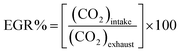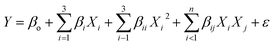A comparative evaluation and optimization of performance and emission characteristics of a DI diesel engine fueled with n-propanol/diesel, n-butanol/diesel and n-pentanol/diesel blends using response surface methodology
B. Rajesh Kumar *a,
T. Muthukkumar*bc,
V. Krishnamoorthyc and
S. Saravanand
*a,
T. Muthukkumar*bc,
V. Krishnamoorthyc and
S. Saravanand
aDepartment of Mechanical Engineering, Jeppiaar Institute of Technology, Chennai, India. E-mail: rajesh_thermal@yahoo.com
bCentre for Research, Sathyabama University, Chennai, India
cDepartment of Mechanical Engineering, Jeppiaar Maamallan Engineering College, Chennai, India. E-mail: tmuthu_me@rediffmail.com
dEngine Research Lab, Department of Mechanical Engineering, Sri Venkateswara College of Engineering, Chennai, India
First published on 23rd June 2016
Abstract
High carbon bio-alcohols have recently grabbed the attention of diesel engine researchers because of higher energy density, higher cetane number and better blend stability than their low carbon counterparts. This study utilizes three high carbon bio-alcohol/diesel blends prepared by mixing 40% by vol. of n-propanol, n-butanol and n-pentanol individually with fossil diesel in a DI diesel engine. Engine performance and emission characteristics were measured under high-load conditions based on a 33 full-factorial experimental design matrix using exhaust gas recirculation (EGR) rate, injection-timing and alcohol type used in the blends as factors for controlling charge-dilution and combustion-phasing. A statistical investigation was then carried out to compare and analyze the effects of these factors on all measured responses like nitrogen oxides (NOx), smoke, hydrocarbons (HC), carbon monoxide (CO), brake thermal efficiency (BTE) and brake specific fuel consumption (BSFC). Multiple regression models were developed for all responses using a response surface methodology (RSM) and were found to be statistically significant at 99% confidence levels. Interactive effects between injection timing and EGR for all blends were compared and analyzed through response surface plots fitted using developed models with high R2 values. Optimization was performed using a desirability approach with an objective to minimize NOx, smoke and BSFC with maximum BTE. n-Propanol/diesel blend injected at 25° CA bTDC under 30% EGR with a desirability of 0.965 was predicted to be optimum for this engine. Similarly n-butanol/diesel and n-pentanol/diesel blends injected at 24° CA bTDC under 10% EGR were found to be optimum in their respective category. Confirmatory tests validated that the developed RSM models were adequate to describe the effects of injection timing and EGR on the engine characteristics as the predicted error is within 5%.
1. Introduction
Diesel engines are attractive energy conversion devices due to their high efficiency, durability and cost effectiveness.1 Though fossil diesel is priced higher than gasoline in many countries, its high energy density implies more energy can be extracted from it than from the same volume of gasoline.2 Despite their ubiquitous role in transportation, heavy duty machinery and power generation, diesel engines are branded ‘dirty’ and face severe opposition from the global community over pollution concerns.3 Alarming pollution levels in mega cities like New Delhi, Paris, Amsterdam and London have forced local governments to impose unprecedented restrictions on diesel engines.4 New Delhi has banned new registration of diesel vehicles above 2000cc and is currently restricting vehicles based on their license plates (odd or even) on road in alternative days.5 Paris has already decided to ban diesel cars by 2020 6 and London expected to follow the suit.7 Amsterdam is already imposing high taxes on diesel vehicles.4With increasing global concerns on diesel engine exhaust, engine researchers are striving to improve this energy-efficient workhorse by (i) modifying engine design, (ii) reformulating fossil diesel with biofuels, (iii) employing new combustion strategies and, (iv) using after-treatment devices like diesel oxidation catalysts, diesel particulate filters and selective catalytic reduction. Diesel reformulation is a simple and straightforward approach which requires minimal modifications in the engine. Biofuels derived from sustainable sources could be viable substitutes for fossil diesel that also can reduce lethal emissions by increasing oxygen availability during combustion with their fuel-bound oxygen.1 Besides the stringent emission norms, rapid diminution of fossil fuel resources and fluctuating crude oil prices also necessitates engine researchers to explore alternative biofuels in diesel engines. Bio-alcohols used in this study can be derived from non-food based sources by microbial fermentation and cellulose de-polymerization.8 Several sustainable pathways have emerged to bio-synthesize high carbon alcohols using engineered micro-organisms like Escherichia coli and Clostridium species.9
Low carbon alcohols like methanol and ethanol were extensively researched in spark-ignition engines due to their better anti-knock characteristics and low CO & UHC emissions.10 However they are incompatible with diesel engine technology due to their low cetane number, less calorific value and poor blend stability11 when compared to fossil diesel. Recently the use of higher carbon alcohols in diesel engines is gaining attention due to their higher energy density, higher cetane number, lower vapor pressure, lower volatility and higher hygroscopicity when compared to low carbon alcohols.12 The properties of some high carbon alcohols in comparison with ethanol and methanol are shown in Table 1. This study utilizes three higher carbon alcohols namely n-propanol, n-butanol and n-pentanol as blend components with fossil diesel. Fig. 1 shows the molecular structure of these alcohols. The oxygen atoms (shown in red) bonded in the functional group of alcohols reduces soot formation by inhibiting soot precursors and increases the availability of oxygen even in fuel-rich zones.13 On the other hand, EGR and retarded injection timing have been the most popular methods already in use to reduce obnoxious NOx emissions.14
| Properties | Diesel | Methanol | Ethanol | Propanol | Butanol | Pentanol |
|---|---|---|---|---|---|---|
| Molecular formula | CxHy | CH3–OH | C2H5–OH | C3H7–OH | C4H9–OH | C5H11–OH |
| Molecular weight (kg kmol−1) | 190–211.7 | 32.04 | 46.07 | 60.09 | 74.12 | 88.15 |
| C (wt%) | 86.13 | 37.48 | 52.14 | 59.96 | 64.82 | 68.13 |
| H (wt%) | 13.87 | 12.48 | 13.02 | 13.31 | 13.49 | 13.61 |
| O (wt%) | 0 | 49.93 | 34.73 | 26.62 | 21.59 | 18.15 |
| Lubricity (μm corrected wear scar) | 315 | 1100 | 1057 | 922 | 591 | 670.5 |
| Cetane number | 52 | 5 | 8 | 12 | 17 | 20 |
| Self-ignition temperature (°C) | 254–300 | 463 | 420 | 350 | 345 | 300 |
| Density (kg m−3) at 15 °C | 835 | 791.3 | 789.4 | 803.7 | 809.7 | 814.8 |
| Viscosity at 40 °C (mm s−2) | 2.72 | 0.58 | 1.13 | 1.74 | 2.22 | 2.89 |
| Lower heating value (MJ kg−1) | 42.49 | 19.58 | 26.83 | 30.63 | 33.09 | 34.65 |
| Latent heat of evaporation (kJ kg−1) | 270–375 | 1162.64 | 918.42 | 727.88 | 684 | 647.1 |
| Vapor pressure (mmHg) | 0.4 | 127 | 55 | 20 | 7 | 6 |
| CFPP (°C) | −17 | <−51 | <−51 | <−51 | <−51 | −40 |
| Boiling point (°C) | 180–360 | 64.7 | 78.3 | 97.1 | 117.5 | 137.9 |
| Flash point (°C) | >55 | 11–12 | 17 | 11.7 | 35–37 | 49 |
The use of n-butanol as blend component with diesel has been extensively researched in diesel engines.15–20 However there are relatively few studies involving n-propanol/diesel21–23 and n-pentanol/diesel blends11,12,24,25 in diesel engines. A general collective conclusion (with reference to fossil diesel) from these studies can be summarized as follows: ignition delay increased with increasing alcohol content in the blends due to their low cetane number. There is an increase in peak combustion pressure and heat release rates with alcohol addition due to an enhanced premixed combustion phase. Smoke consistently decreased with increasing alcohol content in the blends. The presence of fuel-bound oxygen in the blends especially in locally rich zones improved the combustion process resulting in less smoke emissions.16 HC emissions increased with increasing alcohol content in the blends. The slower vaporization and poorer air–fuel mixing due to the higher latent heat of evaporation of alcohol blends is the major reason for HC emissions. BTE increased with increasing n-propanol or n-butanol content in the blends due to enhanced oxygen content while BSFC increased for all alcohols due to low heating value. BTE decreased with n-pentanol/diesel blends due to less combustion efficiency as a result of lower oxygen content when compared to n-butanol and n-propanol. NOx emissions generally decreased with increasing n-propanol or n-butanol content in the blends due to the engine running overall leaner and the temperature lowering effect of n-propanol or n-butanol (due to its lower calorific value and higher heat of evaporation).
Simultaneous reduction of NOx and smoke emissions is a challenge diesel engine researchers face due to the inherent trade-off relationship between them.26 Methods like EGR, retarded injection timing and using low energy alcohols as blend components affect engine performance.27 Hence there is a necessity to obtain an optimal combination of injection timing, EGR and the fuel type to achieve a desired emission and performance targets. The current study sets out to achieve an objective of minimal NOx, smoke, BSFC and maximum BTE in a light-duty diesel engine fueled with three alcohol/diesel blends by optimization using a full factorial experimental design matrix and response surface methodology (RSM). RSM is a widely used technique for optimizing engine parameters like load, speed, compression ratio, injection pressure, injection timing to obtain the desired combustion and performance characteristics.10,28–34
The present investigation also attempts to fill a gap in the existing body of literature of high carbon alcohols by presenting a vis-a-vis study that compares and analyzes three higher alcohol/diesel blends (n-propanol, n-butanol and n-pentanol) in a same light-duty, direct injection diesel engine under similar operating conditions using response surface methodology. To the authors' knowledge, this is the first time that such a comparative evaluation is reported for many high carbon bio-alcohol/diesel blends in the same engine and operating conditions concerning combustion and emissions characteristics of the engine.
The objectives of this study can be summarized as follows: (i) to compare and analyze the effects of EGR and injection timing on performance and emission characteristics of a DI diesel engine when fueled with n-propanol/diesel, n-butanol/diesel and n-pentanol/diesel blends (ii) to predict NOx, smoke, HC, CO, BTE and BSFC for all blends using models developed by RSM for the experimental design. (iii) To determine an optimum combination of alcohol type used in the blends, injection timing and EGR to minimize NOx, smoke, BSFC with maximum BTE in a single cylinder DI diesel engine using RSM based desirability approach.
2. Materials and methods
2.1 Test fuels
Three alcohols viz., n-propanol (CAS no. 71-23-8), n-butanol (CAS no. 71-36-3) and n-pentanol (CAS no. 71-41-0) that were certified to a purity of 99%, 99% and 98% (analytical grade) respectively were procured from a local supplier. Ultra low sulfur diesel (ULSD) used in this study was procured from Shell petroleum, Chennai. Three test fuels were prepared by mixing these three alcohols individually in ULSD with a blending ratio of 40/60 by vol. No cetane improvers/surfactants were added. Solubility of all blends was checked and no phase separation was observed after several weeks. The choice of 40% higher alcohol/diesel blend was made because the variations of combustion and emission parameters would be highly discernible for higher volumes of alcohols in the blends. Such an approach was also used in previous researches involving high carbon alcohols.12,35 Further, this would also enable a higher fraction of renewable biofuels to replace fossil diesel. The properties of the test fuels were measured using ASTM test methods in the Polymeric Materials Research Laboratory at Alagappa College, Tamil Nadu and are presented in Table 2. Since the facility to determine cetane number of the test fuels using ASTM D613 method was not available with this laboratory, the cetane indices of the test fuels were estimated by using the four variable equation as specified by ASTM4737 using the test fuel's density (estimated by ASTM D4052) and T10, T50 and T90 distillation points (estimated by ASTM D86).| Properties | Test method | ULSD | PRO40 | BUT40 | PEN40 |
|---|---|---|---|---|---|
| Oxygen (wt%) | — | 0 | 10.648 | 8.636 | 7.26 |
| Calculated cetane index | ASTM D4737 | 48 | 33.6 | 35.6 | 36.8 |
| Low heating value (MJ kg−1) | ASTM D240 | 41.82 | 34.972 | 35.852 | 36.492 |
| Kinematic viscosity at 40 °C (mm2 s−1) | ASTM D445 | 3.8 | 2.964 | 3.168 | 3.436 |
| Density (kg m−3) | ASTM D4052 | 841 | 826.6 | 828.6 | 831 |
| Flash point (°C) | ASTM D93 | 65 | 12 | 35 | 48 |
2.2 Test engine and facilities
Tests were performed in a naturally-aspirated, constant-speed (1500 rpm), single-cylinder, four-stroke, direct-injection diesel engine which was used in the author's previous works involving iso-butanol/diesel,36 n-pentanol/diesel11 and n-octanol/diesel blends.9 This engine is widely used in the Indian agricultural sector. About 14.42 million of these engines were used to drive pump-sets for irrigation purposes across the country.37 The annual production of these diesel-driven pump-sets is 1.5 million which is expected to grow by 7% every year. In 2012–13, India's agricultural sector has recorded a diesel consumption of 6 MMT (Million Metric Tons) which is about 8.55% of India's total diesel consumption (69 MMT).38 Thus a large population of farmers in India are severely exposed to toxic diesel exhaust arising from these stationary equipment. Hence this engine was chosen for this study. Specifications of this engine are given in Table 3.| Make and model | Kirloskar, TAF1 make |
|---|---|
| Number of cylinders | 1 |
| Combustion chamber | Hemispherical open type |
| Piston | Bowl-in type |
| Bore, mm | 87.5 |
| Stroke, mm | 110 |
| Connecting rod length, mm | 220 |
| Swept volume, cm3 | 661 |
| Clearance volume, cm3 | 36.87 |
| Compression ratio | 17.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| Rated power, kW | 4.4 |
| Rated speed, rpm | 1500 |
| Injection type | Direct injection |
| Number of nozzle holes | 3 |
| Spray-hole diameter, mm | 0.25 |
| Injection pressure, bar | 210 |
| Cone angle, deg | 110 |
| Needle lift, mm | 0.25 |
| Fuel injection timing, ° CA bTDC | 23 |
| Fuel injection duration, ° CA | 20–30 |
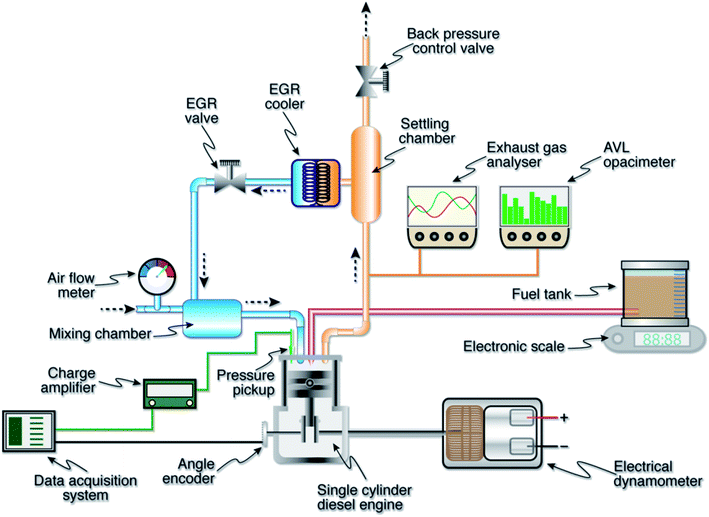
The schematic layout of the engine test bed is shown in Fig. 2. The detailed description of the engine and its related instrumentation can be found in the author's previous works.9,11,12,36 Cooled EGR is used in this study which maintains the exhaust gas at 35 °C. EGR quantity was determined using the relation,
The quantity of CO2 in the exhaust was measured using the gas analyzer by adjusting the control valve to vary the flow rate of the exhaust until the quantity of CO2 in the intake reaches the desired value. Similar method was used earlier to determine the EGR rates in this engine.11,12
Errors and uncertainties in the experiments can arise from instrument selection, condition, calibration, environment, observation, reading and test planning.39 Therefore, it is necessary to establish the accuracy of the experiments by carrying out an error analysis. The magnitudes of error in various measured parameters namely speed, pressure, crank angle and exhaust emissions were estimated from the minimum values of output and accuracy of the instruments using the method proposed by Moffat.40 Table 4 shows the range, accuracy and uncertainties of the instruments used in this study. The maximum possible error in the calculation of BTE and BSFC was determined to be 0.32%.
| Instrument | Measured quantity | Range | Accuracy | Uncertainties, % |
|---|---|---|---|---|
| Gas analyzer | NOx | 0–5000 ppm | ±10 ppm | 0.20 |
| HC | 0–20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm 000 ppm |
±20 ppm | 0.20 | |
| CO | 0–20% | ±0.02% | 0.15 | |
| Smoke meter | Smoke | 0–10 FSN | ±0.01 | 0.10 |
| Speed measuring unit | Engine speed | 0–9999 rpm | ±10 rpm | 0.15 |
| Pressure transducer | Cylinder pressure | 0–250 bar | ±0.1 bar | 0.10 |
| Crank angle encoder | Crank angle | 0–360° | ±1° | 0.20 |
2.3 Test procedure
All the tests were conducted under steady state conditions. The temperature of the lubricating oil was maintained between 85 and 90 °C. Experiments were carried out in the same day at constant ambient temperature to improve the reliability of the recordings. The engine was warmed up for 10 min to allow for stabilization before the readings were recorded. Each test was repeated three times and the results were averaged. This was done to ensure that the recordings are repeatable within the experimental uncertainties. The manufacturer recommended injection timing for this engine is 23° CA bTDC. Start of fuel injection was determined at static conditions using spill method which was also used in the author's earlier works.11,12 Fuel injection pump outlet was disconnected and connected to an annular tube specially designed for this purpose. By slowly rotating the flywheel, fuel was made to spill out from the tube. The flywheel was provided with divisions on its circumference. The distance of the spill out point from the TDC position on flywheel was converted into degrees as start of fuel injection angle. The static injection timing was changed by varying the number of shims under the mounting flange of the fuel injection pump. To advance the fuel injection timing the shims under the pump were removed and to retard the injection timing, shims were added under the fuel injection pump. EGR rates were varied from 10 to 30% by opening the EGR valve gradually. The engine was tested at a constant speed of 1500 rpm at high engine load that corresponds to the brake mean effective pressure (bmep) of 4.0 bar based on the full factorial design with the combination of different levels of influencing factors consisting of 27 trials. Before refueling, the engine was allowed to run for some time in-order for it to consume the fuel that remained in the fuel system during the previous trial.2.4 Experimental design
| Factors | Factor type | Code | Levels | ||
|---|---|---|---|---|---|
| 1 | 2 | 3 | |||
| Injection timing (° CA) | Numeric | A | 21 | 23 | 25 |
| EGR (%) | Numeric | B | 10 | 20 | 30 |
| Alcohol type | Categoric | C | n-Propanol | n-Butanol | n-Pentanol |
In-cylinder temperature and oxygen availability during combustion are the two important factors affecting formation of NOx and smoke in the engine. Variation of fuel injection timing exerts considerable effect on spray formation, atomization and combustion while EGR decreases oxygen availability and reduces flame temperature.27 In this study, three injection timings (21, 23 and 25° CA) were chosen. EGR rates upto 30% were chosen because smoke emissions increased significantly beyond 30% with n-pentanol/diesel and iso-butanol/diesel blends.11,36 The response variables measured were NOx, smoke, HC, CO, BTE and BSFC. Table 6 shows the 27 trials that were performed in the engine and the response values recorded.
| Run | A: Injection timing, ° CA | B: EGR, % | C: Alcohol type | NOx, ppm | Smoke, FSN | HC, ppm | CO, % vol | BTE, % | BSFC, kg kW−1 h−1 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 21 | 10 | n-Propanol | 851 | 0.25 | 53 | 0.02 | 26.2889 | 0.3629 |
| 2 | 21 | 20 | n-Propanol | 732 | 0.41 | 55 | 0.03 | 25.2373 | 0.3780 |
| 3 | 21 | 30 | n-Propanol | 625 | 0.63 | 58 | 0.03 | 24.1858 | 0.3944 |
| 4 | 23 | 10 | n-Propanol | 1069 | 0.18 | 50 | 0.03 | 30.4951 | 0.3128 |
| 5 | 23 | 20 | n-Propanol | 921 | 0.37 | 52 | 0.03 | 29.4435 | 0.3240 |
| 6 | 23 | 30 | n-Propanol | 783 | 0.43 | 55 | 0.03 | 28.3920 | 0.3360 |
| 7 | 25 | 10 | n-Propanol | 1668 | 0.11 | 46 | 0.03 | 34.7013 | 0.2749 |
| 8 | 25 | 20 | n-Propanol | 1454 | 0.21 | 47 | 0.03 | 33.6497 | 0.2835 |
| 9 | 25 | 30 | n-Propanol | 1271 | 0.27 | 49 | 0.03 | 32.5982 | 0.2926 |
| 10 | 21 | 10 | n-Butanol | 829 | 0.29 | 46 | 0.02 | 24.4780 | 0.3792 |
| 11 | 21 | 20 | n-Butanol | 730 | 0.65 | 50 | 0.02 | 23.4581 | 0.3957 |
| 12 | 21 | 30 | n-Butanol | 593 | 0.90 | 53 | 0.03 | 22.4382 | 0.4137 |
| 13 | 23 | 10 | n-Butanol | 1073 | 0.25 | 44 | 0.02 | 26.5179 | 0.3501 |
| 14 | 23 | 20 | n-Butanol | 937 | 0.50 | 47 | 0.02 | 25.4980 | 0.3641 |
| 15 | 23 | 30 | n-Butanol | 803 | 0.66 | 50 | 0.03 | 24.4780 | 0.3792 |
| 16 | 25 | 10 | n-Butanol | 1566 | 0.13 | 40 | 0.02 | 31.6175 | 0.2936 |
| 17 | 25 | 20 | n-Butanol | 1409 | 0.23 | 43 | 0.02 | 30.5975 | 0.3034 |
| 18 | 25 | 30 | n-Butanol | 1239 | 0.33 | 45 | 0.03 | 29.5776 | 0.3139 |
| 19 | 21 | 10 | n-Pentanol | 799 | 0.44 | 26 | 0.02 | 23.0551 | 0.3961 |
| 20 | 21 | 20 | n-Pentanol | 694 | 0.72 | 30 | 0.02 | 22.0528 | 0.4141 |
| 21 | 21 | 30 | n-Pentanol | 591 | 0.97 | 38 | 0.03 | 21.0504 | 0.4338 |
| 22 | 23 | 10 | n-Pentanol | 947 | 0.40 | 23 | 0.02 | 26.0623 | 0.3504 |
| 23 | 23 | 20 | n-Pentanol | 891 | 0.52 | 32 | 0.02 | 25.0599 | 0.3644 |
| 24 | 23 | 30 | n-Pentanol | 771 | 0.90 | 43 | 0.03 | 24.0575 | 0.3796 |
| 25 | 25 | 10 | n-Pentanol | 1473 | 0.20 | 20 | 0.02 | 29.0695 | 0.3142 |
| 26 | 25 | 20 | n-Pentanol | 1362 | 0.27 | 25 | 0.02 | 28.0671 | 0.3254 |
| 27 | 25 | 30 | n-Pentanol | 1189 | 0.36 | 28 | 0.03 | 27.0647 | 0.3374 |
where Y is the response, Xi are numeric values of the factors, terms β0, βi, βii and βij are intercept, linear, quadratic and interaction regression coefficients respectively, and ε is the experimental error.32 The developed equations, if statistically significant, present a correlation between factors and measured responses and can be used to predict the responses. Response surface plots were constructed using these fitted models. The optimal combination of fuel blends, EGR rates and injection timing can be finally obtained by using the desirability approach of RSM.
3. Results and discussion
The raw data for the responses obtained from the 27 trials conducted (as shown in Table 6) was analyzed and discussed in the upcoming sections.3.1 Model analysis and evaluation
Table 7 presents the regression models developed for fitting the experimental data of all responses. These models were validated using analysis of variance (ANOVA) and the results of which were presented in Tables 8–10. The statistical significance of all three considered factors and their possible two-way and three-way interactions on all responses were evaluated from their F-statistic and p-values. The statistical significance of a factor is greater if its F-value is higher. The values of p less than <0.0001 indicate factors and their interactions significant at 99% confidence level.42 Second order polynomial regression models also known as ‘quadratic models’ were found to be appropriate for describing the experimental data obtained for responses NOx, smoke, HC, BTE and BSFC. They were found to be statistically significant at 99% confidence level. Linear model was found to be best fit for CO emissions and is statistically significant at 99% confidence level.| Response | Alcohol type | Regression equationsa |
|---|---|---|
| a t – injection timing in ° CA; e – EGR rate in %. | ||
| NOx, R2 = 0.9972 | n-Propanol | +16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 114.43 − 1505.17 × t + 19.91 × e − 1.41 × t × e + 37.29 × t2 − 0.07 × e2 114.43 − 1505.17 × t + 19.91 × e − 1.41 × t × e + 37.29 × t2 − 0.07 × e2 |
| n-Butanol | +16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 303.18 − 1515.42 × t + 21.18 × e − 1.41 × t × e + 37.29 × t2 − 0.07 × e2 303.18 − 1515.42 × t + 21.18 × e − 1.41 × t × e + 37.29 × t2 − 0.07 × e2 |
|
| n-Pentanol | +16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 430.68 − 1525.58 × t + 23.93 × e − 1.41 × t × e + 37.29 × t2 − 0.07 × e2 430.68 − 1525.58 × t + 23.93 × e − 1.41 × t × e + 37.29 × t2 − 0.07 × e2 |
|
| Smoke, R2 = 0.9725 | n-Propanol | −8.21 + 0.70 × t + 0.11 × e − 4.12 × 10−3 × t × e − 0.01 × t2 − 3.33 × 10−5 × e2 |
| n-Butanol | −7.37 + 0.66 × t + 0.12 × e − 4.17 × 10−3 × t × e − 0.01 × t2 − 3.33 × 10−5 × e2 | |
| n-Pentanol | −6.98 + 0.65 × t + 0.12 × e − 4.17 × 10−3 × t × e − 0.01 × t2 − 3.33 × 10−5 × e2 | |
| HC, R2 = 0.9810 | n-Propanol | −213.48 + 24.22 × t + 0.87 × e − 0.03 × t × e − 0.56 × t2 + 2.78 × 10−3 × e2 |
| n-Butanol | −226.12 + 24.47 × t + 0.96 × e − 0.03 × t × e − 0.56 × t2 + 2.78 × 10−3 × e2 | |
| n-Pentanol | −250.45 + 24.47 × t + 1.32 × e − 0.03 × t × e − 0.56 × t2 + 2.78 × 10−3 × e2 | |
| CO, R2 = 0.6868 | n-Propanol | +0.01 + 2.78 × 10−4 × t + 3.89 × 10−4 × e |
| n-Butanol | +9.17 × 10−3 + 2.78 × 10−4 × t + 3.89 × 10−4 × e | |
| n-Pentanol | +9.17 × 10−3 + 2.78 × 10−4 × t + 3.89 × 10−4 × e | |
| BTE, R2 = 0.9911 | n-Propanol | +50.28 − 3.76 × t − 0.11 × e − 3.56 × 10−15 × t × e + 0.13 × t2 − 1.01 × 10−15 × e2 |
| n-Butanol | +54.61 − 4.08 × t − 0.11 × e − 3.56 × 10−15 × t × e + 0.13 × t2 − 1.01 × 10−15 × e2 | |
| n-Pentanol | +59.58 − 4.36 × t − 0.10 × e − 3.56 × 10−15 × t × e + 0.13 × t2 − 1.01 × 10−15 × e2 | |
| BSFC, R2 = 0.9885 | n-Propanol | +0.66 − 0.01 × t + 5.06 × 10−3 × e − 1.77 × 10−4 × t × e − 1.99 × 10−4 × t2 + 5.42 × 10−6 × e2 |
| n-Butanol | +0.67 − 0.01 × t + 5.25 × 10−3 × e − 1.77 × 10−4 × t × e − 1.99 × 10−4 × t2 + 5.42 × 10−6 × e2 | |
| n-Pentanol | +0.67 − 9.55 × 10−3 × t + 5.36 × 10−3 × e − 1.77 × 10−4 × t × e − 1.99 × 10−4 × t2 + 5.42 × 10−6 × e2 | |
| Source | NOx | Smoke | ||||
|---|---|---|---|---|---|---|
| SS | F | p-Value | SS | F | p-Value | |
| Model | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 627 627![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 935.67 935.67 |
478.81 | <0.0001 | 1.48 | 48.18 | <0.0001 |
| A-Injection timing | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 126 126![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 609.39 609.39 |
4262.12 | <0.0001 | 0.55 | 197.19 | <0.0001 |
| B-EGR | 322![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 672.22 672.22 |
646.70 | <0.0001 | 0.57 | 203.50 | <0.0001 |
| C-Alcohol type | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300.67 300.67 |
25.35 | <0.0001 | 0.21 | 36.82 | <0.0001 |
| A × B | 9520.33 | 19.08 | 0.0006 | 0.08 | 29.81 | <0.0001 |
| A × C | 5002.11 | 5.01 | 0.0215 | 0.03 | 5.81 | 0.0135 |
| B × C | 5060.11 | 5.07 | 0.0208 | 0.02 | 3.44 | — |
| A2 | 133![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 504.17 504.17 |
267.57 | <0.0001 | 0.02 | 7.30 | 0.0164 |
| B2 | 266.67 | 0.53 | 0.476 | 0.00 | 0.02 | — |
| Error | 7484.33 | 0.04 | ||||
| Total | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 635 635![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 420.00 420.00 |
1.52 | ||||
| Source | HC emissions | CO emissions | ||||
|---|---|---|---|---|---|---|
| SS | F | p-Value | SS | F | p-Value | |
| Model | 3057.56 | 70.45 | <0.0001 | 0.0005 | 12.06 | <0.0001 |
| A-Injection timing | 242.00 | 61.33 | <0.0001 | 5.56 × 10−6 | 0.58 | — |
| B-EGR | 280.06 | 70.98 | <0.0001 | 0.0003 | 28.37 | <0.0001 |
| C-Alcohol type | 2430.30 | 307.97 | <0.0001 | 0.0002 | 9.65 | — |
| A × B | 5.33 | 1.35 | — | — | — | — |
| A × C | 1.00 | 0.13 | — | — | — | — |
| B × C | 68.78 | 8.72 | 0.0031 | — | — | — |
| A2 | 29.63 | 7.51 | 0.0152 | — | — | — |
| B2 | 0.46 | 0.12 | — | — | — | — |
| Error | 59.19 | 0.0002 | ||||
| Total | 3116.74 | 0.0007 | ||||
| Source | BTE | BSFC | ||||
|---|---|---|---|---|---|---|
| SS | F | p-Value | SS | F | p-Value | |
| Model | 347.04 | 151.64 | <0.0001 | 0.049072 | 117.20 | <0.0001 |
| A-Injection timing | 232.55 | 1117.79 | <0.0001 | 0.038196 | 1003.45 | <0.0001 |
| B-EGR | 18.90 | 90.83 | <0.0001 | 0.003377 | 88.72 | <0.0001 |
| C-Alcohol type | 89.70 | 215.58 | <0.0001 | 0.007291 | 95.77 | <0.0001 |
| A × B | 0.00 | 0.00 | — | 0.000151 | 3.95 | — |
| A × C | 4.32 | 10.38 | 0.0015 | 2.5 × 10−5 | 0.33 | — |
| B × C | 0.01 | 0.02 | — | 2.7 × 10−5 | 0.35 | — |
| A2 | 1.56 | 7.50 | 0.0152 | 3.82 × 10−6 | 0.10 | — |
| B2 | 0.00 | 0.00 | — | 1.76 × 10−6 | 0.05 | — |
| Error | 3.12 | 0.000571 | ||||
| Total | 350.16 | 0.049643 | ||||
Table 11 lists the values of various terms that were used to evaluate the developed models. The coefficient of determination (R2) indicates how well the experimental data fit the developed statistical models. A value of 1 indicates a perfect fit while a value of 0 indicates that the regression line does not fit the data.43 It can be inferred that the correlation between the experimental and predicted values were found to be excellent for NOx (R2 = 0.9972), smoke (R2 = 0.9725), HC (R2 = 0.9810), BTE (R2 = 0.9911) and BSFC (R2 = 0.9885). CO model was found to correlate only by upto 68.68% (R2 = 0.6868) to the experimental data. Fig. 3 shows the graph plotted between the predicted and the actual values for all the responses. Excellent agreement between the predicted and actual values for all the responses except CO is evident. This is because CO values of the blends remained very low and range of variation is only 0.01% vol.
| Model | NOx | Smoke | HC | CO | BTE | BSFC |
|---|---|---|---|---|---|---|
| Degree | Quadratic | Quadratic | Quadratic | Linear | Quadratic | Quadratic |
| R2 | 0.9972 | 0.9725 | 0.9810 | 0.6868 | 0.9911 | 0.9885 |
| Adj. R2 | 0.9951 | 0.9523 | 0.9671 | 0.6299 | 0.9846 | 0.9801 |
| Pred. R2 | 0.9895 | 0.9120 | 0.9382 | 0.5266 | 0.9734 | 0.9657 |
| Adeq. precision | 69.2563 | 25.6274 | 29.1150 | 10.8356 | 44.8925 | 38.5621 |
| CoV% | 2.21 | 12.33 | 4.67 | 12.30 | 1.69 | 1.76 |
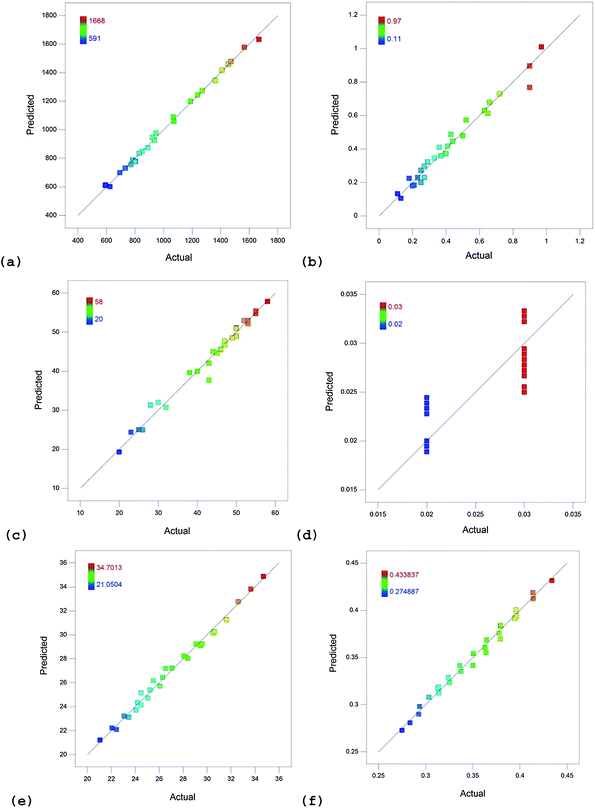 | ||
| Fig. 3 Plots of actual and predicted values for (a) NOx emissions, (b) smoke, (c) HC emissions, (d) CO emissions, (e) BTE and (f) BSFC. | ||
The adjusted R2 value is also high (except for CO model) which indicates that the models are highly accurate.44 The accuracy of the CO model is only 63%. All the Pred. R2 values are in reasonable agreement with the Adj. R2 values because their difference is less than 0.2. Adeq. precision which measures the signal-to-noise ratio of the data is greater than the desirable value of 4. Hence, this model can be used to navigate the design space. Low values of coefficient of variation (CoV) denote that the predicted values are relatively closer to the actual values. Low values of CoV thus indicate that the developed models can be highly reliable.45 CoV values of all the models shown in Table 11 were found to be low for all responses.
3.2 NOx (oxides of nitrogen)
As per ANOVA from Table 8, all the considered factors have significant influence on NOx emissions at 99% confidence intervals. Fig. 4 provides a comparison of emission and performance characteristics for all higher alcohol/diesel blends as a function of injection timings at 30% EGR. The trends of all responses for all alcohol/diesel blends are similar at all EGR rates and hence for the sake of brevity of space, only the highest EGR rate is presented. It can be seen that NOx emissions are low when the injection timing is retarded. Late injection causes more fuel to burn after the TDC and hence the peak cylinder pressure decreases. This in-turn reduces peak cylinder temperature and reduces NOx emissions.46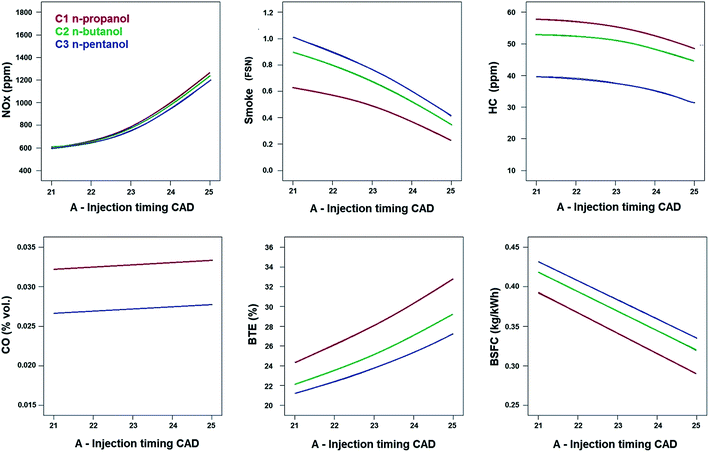 | ||
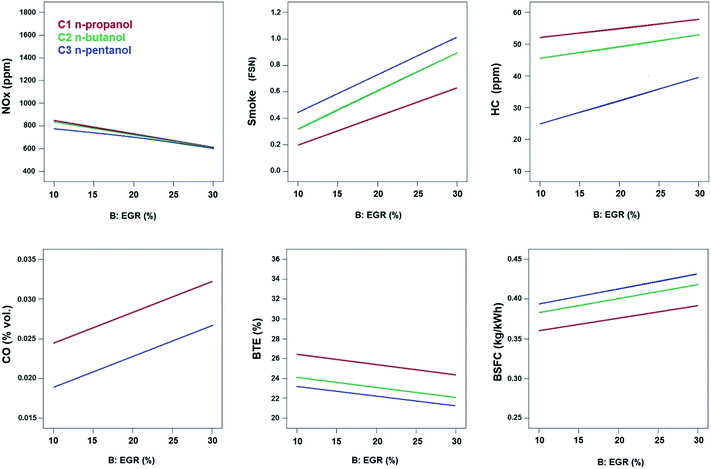
| Fig. 4 Comparison of emission and performance characteristics between higher alcohol/diesel blends as a function of injection timing under EGR = 30%, bmep = 4.0 bar and engine speed = 1500 rpm. | ||
Fig. 5 provides a comparison of emission and performance characteristics for all higher alcohol/diesel blends as a function of EGR rates at retarded injection timing of 21° CA bTDC. The trend of all responses for all higher alcohol/diesel blends are similar at all injection timings and hence again, only retarded injection timing is presented in this study for the sake of brevity of space. Irrespective of the injection timing, introduction of EGR increases the concentration of inert gases which reduces the peak temperatures that favors NOx formation inside the combustion chamber.11 Fig. 5 confirms the reduction in NOx emissions when the EGR rate is increased.
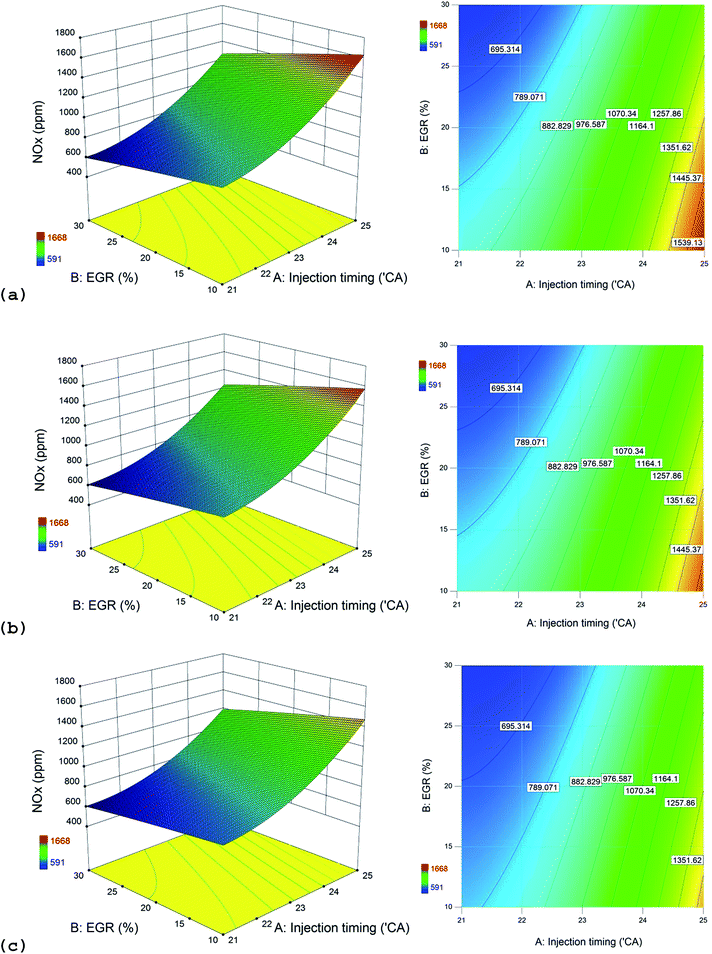
The interactive effect of injection timing and EGR on NOx emissions, when the engine is fueled with three alcohol blends is portrayed in Fig. 6. The bluish area of the contour plots indicates the regions where the NOx emissions are low. This surface is found at the region where the injection timing is latest and the EGR rate is high. In general, a decreasing trend in NOx emissions is evident when the injection timing is retarded from 25° CA bTDC to 21° CA bTDC and when the EGR rate increases from 10 to 30%. The reddish areas of the response surfaces (where NOx emissions are high) are more pronounced with n-propanol/diesel blends followed by n-butanol/diesel and n-pentanol/diesel blends. NOx emission for higher alcohol/diesel blends is usually the result of counter-active effects between the fuel cetane number, oxygen content and latent heat of vaporization. The cetane indices of the blends are of the order (as inferred from Table 2): n-propanol/diesel < n-butanol/diesel < n-pentanol/diesel. Equally from Table 2, the oxygen content of the blends is in the sequence of: n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel. From Table 1, it can be deduced that the latent heat of vaporization is of the order: n-propanol > n-butanol > n-pentanol which could be applied to its diesel blends. Lower cetane number of the fuel causes longer ignition delay periods during which a large quantity of fuel enters the combustion chamber to produce the same power output of the engine as would with pure diesel. The combustion of this large quantity of fuel produces high temperatures which increases NOx emissions. Similarly the high oxygen content of the blends improves combustion efficiency which also increases NOx emissions. Contrarily, high latent heat of vaporization of the blends causes a cooling effect that reduces NOx emissions.11
From Fig. 4–6, it can be concluded that NOx emissions are of the order (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blends at bmep = 4.0 bar and speed = 1500 rpm. n-Propanol/diesel blends produced highest NOx emissions due to its lowest cetane number (causing longest ignition delay) and highest oxygen content than the other blends.
3.3 Smoke
All the considered factors have significant influence on smoke emissions of the blends at 99% confidence level as per ANOVA from Table 8. Smoke intensifies as EGR increases (as seen from Fig. 5) for all alcohol/diesel blends. This is due to the oxygen deficient environment induced by EGR that deteriorates normal combustion. Late injection causes low peak temperatures during the expansion stroke and less time for soot oxidation to occur.46 Hence smoke is high at retarded injection timing as seen in Fig. 4. The interactive effect of injection timing and EGR on smoke, when the engine is fueled with three alcohol blends is shown in Fig. 7. Smoke is generally high at the combination of high EGR rates and retarded injection timing. The combined effect of low EGR and early injection is seen as bluish areas in the contour plot where smoke is low due to more oxygen at low EGR rates and more time for soot oxidation at early injection.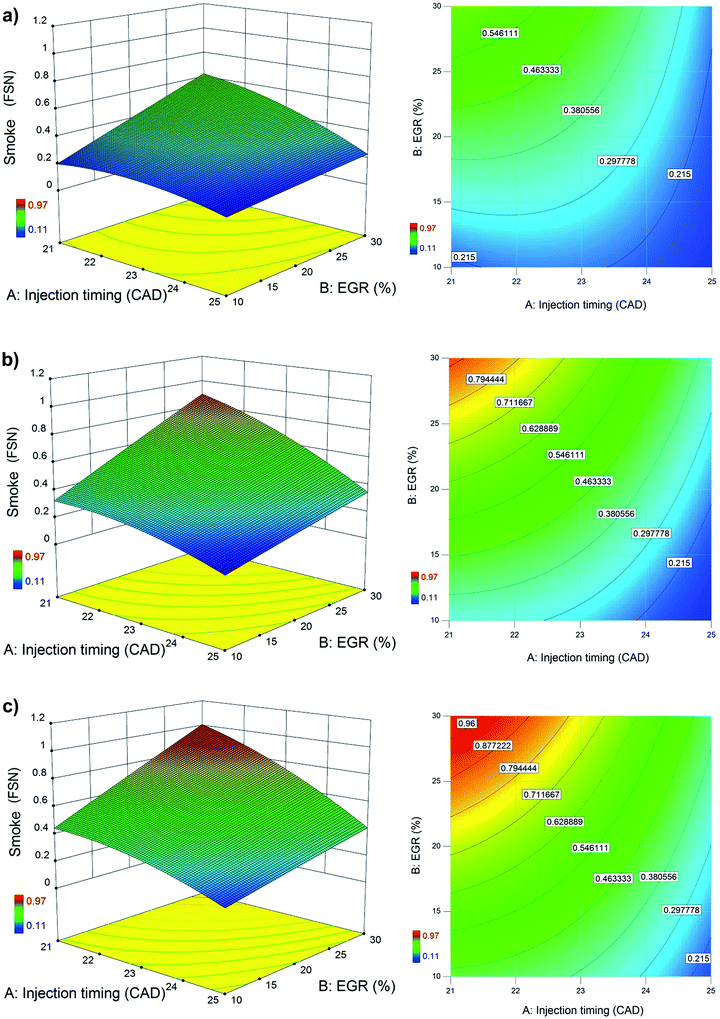 | ||
| Fig. 7 Interactive effect of injection timing and EGR on smoke for (a) n-propanol/diesel blend, (b) n-butanol/diesel blend and (c) n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm. | ||
From Fig. 4, 5 and 7, it can be inferred that smoke is lowest for n-propanol/diesel blend due to its highest oxygen content among the other blends (Table 2). Fuel-bound oxygen content suppresses soot formation by inhibiting soot precursors and increases availability of oxygen even in fuel-rich zones.9 In an earlier work by the authors,47 it was reported that smoke emissions increased with increase in length of the carbon chain in the alcohol fuel molecule. Hence it was found that high carbon alcohols tend to produce more smoke than low carbon alcohols. Eventually it can be observed from Fig. 7 that smoke clearly follows the order of the oxygen content in the blends and the length of carbon chain in the alcohols as follows, n-propanol/diesel < n-butanol/diesel < n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm.
3.4 HC (hydrocarbons)
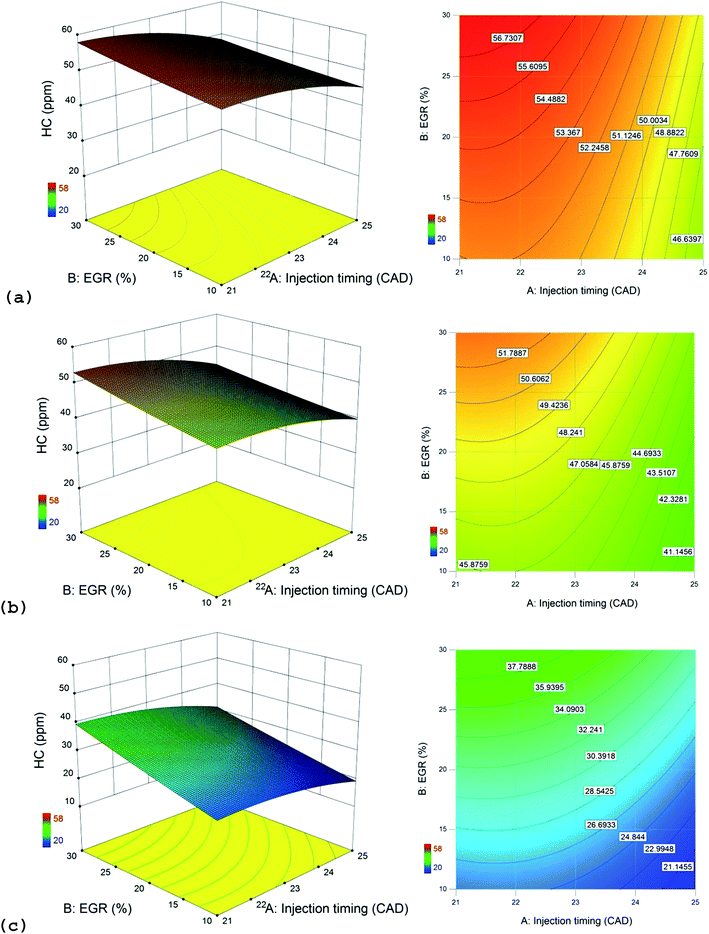
The effects of all the considered factors on HC emissions were found to have statistical significance at 99% confidence level. HC emissions are generally high for alcohols due to the combined effect of lower cetane number and higher latent heat of vaporization when compared to diesel.48 HC emissions are high at high EGR rates due to incomplete combustion as evident from Fig. 5. Late injection causes quenching effect at the cylinder walls which increases HC emissions as seen in Fig. 4. The interactive effect plot between injection timing and EGR in HC emissions is shown in Fig. 8. HC emissions are high in the reddish regions of the response surface and contour plots. These regions correspond to the region where EGR rates are high and the injection timing is retarded.The highest HC emissions is noticed for n-propanol/diesel blend when compared to other blends, as evident from Fig. 4, 5 and 8 which is due to its lowest cetane number and highest latent heat of vaporization. Low cetane number causes longer ignition delays which promotes quenching effect in the leaner mixture zones of the cylinder which increases HC emissions. High latent heat of vaporization causes poorer and slower fuel–air mixing that can increase NOx emissions.49 Thus HC emissions clearly follow the order of cetane index and latent heat of vaporization of the blends as follows (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm.
3.5 CO (carbon monoxide)
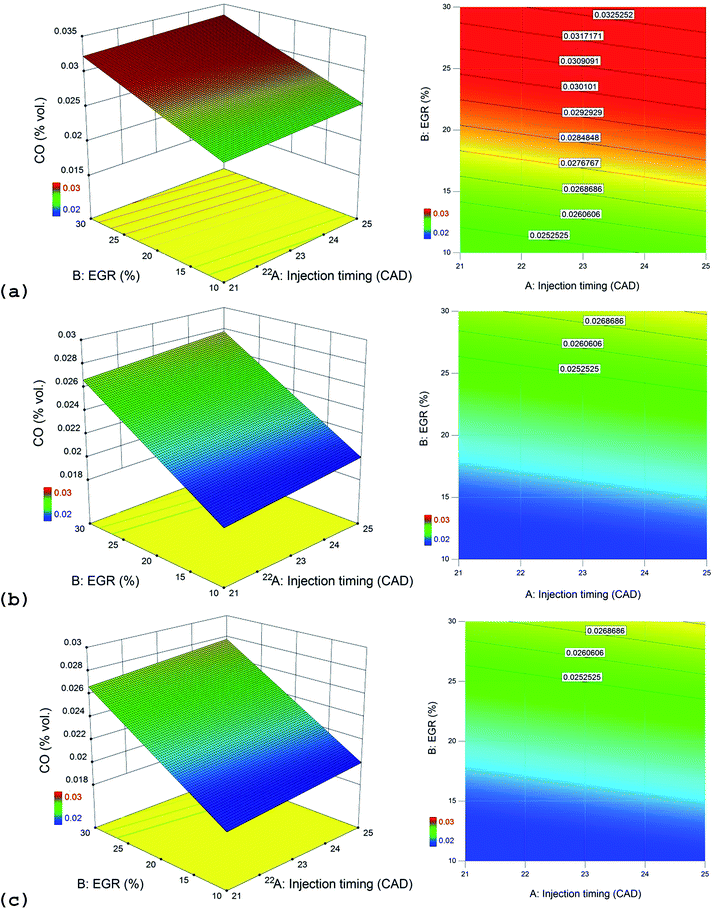
The effect of EGR on CO emissions is found to be significant at 99% confidence level as per ANOVA in Table 9. EGR prevents oxidation of CO due to oxygen deficient conditions inside the cylinder and hence it increases with EGR as seen in Fig. 5. The interactive effect of EGR and injection timing for CO emissions is depicted in Fig. 9. CO emissions also follow the trend of the HC emissions. It could be inferred from the contour plots that CO emissions are generally high at all injection timings under high EGR rates. n-Propanol/diesel blend presents highest CO emissions when compared to other blends. The sequence of CO emissions is as follows (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm.3.6 BTE (brake thermal efficiency)
As per ANOVA (from Table 10), all the factors have significant influence on BTE of the engine when fueled with these blends. It is obvious that EGR deteriorates combustion by decreasing oxygen concentration inside the cylinder and hence BTE decreases with increasing EGR rates as seen in Fig. 5. Advancing the injection timing to 25° CA causes a large amount of evaporated fuel to accumulate in the combustion chamber during the ignition delay period. This leads to rapid burning during the premixed combustion phase causing very high pressure and high heat release rates. Hence the power output increases resulting in improved performance as evident from Fig. 4.The interactive plots between injection timing and EGR for all alcohol/diesel blends BTE were presented in Fig. 10. BTE is high at regions closer to low EGR and early injection timings for all the blends. BTE for alcohol/diesel blends is based on the combined effects of cetane number, calorific value, viscosity, density and oxygen content. Low cetane number of the blends extends the ignition delay which causes more quantity of fuel to be injected into the combustion chamber during this period. When this large quantity of blend ignites, it results in high pressure and heat release rates during the premixed combustion phase thus increasing the power output of the engine and eventually the BTE. High calorific value of the fuel delivers high energy per unit kg of the blends. Viscosity and density affect the fuel atomization characteristics and spray formation. The density and viscosity of the blends is of the sequence (from lowest to highest): n-propanol/diesel < n-butanol/diesel < n-pentanol/diesel blends. High oxygen content in the fuel improves combustion efficiency by increasing oxygen availability even in fuel-rich zones. Similar trends with n-butanol/diesel blends exhibiting better BTE than n-pentanol/diesel blends were reported by Campos-Fernández et al.25 despite the higher energy content of n-pentanol than n-butanol.
 | ||
| Fig. 10 Interactive effect of injection timing and EGR on BTE for (a) n-propanol/diesel blend, (b) n-butanol/diesel blend and (c) n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm. | ||
n-Propanol/diesel blends delivered best performance among the blends due to its highest oxygen content, lowest viscosity and lowest density. The sequence of BTE for the blends is as follows (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm. This sequence is confirmed by the trends seen in Fig. 4, 5 and 10.
3.7 BSFC (brake specific fuel consumption)
ANOVA (from Table 10) confirms significant influence for all considered factors on BSFC of the engine like BTE. BSFC increases with increasing EGR rates as seen in Fig. 5 due to the degradation of combustion quality as result of less oxygen and high inert gas concentration. Late injection also increases BSFC due to deterioration of combustion efficiency as seen in Fig. 4. Fig. 11 depicts the interactive effect of EGR and injection timing on BSFC of the engine when fueled with alcohol/diesel blends. It can be seen that the reddish high consumption areas are predominant when the EGR rate is high and when the injection timing is retarded. | ||
| Fig. 11 Interactive effect of injection timing and EGR on BSFC for (a) n-propanol/diesel blend, (b) n-butanol/diesel blend and (c) n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm. | ||
BSFC is generally very high for n-pentanol/diesel blend as a result of less oxygen content, high viscosity and density when compared to the other blends. This directly affects the combustion efficiency requiring more quantity of fuel blends to be burned to produce the same power output in the engine. It could be noted from Table 2 that the low heating value of n-pentanol/diesel blends is only slightly higher than n-butanol/diesel and n-propanol/diesel blends which has less effect on combustion efficiency when compared to the effects of high oxygen content, low viscosity and density of the other fuel blends. In an earlier work by the authors,12 40% by vol. of iso-butanol/diesel blend has presented better BSFC when compared to 40% by vol. of n-pentanol/diesel blend despite the slightly lower energy content of iso-butanol than n-pentanol. Campos-Fernández et al.25 also have reported n-butanol/diesel blends to have better BSFC than n-pentanol/diesel blends. This trend is here confirmed by Fig. 4, 5 and 11. The sequence of BSFC for the blends is as follows (from lowest to highest): n-propanol/diesel < n-butanol/diesel < n-pentanol/diesel blend at bmep = 4.0 bar and speed = 1500 rpm.
3.8 Optimization
One of the objectives of this study is to optimize injection timing, EGR and the type of alcohol used to achieve minimum NOx, smoke emissions, BSFC with maximum BTE. The criteria for optimization for the responses based on the objective stated above with their lower and upper limits, the weightages used and their importance is shown in Table 12. Weights range from 0.1 to 10. A weight greater than 1 gives more emphasis to the goal while a weight less than 1 gives less emphasis. The desirability value varies in a linear mode with weight equal to 1.| Factors | Target | Limits | Weight | Importance | ||
|---|---|---|---|---|---|---|
| Lower | Upper | Lower | Upper | |||
| A: Injection timing | Is in range | 21 | 25 | 1 | 1 | 5 |
| B: EGR | Is in range | 10 | 30 | 1 | 1 | 5 |
| C: Alcohol type | Is in range | n-Propanol | n-Pentanol | 1 | 1 | 5 |
| NOx | Minimize | 591 | 1668 | 1 | 0.1 | 5 |
| Smoke | Minimize | 0.11 | 0.97 | 1 | 0.1 | 5 |
| BTE | Maximize | 21.0504 | 34.7013 | 0.1 | 1 | 5 |
| BSFC | Minimize | 0.2749 | 0.4338 | 1 | 0.1 | 5 |
Table 13 lists the optimal settings of the considered factors for minimizing smoke and NOx emissions with best BTE and low BSFC. Desirability based approach was used to obtain various solutions for the set optimization criteria and eventually the optimal conditions were generated using Design Expert®. Solutions with high desirability are close to the set optimization criteria and are preferred. From this approach, n-propanol/diesel blend injected at 25° CA bTDC at 30% EGR is considered to be optimum for this particular engine as it has a maximum desirability of 0.965. n-Butanol/diesel blend injected at 24° CA bTDC at 10% EGR is found to be best for the set objective among the various settings for n-butanol. Equally, n-pentanol/diesel blend injected at 24° CA bTDC at 10% EGR is found to be best for the set objective among the various settings for n-pentanol.
| Best solution | Injection timing, ° CA | EGR, % | Alcohol type, - | NOx, ppm | Smoke, FSN | HC, ppm | CO, % vol. | BTE, % | BSFC, kg kW−1 h−1 | Desirability, - |
|---|---|---|---|---|---|---|---|---|---|---|
| Optimum among all blends | 25 | 30 | n-Propanol | 1280.55 | 0.23 | 48.44 | 0.03 | 32.81 | 0.290 | 0.965 |
| Optimum for n-butanol | 24 | 10 | n-Butanol | 1232.46 | 0.22 | 43.50 | 0.02 | 28.73 | 0.324 | 0.952 |
| Optimum for n-pentanol | 24 | 10 | n-Pentanol | 1239.77 | 0.27 | 21.88 | 0.02 | 27.69 | 0.329 | 0.945 |
Fig. 12 shows the optimized values of the factors considered for each alcohol/blend graphically with their desirability values to achieve minimal smoke and NOx emissions with maximum BTE and minimum BSFC. The red cross in the graphic indicates the optimum level of the factor for a particular response for a particular alcohol/diesel blend. Since the NOx emissions of n-propanol/diesel blends are very high, it can be seen high amount of EGR (30%) is required to bring it down when compared to the other blends.
 | ||
| Fig. 12 Main effects plot of factors at optimized conditions for (a) n-propanol/diesel (b) n-butanol/diesel and (c) n-pentanol diesel blends. | ||
Graphical optimization displays the areas where the optimization criteria can be met by the response values.50 Fig. 13 shows the overlay plots obtained by superimposing the contours of all the response surfaces developed for NOx, smoke, BTE and BSFC for all blends. This plot reveals a “sweet spot” (shown in yellow) where all the criteria set for optimization are met by the responses within the factor space.
3.9 Validation
Confirmatory tests were carried out using the optimal settings of the factors considered in order to validate the solutions generated by Design Expert® software using desirability approach. Three trials were conducted at the optimum factor levels and the average value was recorded. Table 14 shows the results of the confirmatory experiments with the predicted values from RSM models and the averaged actual values from the confirmatory trials. The percentages of error in predicting the optimized parameters (NOx, smoke, BTE and BSFC) were given alongside. It can be seen that the models developed using RSM were adequate to describe the effect of the injection timing and EGR on performance and emissions for all blends and the error in prediction was found to be within 5%.| # | Injection timing, ° CA | EGR, % | Alcohol type | Error | NOx, ppm | Smoke, FSN | BTE, % | BSFC, kg kW−1 h−1 | HC, ppm | CO, % vol. |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 25 | 30 | n-Propanol | Predicted | 1280.545 | 0.231 | 32.806 | 0.290 | 48.444 | 0.033 |
| Actual | 1265 | 0.24 | 32.671 | 0.288 | 50 | 0.03 | ||||
| % error | 1.21 | 3.89 | 0.41 | 0.68 | 3.21 | 9.09 | ||||
| 2 | 24 | 10 | n-Butanol | Predicted | 1232.456 | 0.22 | 28.730 | 0.324 | 43.503 | 0.020 |
| Actual | 1255 | 0.21 | 29.546 | 0.333 | 41 | 0.02 | ||||
| % error | 1.82 | 4.54 | 2.84 | 2.77 | 5.75 | 0.00 | ||||
| 3 | 24 | 10 | n-Pentanol | Predicted | 1239.765 | 0.271 | 27.689 | 0.329 | 21.884 | 0.020 |
| Actual | 1224 | 0.28 | 26.924 | 0.320 | 19 | 0.02 | ||||
| % error | 1.27 | 3.70 | 2.76 | 2.73 | 13.18 | 0.00 |
The contour plots at optimum conditions (low NOx, low smoke, high BTE and low BSFC) for all alcohol/diesel blends are presented in Fig. 14. Reddish zones indicate high values of responses while the bluish zones indicate low values.
 | ||
| Fig. 14 Contour plots at optimum conditions (low NOx, low smoke, high BTE and low BSFC) for (a) n-propanol/diesel blend, (b) n-butanol/diesel blend and (c) n-pentanol/diesel blend. | ||
4. Conclusions
This study employed three high carbon bio-alcohols namely n-propanol, n-butanol and n-pentanol as blend components (40% by vol.) with fossil diesel. A statistical investigation was carried out using response surface methodology to compare and analyze the interactive effects of injection timing and EGR on the emission and performance characteristics of the engine fueled with these three high carbon bio-alcohol/diesel blends. RSM was used to model, predict and optimize the experimental data of the responses obtained as per the full-factorial experimental design matrix at bmep = 4.0 bar and engine speed = 1500 rpm. The following conclusions were drawn from the analysis:1. As per ANOVA,
(i) All the developed regression models for NOx, smoke, HC, CO, BTE and BSFC were found to be statistically significant at 99% confidence level.
2. From the response surface and contour plots, it can be concluded that
(i) NOx emissions of the high carbon bio-alcohol/diesel blends are of the order (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blends. n-Propanol/diesel blend produced highest NOx emissions due to its lowest cetane number (causing longest ignition delay) and highest oxygen content than the other blends.
(ii) Smoke emissions for the blends follow the order as follows (from lowest to highest): n-propanol/diesel < n-butanol/diesel < n-pentanol/diesel blend. Smoke is lowest for n-propanol/diesel blend due to its highest oxygen content among the other blends.
(iii) HC emissions clearly follow the order of cetane number and latent heat of vaporization of the blends as follows (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel.
(iv) n-Propanol/diesel blend presented highest CO emissions than the other blends. The sequence of CO emissions is as follows (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blend.
(v) n-Propanol/diesel blends delivered best performance among the blends due to its highest oxygen content, lowest viscosity and lowest density. The sequence of BTE for the blends is as follows (from highest to lowest): n-propanol/diesel > n-butanol/diesel > n-pentanol/diesel blend.
(vi) BSFC is generally very high for n-pentanol/diesel blend as a result of less oxygen content, high viscosity and density of the blends. The sequence of BSFC for the blends is as follows (from lowest to highest): n-propanol/diesel < n-butanol/diesel < n-pentanol/diesel blend.
3. The optimization criterion is to minimize smoke, NOx and BSFC of the engine with maximum BTE. Solutions that are closer to this criterion were obtained using desirability approach. From this approach, n-propanol/diesel blend injected at 25° CA bTDC at 30% EGR is considered to be optimum for this particular engine. n-Butanol/diesel and n-pentanol/diesel blends injected at 24° CA bTDC at 10% EGR were found to be best for the respective alcohol types according to the set objective.
4. Confirmatory experiments using the optimal factor settings indicated that the models developed using RSM were adequate to describe the interactive effect of the injection timing and EGR on performance and emissions for all blends within 5% of error in prediction.
Abbreviations
| a(b)BDC | After(before) bottom dead centre |
| a(b)TDC | After(before) top dead centre |
| ANOVA | Analysis of variance |
| ASTM | American society for testing and materials |
| bmep | Brake mean effective pressure |
| BUT40 | 40% n-butanol + 60% ULSD blend |
| BSFC | Brake specific fuel consumption |
| BTE | Brake thermal efficiency |
| CA | Crank angle |
| CAS | Chemical abstract service |
| CFPP | Cold filter plugging point |
| CO | Carbon monoxide |
| CO2 | Carbon dioxide |
| CoV | Coefficient of variation |
| DI | Direct injection |
| EGR | Exhaust gas recirculation |
| FSN | Filter smoke number |
| HC | Hydrocarbons |
| MMT | Million metric tons |
| NOx | Nitrogen oxides |
| PEN40 | 40% n-pentanol + 60% ULSD blend |
| PM | Particulate matter |
| PRO40 | 40% n-propanol + 60% ULSD blend |
| RSM | Response surface methodology |
| ULSD | Ultra-low sulfur diesel |
| UHC | Un-burnt hydrocarbons |
Symbols
| β | Regression coefficient |
| e | EGR rates |
| ε | Experimental error |
| wt% | Percentage per weight |
| Adj. R2 | Adjusted R2 value |
| F | Value of F-statistic test |
| P | Percentage contribution |
| Pred. R2 | Predicted R2 value |
| p-Value | Probability value |
| R2 | Coefficient of determination |
| SS | Sum of squares |
| t | Injection timing |
| X | Numeric value of the factors |
| Y | Value of the response |
Acknowledgements
The authors appreciate the support of the respective Managements of Sathyabama University, Jeppiaar Maamallan Institute of Technology, Jeppiaar Institute of Technology and Sri Venkateswara College of Engineering, Sriperumbudur, Chennai for facilitating this research. The authors gratefully thank the assistance rendered by Dr A. Nagendran, PG & Research Department of Chemistry, Polymeric Materials Research Lab at Alagappa College, Karaikudi for estimating fuel properties. The authors would also like to acknowledge Dr D. Rana, Department of Chemical and Biological Engineering, University of Ottawa for his technical assistance.References
- B. Rajesh Kumar and S. Saravanan, Renewable Sustainable Energy Rev., 2016, 60, 84–115 CrossRef CAS.
- Z. Wadud, Appl. Energy, 2016, 165, 849–857 CrossRef.
- A. Schaeffer, Auto Tech Review, 2016, 5, 12–13 CrossRef.
- H. Carvalho, Lancet Respir. Med., 2016, 4, e2–e3 CrossRef PubMed.
- B. Rajesh Kumar and S. Saravanan, Fuel, 2016, 180, 396–406 CrossRef CAS.
- L. Van Eeckhout, Comment Anne Hidalgo veut réduire la pollution à Paris, Le Monde, Paris, Jan 27, 2015 Search PubMed.
- G. Graham, London will follow Paris and ban diesel cars, campaigners warn, The Telegraph, London, Dec 8, 2014 Search PubMed.
- J. Fan, M. De bruyn, V. Budarin, M. Gronnow, P. Shuttleworth, S. Breeden, D. Macquarrie and J. Clark, J. Am. Chem. Soc., 2013, 135, 11728–11731 CrossRef CAS PubMed.
- B. Rajesh Kumar, S. Saravanan, D. Rana, V. Anish and A. Nagendran, Energy Convers. Manage., 2016, 118, 275–286 CrossRef CAS.
- G. Najafi, B. Ghobadian, T. Yusaf, S. Safieddin Ardebili and R. Mamat, Energy, 2015, 90, 1815–1829 CrossRef CAS.
- B. Rajesh kumar and S. Saravanan, Fuel, 2015, 160, 217–226 CrossRef CAS.
- B. Rajesh Kumar and S. Saravanan, Fuel, 2016, 170, 49–59 CrossRef CAS.
- L. Siwale, L. Kristóf, T. Adam, A. Bereczky, M. Mbarawa, A. Penninger and A. Kolesnikov, Fuel, 2013, 107, 409–418 CrossRef CAS.
- S. Kook, C. Bae, P. Miles, D. Choi and L. Pickett, SAE Tech. Pap. Ser., 2005, DOI: 10.4271/2005-01-3837 Search PubMed.
- M. Yao, H. Wang, Z. Zheng and Y. Yue, Fuel, 2010, 89, 2191–2201 CrossRef CAS.
- D. Rakopoulos, C. Rakopoulos, E. Giakoumis, A. Dimaratos and D. Kyritsis, Energy Convers. Manage., 2010, 51, 1989–1997 CrossRef CAS.
- O. Armas, R. García-Contreras and Á. Ramos, Fuel Process. Technol., 2012, 100, 63–72 CrossRef CAS.
- Z. Chen, J. Liu, Z. Han, B. Du, Y. Liu and C. Lee, Energy, 2013, 55, 638–646 CrossRef CAS.
- C. D. Rakopoulos, A. M. Dimaratos, E. G. Giakoumis and D. C. Rakopoulos, Appl. Energy, 2011, 88, 3905–3916 CrossRef CAS.
- B. Choi, X. Jiang, Y. Kim, G. Jung, C. Lee, I. Choi and C. Song, Appl. Energy, 2015, 146, 20–28 CrossRef CAS.
- A. Singh, C. Mishra, V. Vibhanshu and N. Kumar, SAE Tech. Pap. Ser., 2013 DOI:10.4271/2013-01-1132.
- T. Balamurugan and R. Nalini, Energy, 2014, 78, 356–363 CrossRef CAS.
- H. Hazar and M. Uyar, International Journal of Automotive Engineering and Technologies, 2015, 4, 1 CrossRef.
- L. Wei, C. Cheung and Z. Huang, Energy, 2014, 70, 172–180 CrossRef CAS.
- J. Campos-Fernández, J. Arnal, J. Gómez and M. Dorado, Appl. Energy, 2012, 95, 267–275 CrossRef.
- S. Subramani and R. Babu, J. Braz. Soc. Mech. Sci. Eng., 2015 DOI:10.1007/s40430-015-0361-1.
- S. Saravanan, G. Nagarajan and S. Sampath, Fuel, 2013, 104, 409–416 CrossRef CAS.
- M. Pandian, S. Sivapirakasam and M. Udayakumar, Appl. Energy, 2011, 88, 2663–2676 CrossRef CAS.
- B. Hirkude and A. Padalkar, Fuel, 2014, 119, 266–273 CrossRef.
- T. Ganapathy, R. Gakkhar and K. Murugesan, Int. J. Sustain. Energ., 2011, 30, S76–S90 CrossRef.
- E. Ileri, A. Karaoglan and A. Atmanli, J. Renewable Sustainable Energy, 2013, 5, 033132 CrossRef.
- A. Atmanlı, B. Yüksel, E. İleri and A. Deniz Karaoglan, Energy Convers. Manage., 2015, 90, 383–394 CrossRef.
- S. Dhingra, G. Bhushan and K. K. Dubey, J. Renewable Sustainable Energy, 2013, 5, 063117 CrossRef.
- Z. Win, R. Gakkhar, S. Jain and M. Bhattacharya, Proc. Inst. Mech. Eng., Part D, 2005, 219, 1181–1192 CrossRef.
- Z. Zheng, C. Li, H. Liu, Y. Zhang, X. Zhong and M. Yao, Fuel, 2015, 141, 109–119 CrossRef CAS.
- B. Rajesh Kumar and S. Saravanan, Proc. Inst. Mech. Eng., Part A, 2015, 230, 112–125 CrossRef.
- GOI (Government of India), Annual Report (2013–2014), Ministry of Petroleum & Natural Gas, New Delhi, India, 2014, http://petroleum.nic.in/docs/Annual_Report/AR13-14.pdf, accessed 21 Jun 2016 Search PubMed.
- GOI (Government of India), Indian Petroleum and Natural Gas Statistics, Economics and Statistics Division, Ministry of Petroleum & Natural Gas, New Delhi, India, 2014, http://petroleum.nic.in/docs/pngstat.pdf, accessed 21 Jun 2016 Search PubMed.
- J. Devaraj, Y. Robinson and P. Ganapathi, Energy, 2015, 85, 304–309 CrossRef CAS.
- R. Moffat, J. Fluids Eng., 1985, 107, 173 CrossRef CAS.
- A. Fayyazbakhsh and V. Pirouzfar, Fuel, 2016, 171, 167–177 CrossRef CAS.
- J. Avramović, O. Stamenković, Z. Todorović, M. Lazić and V. Veljković, Fuel Process. Technol., 2010, 91, 1551–1557 CrossRef.
- D. Gangadharan, S. Sivaramakrishnan, K. Nampoothiri, R. Sukumaran and A. Pandey, Bioresour. Technol., 2008, 99, 4597–4602 CrossRef CAS PubMed.
- T. Mutuk and B. Mesci, J. Cleaner Prod., 2014, 69, 128–132 CrossRef CAS.
- R. McAuliffe, Wiley Encyclopedia of Management, 2015, p. 1 Search PubMed.
- C. Sayin, M. Ilhan, M. Canakci and M. Gumus, Renewable Energy, 2009, 34, 1261–1269 CrossRef CAS.
- B. Rajesh Kumar, S. Saravanan, D. Rana and A. Nagendran, Energy Convers. Manage., 2016, 119, 246–256 CrossRef CAS.
- O. Doğan, Fuel, 2011, 90, 2467–2472 CrossRef.
- A. Ozsezen, A. Turkcan, C. Sayin and M. Canakci, Energy Explor. Exploit., 2011, 29, 525–541 CrossRef CAS.
- S. Ghafari, H. Aziz, M. Isa and A. Zinatizadeh, J. Hazard. Mater., 2009, 163, 650–656 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |