Dual functional polylactide–hydroxyapatite nanocomposites for bone regeneration with nano-silver being loaded via reductive polydopamine†
Abstract
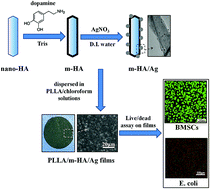
In bone tissue engineering, failure might take place in treating infected bone defects. Scaffolding materials with antibacterial function should be a potential solution for this problem. Herein, poly(L-lactide) (PLLA)–hydroxyapatite (HA) nanocomposites containing antibacterial silver nanoparticles (AgNPs) were prepared. In the preparation, surface coating of polydopamine (PDA) on HA nano-rods was a critical step to ensure both the homogeneous dispersion of HA nano-rods in the PLLA matrix and the successful loading of AgNPs onto HA nano-rods. With the total content of HA being fixed at 30 wt%, PLLA/HA nanocomposites containing different amounts of AgNPs (∼0.54 wt%, ∼0.27 wt% and ∼0.18 wt%) were prepared by mixing PDA-coated HA nano-rods (m-HA) with or without AgNPs at different ratios into PLLA. The resulting PLLA/HA/Ag nanocomposites demonstrated strong antibacterial activity against Escherichia coli at low content of AgNPs being incorporated, while they did not show significant cytotoxicity to bone mesenchymal stromal cells (BMSCs). In comparison with those cells cultured on pure PLLA film and tissue culture polystyrene, on the contrary, osteogenic differentiation of BMSCs was enhanced on PLLA/HA/Ag nanocomposite films benefiting from the HA component. The results suggested that PLLA/HA/Ag nanocomposites are anticipated to be good scaffolding materials for bone regeneration with dual functions of both antibacterial activity and osteocompatibility.

 Please wait while we load your content...
Please wait while we load your content...