Fabrication of a monolithic cryogel from the cyclohexane organogel of a coordination polymer based on a phosphoester†
Abstract
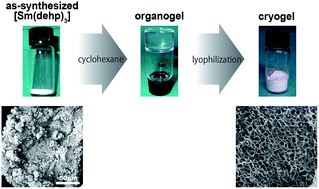
Since coordination polymers (CPs) are generally obtained as fine powders, the fabrication of a CP is an issue that should be addressed. A monolithic CP was successfully prepared through the lyophilization of a cyclohexane organogel of [Sm(dehp)3] consisting of Sm3+ and di-(2-ethylhexyl)phosphoric acid. The crystalline domains of the CP possibly act as cross-linking points, and the crystallization of cyclohexane is likely to generate the void space in the resulting cryogels. The architecture of the material can be varied by changing the volume ratio of cyclohexane to [Sm(dehp)3]. While the morphology observed through the scanning electron microscope was completely different from the as-synthesized material, powder X-ray diffraction analysis revealed that the crystalline structure did not change. The distribution coefficient and kinetic constant of ion exchange in the lanthanide series were improved by cryogel formation.

 Please wait while we load your content...
Please wait while we load your content...