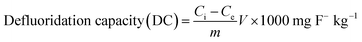A facile synthesis of metal ion-imprinted graphene oxide/alginate hybrid biopolymeric beads for enhanced fluoride sorption
Kalimuthu Pandi and
Natrayasamy Viswanathan*
Department of Chemistry, Anna University, University College of Engineering – Dindigul, Reddiyarchatram, Dindigul – 624 622, Tamilnadu, India. E-mail: drnviswanathan@gmail.com; Fax: +91-451-2554066; Tel: +91-451-2554066
First published on 22nd July 2016
Abstract
The enhanced surface properties of graphene oxide (GO) illustrate its vital role in environmental remediation. However, it is difficult to use it in column studies as it causes a pressure drop during field applications. To overwhelm its drawbacks and to improve its adsorption feature, GO was synthesized in a usable bead form by interspersing GO within an alginate (Alg) polymeric matrix and cross-linked with La(III) ions (GOAlgLa). The synthesized GOAlgLa composite beads not only display good field applications but also demonstrate an extremely enhanced defluoridation capacity (DC) compared with GO and calcium cross-linked alginate beads (AlgCa). The DC of GOAlgLa, AlgCa composite beads and GO was found to be 6617, 618 and 2438 mg F− kg−1, respectively. To determine the effect of different influencing parameters, such as pH, contact time, competitor co-anions, temperature and initial fluoride concentration, studies were conducted in batch mode. The sorbents were analyzed using various characterization techniques such as FTIR, TEM, SEM, EDAX, and Raman analysis. The characteristics of the sorption process were investigated using Freundlich, Langmuir, and D–R isotherms. The value of the thermodynamic parameters indicates that the fluoride sorption onto GOAlgLa composite beads was endothermic and spontaneous in nature. GOAlgLa composite beads also reveal a good regenerability over repeated adsorption/desorption processes. The applicability of hybrid beads to field water sample indicate its adaptable nature at field conditions.
1. Introduction
Removal of toxic ions from water has attracted considerable concern in worldwide, due to their adverse effects on human beings and environment. Fluoride causes adverse effects to human health when its concentration exceeds >1.5 mg L−1 in drinking water. The excess fluoride levels in drinking water leads to different forms of fluorosis.1 One of the foremost sources of fluoride ingestion by a human being is drinking water. The major sources of fluoride contaminants are fluoride bearing rocks/minerals and various industrial activities. Minerals such as fluorite, cryolite, ralstonite, and mica that contains fluoride prevalent components of sediments.2 In developing countries, ground water is used for drinking, cooking, and domestic purposes; however, unfortunately, the fluoride concentrations are more than 1.5 mg L−1 in several places in India and other foreign countries. Hence, it is necessary to provide water with safe fluoride levels to the common people to prevent them from devouring water contaminated with an excess amount of fluoride.Numerous fluoride remediation technologies, including precipitation, ion-exchange, electrocoagulation, electrodialysis, nanofiltration, reverse osmosis, and adsorption,3–9 have been developed. Among them, adsorption is believed to be a more simple, efficient, and universal option than the other reported techniques. The recycling of the adsorbent and the low generation of residues in this technique are some of the advantages over the other fluoride removal methods.10–12 Researchers all over the world have paid plentiful efforts to developing a novel and efficient adsorbent for the removal of fluoride from aqueous solutions.
Graphene oxide has received considerable attention in recent years and shows a promising adsorption performance towards toxic ions owing its huge surface area, copious surface functional groups (hydroxyl and carboxyl groups), incredible dispersive properties in water, and easy modification.13–19 Although GO possesses good adsorption efficiency for the removal of toxic ions, the application of GO in column studies is restricted due to its powdered nature, brittleness, and poor mechanical strength. To overcome such an industrial blockage, polymer assisted GO composite beads were prepared. The synthesized polymeric composite beads have unique properties such as hardness, inflexibility, and mold shrinkage, which help in the development of modern technology.20–22
Recently, researchers have paid more attention to the biosorption process for environmental remediation because of its eco-friendly, non-toxic, biofunctional, biocompatible, and biodegradable properties.23–25 Alginate is a biopolymer and it is a collection of two different monomers viz. (1 → 4) β-D-mannuronate and (1 → 4) α-L-guluronate and it is obtained from brown seaweeds. Alginate assisted composite adsorbents were extensively studied for toxic ion removal due its non-toxic, biodegradable and biocompatible nature.26–30 It possesses very good affinity towards different metal ions to form ionotropic alginate metal complex beads. However, divalent cation cross-linked alginate composites beads are unstable in an acidic medium because the divalent metal ions can be easily replaced by H+ ions.31 To improve the adsorption capacity, reusability, and solidity, high valence metal ions (trivalent and tetravalent) are utilized as cross-linkers to develop the hybrid beads. Recently, the development of new adsorbents with selectivity and high performance towards fluoride was accomplished by the incorporation of high valence metal ions into the adsorbent materials.32–40 Hence, we synthesized La(III) ion cross-linked alginate/graphene oxide composite beads for the sorption of fluoride.
The present investigation is aimed at the synthesis of economical, efficient, and eco-friendly composite beads by interspersing GO in Alg polymeric matrix and cross-linked with La(III) ion. The developed GOAlgLa composite beads were effectively utilized for fluoride sorption. To determine the effect of different influencing parameters such as competitor co-anions, pH, contact time, initial fluoride concentration, and temperature, batch mode was carried out. A comparative study was conducted for GO, AlgCa, and GOAlgLa composite beads to estimate the defluoridation capacity. The experimental results were fitted with various isotherm and thermodynamic parameters. The effective utilization of GOAlgLa composite beads in the field water sample was done at a nearby fluoride rife area in Dindigul district of Tamilnadu.
2. Experimental section
2.1. Materials
Sodium alginate was purchased from Himedia (India). Graphite powder was purchased from Central Drug House (India). Conc. H2SO4, NaNO3, KMnO4, H2O2, CaCl2·2H2O, LaCl3·7H2O and all other chemicals were purchased from Merck (India) and used as such without further purification. A fluoride solution containing 1000 mg L−1 was prepared by dissolving 2.21 g of NaF (AR grade) in 1000 mL of double distilled water. The working solution of 20 mg L−1 for the batch fluoride sorption process was then prepared by appropriate dilution of the stock solution.2.2. Synthesis of GO
Graphene oxide was synthesized from natural graphite powder by a modified Hummers method.41 Briefly, 24 mL of conc. H2SO4 was added to 1 g of graphite powder. Then, 0.5 g of NaNO3 was added into the reaction mixture, which was then cooled to 0 °C. 1.5 g of KMnO4 was slowly added to the reaction mixture, and then the temperature was maintained at 20 °C. The content was heated to 35 °C and maintained for 45 min. Then, 46 mL distilled water was added to the reaction mixture and the temperature of the content was maintained at 98 °C for 15 min. The reaction was quenched by the addition of 144 mL of distilled water and 1 mL of a 30% H2O2 solution. The final product was cooled to room temperature and washed with a 0.1 M HCl solution several times and distilled water to remove metal ions and then dried.2.3. Synthesis of AlgCa and GOAlgLa composite beads
About 0.5 g of GO was homogeneously dispersed in 100 mL of distilled water. To avoid agglomeration of GO in water during dispersed, the process was carried out by sonication and followed by mechanical stirring for about 3 h. To the dispersion solution, 2 g of sodium alginate was added and stirred vigorously for 3 h. This homogeneous solution was used to prepare the composite beads by dropping into 0.2 mol L−1 of a LaCl3·7H2O aqueous solution to obtain GOAlgLa composite beads. AlgCa composite beads were synthesized by adding 2 g of sodium alginate to 100 mL of distilled water under vigorous stirring for 3 h. Then, the homogeneous alginate solution was dropped into 0.2 mol L−1 of a CaCl2·2H2O solution to obtain AlgCa composite beads. Then, the formed AlgCa and GOAlgLa composite beads were kept in the mother solution for about 24 h to allow the cross-linking reaction to take place. The prepared AlgCa and GOAlgLa composite beads were separated from the solution, washed with distilled water and dried in a hot-air oven at 80 °C for 10 h. Finally, the dried AlgCa and GOAlgLa composite beads were used for defluoridation studies.2.4. Adsorption experiments
Fluoride adsorption experiments were carried out in a batch mode in duplicate. About 0.1 g of GOAlgLa composite beads were added to an iodine flask containing 50 mL of a fluoride solution with an initial concentration of 20 mg L−1 at 303 K. The contents were shaken in a thermostat shaker with a constant speed of 200 rpm. To evaluate the effect of contact time, the contents of the iodine flask were taken out at 10 min intervals from 10–100 min and then the solution was filtered and the final fluoride concentration was measured. The impact of the solution pH on the fluoride adsorption process was carried out by adjusting the solution pH to 3, 5, 7, 9, and 11 with a 0.1 M HCl/NaOH solution. The initial fluoride concentrations varied from 18, 20, 22, and 24 mg L−1 at three various temperatures viz. 303, 313 and 323 K. The amount of fluoride removed by GOAlgLa composite beads was taken as the difference between the initial and equilibrium concentrations of fluoride in the solution, and the defluoridation capacity (mg F− kg−1) of GOAlgLa composite beads was obtained by the following equation.where Ci is the initial fluoride concentration (mg L−1), Ce is the equilibrium fluoride concentration (mg L−1), V is the volume of the solution (L), and m is the mass of the adsorbent (g).
2.5. Analytical methods
The fluoride concentration present in the solution was measured using a Thermo Orion Benchtop multiparameter kit (Model: VERSA STAR92) using a fluoride ion selective electrode having a relative accuracy of ±1 significant digit, detection limit of 0.02 mg L−1 and reproducibility of ±2%. The pH measurements were done with the same instrument (Model: VERSA STAR92) using a pH electrode (Orion 8157BNUMD). All other water quality parameters were investigated using standard methods.422.6. Instrumentation studies
The FTIR spectra of the sorbents were obtained using a JASCO-460 plus spectrometer operated at a 1 cm−1 resolution in the range of 400–4000 cm−1 using KBr pellets. TEM images were recorded using a TEM CM 200 (Philips) model. The surface morphology of the hybrid beads were imagined by a Vega3 Tescan model scanning electron microscope (SEM). The change in the surface morphology of the fresh and fluoride sorbed composite beads was obtained by the SEM analysis. The energy dispersive X-ray analyzer (EDAX) of the composite beads was determined using a Bruker Nano GMBH model. The pH at the zero point of charge (pHzpc) of the composite beads was measured using the pH drift method.432.7. Statistical tools
The computations of the obtained experimental results were carried out using the Microcal Origin (Version 8.0) software. The best model and goodness of the fit were determined using the regression correlation coefficient (r), standard deviation (sd), and chi-square analysis (χ2).3. Results and discussion
3.1. Effect of contact time on the DCs of the sorbents
The sorption performance of GO, AlgCa composite beads and GOAlgLa composite beads for fluoride removal in relation to contact time was carried out by varying the contact time from 10 to 100 min for a 50 mL of 20 mg L−1 fluoride solution with a sorbent dosage of 0.1 g at a neutral pH.The results are shown in Fig. 1. It was found that the equilibrium time for the sorption of fluoride onto AlgCa and GOAlgLa composite beads was 50 min, whereas the equilibrium time was 60 min for GO. The maximum DCs obtained at the equilibrium time for AlgCa, GO and GOAlgLa composite beads was found to be 618, 2438 and 6617 mg F− kg−1, respectively. Among the sorbents, GO and GOAlgLa composite beads possess higher DCs than AlgCa composite beads. Therefore, further studies were limited to GO and GOAlgLa composite beads.
3.2. Impact of solution pH on the fluoride sorption
The solution pH is one of the major factors that alter the net charge of the adsorbent, influencing the adsorption properties. Fig. 2 shows the impact of solution pH on fluoride removal by GO and GOAlgLa composite beads at various initial pH values between 3 and 11.The maximum DCs of GO and GOAlgLa composite beads was found to be 3085 and 7254 mg F− kg−1, respectively, at pH 3, after which the fluoride sorption efficiency of the sorbents decreased with increasing pH. In general, the adsorbent surface is negatively charged at pH > pHzpc, and positively charged at pH < pHzpc. The pHzpc value of GOAlgLa composite beads was found to be 3.83 (inserted in Fig. 2), below which the surface of the sorbent possess a positive charge, favoring fluoride sorption. When the pH increases, the surface of the sorbent becomes less positively charged and the interaction between the fluoride and sorbent becomes less, changing to a repulsive force at pH > pHzpc, resulting in a significant decrease in fluoride sorption. The change in the electrostatic force between the fluoride and the sorbent explains the impact of solution pH on fluoride removal.
3.3. Competitive sorption of fluoride on the sorbents
The co-existing competitive anions such as chloride, nitrate, sulphate, and bicarbonate ions that are usually present in drinking water might interfere with the fluoride adsorption process. Therefore, to analyze the effect of these anions on the DCs of GO and GOAlgLa composite beads, studies were carried out by taking 0.1 g of the sorbents, which were added to a 20 mg L−1 fluoride solution along with 200 mg L−1 of the competitive anions, and the results are presented in Fig. 3. The results reveal that the co-exiting anions, such as chloride, nitrate, and sulphate ions, slightly interfere with GO and GOAlgLa composite beads during fluoride removal, whereas bicarbonate shows a momentous effect on the DCs of GO and GOAlgLa hybrid beads.The decline in the DCs of GO and GOAlgLa composite beads in the presence of HCO3− ions could be a competition between the HCO3− and fluoride ion for the active sites on the adsorbent surface, which is decided by the concentration, charge and size of the anions. In addition to this, HCO3− ions will increase the solution pH, which diminishes the active sites for fluoride sorption. Among the sorbents, GOAlgLa composite beads possess an enhanced DC compared with GO. Hence, the isotherm and field studies were restricted to GOAlgLa composite beads.
3.4. Characterization of GOAlgLa composite beads
FTIR spectra of GO, GOAlgLa composite beads, and fluoride sorbed GOAlgLa composite beads are shown in Fig. 4a–c, respectively. The FTIR spectrum of GO displays a peak at 1723 cm−1, which is attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of the –COOH group, and the bands present at 1410 and 1625 cm−1 are due to the stretching vibration of C–O–H of phenolic groups and the (C
O stretching of the –COOH group, and the bands present at 1410 and 1625 cm−1 are due to the stretching vibration of C–O–H of phenolic groups and the (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) of aromatic rings, respectively.44
C) of aromatic rings, respectively.44
 | ||
| Fig. 4 FTIR spectra of (a) GO, (b) GOAlgLa composite beads and (c) fluoride sorbed GOAlgLa composite beads. | ||
In GOAlgLa composite beads, bands appeared at 1036, 1424, 1622, 2926, and 3425 cm−1 are accredited to the C–O–C stretching, –CH2 bending, –COO– asymmetric stretching, –CH2 stretching, and –OH stretching vibrations, respectively.20 The decrease in the intensity of the band at 3409 cm−1 in the fluoride sorbed GOAlgLa composite beads is due to the exchangeable hydroxyl ions present in the composite beads being replaced by fluoride ions from the aqueous solution.
Raman spectroscopy is a widely used and definitive technique in the examination of the structural and electronic properties of GO. Fig. 5a and b displays the Raman spectra of GO and GOAlgLa composite beads, and the spectra possess two major peaks, namely, the D and G bands. The strong bands at ∼1330 and 1603 cm−1 correspond to D and G bands, respectively. The D band is the edge-induced disordered state, related to the presence of sp3 defects, and the G band is associated with the in-plane vibration of the sp2 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) aromatic carbon structure.45 Both the FTIR and Raman analysis results show that GO is highly dispersed in the alginate polymeric matrix.
C) aromatic carbon structure.45 Both the FTIR and Raman analysis results show that GO is highly dispersed in the alginate polymeric matrix.
The surface morphology of GO, full image of a GOAlgLa composite bead, close view of GOAlgLa composite bead and fluoride sorbed GOAlgLa composite beads were studied by SEM and the results are presented in Fig. 6a–d. The micrograph images of GO show a loosening of GO nano-sheets and their porous structure due to the opening of the planner carbon network. In GOAlgLa composite beads, alginate possesses a wide surface and acted as a bed/support for GO. The SEM image of GOAlgLa composite beads indicates that distinct porous structures are present in the composite beads and are expected to have more adsorption sites for the removal of fluoride. After fluoride sorption, the porous structure and grooves decreased in GOAlgLa composite beads, indicating fluoride sorption.
 | ||
| Fig. 6 SEM images of (a) GO and (b) overall shape of GOAlgLa composite bead. Close-view SEM images of (c) GOAlgLa composite beads and (d) fluoride sorbed GOAlgLa composite beads. | ||
The fluoride sorption on GOAlgLa composite beads was also confirmed by EDAX analysis. GO contains C and H peaks (cf. Fig. 7a), GOAlgLa composite beads possesses C, H, and La peaks (cf. Fig. 7b) and the fluoride sorbed GOAlgLa composite beads possesses C, H, La, and F peaks, which confirms the adsorption of fluoride onto GOAlgLa composite beads (cf. Fig. 7c). The mapping image results are also given to confirm the fluoride sorption onto GOAlgLa composite beads. The corresponding colors represent the particular elements present in GOAlgLa composite beads. After fluoride sorption, the presence of the fluoride color with the other elements indicates that the fluoride sorption occurred on GOAlgLa composite beads.
 | ||
| Fig. 7 EDAX spectra of (a) GO, (b) GOAlgLa composite beads and (c) fluoride sorbed GOAlgLa composite beads. | ||
The surface morphology of GOAlgLa composite beads was also analyzed by TEM analysis and the results are illustrated in Fig. 8. It indicates that the composite beads exhibited transparent and slightly aggregated morphologies with crinkles. In addition, GO was unidirectionally dispersed in the alginate polymeric matrix.
3.5. Sorption isotherms
Sorption isotherm models were used to describe the solute–sorbent interaction and optimizing conditions for maximum sorption. To estimate the fluoride sorption efficiency of GOAlgLa composite beads, three significant isotherms viz. Freundlich,46 Langmuir,47 and Dubinin–Radushkevich (D–R)48 were used. The linear plot of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe vs. log
qe vs. log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce signifies the applicability of Freundlich isotherm. The obtained 1/n, n and kF values are presented in Table 1. The n values lie between 1 and 10 and 1/n values lies between 0 and 1, corresponding to favorable conditions for fluoride sorption. A linear plot of a Langmuir isotherm is acquired for GOAlgLa composite beads when Ce/qe is plotted against Ce, which gives b and Qo values from the intercept and slope, respectively. The calculated values of Qo and b are listed in Table 1. The RL values lie in the range between 0 and 1 and indicates a favorable sorption. The linear plot of ln
Ce signifies the applicability of Freundlich isotherm. The obtained 1/n, n and kF values are presented in Table 1. The n values lie between 1 and 10 and 1/n values lies between 0 and 1, corresponding to favorable conditions for fluoride sorption. A linear plot of a Langmuir isotherm is acquired for GOAlgLa composite beads when Ce/qe is plotted against Ce, which gives b and Qo values from the intercept and slope, respectively. The calculated values of Qo and b are listed in Table 1. The RL values lie in the range between 0 and 1 and indicates a favorable sorption. The linear plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe vs. ε2 indicates the applicability of D–R isotherm.
qe vs. ε2 indicates the applicability of D–R isotherm.
| Isotherms | Parameters | Temperature | ||
|---|---|---|---|---|
| 303 K | 313 K | 323 K | ||
| Freundlich | 1/n | 0.328 ± 0.020 | 0.379 ± 0.019 | 0.437 ± 0.008 |
| n | 3.072 ± 0.005 | 3.416 ± 0.006 | 3.498 ± 0.005 | |
| kF (mg g−1) (L mg−1)1/n | 4.897 ± 0.003 | 5.049 ± 0.010 | 5.169 ± 0.012 | |
| r | 0.991 ± 0.006 | 0.984 ± 0.015 | 0.987 ± 0.010 | |
| sd | 0.065 ± 0.007 | 0.071 ± 0.009 | 0.079 ± 0.004 | |
| χ2 | 1.6 × 10−3 ± 0.008 | 2.0 × 10−3 ± 0.011 | 2.1 × 10−3 ± 0.007 | |
| Langmuir | Qo (mg g−1) | 6.701 ± 0.002 | 6.869 ± 0.008 | 7.005 ± 0.005 |
| b (L g−1) | 1.864 ± 0.005 | 1.919 ± 0.008 | 2.175 ± 0.003 | |
| RL | 0.167 ± 0.003 | 0.176 ± 0.001 | 0.183 ± 0.008 | |
| r | 0.998 ± 0.001 | 0.997 ± 0.002 | 0.999 ± 0.001 | |
| sd | 0.009 ± 0.006 | 0.017 ± 0.009 | 0.011 ± 0.014 | |
| χ2 | 1.1 × 10−3 ± 0.002 | 1.7 × 10−4 ± 0.006 | 2.58 × 10−4 ± 0.005 | |
| Dubinin–Radushkevich (D–R) | Xm (mg g−1) | 5.649 ± 0.022 | 5.764 ± 0.015 | 6.194 ± 0.017 |
| E (kJ mol−1) | 8.698 ± 0.031 | 9.064 ± 0.120 | 9.341 ± 0.045 | |
| r | 0.994 ± 0.005 | 0.990 ± 0.002 | 0.993 ± 0.007 | |
| sd | 0.038 ± 0.004 | 0.029 ± 0.009 | 0.047 ± 0.006 | |
| χ2 | 1.4 × 10−3 ± 0.013 | 1.8 × 10−3 ± 0.007 | 1.9 × 10−3 ± 0.014 | |
The values of KDR, Xm, and E are shown in Table 1. The E value ranging from 1.0 to 8.0 kJ mol−1 indicates physical sorption, whereas E value ranging from 9.0 to 16.0 kJ mol−1 indicates chemical sorption. The obtained E values are 8.698, 9.064 and 9.341 kJ mol−1 for 303, 313 and 323 K, respectively, which indicate that the defluoridation mechanism of GOAlgLa composite beads is purely chemical in nature. The higher r values and lower sd values designate a suitable isotherm.
The best isotherm fit was identified using low chi-square (χ2) values. The calculated chi-square values are presented in Table 1. The best isotherm fit model follows the order: Langmuir > D–R > Freundlich for fluoride sorption onto GOAlgLa composite beads.
3.6. Thermodynamic studies
The thermodynamic parameters, namely, the standard free energy change (ΔG°), standard entropy change (ΔS°) and standard enthalpy (ΔH°), are commonly used to estimate the nature and feasibility of adsorption and are calculated by Khan and Singh method.49 The obtained results are shown in Table 2. The ΔG° values are negative and increases with increasing adsorption temperature, which signifies the feasibility and spontaneity of fluoride sorption on GOAlgLa composite beads. The positive values of ΔH° indicate the endothermic nature of fluoride sorption. The positive value of ΔS° reveals the increase in the number of species and randomness at the solid–liquid interface.| Thermodynamic parameters | Temperature | |
|---|---|---|
| ΔG° (kJ mol−1) | 303 K | −4.28 |
| 313 K | −3.84 | |
| 323 K | −3.16 | |
| ΔH° (kJ mol−1) | 16.67 | |
| ΔS° (J K−1 mol−1) | 57.00 | |
3.7. Mechanism of fluoride uptake on GOAlgLa composite beads
The graphical representation of fluoride removal by GOAlgLa hybrid beads is shown in Fig. 9. It shows that the fluoride removal by GOAlgLa composite beads is governed by both ion-exchange and electrostatic adsorption, and complexation mechanism. The surface of GO is positively charged with hydroxide as the negative counter anions. Therefore, the more electronegative fluoride ion easily replaces the hydroxyl anion by an ion-exchange mechanism. Second, the positively charged La3+ ions present in GOAlgLa composite beads attract the negatively charged fluoride ions by means of electrostatic adsorption and complexation mechanism.3.8. Desorption and reusability studies of GOAlgLa composite beads
In practical applications, the regeneration and recycling of the adsorbent are indispensible. After fluoride sorption studies, GOAlgLa composite beads were separated by simple filtration, washed with double distilled water, dried and reused for further adsorption–desorption cycles. During adsorption studies, 0.1 g of GOAlgLa composite beads were added to 50 mL of fluoride solution with an initial concentration of 20 mg L−1 for 50 min at a neutral pH. After that, the fluoride sorbed GOAlgLa composite beads (13.30 mg L−1) were separated by simple filtration, washed with double distilled water and left to dry. The dried GOAlgLa composite beads were then added to dilute NaOH solution (50 mL) with various concentrations ranging from 0.02 to 0.1 M. Desorption of fluoride from GOAlgLa composite beads was improved from 0.02 to 0.1 M. A maximum desorption efficiency of 92.6% (12.32 mg L−1) of the composite beads was achieved with 0.1 M NaOH solution as the regenerant. From Fig. 10, it can be clearly observed that the fluoride sorption efficiency of GOAlgLa composite beads was decreased and was found to be 92.6% (12.32 mg L−1), 84.4% (11.23 mg L−1), 70.8% (9.42 mg L−1), and 52.1% (6.93 mg L−1) in fresh to four cycles, respectively. In each cycle, the fluoride removal efficiency was decreased gradually and after four cycles, the fluoride removal reached a minimum. It suggests that GOAlgLa composite beads could be regenerated and effectively used for upto four cycles.3.9. Removal of fluoride from field water sample
The application of GOAlgLa composite beads at field conditions was tested by collecting a bore-well water sample from a nearby fluoride widespread area. About 50 mL of fluoride contaminated water sample was shaked with 0.1 g of GOAlgLa composite beads for 60 min at room temperature and the results are shown in Table 3. The observed concentration of fluoride in the field water sample was 3.28 mg L−1, which is higher than the value recommended by WHO i.e., >1.5 mg L−1. After the treatment, fluoride concentration was greatly reduced to below the tolerance limit, showing the efficiency of GOAlgLa composite beads (cf. Table 3). The results also reveal the selectivity of the GOAlgLa composite beads towards fluoride, as the field water sample also contains a high concentration of other anions other than fluoride. It is interesting to note that the level of other water quality parameters has also been reduced to some extent. Hence, the GOAlgLa composite beads can be efficiently employed for defluoridation of water.| Water quality parameters | Before treatment | After treatment |
|---|---|---|
| F− (mg L−1) | 3.28 | 0.72 |
| pH | 7.68 | 7.75 |
| Cl− (mg L−1) | 617 | 486 |
| Total hardness (mg L−1) | 376 | 351 |
| Total dissolved solids (mg L−1) | 709 | 658 |
3.10. Performance comparison of GOAlgLa composite beads with other adsorbents
A comparative review was carried out on GOAlgLa composite beads with other previous reported adsorbents. Table 4 shows that the fluoride adsorption capacity of GOAlgLa composite beads have appreciable adsorption capacities with the reported adsorbents.| S. No. | Name of the adsorbent | Adsorption capacity (mg g−1) | Reference |
|---|---|---|---|
| 1 | GOAlgLa composite beads | 6.62 | Present study |
| 2 | Bauxite clay | 5.16 | 50 |
| 3 | Zn/Al and Mg/Al intercalated sodium alginate | 5.10 | 51 |
| 4 | Al–Zr in cellulose matrix | 5.76 | 52 |
| 5 | Corn stover biochar (CSBC) | 6.42 | 53 |
| 6 | Magnetic corn stover biochar (MCSBC) | 3.61 | 53 |
| 7 | La(III) loaded hematite | 0.36 | 54 |
| 8 | La loaded zeolite | 0.36 | 54 |
| 9 | Fe–Al-impregnated granular ceramic | 3.56 | 55 |
| 10 | Calcium chloride modified natural zeolite | 1.77 | 56 |
| 11 | Magnesia-loaded fly ash cenospheres | 6.00 | 57 |
| 12 | La(III) incorporated carboxylated chitosan beads | 4.71 | 58 |
| 13 | Waste carbon slurry | 4.31 | 59 |
| 14 | La(III) cross-linked nano-hydroxyapatite alginate composite beads | 3.72 | 21 |
| 15 | Fe3O4 coated nano-hydroxyapatite gelatin composite | 5.01 | 10 |
| 16 | Chitosan supported lanthanum/zirconium mixed oxide composite | 4.97 | 60 |
| 17 | Activated carbon (Acacia farnesiana) | 2.62 | 61 |
| 18 | Siderite (modified) | 5.46 | 62 |
| 19 | Stilbite zeolite modified with Fe(III) | 2.31 | 63 |
| 20 | Cellulose@HAP nanocomposites | 2.76 | 64 |
| 21 | Hydrous bismuth oxides | 1.93 | 65 |
| 22 | HFO doped alginate beads | 8.90 | 66 |
| 23 | Zirconium–iron oxide | 9.80 | 67 |
| 24 | Zirconium phosphate | 4.27 | 68 |
| 25 | Aluminum (hydr)oxide coated pumice | 7.87 | 69 |
| 26 | Alginate impregnated alumina | 17.00 | 34 |
| 27 | KMnO4 modified carbon | 15.90 | 70 |
| 28 | Granular ceramic | 12.12 | 71 |
| 29 | Chemical treated laterite | 37.90 | 72 |
| 30 | Magnetic chitosan | 22.49 | 73 |
4. Conclusions
The hybrid biopolymeric composite beads were synthesized by incorporating GO into Alg polymeric matrix and followed by cross-linking with La(III) metal ions. The developed GOAlgLa composite beads showed a potential efficiency for the removal of fluoride from an aqueous solution. The DC of GOAlgLa composite beads decreased drastically when the solution pH was increased from 3 to 11. The presence of competitive anions such as chloride, nitrate, and sulphate ions do not considerably decrease the DC of GOAlgLa composite beads. However, bicarbonate ions retard the DC of GOAlgLa composite beads. The sorption isotherm process was better fit with the Langmuir isotherm than D–R and Freundlich models. GOAlgLa composite beads possess a higher DC than GO because they remove fluoride by ion-exchange, electrostatic adsorption and complexation. The thermodynamic parameter values indicate a spontaneous and endothermic nature for fluoride removal. The regeneration and reusability studies indicate that the performance of GOAlgLa composite beads for continuous applications. The results of the field application of GOAlgLa composite beads illustrates that the biopolymeric composite beads can be efficiently utilized for fluoride sorption and will make a platform to develop the defluoridation technology.Acknowledgements
The authors are thankful to the Department of Science and Technology-Science and Engineering Research Board (No. SR/FT/CS-43/2011 dt. 24-05-2012), New Delhi, India for the provision of financial support to carry out this research study. The first author (K. Pandi) would like to thank the Council of Scientific and Industrial Research (CSIR), New Delhi, India for awarding the Senior Research Fellowship.References
- WHO Report, Fluoride and Fluorides: Environmental Health Criteria, World Health Organization, Geneva, 1984 Search PubMed.
- M. Edmunds and P. Smedley, Fluoride in natural waters. Essentials of medical geology, impacts of natural environment on public health, Elsevier Academic Press, 2005 Search PubMed.
- B. D. Turner, P. Binning and S. L. S. Stipp, Environ. Sci. Technol., 2005, 39, 9561–9568 CrossRef CAS PubMed.
- S. Meenakshi and N. Viswanathan, J. Colloid Interface Sci., 2007, 308, 438–450 CrossRef CAS PubMed.
- S. Vasudevan, J. Lakshmi and G. Sozhan, Clean, 2009, 37, 372–378 CAS.
- Z. Amor, B. Bariou, N. Mameri, M. Taky, S. Nicolas and A. Elmidaoui, Desalination, 2001, 133, 215–223 CrossRef CAS.
- F. Elazhar, M. Tahaikt, A. Achatei, F. Elmidaoui, M. Taky, F. El Hannouni, I. Laaziz, S. Jariri, M. El Amrani and A. Elmidaoui, Desalination, 2009, 249, 154–157 CrossRef CAS.
- S. Chatterjee and S. De, Sep. Purif. Technol., 2014, 125, 223–238 CrossRef CAS.
- S. Muthu Prabhu and S. Meenakshi, Chem. Eng. J., 2015, 262, 224–234 CrossRef CAS.
- K. Pandi and N. Viswanathan, J. Chem. Eng. Data, 2016, 61, 571–578 CrossRef CAS.
- B. Pan, J. Xu, B. Wu, Z. Li and X. Liu, Environ. Sci. Technol., 2013, 47, 9347–9354 CrossRef CAS PubMed.
- C. Jing, J. Cui, Y. Huang and A. Li, ACS Appl. Mater. Interfaces, 2012, 4, 714–720 CAS.
- Y. Li, P. Zhang, Q. Du, X. Peng, T. Liu, Z. Wang, Y. Xia, W. Zhang, K. Wang, H. Zhu and D. Wu, J. Colloid Interface Sci., 2011, 363, 348–354 CrossRef CAS PubMed.
- C. J. Madadrang, H. Y. Kim, G. Gao, N. Wang, J. Zhu, H. Feng, M. Gorring, M. L. Kasner and S. Hou, ACS Appl. Mater. Interfaces, 2012, 4, 1186–1193 CAS.
- J. Wang and B. Chen, Chem. Eng. J., 2015, 281, 379–388 CrossRef CAS.
- Y. Li, Q. Du, J. Wang, T. Liu, J. Sun, Y. Wang, Z. Wang, Y. Xia and L. Xia, J. Fluorine Chem., 2013, 148, 67–73 CrossRef CAS.
- F. Zhang, B. Wang, S. He and R. Man, J. Chem. Eng. Data, 2014, 59, 1719–1726 CrossRef CAS.
- C. Jiao, J. Xiong, J. Tao, S. Xu, D. Zhang, H. Lin and Y. Chen, Int. J. Biol. Macromol., 2016, 83, 133–141 CrossRef CAS PubMed.
- G. Sheng, Y. Li, X. Yang, X. Ren, S. Yang, J. Hu and X. Wang, RSC Adv., 2012, 2, 12400–12407 RSC.
- W. M. Algothmi, N. M. Bandaru, Y. Yu, J. G. Shapter and A. V. Ellis, J. Colloid Interface Sci., 2013, 397, 32–38 CrossRef CAS PubMed.
- K. Pandi and N. Viswanathan, J. Appl. Polym. Sci., 2015, 132, 41937–41945 CrossRef.
- H. Zhu, Y. Fu, R. Jiang, J. Yao, L. Xiao and G. Zeng, Ind. Eng. Chem. Res., 2014, 53, 4059–4066 CrossRef CAS.
- N. Viswanathan and S. Meenakshi, Colloids Surf., B, 2009, 72, 88–93 CrossRef CAS PubMed.
- S. Muthu Prabhu and S. Meenakshi, Carbohydr. Polym., 2015, 120, 60–68 CrossRef PubMed.
- R. Jayakumar, M. Rajkumar, H. Freitas, N. Selvamurugan, S. V. Nair, T. Furuike and H. Tamura, Int. J. Biol. Macromol., 2009, 44, 107–111 CrossRef CAS PubMed.
- M. G. Sujana, A. Mishra and B. C. Acharya, Appl. Surf. Sci., 2013, 270, 767–776 CrossRef CAS.
- K. Pandi and N. Viswanathan, Carbohydr. Polym., 2014, 112, 662–667 CrossRef CAS PubMed.
- M. Jain, V. K. Garg, K. Kadirvelu and M. Sillanpaa, Ind. Eng. Chem. Res., 2015, 54, 1419–1425 CrossRef CAS.
- S. Cataldo, A. Gianguzza, A. Pettignano and I. Villaescusa, React. Funct. Polym., 2013, 73, 207–217 CrossRef CAS.
- S. Cataldo, A. Gianguzza and A. Pettignano, Arabian J. Chem. DOI:10.1016/j.arabjc.2014.10.031.
- N. Jiang, Y. Xu, Y. Dai, W. Luo and L. Dai, J. Hazard. Mater., 2012, 215–216, 17–24 CrossRef CAS PubMed.
- S. A. Wasay, M. J. Haron and S. Tokunaga, Water Environ. Res., 1996, 68, 295–300 CrossRef CAS.
- H. Paudyal, B. Pangeni, K. Inoue, H. Kawakita, K. Ohto, K. N. Ghimire and S. Alam, Bioresour. Technol., 2013, 148, 221–227 CrossRef CAS PubMed.
- H. Basu, R. K. Singhal, M. V. Pimple and A. V. R. Reddy, Water, Air, Soil Pollut., 2013, 224, 1572–1583 CrossRef.
- J. Ma, Y. Shen, C. Shen, Y. Wen and W. Liu, Chem. Eng. J., 2014, 248, 98–106 CrossRef CAS.
- N. Thakur, S. A. Kumar, H. Parab, A. K. Pandey, P. Bhatt, S. D. Kumar and A. V. R. Reddy, RSC Adv., 2014, 4, 10350–10357 RSC.
- K. Pandi and N. Viswanathan, Carbohydr. Polym., 2015, 118, 242–249 CrossRef CAS PubMed.
- E. Vences-Alvarez, L. H. Velazquez-Jimenez, L. F. Chazaro-Ruiz, P. E. Diaz-Flores and J. R. Rangel-Mendez, J. Colloid Interface Sci., 2015, 455, 194–202 CrossRef CAS PubMed.
- S. Dong and Y. Wang, Water Res., 2016, 88, 852–860 CrossRef CAS PubMed.
- S. Dong and Y. Wang, Water Res., 2016, 88, 852–860 CrossRef CAS PubMed.
- N. I. Kovtyukhova, P. J. Ollivier, B. R. Martin, T. E. Mallouk, S. A. Chizhik, E. V. Buzaneva and A. D. Gorchinskiy, Chem. Mater., 1999, 11, 771–778 CrossRef CAS.
- APHA, Standard methods for the examination of water and waste water, American Public Health Association, Washington, DC, USA, 2005 Search PubMed.
- M. V. Lopez-Ramon, F. Stoeckli, C. Moreno-Castilla and F. Carrasco-Marin, Carbon, 1999, 37, 1215–1221 CrossRef CAS.
- P. Sharma, N. Hussain, D. J. Borah and M. R. Das, J. Chem. Eng. Data, 2013, 58, 3477–3488 CrossRef CAS.
- G. Bharath, R. Madhu, S. Chen, V. Veeramani, D. Mangalaraj and N. Ponpandian, J. Mater. Chem. A, 2015, 3, 15529–15539 CAS.
- H. M. F. Freundlich, Z. Phys. Chem., 1906, 57A, 385–470 Search PubMed.
- I. Langmuir, J. Am. Chem. Soc., 1916, 38, 2221–2295 CrossRef CAS.
- S. Karahan, M. Yurdakoc, Y. Seki and K. Yurdakoc, J. Colloid Interface Sci., 2006, 293, 36–42 CrossRef CAS PubMed.
- A. A. Khan and R. P. Singh, Colloids Surf., 1987, 24, 33–42 CrossRef CAS.
- M. G. Sujana and S. Anand, Desalination, 2011, 267, 222–227 CrossRef CAS.
- S. Mandal, V. S. Patil and S. Mayadevi, Microporous Mesoporous Mater., 2011, 158, 241–246 CrossRef.
- M. Barathi, A. Santhana Krishna Kumar and N. Rajesh, J. Environ. Chem. Eng., 2013, 1, 1325–1335 CrossRef CAS.
- D. Mohan, S. Kumar and A. Srivastava, Ecol. Eng., 2014, 73, 798–808 CrossRef.
- A. Teutli-Sequeira, V. Martinez-Miranda, M. Solache-Rios and I. Linares-Hernandez, J. Fluorine Chem., 2013, 148, 6–13 CrossRef CAS.
- N. Chen, C. Feng and M. Li, Clean Technol. Environ. Policy, 2014, 16, 609–617 CrossRef CAS.
- Z. Zhang, Y. Tan and M. Zhong, Desalination, 2011, 276, 246–252 CrossRef CAS.
- X. Xu, Q. Li, H. Cui, J. Pang, L. Sun, H. An and J. Zhai, Desalination, 2011, 272, 233–239 CrossRef CAS.
- N. Viswanathan and S. Meenakshi, J. Colloid Interface Sci., 2008, 322, 375–383 CrossRef CAS PubMed.
- V. K. Gupta, I. Ali and V. K. Saini, Water Res., 2007, 41, 3307–3316 CrossRef CAS PubMed.
- S. Muthu Prabhu and S. Meenakshi, Journal of Water Process Engineering, 2014, 2, 96–104 CrossRef.
- Y. Hanumantharao, M. Kishore and K. Ravindhranath, Int. J. Plant, Anim. Environ. Sci., 2011, 1, 209–223 CAS.
- Y. Shan and H. Guo, Chem. Eng. J., 2015, 223, 183–191 CrossRef.
- Y. Sun, Q. Fang, J. Dong, X. Cheng and J. Xu, Desalination, 2011, 277, 121–127 CrossRef CAS.
- X. Yu, S. Tong, M. Ge and J. Zuo, Carbohydr. Polym., 2013, 92, 269–275 CrossRef CAS PubMed.
- L. Chai, Y. Wang, N. Zhao, W. Yang and X. You, Water Res., 2013, 47, 4040–4049 CrossRef CAS PubMed.
- M. G. Sujana, A. Mishra and B. C. Acharya, Appl. Surf. Sci., 2013, 270, 767–776 CrossRef CAS.
- X. Dou, Y. Zhang, H. Wang, T. Wang and Y. Wang, Water Res., 2011, 45, 3571–3578 CrossRef CAS PubMed.
- S. K. Swain, T. Patnaik, V. K. Singha, U. Jha, R. K. Patel and R. K. Dey, Chem. Eng. J., 2011, 171, 1218–1226 CrossRef CAS.
- A. Salifu, B. Petrusevski, K. Ghebremichael, L. Modestus, R. Buamah, C. Aubry and G. L. Amy, Chem. Eng. J., 2013, 228, 63–74 CrossRef CAS.
- A. A. M. Daifullah, S. M. Yakout and S. A. Elreefy, J. Hazard. Mater., 2007, 147, 633–643 CrossRef CAS PubMed.
- N. Chen, Z. Zhang, C. Feng, N. Sugiura, M. Li and R. Chen, J. Colloid Interface Sci., 2010, 348, 579–584 CrossRef CAS PubMed.
- A. Maiti, J. K. Basu and S. De, Desalination, 2011, 265, 28–36 CrossRef CAS.
- W. Ma, F.Q. Ya, M. Han and R. Wang, J. Hazard. Mater., 2007, 143, 296–302 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |