Propene adsorption on gold–palladium nanoalloys supported on bundle nanotubes†
Hamed Akbarzadeh*,
Mohsen Abbaspour and
Elham Khomarian
Department of Chemistry, Faculty of Basic Sciences, Hakim Sabzevari University, 96179-76487 Sabzevar, Iran. E-mail: akbarzadehhamed@yahoo.com; Fax: +98 51 44410104; Tel: +98 915 3008670
First published on 6th July 2016
Abstract
Propene adsorption on (Pd–Au)N nanoalloys supported on carbon nanotube (CNT) bundles has been investigated using molecular dynamics (MD) simulations at 300 K. We have examined many of the possible effects, including the effects of the nanocluster size, cluster mole fraction, CNT bundle diameter, CNT bundle chirality, and effect of the different supports (such as graphene, BNNT bundle, and SiCNT bundle) on the thermodynamic, structural, and dynamic properties of propene adsorption on the deposited nanoalloys. Our results indicate that the nanocluster size and mole fraction have greater influence on the adsorption phenomena. The results also show that the smaller nanoclusters with a greater molar fraction of Pd supported on smaller CNT bundles have more coverage, higher enthalpy changes of adsorption, greater cluster-nanotube radial distribution functions (RDFs), and smaller self-diffusion coefficients than the other clusters. Our results also indicate that the chirality of the CNT bundle has a small effect on the different properties. The investigation of support effect shows that the following trend: graphene < CNT < BNNT < SiCNT exists for the adsorption coverage, enthalpy changes, RDF values, but the reverse trend exists for the self-diffusion coefficients of the nanoclusters.
1. Introduction
Bimetallic nanoclusters have recently received considerable attention for their unique optical, magnetic, and catalytic properties, which are very different from those of their mono-metallic components.1,2 Among the different bimetallic nanoclusters, gold–palladium (Au–Pd) is one of the most attractive nanoclusters because of its promising use as a catalyst in CO and benzyl oxidation, hydrogen peroxide and vinyl acetate monomer synthesis, hydrodechlorination of CClF2, hydrogenation of hydrocarbon, and cyclotrimerization of acetylene.3–9 It is also demonstrated that the chemical activity of the Au–Pd nanoclusters is strongly dependent not only on the particle size but also on the Au–Pd ratio at the surface. Much research has also been performed to determine the atomic structure of Au–Pd core–shell nanoclusters and their structural transformation upon annealing.11–13 Theoretical calculations predicted that the stable ordered structures of the bimetallic nanoclusters have a Pd core and a Au shell. Although nanoclusters can form a Au core and a Pd shell at low temperatures, but the Au atoms prefer to migrate to the outer shell with increasing temperature.10,14–16Propene (also known as propylene) is the second most important starting product in the petrochemical industry after ethylene. It is the raw material for a wide variety of industrial productions. Propene is also used for the production of important chemicals such as propylene oxide, acrylonitrile, cumene, butyraldehyde, and acrylic acid.17,18
The adsorption of propene on the nanoclusters and nanotubes has been a subject of some experimental and theoretical studies.19–22 For example Ajo et al.19 studied the adsorption of propene on TiO2(110) surface and on gold nanoparticles dispersed on TiO2(110), using temperature programmed desorption (TPD), X-ray photoelectron spectroscopy (XPS), and He+ low energy ion scattering spectroscopy (LEIS). Janssens et al.20 investigated the adsorption of propene on neutral gold clusters by pressure-dependent kinetic measurements and delayed unimolecular dissociation of the excited Aun–propene complexes. They also analyzed the cluster size (n = 9–25) and temperature (T = 90–300 K) dependence of the propene adsorption and showed a strong size dependences of the absorption reaction; a larger propene adsorption probability was found for gold clusters composed of an even number of atoms. Cruz et al.21 carried out configurational-bias Grand Canonical Monte Carlo (GCMC) simulations of propylene adsorption onto homogeneous bundles of single-walled carbon nanotubes at ambient temperature (T = 298.15 K) and over a pressure range of 0.1 bar ≤ p ≤ 10.4 bar. They also individually addressed distinct contributions from external sites (grooves and external surface) and endohedral volume (inter- and intra-tubular) for the bundles of nanotubes with different diameters. Zaporotskova et al.22 researched the interaction mechanism between single-walled carbon nanotubes and most common polymers that results in the formation of stable polymer based nanotube reinforced composites (such as polypropylene) by applying DFT method and B3LYP functional.
The catalytic properties of a metal nanocluster can be modified substantially by anchoring it to support surfaces.23,24 The thermal, structural and dynamic properties of metallic clusters supported on surfaces have also attracted much attention lately.23–32 Supported gold and gold alloy nanoparticles have been found to be highly active and selective in a variety of important catalytic reactions, particularly when the particles are only a few nm in size.33–40 Also, the large surface area of carbon nanotubes (CNTs) provides the possibility to deposit metallic nanoparticles on their surface in order to enhance their properties.41,42 The CNTs are considered as one of the important nanomaterials, which are of interest to many theoretical and experimental scientists due to their key physical and chemical properties.43–46 Therefore, both of CNTs and metallic nanoclusters have unique and interesting properties and integration of CNTs with metallic nanoclusters can lead to novel properties which may create major revolutions in relevant technologies.25
There are many theoretical investigations on the adsorption of different gases on the nanoclusters supported on the CNTs and their composites. Shamkhali and Akbarzadeh32 studied the adsorption of H2 on a bundle of single-walled carbon nanotubes (SWCNTs) and an Ag–Cu bimetallic nanoalloy located on the groove site of the bundle by molecular dynamics (MD) simulation. Their simulations were performed in order to study the effect of the Ag mole fraction and the diameter of the nanotube on the H2 adsorption. Akbarzadeh et al.31 performed MD simulations to study CO adsorption on Ag nanoclusters supported on carbon nanotube under various gas pressures at different temperatures. They found that the nanocluster structural changes are irreversible in such a way that by gradually decreasing the gas pressure to zero, the nanocluster geometry is not reversed to its initial structure in vacuum conditions. Recently, Akbarzadeh and Shamkhali47 studied H2 physisorption on AgN/carbon-nanotube composites by MD simulation in order to investigate the effect of nanocluster size, diameter and chirality of nanotube on the adsorption phenomena. Their calculations indicated that the increase of nanocluster size, decreases the H2 adsorption. More recently, Akbarzadeh et al.25 investigated CO adsorption on the (Ag–Au)N nanoalloys supported on carbon nanotubes (CNTs) at different temperatures and pressures via MD simulation. They studied effects of the nanoalloy size, nanocluster mole fraction, CNT diameter, and CNT chirality, and the effect of the support on the thermodynamic, structural, and dynamic properties of the CO adsorption on the supported nanoalloys. Their results indicated that the nanoalloy size and mole fraction have considerable influences on the adsorption phenomena.
In this work, we have performed MD simulation to investigate propene adsorption on (Au–Pd)N (with N = 108, 256, and 500) nanoalloys supported on the carbon nanotube bundles at 300 K. We have studied many of the possible effects; the effects of the nanoalloy size, nanoalloy mole fraction, CNT bundle diameter, CNT bundle chirality, and effect of the support on the thermodynamic, structural, and dynamic properties of the propene adsorption on the supported nanoalloy. The investigation of different properties of bimetallic nanoparticles in presence of gas is vitally important if real-world applications are to be achieved.48,49 Although the adsorption of propene on nanoclusters and nanotubes has a wide variety of industrial applications19–22 but there is not any experimental work about the propene adsorption on the supported Pd–Au nanoalloy on the CNT bundle in the literature. This work which is an atomistic simulation of propene adsorption on the supported Pd–Au nanoclusters tries to examine many effects to gain the insight into the special catalytic properties of supported Ag–Au nanoclusters in experimental interaction with propene.
2. Simulation details
In order to investigate the effect of the size of the Pd–Au nanocluster, three fcc-like (Au–Pd)N nanoclusters with N = 108, 256, and 500 atoms were placed on the support. Also (0.1, 0.3, 0.5, 0.7, and 0.9) were selected for Au mole fractions of the mentioned nanoclusters. In order to investigate the effect of the diameter of bundle nanotube, we have chosen three armchair nanotubes ((10,10), (15,15), and (20,20)). Moreover, in order to investigate the effect of chirality, (15,15), (26,0), and (18,12) bundle carbon nanotube with the same diameters were chosen. In addition to bundle carbon nanotube, boron nitride nanotubes, silicon carbide nanotubes and graphite were used in order to study the effect of support type.The initial simulation box was composed of the annealed nanocluster on the support, together with propene molecules at random positions. Periodic boundary conditions added in all directions. Furthermore, various pressures were applied by deleting some gas atoms at each temperature. By using this method, pressure reduced near the zero at several steps, and after each step, the final configuration was used as initial configuration for the next step. The values of pressure were calculated using the van der Waals equation of state.37,41 The MD simulations were performed in canonical ensemble (NVT) using Nóse–Hoover thermostat.50,51 The equations of motion were integrated using the Verlet-leapfrog algorithm52 with time steps of 1 fs. The simulations were carried out for 1 ns of equilibration followed by production time of 2 ns for calculated properties using the DLPOLY 4.03.53 All interatomic interactions between the atoms in the simulation box were calculated within the cutoff distance of 12 Å. For Pd–Pd, Au–Pd, and Au–Au interactions, quantum Sutten–Chen potentials (QSCs) were used.54,55 Also, the Lennard-Jones (12,6) potentials with Lorentz–Bertholet combination rules were used for support-nanocluster, support-propene, nanocluster–propene interactions.56,57 The support atoms were fixed in their positions. We have presented the snapshots of the propene adsorption of the (Au–Pd)256 nanocluster with the xAu = 0.5 supported on the (18,0) bundle carbon nanotube at 300 K in the Fig. 1.
 | ||
| Fig. 1 The snapshot of the propene adsorption on the (Pd–Au)256 nanocluster supported on the CNT bundle at 300 K. | ||
In order to examine the force fields used in the adsorption process, we have performed a DFT calculation with B3LYP level of theory on adsorption of one molecule propene on Au5Pd5 nanocluster. Our calculation resulted the adsorption energy of 0.413 eV which is in good agreement with the typical binding energy of propene to metal surfaces (0.35–0.4 eV).58–60 We have also put the Au5Pd5 nanocluster in an atmosphere of propene and performed a MD simulation to obtain the propene adsorption energy. Our simulation resulted the adsorption energy of 0.399 eV which is in good agreement with the DFT calculation.
3. Results and discussion
3.1 Effect of the nanocluster size
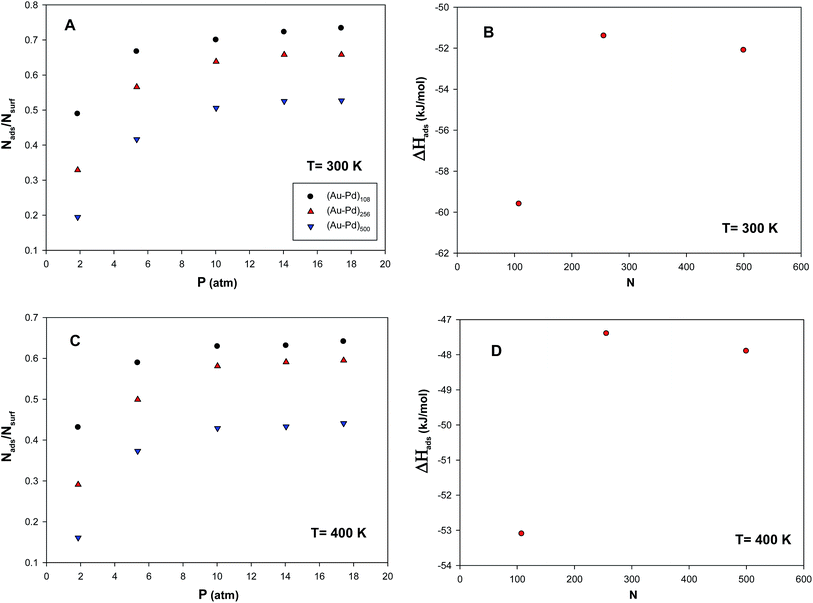
We have calculated the adsorption isotherms (Nads/Nsurf) versus pressure for propane adsorption on the (Au–Pd)N nanoalloys (with N = 108, 256, and 500) with the xAu = 0.5 supported on the (15,15) CNT at 300 K and presented in plot A in Fig. 2. In this plot, Nads is the number of adsorbates and Nsurf is the total number of nanoalloy surface atoms. According to plot A in Fig. 2, at constant temperature and pressure, the smaller nanoclusters have more coverage than larger ones which is due to the more number of surface atoms and adsorption sites in smaller nanoalloys than the bigger ones. The similar results have been also reported by Akbarzadeh et al.25,31,47 for CO and H2 adsorptions on the Ag and Ag–Au nanoclusters with different number of atoms supported on the SWCNT. They also observed that the increase of the supported nanocluster size, decreases the gas adsorption.We have also calculated the enthalpy of propene adsorption on the supported nanoclusters using the Clausius–Clapeyron equation and presented in plot B in Fig. 2. According to this plot, the enthalpy of adsorption increases as the nanocluster size decreases. It is also shown that the absolute value of adsorption enthalpy increases sharply with decreasing cluster size from 256 to 108. This is due to the fact that the smaller cluster has the larger surface/volume ratio and so it has lower cohesive energy than the bigger cluster. Therefore, the surface atoms in the smaller cluster have more tendencies to adsorb propene molecules in order to increase their cohesive energy. It is also shown in plot B in Fig. 2 that the enthalpy of adsorption has not a linear trend for the different cluster sizes. This can be due to the presence the different number and different kind of atoms in the cluster surface in the different sizes.
In order to examine the adsorption and enthalpy change trends, we have also calculated the adsorption isotherms and the enthalpy of propene adsorption on the (Au–Pd)N nanoalloys (with N = 108, 256, and 500) with the xAu = 0.5 supported on the (15,15) CNT at 400 K and presented in plots C and D in Fig. 2. According to these plots, the adsorption and enthalpy change values at 400 K show the similar trends of the corresponding values at 300 K. It is also expected and shown that the adsorption and enthalpy change values at 400 K are smaller than those values at 300 K.
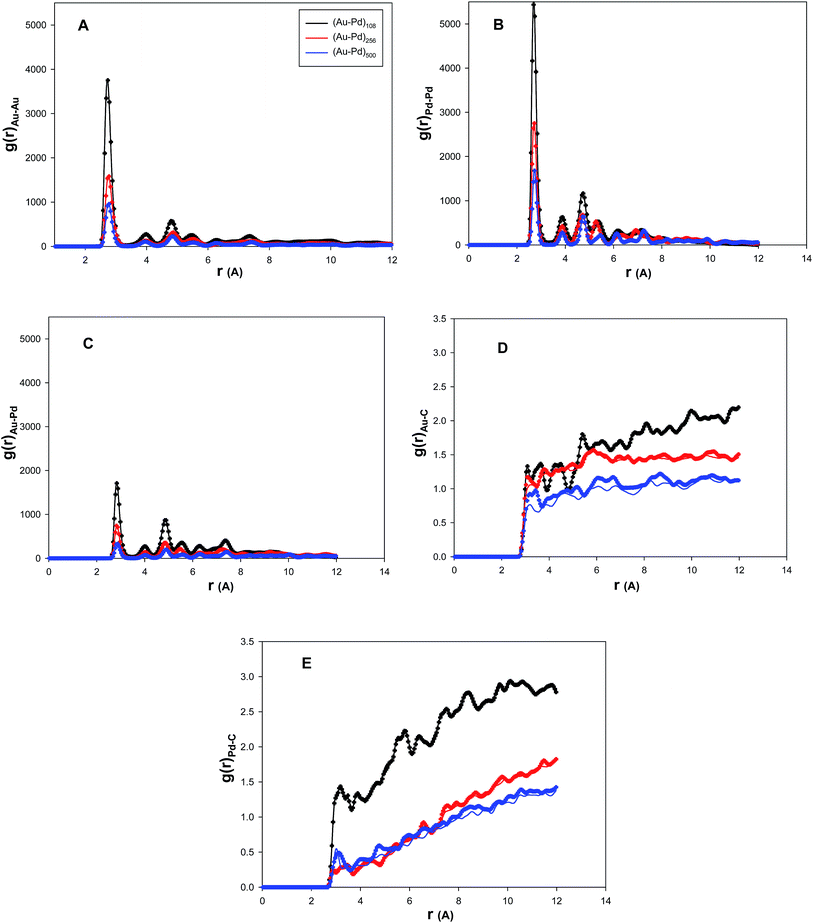
In order to investigate the structural changes of the nanocluster during the propene adsorption, we have calculated the Au–Au, Pd–Pd, Au–Pd, Au–C(CNT), and Pd–C(CNT) radial distribution functions (RDFs) of the (Au–Pd)N nanoalloy with the xAu = 0.5 supported on the (15,15) CNT bundle at 300 K in vacuum and in vacuum after removing the propene molecules and presented in the plots A, B, C, D, and E in Fig. 3. According to this figure, the RDFs of the smaller nanocluster are greater than those of the bigger one. This can be due to the greater interactions into the smaller nanocluster and also between the smaller cluster and the CNT bundle than those in the bigger one. In the other words, the smaller nanocluster has lower cohesive energy but has greater adhesive energy with the CNT bundle. This can be also due to the fact that for a small cluster, the proportion of atoms in contact with the nanotube wall is higher than that of the large cluster.
It is also shown that the first peaks of the Pd–Pd and Pd–C RDFs in the plots B and E are greater than the Au–Au and Au–C RDFs in the plots A and D for all of the nanoclusters. The first RDF peaks are also greatest for the smallest cluster. This is due to the greater Pd–Pd and Pd–C interactions than the Au–Au and Au–C interactions. It is also shown in plot E that the first peak of Pd–C in (Au–Pd)500 cluster is greater than that of the (Au–Pd)256 cluster. This can be due to the greater Pd–C interactions in the (Au–Pd)500 cluster than that of the (Au–Pd)256 cluster which means that the Au atoms tend to lie in the surface of the (Au–Pd)500 cluster more than the Pd atoms. In the other words, the Au/Pd proportion in the surface of the (Au–Pd)500 cluster is more than the (Au–Pd)256 cluster.
According to plots D and E, after removing the propene molecules, the g(r)Au–C and g(r)Pd–C are larger than those RDFs in the initial state (vacuum) especially for bigger clusters. This is due to the fact that the gas phase causes the nanocluster to roll around the nanotube and the interface structure between the nanocluster and the nanotube is expanded. Therefore, the interaction between the nanocluster surface atoms and the gas tends to stabilize the surface atoms on the nanocluster. This phenomenon can be better found in Fig. 4 in which we have presented the snapshots of the both smallest and biggest nanoclusters on the CNT bundles at 300 K. It can be observed in Fig. 4 that the cluster in contact with the CNT walls has expanded after removing the propene gas and it has a larger area than the initial state.
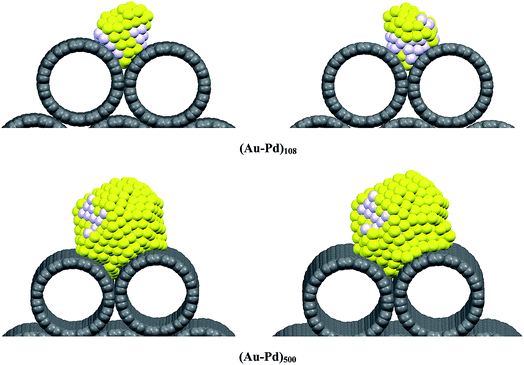 | ||
| Fig. 4 The snapshots of the both smallest and biggest nanoclusters on the (15,15) CNT bundles in vacuum (the left side) and in vacuum after removing the propene molecules (the right side) at 300 K. | ||
In order to examine the nanocluster diffusion during the gas adsorption, we have calculated the self-diffusion coefficient (D) for the different nanoalloys with the xAu = 0.5 supported on the (15,15) CNT bundle at 300 K and presented in Table 1. According to this table, the smaller cluster has slower dynamics than the larger one. This result is in agreement with the RDF results. It means that the smaller nanoalloy has greater interaction with the CNT bundle and so, it has greater RDF and smaller self-diffusion coefficients. It is also found that after removing the propene gas, the self-diffusion coefficients of the nanoclusters are lower than those values in the initial state (vacuum). This result is also in agreement with our Au–C and Pd–C RDFs.
| Nanoalloy size | D (109 m2 s−1) (vacuum, at initial) | D (109 m2 s−1) (vacuum, after exposing to propene) |
|---|---|---|
| 108 | 0.0076 | 0.00038 |
| 256 | 0.0151 | 0.00307 |
| 500 | 0.0210 | 0.00670 |
3.2 Effect of the nanotube bundle diameter
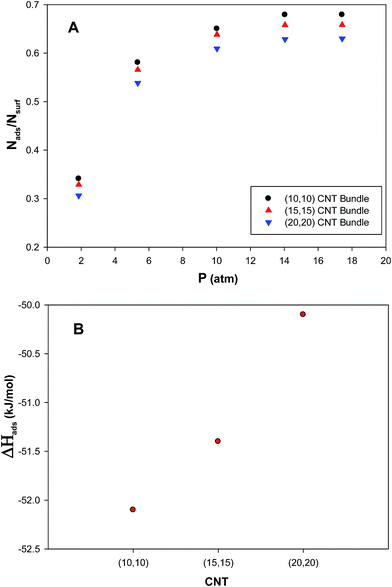
The isotherms for propene adsorption on the (Au–Pd)256 nanocluster with the xAu = 0.5 supported on the CNT bundles with the different diameters have been calculated at 300 K and presented in plot A in Fig. 5. According to this plot, the saturation coverage on the nanocluster surface increases with decreasing the diameter of the nanotube bundle. This can be due to the fact that the nanoalloy experiences more wetting on the smaller CNT bundle (due to the more curvature of the smaller nanotube than the bigger one). Therefore, the interface between the CNT bundle and the nanocluster increases by colliding the propene molecules to the nanoalloy surface which increases the interface between the nanoalloy and the gas phase and so the coverage increases. The increase in the interface is more for the smaller CNT bundle and so, the propene adsorption increases. The similar results have been also obtained by Akbarzadeh et al.25,31,47 for gas adsorptions on the Ag and Ag–Au nanoclusters on the SWCNT. They observed that as the diameter of nanotube is increased, the amount of the gas adsorption on the nanocluster is decreased.We have also calculated the ΔHads of the propene gas on the nanocluster with xAu = 0.5 supported on the CNT bundles with the different diameters and presented in plot B in Fig. 5. According to this plot, the absolute value of enthalpy change increases with decreasing the CNT bundle diameter which is due to the increasing of the interface (and interaction) between the smaller CNT bundle and the nanocluster. Therefore, the gas adsorption and the adsorption energy increases.
In order to investigate the effect of CNT bundle diameter on the RDF during the gas adsorption, we have calculated the different RDFs of the (Au–Pd)256 nanoalloy with the xAu = 0.5 supported on the CNT bundles with the different diameters at 300 K in vacuum and in vacuum after removing the propene molecules and presented in plots A–E in Fig. S1.† According to plots D and E, the Pd–C and Au–C RDFs in the smallest nanotube bundle are much greater than the RDFs in the bigger ones. This result is in agreement with our adsorption isotherm and energy results. In the other words, the interface between the smallest nanotube bundle and the nanocluster is greater than the bigger CNT bundles. Therefore, there are more number of the nanoalloy atoms surrounding the nanotube atoms of the smallest bundle than the bigger ones. We have also presented the snapshots of the (Au–Pd)256 nanocluster on the smallest and biggest CNT bundles at 300 K in Fig. 6. According to this figure, the cluster on the smallest bundle has expanded (after removing the propene gas) more than that on the biggest one. It can be also observed that the cluster on the smallest bundle has been achieved more stable state in which there is more interactions between the Au and Pd atoms than the initial state. This phenomena has been also observed by increasing the Au–Pd RDF of the nanocluster on the smallest bundle in plot C in Fig. S1.†
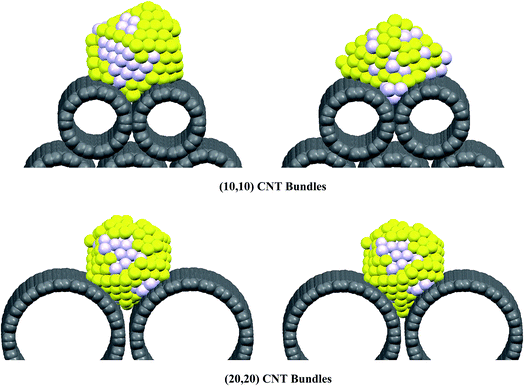 | ||
| Fig. 6 The snapshots of the (Au–Pd)256 nanocluster on the smallest and biggest CNT bundles in vacuum (the left side) and in vacuum after removing the propene molecules (the right side). | ||
In order to examine the effect of the bundle diameter on the nanocluster dynamics, we have calculated the self-diffusion coefficient for the cluster with xAu = 0.5 supported on the nanotube bundles with the different diameters and presented in Table 2. According to this table, the nanoalloy on the smaller bundle has slower dynamics than the bigger ones. This result is in agreement with our adsorption isotherm, enthalpy change, and RDF results. In the other words, when the nanocluster is supported on the smallest bundle, its interface and interaction with the nanotube increases which decreases the self diffusion coefficient of the cluster.
| CNT bundle | D (109 m2 s−1) (vacuum, at initial) | D (109 m2 s−1) (vacuum, after exposing to propene) |
|---|---|---|
| (10,10) | 0.00954 | 0.00088 |
| (15,15) | 0.0151 | 0.00307 |
| (20,20) | 0.0231 | 0.0051 |
3.3 Effect of the nanotube bundle chirality
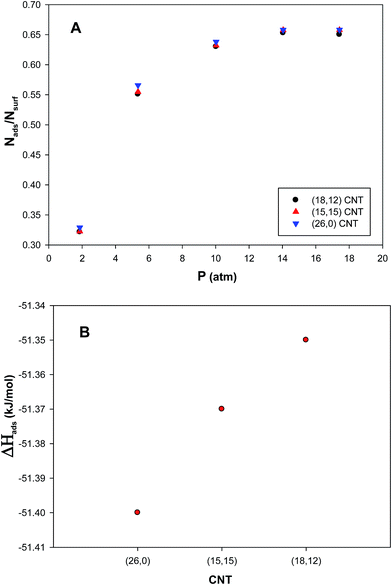
The propene adsorption isotherms on the (Au–Pd)256 nanocluster with the xAu = 0.5 supported on the zigzag (26,0), armchair (15,15), and chiral (18,12) CNT bundles have been computed at 300 K and presented in plot A in Fig. 7. According to this plot, there are small differences between the results of the different chiralities. It is also shown that the saturation value of coverage is more for the zigzag bundle. This can be due to the greater interaction of the nanocluster supported on the zigzag bundle and the propene gas than the armchair one. It means that the interface between the zigzag bundle and the cluster is (a little) more increased than that of the armchair bundle which increases the interface between the cluster and the gas molecules and so the interaction and the gas adsorption increases. Recently, Akbarzadeh et al.25 observed that the Ag–Au nanocluster supported on a zigzag SWCNT adsorbs more CO gas than an armchair nanotube. Akbarzadeh et al.61 also concluded that the confined molecules of an ionic liquid have greater interactions with a zigzag CNT than an armchair one.The enthalpy change of propene adsorption on the supported nanoclusters have been also presented in plot B in Fig. 7. It is shown that the differences between the energy values are small. According to this plot, the absolute value of ΔHads for the zigzag bundle is greater than that of the armchair and chiral ones. This can be also due to the greater interaction of the propene molecules and the cluster supported on the zigzag bundle than the armchair and chiral ones.
To investigate the chirality effect on the RDF during the gas adsorptions, we have computed the different RDFs of the nanoalloys with xAu = 0.5 supported on the nanotube bundles with the different chiralities at 300 K and presented in plots A–E in Fig. S2.† According to plot E, the peaks are greater for the zigzag bundle than the armchair and chiral configurations which is due to the greater interaction between the nanoalloy and the zigzag bundle which increases the interface between the cluster and the bundle. In the other words, there are more probability of finding number of the nanoalloy atoms surrounding the carbon atoms of the zigzag bundle than the armchair and chiral ones. The differences between the RDF peaks are smaller for the Au–C RDF (plot D) than the Pd–C values (plot E) which is due to the greater Pd–C interactions than the Au–C interactions. We have also presented the snapshots of the (Au–Pd)256 nanocluster on the CNT bundles with the different chiralities at 300 K in Fig. 8.
 | ||
| Fig. 8 The snapshots of the (Au–Pd)256 nanocluster on the CNT bundles with the different chiralities. | ||
In order to examine the effect of the bundle chirality on the nanoalloy dynamics, we have calculated the self-diffusion coefficient for the nanocluster with xAu = 0.5 supported on the bundles with the different chiralities at 300 K and presented in Table 3. It is also shown that there are small differences between the values corresponding to the different chiralities. It is observed that the nanoalloy on the zigzag bundle has slower dynamics than the armchair and chiral ones. This result is in agreement with our adsorption isotherms, the enthalpy changes, and the RDF values. It means when the nanocluster is supported on the zigzag bundle, its interface and interaction with the nanotube increases which decreases the diffusion of the deposited cluster.
| CNT bundle | D (109 m2 s−1) (vacuum, at initial) | D (109 m2 s−1) (vacuum, after exposing to propene) |
|---|---|---|
| (18,12) | 0.0198 | 0.00397 |
| (15,15) | 0.0161 | 0.00333 |
| (26,0) | 0.0151 | 0.00307 |
3.4 Effect of the nanocluster mole fraction
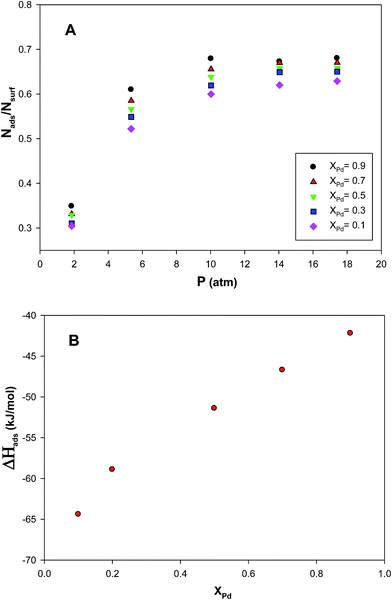
The propene adsorption isotherms of the (Au–Pd)256 nanoclusters with the different mole fractions (xPd = 0.1, 0.3, 0.5, 0.7, and 0.9) supported on the (15,15) nanotube bundle have been computed at 300 K and presented in plot A in Fig. 9. According to this plot, the saturation value of coverage increases as the mole fraction of Pd is increased. This can be due to the greater Pd–propene interaction than the Au–propene interaction. It should be also noted that the adsorption of propene on the nanocluster is a physical phenomena and this physisorption is not usually site dependent. Therefore, the isotherms for the different Pd mole fractions are almost similar.The enthalpy change of propene adsorption on the supported nanoclusters with the different Pd mole fractions have been also presented in plot B in Fig. 9. It is shown that the absolute value of ΔHads increases with increasing the xPd which is due to the greater Pd–propene interaction than the Au–propene interaction. This result is also in agreement with the adsorption isotherms.
To examine the effect of the Pd mole fraction on the RDF during the propene adsorption, we have calculated the different RDFs for the nanoalloys with the different mole fractions and presented in plots A–E in Fig. S3.† According to plot A, the Au–Au RDF of the cluster with xAu = 0.9 (or xPd = 0.1) is greater than the cluster with xAu = 0.1 which means that there is more probability of finding the Au atoms in the nanocluster with xAu = 0.9 than other clusters around a reference atom. The same situation exists for the g(r)Au–C for the nanocluster with xAu = 0.9 in plot D. These results are due to the competitive adsorption of the Au and Pd atoms on the bundle surface; the g(r)Pd–C decreases when the g(r)Au–C increases. The same situations also exist for the g(r)Pd–Pd and g(r)Pd–C for the cluster with xPd = 0.9 in plots B and E. We have also presented the snapshots of the (Au–Pd)256 nanoclusters with the different mole fractions supported on the (15,15) nanotube bundle at 300 K in Fig. 10.
 | ||
| Fig. 10 The snapshots of the (Au–Pd)256 nanoclusters with the different mole fractions supported on the (15,15) CNT bundle. | ||
In order to investigate the nanoalloy self-diffusion during the propene adsorption, we have compared the self-diffusion coefficient of the nanoclusters with the different mole fractions at 300 K and presented in Table 4. According to this table, the nanoalloy with xPd = 0.9 has lower self-diffusion than that of the xPd = 0.1. This is in agreement with our previous results and is due to the greater Pd–Pd and Pd–C interactions than the Au–Au and Au–C interactions.
| xPd | D (109 m2 s−1) (vacuum, at initial) | D (109 m2 s−1) (vacuum, after exposing to propene) |
|---|---|---|
| 0.1 | 0.0294 | 0.0086 |
| 0.5 | 0.0151 | 0.00307 |
| 0.9 | 0.00312 | 0.000187 |
3.5 Effect of the support
In order to examine the support effect on the propene adsorption, we have calculated the gas adsorption isotherms on the (Ag–Au)256 nanoclusters with the xAu = 0.5 supported on graphene, (15,15) CNT, (15,15) BNNT, and (15,15) SiCNT at 300 K and presented in plot A in Fig. 11. According to this plot, the amount of gas adsorption obeys the following trend: graphene < CNT < BNNT < SiCNT. In the other words, the nanoalloy supported on the SiCNT bundle has more saturation value than the other supports. This can be due to the greater interaction between the nanoalloy and the SiCNT bundle than the other supports. This leads to exposing more atoms on the surface of the cluster deposited on the SiCNT than the other supports. Therefore, the interface between the SiCNT bundle and the cluster increases which increases the interface between the cluster and the gas phase and so the coverage increases more than the other supports.The enthalpy change of the gas adsorption on the nanocluster deposited on the different supports have been also presented in plot B in Fig. 11. It is shown that the absolute value of ΔHads increases as: graphene < CNT < BNNT < SiCNT. This trend is due to the same order of the interactions between the different supports and the nanocluster. In the other words, due to the more interface between the cluster and gas molecules, there is greater interaction between the propene molecules and the cluster supported on the SiCNT bundle than other supports.
According to the RDF plots in Fig. S4,† the peaks of the Au–Au, Pd–Pd, Au–Pd, Au–nanotube, and Pd–nanotube RDFs in plots A–E are greater for the cluster deposited on the SiCNT bundle than the other supports which is also due to the greater interaction between the nanoalloy and the SiCNT bundle which increases the interface between the cluster and the bundle. We have also presented the snapshots of the (Au–Pd)256 nanocluster deposited on the different supports at 300 K in Fig. 12. It can be also observed that the interface between the nanoalloy and the SiCNT bundle has been increased more than the other supports.
According to the self-diffusion coefficients in Table 5, the nanoalloy deposited on the SiCNT bundle has slower dynamics than the other supports. This result is in agreement with our adsorption isotherms, the enthalpy changes, and the RDF values. It means when the nanocluster is supported on the SiCNT bundle, its interface and interaction with the nanotube increases which decreases the diffusion of the deposited cluster.
| Support | D (109 m2 s−1) (vacuum, at initial) | D (109 m2 s−1) (vacuum, after exposing to propene) |
|---|---|---|
| Graphite | 0.0492 | 0.00739 |
| (15,15) CNT | 0.0283 | 0.00504 |
| (15,15) BNNT | 0.0151 | 0.00307 |
| (15,15) SiCNT | 0.0090 | 0.00150 |
4. Conclusions
We have investigated propene adsorption on the (Au–Pd)N nanoalloys supported on the nanotube bundles at 300 K using the MD simulations. We have investigated many of the possible effects; the effects of the nanocluster size, cluster mole fraction, bundle diameter, bundle chirality, and effect of the support on the thermodynamic, structural, and dynamic properties of the gas adsorption on the deposited nanoalloys.Our cluster size investigations indicated that the smaller supported nanoclusters have more coverage than larger ones which is due to the more number of surface atoms and in smaller nanoalloys than the bigger ones. Also, the enthalpy of adsorption increases as the nanocluster size decreases. This is due to the fact that the smaller cluster has lower cohesive energy and so, it has more tendency to adsorb propene molecules. The structural results showed that after removing the propene molecules, the interface structure between the nanocluster and the nanotube has been expanded. Our results also showed that the smaller cluster has slower dynamics than the larger one. This is in agreement with the adsorption isotherms, enthalpy changes, and RDF results which is due to the greater interaction of the smaller cluster and the CNT bundle than the bigger one.
Our CNT bundle diameter analysis showed that the saturation coverage on the nanocluster surface increases with decreasing the diameter of the nanotube bundle. This is due to the fact that the nanoalloy experiences more wetting on the smaller CNT bundle. Also, the absolute value of enthalpy change increases with decreasing the CNT bundle diameter which is due to the increasing of the interface between the smaller CNT and the nanocluster. The structural results also showed that the interface between the smallest nanotube bundle and the nanocluster is greater than the bigger ones. The nanoalloy on the smaller bundle has also slower dynamics than the bigger ones.
Our investigations showed that there are small differences between the results obtained from the CNT bundles with the different chiralities. It is also shown that the saturation value of coverage is more for the zigzag bundle. Also, the absolute value of ΔHads for the zigzag bundle is greater than that of the armchair and chiral ones. This can be also due to the greater interaction of the propene molecules and the cluster supported on the zigzag bundle than the armchair and chiral ones. The structural results also confirmed these findings. It is also observed that the nanoalloy on the zigzag bundle has slower dynamics than the armchair and chiral ones.
Our cluster mole fraction investigations showed that the saturation value of coverage increases as the mole fraction of Pd is increased. Also, the absolute value of ΔHads increases with increasing the xPd but the self-diffusion coefficient decreases with increasing the xPd. These results can be due to the greater Pd–propene and Pd–Pd interactions than the Au–propene and Au–Au interactions. Our structural results also showed the competitive adsorption of the gas molecules by the Au and Pd atoms on the bundle surface.
The support investigations indicated that the nanoalloy supported on the SiCNT bundle has more saturation value than the other supports. This can be due to the greater interaction between the nanoalloy and the SiCNT bundle than the other supports. For the same reason, the nanoalloy deposited on the SiCNT bundle has slower dynamics than the other supports. The amount of gas adsorption and the ΔHads obey the following trend: graphene < CNT < BNNT < SiCNT. Our structural results also confirmed these findings.
The comparison between the different effects indicates that the nanoalloy size and mole fractions have stronger effects on the propene adsorption. Also, the bundle chirality and diameter have weaker effects than other ones.
This study offers an atomistic simulation of propene adsorption on the supported Pd–Au nanoclusters and investigated the many effects (size, composition, CNT bundle diameter and chirality, and support) to enhance the insight into the special catalytic properties of supported Ag–Au nanoclusters for the different reactions of propene (such as epoxidation).
References
- Y. Ding, F. Fan, Z. Tian and Z. L. Wang, J. Am. Chem. Soc., 2010, 132, 12480 CrossRef CAS PubMed.
- C. Burda, X. B. Chen, R. Narayanan and M. A. El-Sayed, Chem. Rev., 2005, 105, 1025 CrossRef CAS PubMed.
- M. S. Chen, D. Kumar, C. W. Yi and D. W. Goodman, Science, 2005, 310, 291 CrossRef CAS PubMed.
- C. W. Yi, K. Luo, T. Wei and D. W. Goodman, J. Phys. Chem. B, 2005, 109, 18535 CrossRef CAS PubMed.
- D. Wang, A. Villa, F. Porta, D. S. Su and L. Prati, Chem. Commun., 2006, 1956 RSC.
- A. A. Herzing, A. F. Carley, J. K. Edwards, G. J. Hutchings and C. J. Kiely, Chem. Mater., 2008, 20, 1492 CrossRef CAS.
- H. Heinz, B. L. Farmer, R. B. Pandey, J. M. Slocik, S. S. Patnaik, R. Pachter and R. R. Naik, J. Am. Chem. Soc., 2009, 131, 9704 CrossRef CAS PubMed.
- J. Naughton, A. F. Lee, S. Thompson, C. P. Vinod and K. Wilson, Phys. Chem. Chem. Phys., 2010, 12, 2670 RSC.
- M. Yang, Z. Wang, W. Wang and C. Liu, Nanoscale Res. Lett., 2014, 9, 405 CrossRef PubMed.
- H. B. Liu, U. Pal, A. Medina, C. Maldonado and J. A. Ascencio, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 71, 075403 CrossRef.
- C. J. Baddeley, C. J. Barnes, A. Wander, R. M. Ormerod, D. A. King and R. M. Lambert, Surf. Sci., 1994, 314, 1 CrossRef CAS.
- I. Atanasov and M. Hou, Surf. Sci., 2009, 603, 2639 CrossRef CAS.
- F. Liu, D. Wechsler and P. Zhang, Chem. Phys. Lett., 2008, 461, 254 CrossRef CAS.
- J. A. Boscoboinik, C. Plaisance, M. Neurock and W. T. Tysoe, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 77, 045422 CrossRef.
- D. W. Yuan, X. G. Gong and R. Q. Wu, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 78, 035441 CrossRef.
- H. B. Liu, U. Pal, R. Perez and J. A. Ascencio, J. Phys. Chem. B, 2006, 110, 5191 CrossRef CAS PubMed.
- Market Study: Propylene, Ceresana, 2nd edn, 2015, http://www.ceresana.com Search PubMed.
- S. Budavari, 8034. Propylene, The Merck Index, Merck & Co, New Jersey, 12th edn, 1996 Search PubMed.
- H. M. Ajo, V. A. Bondzie and C. T. Campbell, Catal. Lett., 2002, 78, 359 CrossRef CAS.
- E. Janssens, H. T. Le and P. Lievens, Chemistry, 2015, 21, 15256 CrossRef CAS PubMed.
- F. J. A. L. Cruz, I. A. A. C. Esteves, J. P. B. Mota, S. Agnihotri and E. A. Müller, J. Nanosci. Nanotechnol., 2010, 10, 2537 CrossRef CAS PubMed.
- I. V. Zaporotskova, A. A. Krutoyarov and N. P. Polikarpova, Comput. Modell. New Technol., 2015, 19, 19 Search PubMed.
- A. R. Harutyunyan, T. Tokune and E. Mora, Appl. Phys. Lett., 2005, 87, 051919 CrossRef.
- J. C. Jimenez-Saez, A. M. C. Perez-Martin and J. J. Jimenez-Rodrıguez, Nucl. Instrum. Methods Phys. Res., Sect. B, 2006, 248, 816 CrossRef.
- H. Akbarzadeh, M. Abbaspour and S. Salemi, New J. Chem., 2016, 40, 310 RSC.
- Y. Shibuta and S. Maruyama, Chem. Phys. Lett., 2007, 437, 218 CrossRef CAS.
- C. F. Sanz-Navarro, P. O. Astrand, D. Chen, M. Ronning, A. C. T. van Duin, J. E. Mueller and W. A. Goddard, J. Phys. Chem. C, 2008, 112, 12663 CAS.
- C. F. Sanz-Navarro, P. Astrand, D. Chen, M. Ronning, A. C. T. van Duin and W. A. Goddard, J. Phys. Chem. C, 2010, 114, 3522 CAS.
- S. Jalili, C. Mochani, M. Akhavan and J. Schofield, Mol. Phys., 2012, 110, 267 CrossRef CAS.
- H. Akbarzadeh and H. Yaghoubi, J. Colloid Interface Sci., 2014, 418, 178 CrossRef CAS PubMed.
- H. Akbarzadeh, H. Yaghoubi, A. N. Shamkhali and F. Taherkhani, J. Phys. Chem. C, 2014, 118, 9187 CAS.
- A. N. Shamkhali and H. Akbarzadeh, RSC Adv., 2014, 4, 60866 RSC.
- M. S. Chen, D. Kumar, C. W. Yi and D. W. Goodman, Science, 2005, 310, 291 CrossRef CAS PubMed.
- M. S. Chen and D. W. Goodman, Acc. Chem. Res., 2006, 39, 739 CrossRef CAS PubMed.
- A. S. K. Hashmi and G. J. Hutchings, Angew. Chem., 2006, 45, 7896 CrossRef PubMed.
- C. D. Pina, E. Falletta, L. Prati and M. Rossi, Chem. Soc. Rev., 2008, 37, 2077 RSC.
- C. L. Bracey, P. R. Ellis and G. J. Hutchings, Chem. Soc. Rev., 2009, 38, 2231 RSC.
- J. A. Rodriguez, J. Evans, J. Graciani, J. B. Park, P. Liu, J. Hrbek and J. F. Sanz, J. Phys. Chem. C, 2009, 113, 7364 CAS.
- J. Pritchard, L. Kesavan, M. Piccinini, Q. A. He, R. Tiruvalam, N. Dimitratos, J. A. Lopez-Sanchez, A. F. Carley, J. K. Edwards, C. J. Kiely and G. J. Hutchings, Langmuir, 2010, 26, 16568 CrossRef CAS PubMed.
- L. Kesavan, R. Tiruvalam, M. H. Ab Rahim, M. I. Bin Saiman, D. I. Enache, R. L. Jenkins, N. Dimitratos, J. A. Lopez-Sanchez, S. H. Taylor, D. W. Knight, C. J. Kiely and G. J. Hutchings, Science, 2011, 331, 195 CrossRef CAS PubMed.
- J. H. Lee, N. R. Kim, B. J. Kim and Y. C. Joo, Carbon, 2012, 50, 98 CrossRef CAS.
- Y. Shi, Z. Liu, B. Zhao, Y. Sun, F. Xu, Y. Zhang, Z. Wen, H. Yang and Z. Li, J. Electroanal. Chem., 2011, 656, 29 CrossRef CAS.
- S. Peng and K. Cho, Nano Lett., 2003, 4, 513 CrossRef.
- A. G. Mamalis, L. O. G. Vogtlander and A. Markopoulos, Precis. Eng., 2004, 28, 16 CrossRef.
- K. C. Chin, A. Gohel, H. I. Elim, W. Chen, W. Ji, G. L. Chong, C. H. Sow and A. T. S. Wee, J. Mater. Res., 2006, 21, 2758 CrossRef CAS.
- K. Kostareos, L. Lacerda, G. Patorin, W. Wu, S. Weickowski, J. Luangsivilay, S. Godefroy, D. Pantarotto, J. P. Briand, S. Muller, M. Prato and A. Bianco, Nat. Nanotechnol., 2007, 2, 108 CrossRef PubMed.
- H. Akbarzadeh and A. N. Shamkhali, J. Comput. Chem., 2015, 36, 433 CrossRef CAS PubMed.
- J. F. Sanchez-Ramirez, U. Pal, L. Nolasco-Hernandez, J. Mendoza-Alvarez and J. A. J. Pescador-Rojas, Nanomaterials, 2008, 1 CrossRef.
- S. Link and M. A. El-Sayed, J. Phys. Chem. B, 1999, 103, 8410 CrossRef CAS.
- S. Nóse, J. Phys.: Condens. Matter, 1990, 2, 115–119 CrossRef.
- W. G. Hoover, Phys. Rev. A, 1985, 31, 1695–1697 CrossRef.
- M. P. Allen, and D. J. Tildesley, Computer Simulation of Liquids, Clarendon, Oxford, 1997 Search PubMed.
- W. Smith and I. T. Todorov, Mol. Simul., 2006, 32, 935–943 CrossRef CAS.
- Y. K. T. Cagin, Y. Qi, H. Ikeda, W. L. Johnson and W. A. Goddard III, Mater. Res. Soc. Symp. Proc., 1999, 554, 43–44 CrossRef.
- Y. Qi, T. Çağin, Y. Kimura and W. A. Goddard III, J. Comput.-Aided Mater. Des., 2001, 8, 233–243 CrossRef CAS.
- M. Neek-Amal, R. Asgari and M. R. Rahimi Tabar, Nanotechnology, 2009, 20, 135602–135608 CrossRef CAS PubMed.
- M. Malek and M. Sahimi, J. Chem. Phys., 2010, 132, 014310 CrossRef PubMed.
- S. M. Wetterer, D. J. Lavrich, T. Cummings, S. L. Bernasek and G. Scoles, J. Phys. Chem. B, 1998, 102, 9266–9275 CrossRef CAS.
- D. Dalucu, J. E. Klemberg-Sapieha and L. Martinu, Surf. Sci., 2001, 472, 33 CrossRef.
- A. M. Venezia, V. La Parola, G. Deganello, B. Pawelec and J. L. G. Fierro, J. Catal., 2003, 215, 317 CrossRef CAS.
- H. Akbarzadeh, M. Abbaspour, S. Salemi and M. Dastoorani, ChemPhysChem, 2015, 16, 1676 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra12350c |
| This journal is © The Royal Society of Chemistry 2016 |