7506 CrossRef CAS PubMed; (b) I. Paterson and M. M. Mansuri, Tetrahedron, 1985, 41, 3569 CrossRef CAS; (c) S. Masamune, G. S. Bates and J. W. Corcoran, Angew. Chem., Int. Ed., 1977, 16, 585 CrossRef CAS PubMed; (d) R. Mahrwald, Modern Aldol Reactions, Wiley-VCH, 2004, vol. 1, p. 2 Search PubMed.
- (a) B. Wang, X. W. Liu, L. Y. Liu, W. X. Chang and J. Li, Eur. J. Org. Chem., 2010, 31, 5951 CrossRef; (b) B. List, R. A. Lerner and C. F. Barbas III, J. Am. Chem. Soc., 2000, 122, 2395 CrossRef CAS; (c) B. List, P. Pojarliev and C. Castello, Org. Lett., 2001, 3, 573 CrossRef CAS PubMed; (d) R. S. Rambo and P. H. Schneider, Tetrahedron: Asymmetry, 2010, 21, 2254 CrossRef CAS; (e) M. Rachwalski, S. Lesniak and P. Kiełbasinski, Tetrahedron: Asymmetry, 2011, 22, 1325 CrossRef CAS; (f) J. Paradowska, M. Pasternak, B. Gut, B. Gryzło and J. Mlynarski, J. Org. Chem., 2012, 77, 173 CrossRef CAS PubMed.
- (a) Z. Guan, L. Y. Li and Y. H. He, RSC adv., 2015, 5, 16801 RSC; (b) Y. Miao, M. Rahimi, E. M. Geertsema and G. J. Poelarends, Curr. Opin. Chem. Biol., 2014, 25, 115 CrossRef PubMed; (c) S. M. Dean, W. A. Greenberg and C. H. Wong, Adv. Synth. Catal., 2007, 349, 1308 CrossRef CAS; (d) Z. B. Xie, N. Wang, L. H. Zhou, F. Wan, T. He, Z. G. Le and X. Q. Yu, ChemCatChem, 2013, 5, 1935 CrossRef CAS; (e) Z. Q. Liu, Z. W. Xiang, Z. Shen, Q. Wu and X. F. Lin, Biochimie, 2014, 101, 156 CrossRef CAS PubMed.
- (a) X. W. Feng, C. Li, N. Wang, K. Li, W. W. Zhang and X. Q. Yu, Green Chem., 2009, 11, 1933 RSC; (b) W. W. Zhang, N. Wang, X. W. Feng, Y. Zhang and X. Q. Yu, Appl. Biochem. Biotechnol., 2014, 173, 535 CrossRef CAS PubMed.
- (a) A. S. Evitt and U. T. Bornscheuer, Green Chem., 2011, 13, 1141 RSC; (b) M. Kapoor, A. B. Majumder, J. Mukherjee and M. N. Gupta, Biocatal. Biotransform., 2012, 30, 399 CrossRef CAS.
- (a) M. S. Nair and S. Joly, Tetrahedron. Asymmetry., 2000, 11, 2049 CrossRef CAS; (b) S. Joly and M. S. Nair, J. Mol. Catal. B: Enzym., 2003, 22, 151 CrossRef CAS; (c) M. Edin, J. E. Backvall and C. Armando, Tetrahedron Lett., 2004, 45, 7697 CrossRef CAS.
- (a) D. E. Fogg and E. N. dos Santos, Coord. Chem. Rev., 2004, 248, 2365 CrossRef CAS; (b) A. Ajamian and J. L. Gleason, Angew. Chem., Int. Ed., 2004, 43, 3754 CrossRef CAS PubMed; (c) J. C. Wasilke, S. J. Obrey, R. T. Baker and G. C. Bazan, Chem. Rev., 2005, 105, 1001 CrossRef CAS PubMed; (d) H. C. Guo and J. A. Ma, Angew. Chem., Int. Ed., 2006, 45, 354 CrossRef CAS PubMed; (e) M. M. Hussain and P. J. Walsh, Acc. Chem. Res., 2008, 41, 883 CrossRef CAS PubMed; (f) F. X. Felpin and E. Fouquet, ChemSusChem, 2008, 1, 718 CrossRef CAS PubMed; (g) J. Zhou, Chem.–Asian J., 2010, 5, 422 CrossRef CAS PubMed; (h) J. Aleman and S. Cabrera, Chem. Soc. Rev., 2013, 42, 774 RSC; (i) C. A. Denard, J. F. Hartwig and H. M. Zhao, ACS Catal., 2013, 3, 2856 CrossRef CAS; (j) J. Muschiol, C. Peters, N. Oberleitner, M. D. Mihovilovic, U. T. Bornscheuer and F. Rudroff, Chem. Commun., 2015, 51, 5798 RSC.
- M. Edin, J. E. Backvall and C. Armando, Tetrahedron Lett., 2004, 45, 7697 CrossRef CAS.
- (a) F. Xu, J. L. Wang, B. K. Liu, Q. Wu and X. F. Lin, Green Chem., 2011, 13, 2359 RSC; (b) F. Xu, Q. S. Wu, X. Y. Chen, X. F. Lin and Q. Wu, Eur. J. Org. Chem., 2015, 24, 5393 CrossRef.
- (a) E. Krieger, T. Darden, S. B. Nabuurs, A. Finkelstein and G. Vriend, Proteins: Struct., Funct., Bioinf., 2004, 57, 678 CrossRef CAS PubMed; (b) Y. Duan, C. Wu, S. Chowdhury, M. C. Lee, G. Xiong, W. Zhang, R. Yang, P. Cieplak, R. Luo, T. Lee, J. Caldwell, J. Wang and P. Kollman, J. Comput. Chem., 2003, 24, 1999 CrossRef CAS PubMed; (c) E. Krieger, J. E. Nielsen, C. A. Spronk and G. Vriend, J. Mol. Graphics Modell., 2006, 25, 481 CrossRef CAS PubMed; (d) A. Jakalian, D. B. Jack and C. I. Bayly, J. Comput. Chem., 2002, 23, 1623 CrossRef CAS PubMed; (e) J. Uppenberg, M. T. Hansen, S. Patkar and T. A. Jones, Structure, 1994, 2, 293 CrossRef CAS PubMed; (f) U. Essmann, L. Perera and M. L. Berkowitz, J. Chem. Phys., 1995, 103, 8577 CrossRef CAS.
- B. M. Choudary, L. Chakrapani, T. Ramani, K. V. Kumar and M. L. Kantam, Tetrahedron, 2006, 62, 9571 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: Supplementary tables and figures; synthesis procedure of substrates and all the racemic standards; 1H and 13C NMR spectra for all β-hydroxy ketones and acylated β-hydroxy ketone derivatives; chiral HPLC conditions for all the related compounds. See DOI: 10.1039/c6ra12729k |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |