Base free regioselective synthesis of α-triazolylazine derivatives†
Mysore Bhyrappa Harishaab,
Muthupandi Nagaraja,
Shanmugam Muthusubramanian*a and
Nattamai Bhuvaneshc
aDepartment of Organic Chemistry, School of Chemistry, Madurai Kamaraj University, Madurai – 625 021, Tamil Nadu, India. E-mail: muthumanian2001@yahoo.com
bAdvinus Therapeutics Limited, A TATA Enterprise, Bangalore – 560 058, India
cX-ray Diffraction Laboratory, Department of Chemistry, Texas A & M University, College Station, TX 77842, USA
First published on 13th June 2016
Abstract
A regioselective α-heteroarylation followed by deoxygenation towards the synthesis of variety of azine triazole from simple azine N-oxides derivatives and N-tosyl-1,2,3-triazoles has been described. The reaction is metal free and base free with shorter reaction time, high yields and a broad substrate scope.
Introduction
(Hetero)aryl substituted azines have a wide range of medicinal properties. Pyridotriazoles and quinolinotriazoles1 are particularly interesting, as they exhibit significant biological properties including control of arthropod tests,2 substance-related disorders,3 ATP-competitive inhibition of vascular endothelial growth factor receptors I and II4 and other biological activities.5 A number of derivatives of 1-(pyridin-2-yl)-1,2,3-triazole have been used as ligands.6 In recent years, direct (hetero)arylation has emerged as an attractive alternative to the commonly employed cross coupling reactions as it avoids the preliminary preparation of the metallated or halogenated arene. Several reviews highlight the broad scope of this strategy involving high functional group tolerance, atom economy and mild reaction conditions.7Functionalized azines are to be designed due to their biological importance and though significant progress has been achieved in the development of catalytic alkylation, alkenylation and acylation reactions at the pyridine nucleus,8 the arylation/heteroarylation9 and alkenylation10 at pyridine N-oxides could be achieved only with transition metal catalysts. The regioselective arylation of azines has been achieved through Pd-catalyzed direct arylation of the corresponding N-oxides and N-iminopyridinium-ylides.11 New metal free methods of arylation with regiocontrol are thus invaluable. We report a new C–N bond forming reaction via regioselective α-heteroarylation followed by deoxygenation towards the synthesis of variety of azine triazole from simple azine N-oxides derivatives and N-tosyl-1,2,3-triazole.
Results and discussion
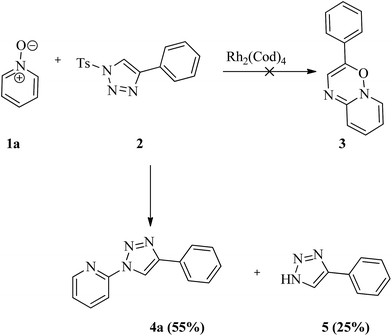
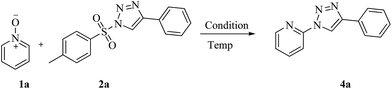
It has been planned to investigate the reactivity of tosyl substituted triazole with pyridine-N-oxide in the presence of rhodium catalyst anticipating a [3 + 3] addition reaction. It can be noticed that such additions are popular towards the construction of six membered rings.12 Tosyl azide is well known to generate active carbene in presence of Rh complex, which can undergo [3 + 3] addition reactions.13 With this idea, tosyl triazole was made to react with pyridine-N-oxide 1 in the presence metal catalyst. The selected substrates 1a–y were prepared by the oxidation of various pyridine with m-chloroperbenzoic acid under basic conditions in dichloromethane.14 The sulfonyl triazole 2 was synthesised by the reaction of 1 eq. of tosyl azide and 1.2 eq. of phenyl acetylene in the presence of cuprous oxide in water at room temperature.15 The investigation was started with the reaction between pyridine-N-oxide 1a and sulfonyl triazole 2a with the hope of getting 3-phenylpyrido[1,2-b][1,2,4]oxadiazine moiety via formal [3 + 3] cycloaddition of pyridine 1a with the sulfonyl triazole 2, but the reaction did not proceed in the expected pathway. The unexpected product 2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine, 4a was obtained in 55% yield along with a by-product 5 in 25% yield (Scheme 1). The by-product was identified as 4-phenyl-1H-1,2,3-triazole.Puzzled by this, the reaction conditions were optimized (Table 1) and it is found under no circumstances, the six membered ring is generated. It was also realized that the presence of metal catalyst or base is not necessary for the reaction to yield 4. It is found that this is the first report of the α-heteroarylation with simultaneous deoxygenative reaction without a catalyst or a base. It should be mentioned that previously stronger bases were employed for the related conversion studied. Nevertheless, this protocol is a new method of preparing triazole at the 2-position of pyridine ring. The advantages of the present method are that it is free from metal catalyst and base with reduced reaction time and high yields. The substrate scope is broadened in the present investigation (Scheme 2).
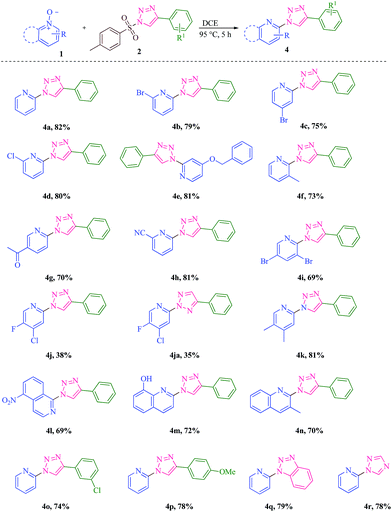 | ||
| Scheme 2 Scope of the substrates (4a–4r). Reaction conditions: 1 (1 mmol), 2 (1 mmol), DCE (2 mL), 95 °C, 5 h. Isolated yields refer to 1. | ||
| Entry | Solvent | Catalyst (mol%) | Base | t (°C) | Time (h) | Yield of 4a (%) |
|---|---|---|---|---|---|---|
| a Reagent and conditions: pyridine N-oxide 1a (1 mmol), sulfonyltriazole 2a (1 mmol), solvents (2 mL).b No reaction.c Reactions carried out without catalyst.d Isolated yield. | ||||||
| 1 | CHCl3 | Rh2(Cod)4 (20) | 90 | 1.5 | 55 | |
| 2 | DCE | Rh2(OAc)4 (20) | 90 | 7 | 43 | |
| 3 | CHCl3 | Rh2(OAc)4 (20) | 90 | 2.5 | 52 | |
| 4 | DCE | Rh2(OAc)4 (20) | rt | 18 | <2 | |
| 5 | THF | Ni(COD)2 (20) | 90 | 18 | NRb | |
| 6 | DCE | CuI (20) | 90 | 12 | 8 | |
| 7 | Toluene | —c | 110 | 6 | 68 | |
| 8 | Dioxane | — | 100 | 12 | <10 | |
| 9 | DCM | — | 50 | 12 | 48 | |
| 10 | ACN | — | 80 | 12 | 54 | |
| 11 | Xylene | — | 130 | 12 | 10 | |
| 12 | DMF | — | 140 | 20 | NRb | |
| 13 | DMSO | — | 150 | 20 | NRb | |
| 14 | CHCl3 | — | 80 | 8 | 58 | |
| 15 | THF | — | 65 | 18 | <5 | |
| 16 | DCE | — | 95 | 5 | 82d | |
| 17 | DCE | — | rt | 18 | <2 | |
| 18 | DCE | — | DIPA | 90 | 12 | <5 |
| 19 | DCE | — | DIPEA | 90 | 12 | <5 |
| 20 | DCE | — | Et3N | 90 | 12 | <2 |
| 21 | DIPA | — | 95 | 8 | 62 | |
| 22 | DIPEA | — | 95 | 8 | 58 | |
| 23 | Et3N | — | 95 | 8 | 20 | |
The reaction has led to the formation of a single product 4 in many cases. It must be mentioned that it is certain that the product 4 formed is triazole connected to the pyridine ring at 2 position, but the regiochemistry in the triazole ring has to be established unambiguously. Previously a mixture of regioisomers have been obtained while constructing these skeletons.16 The NMR features have been analyzed carefully in these cases. A NOESY spectrum of 4c has also been recorded which gives stronger contour between the triazole ring singlet and the ortho-hydrogens of the aryl substituent. All the three isomers A, B and C (Fig. 1) will show such contour. A, B and C are quite possible as the reaction is supposed to take place after the generation of free triazolyl anion, which may be resonating between different forms. However, if the regiochemistry of the starting tosyl triazole is to be taken into account, by memory effect and the stability of the triazolyl anion, the isomer A may be the favourable one. B is sterically hindered and hence its formation is not quite feasible. C could be the other regioisomer that might have been formed in low yield in some cases.
In the major/only isomer of 4 prepared in the present investigation, the triazole hydrogen appears at 9.30 ppm (DMSO-d6). This hydrogen is said to have observed at 8.88 in CDCl3 and 9.37 in DMF for the structure A.17 Hence the above compounds have been said to assume the structure where the 2-pyridyl ring is 1 position and the aryl at 4 position. This is confirmed by two dimensional NMR studies (Fig. 2 shows correct assignment based on 2D connectivities for 4c) and single crystal X-ray analysis of 4d (Fig. 3).
Interestingly in 4f, the coupling has occurred ortho to methyl in the pyridyl ring and not para to methyl (no one hydrogen singlet in the 1H NMR spectrum), though it is sterically placed. Thus the product is 3-methylpyrid-2-yl triazole and not 5-methylpyrid-2-yl triazole. In contrast, in 4g, the coupling has occurred para to acetyl and not ortho to acetyl (two singlets and two doublets in the 1H NMR spectrum apart from the singlet of triazole ring). Inductive electron withdrawing could have assisted to have more electron deficiency in the carbon alpha to methyl (4f), while mesomeric electron withdrawing would have telling effect at the para to acetyl group (4g), avoiding the steric conjunction, if entered at ortho to acetyl.
With 4h, it seems to be a mixture (81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19). Considerable amount of another regioisomer has been formed and it could not be possible to separate that from the major one. The major isomer has the triazolyl ring hydrogen at 9.4 ppm as in the case of 4a to 4g. The minor isomer has the triazolyl singlet appearing at 8.8 ppm. The structure of this minor isomer may be C, 2-pyridyl-4-phenyl-2H-1,2,3-triazole. This is confirmed with the related isomer 4ja (vide infra).
19). Considerable amount of another regioisomer has been formed and it could not be possible to separate that from the major one. The major isomer has the triazolyl ring hydrogen at 9.4 ppm as in the case of 4a to 4g. The minor isomer has the triazolyl singlet appearing at 8.8 ppm. The structure of this minor isomer may be C, 2-pyridyl-4-phenyl-2H-1,2,3-triazole. This is confirmed with the related isomer 4ja (vide infra).
In 4f and 4i, the triazolyl ring hydrogen is appearing slightly shielded at 9.1 ppm. The presence of ortho substituent would have made the triazole ring to go out plane with the pyridyl ring and the deshielding ring current effect may be reduced.
With 3-fluoro-4-chloropyridine-N-oxide, the course is different. Here two isomers have been obtained in relatively good yield (38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) labelled as 4j and 4ja. With 4j, the triazole ring hydrogen comes at 9.4 ppm as with other cases, suggesting the structure to be A. In the case of 4ja, the triazole ring hydrogen appears at 8.8 ppm suggesting the structure to be different. It is found to be C, 2-pyridyl-4-aryl-2H-1,2,3-triazole. It is possible to grow a single crystal in this case and the single crystal X-ray data confirm the structure (Fig. 4).
20) labelled as 4j and 4ja. With 4j, the triazole ring hydrogen comes at 9.4 ppm as with other cases, suggesting the structure to be A. In the case of 4ja, the triazole ring hydrogen appears at 8.8 ppm suggesting the structure to be different. It is found to be C, 2-pyridyl-4-aryl-2H-1,2,3-triazole. It is possible to grow a single crystal in this case and the single crystal X-ray data confirm the structure (Fig. 4).
With 4k, the isomer is found to be 4,5-dimethylpyrid-2-yl triazole and not 3,4-dimethylpyrid-2-yltriazole. There are three one hydrogen singlets in the 1H NMR spectrum proving that the compound is the former and not the latter with the triazole group getting bonded to pyridine at para to the methyl at 3 position and not at ortho to that methyl group. This is surprising as 2-methylpyridine-N-oxide has behaved differently giving 4f, where the triazole group is attached to the C-2 position of the pyridyl ring.
After varying the substituents in pyridine-N-oxide, the scope is extended to fused pyridine ring. With 5-nitroisoquinoline oxide, the reaction goes successfully coupling with the triazole ring in the 1-position of isoquinoilne and not at the 3-position giving 4l (four doublets and one triplet). This is not surprising as the nucleophilicity of C-1 position is more compared C-3 position, in spite of the peri interaction.17 Obviously, due to peri interaction, the triazole ring goes out of plane feeling an upfield shift for the triazolyl hydrogen, appearing at 8.8 ppm (cf.: 4f and 4i).
The reaction takes with equal ease in quinoline system as well (4m). The substrate scope has also been extended by introducing the substituents in the 4-aryl ring of triazole (4o and 4p). The starting triazole sulphonyl compounds for 4q and 4r have been prepared by reported procedures.18
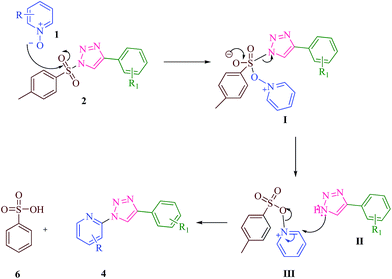
The plausible mechanism of the reaction is provided in Scheme 3. It must be admitted that the reaction is not taking place with alkyl azides.
Conclusions
We have presented a new C–N bond forming reaction via regioselective α-heteroarylation followed by deoxygenation towards the synthesis of variety of azinetriazole from simple azine N-oxides derivatives and N-tosyl-1,2,3-triazoles. The reaction is metal free and base free with lesser reaction time, high yields and a broad substrate scope. Out of the synthesised compounds, 4b, 4c, 4e–n & 4q are new.Experimental section
General consideration
The melting points reported in the work are uncorrected. Unless stated otherwise, solvents and chemicals were obtained from commercial sources and used without further purification. The 1H and 13CNMR spectra of the new compounds were measured at 400 MHz or 300 MHz and 100 MHz or 75 MHz (mentioned in respective NMR data itself) respectively using Bruker NMR instrument in DMSO-d6 or CDCl3. Chemical shifts are reported in parts per million (δ), coupling constants (J values) are reported in Hertz (Hz) relative to tetramethylsilane. Spin multiplicities are indicated by the following symbols: s (singlet), d (doublet), t (triplet), m (multiplet) dd (doublet of doublets), bs (broad singlet). Infrared spectra were recorded on a FT-IR instrument and only major peaks are reported in cm−1. Column chromatography was carried out in silica gel (60–120 mesh) using petroleum ether–ethyl acetate as eluent.General procedure for the preparation of 4
To a solution of pyridine-N-oxide 1 (1 mmol) in 1,2-dichloroethane (2 mL) was added sulfonyl triazole 2 (1 mmol). The reaction mixture was stirred magnetically and refluxed at 95 °C for the indicated time (Table 1). After completion of the reaction (monitored by TLC), the product was extracted with ethyl acetate (15 mL) and purified by column chromatography (silica gel) using ethyl acetate/petroleum ether mixture as a gradient elution to afford product (4a–r).2-(4-Phenyl-1H-1,2,3-triazol-1-yl)pyridine 4a1,6f,19
Isolated yield 0.19 g (82%); off white solid; mp 128–130 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.39 (t, J = 7.6 Hz, 1H), 7.50 (t, J = 7.2 Hz, 2H), 7.56–7.59 (m, 1H), 8.04 (d, J = 7.2 Hz, 2H), 8.12–8.19 (m, 2H), 8.63 (d, J = 7.6 Hz, 1H), 9.32 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 114.2, 118.7, 124.8, 126.1, 128.9, 129.4, 130.4, 140.6, 147.9, 149.0, 149.5; 1H NMR (300 MHz, CDCl3): δH: 7.35–7.40 (m, 2H), 7.49 (t, J = 7.2 Hz), 7.94–7.97 (m, 3H), 8.26 (d, J = 8.4 Hz, 1H), 8.53–8.54 (m, 1H), 8.82 (s, 1H); 13C NMR (75 MHz, CDCl3) δC: 113.8, 116.8, 123.5, 125.9, 128.4, 128.9, 130.3, 139.1, 148.1, 148.5, 149.3; IR (ATR KBr cell, cm−1) 1053, 1560, 3078, 3125.2-Bromo-6-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4b
Isolated yield 0.14 g (79%); off white solid; mp 180–182 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.40 (t, J = 7.2 Hz, 1H), 7.50 (t, J = 7.2 Hz, 2H), 7.83 (d, J = 7.6 Hz, 1H), 8.05–8.10 (m, 3H), 8.20 (d, J = 8 Hz, 1H), 9.33 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 113.6, 118.9, 126.1, 128.8, 128.9, 129.4, 130.2, 140.2, 143.3, 147.8, 148.8; IR (ATR KBr cell, cm−1) 550, 1280, 3042, 3145. LC-MS calcd m/z 301, found 302 [(M + 1)]+. Anal. calcd for C13H9BrN4: C, 51.85; H, 3.01; N, 18.60; found: C, 51.80; H, 2.98; N, 18.54.4-Bromo-2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4c
Isolated yield 0.13 g (75%); off white solid; mp 118–120 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.41 (t, J = 7.4 Hz, 1H), 7.51 (t, J = 7.44 Hz, 2H), 7.87 (dd, J = 1.48 Hz, 5.2 Hz, 1H), 8.04 (d, J = 7.36 Hz, 2H), 8.39 (d, J = 1.48 Hz, 1H), 8.55 (d, J = 5.32 Hz, 1H), 9.40 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 117.2, 119.0, 126.1, 127.8, 128.9, 129.4, 130.2, 135.2, 147.8, 149.6, 150.5; IR (ATR KBr cell, cm−1) 570, 1280, 3100, 3126. LC-MS calcd m/z 301, found 302 [(M + 1)]+. Anal. calcd for C13H9BrN4: C, 51.85; H, 3.01; N, 18.60; found: C, 51.80; H, 2.94; N, 18.53.2-Chloro-6-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4d6f
Isolated yield 0.16 g (80%); off white solid; mp 136–138 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.40 (t, J = 7.2 Hz, 1H), 7.49 (t, J = 7.2 Hz, 2H), 7.69 (dd, J = 1.6 Hz, 6.8 Hz, 1H), 8.06 (d, J = 8.4 Hz, 2H), 8.15–8.21 (m, 2H), 9.32 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 113.3, 119.0, 125.0, 126.1, 129.0, 129.4, 130.2, 143.8, 147.9, 148.7, 149.5; IR (ATR KBr cell, cm−1) 682, 733, 1474, 1593, 3131.4-(Benzyloxy)-2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4e
Isolated yield 0.13 g (81%); off white solid; mp 155–157 °C; 1H NMR (400 MHz, DMSO-d6): δH: 5.36 (s, 2H), 7.20–7.22 (m, 1H), 7.37–7.52 (m, 8H), 7.74 (d, J = 2 Hz, 1H), 8.03 (d, J = 7.2 Hz, 2H), 8.44 (d, J = 5.6 Hz, 1H), 9.33 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 70.5, 100.3, 112.3, 119.0, 126.0, 128.5, 128.8, 128.9, 129.1, 129.4, 130.4, 136.1, 147.6, 150.5, 150.6, 167.2; IR (ATR KBr cell, cm−1) 1580, 3080, 3125; LC-MS calcd m/z 328, found 329 [(M + 1)]+. Anal. calcd for C20H16N4O: C, 73.15; H, 4.91; N, 17.06; found: C, 73.11; H, 4.84; N, 17.03.3-Methyl-2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4f
Isolated yield 0.16 g (73%); off white solid; mp 148–150 °C; 1H NMR (400 MHz, DMSO-d6): δH: 2.44 (s, 3H), 7.40 (t, J = 3.8 Hz, 1H), 7.51 (t, J = 7.6 Hz, 2H), 7.56–7.59 (m, 1H), 8.00–8.05 (m, 3H), 8.51–8.52 (m, 1H), 9.14 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 18.5, 122.1, 125.5, 125.9, 128.2, 128.7, 129.5, 130.7, 142.3, 146.6, 147.0, 148.2; IR (ATR KBr cell, cm−1) 1476, 1537, 2998, 3120; LC-MS calcd m/z 236, found 237 [(M + 1)]+. Anal. calcd for C14H12N4: C, 71.17; H, 5.12; N, 23.71; found: C, 71.12; H, 5.09; N, 23.64.1-(6-(4-Phenyl-1H-1,2,3-triazol-1-yl)pyridin-3-yl)ethanone 4g
Isolated yield 0.13 g (70%); off white solid; mp 198–200 °C; 1H NMR (400 MHz, DMSO-d6): δH: 2.69 (s, 3H), 7.51–7.56 (m, 3H), 8.05–8.23 (m, 3H), 8.58–8.81 (m, 2H), 9.16 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 27.5, 114.0, 126.8, 129.4, 129.7, 130.1, 131.8, 136.2, 139.8, 150.2, 150.5, 152.6, 196.7; IR (ATR KBr cell, cm−1) 1402, 1570, 1645, 2384, 3042; LC-MS calcd m/z 264, found 265[(M + 1)]+. Anal. calcd for C15H12N4O: C, 68.17; H, 4.58; N, 21.20; found: C, 68.11; H, 4.55; N, 21.15.6-(4-Phenyl-1H-1,2,3-triazol-1-yl)picolinonitrile 4h
Isolated yield 0.17 g (81%); off white solid; mp 176–178 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.39 (t, J = 7.2 Hz, 1H), 7.46–7.53 (m, 2H), 8.03–8.06 (m, 2H), 8.19 (d, J = 7.6 Hz, 1H), 8.35–8.41 (m, 1H), 8.49 (d, J = 8.4 Hz, 1H), 9.43 (s, 1H); 13C NMR (75 MHz, DMSO-d6) δC: 116.5, 118.2, 125.9, 126.5, 128.6, 129.0, 129.9, 131.6, 135.4, 141.9, 148.0, 149.5; IR (ATR KBr cell, cm−1) 1570, 2928, 3042; LC-MS calcd m/z 247, found 248 [(M + 1)]+. Anal. calcd for C14H9N5: C, 68.01; H, 3.67; N, 28.32; found: C, 67.99; H, 3.60; N, 28.27.3,5-Dibromo-2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4i
Isolated yield 0.10 g (69%); off white solid; mp 128–130 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.41 (t, J = 7.6 Hz, 1H), 7.51 (t, J = 7.2 Hz, 2H), 7.96 (d, J = 7.2 Hz, 2H), 8.86–8.89 (m, 2H), 9.12 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 116.7, 122.3, 122.9, 126.0, 128.9, 129.5, 130.3, 146.1, 146.6, 146.9, 149.5; IR (ATR KBr cell, cm−1) 557, 1462, 1552, 1949, 3129. LC-MS calcd m/z 380, found 381 [(M + 1)]+. Anal. calcd for C13H8Br2N4: C, 41.09; H, 2.12; N, 14.74; found: C, 41.02; H, 2.09; N, 14.70.4-Chloro-5-fluoro-2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4j
Isolated yield 0.07 g (38%); off white solid; mp 158–160 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.40 (t, J = 7.2 Hz, 1H), 7.50 (t, J = 7.6 Hz, 2H), 8.02 (d, J = 7.6 Hz, 2H), 8.44 (d, J = 5.2 Hz, 1H), 8.83 (s, 1H), 9.35 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 116.3, 126.7, 129.1, 129.7, 131.1, 132.5, 135.6, 137.9, 147.2, 150.1, 154.5; IR (ATR KBr cell, cm−1) 1128, 1570, 3071; LC-MS calcd m/z 274, found 275 [(M + 1)]+. Anal. calcd for C13H8ClFN4: C, 56.84; H, 2.94; N, 20.40; found: C, 56.78; H, 2.91; N, 20.35.4-Chloro-5-fluoro-2-(4-phenyl-2H-1,2,3-triazol-2-yl)pyridine 4ja
Isolated yield 0.03 g (20%); off white solid; mp 170–172 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.47 (t, J = 7.2 Hz, 1H), 7.54 (t, J = 7.2 Hz, 2H), 8.01 (d, J = 7.6 Hz, 2H), 8.34 (d, J = 5.2 Hz, 1H), 8.73 (s, 1H), 8.76 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 116.4, 119.2, 126.1, 129.0, 129.5, 130.2, 133.0, 138.2, 145.6, 147.2, 154.8; IR (ATR KBr cell, cm−1) 700, 1135, 1499, 3012; LC-MS calcd m/z 274, found 275 [(M + 1)]+. Anal. calcd for C13H8ClFN4: C, 56.84; H, 2.94; N, 20.40; found: C, 56.80; H, 2.92; N, 20.36.4,5-Dimethyl-2-(4-phenyl-1H-1,2,3-triazol-1-yl)pyridine 4k
Isolated yield 0.16 g (81%); off white solid; mp 148–150 °C; 1H NMR (400 MHz, DMSO-d6): δH: 2.31 (s, 3H), 2.41 (s, 3H), 7.39 (t, J = 7.2 Hz, 1H), 7.49 (d, J = 7.2 Hz, 2H), 7.99 (s, 1H), 8.03 (d, J = 7.2 Hz, 2H), 8.35 (s, 1H), 9.32 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 16.1, 19.6, 114.4, 118.6, 126.0, 128.8, 129.4, 130.6, 133.7, 147.4, 147.5, 148.5, 150.4; IR (ATR KBr cell, cm−1) 1041, 1466, 1601, 3173; LC-MS calcd m/z 250, found 251 [(M + 1)]+. Anal. calcd for C15H14N4: C, 71.98; H, 5.64; N, 22.38; found: C, 71.93; H, 5.60; N, 22.31.5-Nitro-1-(4-phenyl-1H-1,2,3-triazol-1-yl)isoquinoline 4l
Isolated yield 0.12 g (69%); off white solid; mp 188–190 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.49 (t, J = 7.2 Hz, 1H), 7.56 (t, J = 7.2 Hz, 2H), 7.99 (d, J = 8 Hz, 1H), 8.04 (t, J = 8.4 Hz, 2H), 8.52 (d, J = 7.6 Hz, 1H), 8.69 (d, J = 8.4 Hz, 1H), 8.75 (d, J = 6.8 Hz, 1H), 8.79 (d, J = 6 Hz, 1H), 8.87 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 118.0, 122.7, 126.7, 128.6, 129.5, 129.6, 129.7, 129.9, 130.5, 132.7, 135.2, 144.4, 145.4, 149.7, 149.9; IR (ATR KBr cell, cm−1) 1520, 1625, 3106. LC-MS calcd m/z 317, found 318 [(M + 1)]+. Anal. calcd for C17H11N5O2: C, 64.35; H, 3.49; N, 22.07; found: C, 64.31; H, 3.41; N, 22.01.2-(4-Phenyl-1H-1,2,3-triazol-1-yl)quinolin-8-ol 4m
Isolated yield 0.13 g (72%); off white solid; mp 156–158 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.49 (t, J = 7.2 Hz, 1H), 7.56 (t, J = 7.2 Hz, 2H), 7.99 (d, J = 8 Hz, 2H), 8.04 (t, J = 8.4 Hz, 2H), 8.52 (d, J = 7.6 Hz, 1H), 8.69 (d, J = 8.4 Hz, 1H), 8.75 (d, J = 6.8 Hz, 1H), 8.79 (d, J = 6 Hz, 1H), 8.87 (s, 1H); 13C NMR (75 MHz, DMSO-d6) δC: 112.6, 113.3, 118.4, 119.3, 122.8, 123.7, 125.9, 129.0, 129.6, 130.5, 136.5, 144.0, 146.1, 147.7, 153.5; IR (ATR KBr cell, cm−1) 1598, 3045, 3200. LC-MS calcd m/z 288, found 289 [(M + 1)]+. Anal. calcd for C17H12N4O: C, 70.82; H, 4.20; N, 19.43; found: C, 70.79; H, 4.14; N, 19.40.3-Methyl-2-(4-phenyl-1H-1,2,3-triazol-1-yl)quinoline 4n
Isolated yield 0.13 g (70%); off white solid; mp 148–150 °C; 1H NMR (400 MHz, DMSO-d6): δH: 2.58 (s, 3H), 7.41 (t, J = 7.2 Hz, 1H), 7.52 (t, J = 7.2 Hz, 2H), 7.73 (t, J = 8.4 Hz, 1H), 7.85 (t, J = 8 Hz, 1H), 8.06 (t, J = 7.6 Hz, 4H), 8.58 (s, 1H), 9.27 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 19.2, 122.3, 126.0, 126.1, 127.8, 128.4, 128.5, 128.7, 128.8, 129.5, 130.6, 130.8, 141.2, 145.0, 146.6, 148.2; IR (ATR KBr cell, cm−1) 1496, 1599, 2923. LC-MS calcd m/z 286, found 287 [(M + 1)]+. Anal. calcd for C18H14N4: C, 75.50; H, 4.93; N, 19.57; found: C, 75.46; H, 4.86; N, 19.53.2-(4-(3-Chlorophenyl)-1H-1,2,3-triazol-1-yl)pyridine 4o6f
Isolated yield 0.20 g (74%); off white solid; mp 138–140 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.44 (d, J = 8 Hz, 1H), 7.53 (t, J = 7.6 Hz, 1H), 7.57–7.60 (m, 1H), 8.02 (d, J = 7.6 Hz, 1H), 8.11–8.18 (m, 2H), 8.64 (d, J = 4.4 Hz, 1H), 9.50 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 114.3, 119.6, 124.6, 125.0, 125.7, 128.6, 131.4, 132.6, 134.3, 140.6, 146.3, 148.9, 149.4; IR (ATR KBr cell, cm−1) 774, 1400, 1590, 3108.2-(4-(4-Methoxyphenyl)-1H-1,2,3-triazol-1-yl)pyridine 4p6f
Isolated yield 0.21 g (78%); off white solid; mp 110–112 °C; 1H NMR (400 MHz, DMSO-d6): δH: 3.82 (s, 3H), 7.06 (t, J = 8.8 Hz, 2H), 7.56–7.59 (m, 1H), 7.97 (t, J = 8.4 Hz, 2H), 8.12–8.19 (m, 2H), 8.64 (t, J = 4.8 Hz, 1H), 9.27 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 55.7, 114.2, 114.8, 117.7, 123.0, 124.7, 127.5, 140.6, 147.6, 149.1, 149.4, 159.9; IR (ATR KBr cell, cm−1) 1437, 1476, 1645, 3085.1-(Pyridin-2-yl)-1H-benzo[d][1,2,3]triazole 4q
Isolated yield 0.16 g (79%); off white solid; mp 108–110 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.53–7.58 (m, 2H), 7.70–7.74 (m, 1H), 8.17 (td, J = 1.6 Hz, 7.2 Hz, 1H), 8.21 (d, J = 8 Hz, 1H), 8.26 (d, J = 8.4 Hz, 1H), 8.58 (d, J = 7.6 Hz, 1H), 8.70–8.72 (m, 1H); 13C NMR (100 MHz, DMSO-d6) δC: 114.8, 114.9, 120.1, 123.6, 125.8, 129.7, 131.3, 140.4, 146.5, 149.2, 151.2; IR (ATR KBr cell, cm−1) 1579, 2380, 3141. LC-MS calcd m/z 196, found 197 [(M + 1)]+. Anal. calcd for C11H8N4: C, 67.34; H, 4.11; N, 28.55; found: C, 67.29; H, 4.08; N, 28.51.2-(1H-1,2,4-Triazol-1-yl)pyridine 4r16
Isolated yield 0.12 g (78%); off white solid; mp 84–86 °C; 1H NMR (400 MHz, DMSO-d6): δH: 7.49 (t, J = 5.2 Hz, 1H), 7.87 (d, J = 8 Hz, 1H), 8.09 (t, J = 7.2 Hz, 1H), 8.29 (s, 1H), 8.54 (d, J = 4.4 Hz, 1H), 8.36 (s, 1H); 13C NMR (75 MHz, DMSO-d6) δC: 113.3, 124.1, 140.5, 142.4, 149.1, 149.3, 153.4; IR (ATR KBr cell, cm−1) 1448, 1556, 3121.Acknowledgements
M. B. H. is grateful to Advinus Therapeutics Limited, Bangalore for support. S. M. thanks CSIR, New Delhi for the financial assistance under a major research project.Notes and references
- B. Chattopadhyay, C. I. R. Vera, S. Chuprakov and V. Gevorgyan, Org. Lett., 2010, 12, 2166 CrossRef CAS PubMed.
- T. Bretschneider, E.-M. Franken, U. Goergens, M. Fuesslein, A. Hense, J. Kluth, H.-G. Schwarz, A. Koehler, O. Malsam and A. Voerste, WO 2010006713 A2 20100121, PCT Int. Appl., 2010.
- V. Garzya and S. P. Watson, WO 2009115486 A1 20090924, PCT Int. Appl., 2009.
- A. S. Kiselyov, M. Semenova and V. V. Semenov, Bioorg. Med. Chem. Lett., 2009, 19, 1344 CrossRef CAS PubMed.
- (a) M. F. Gordeev, Z. Yuan and J. Liu, WO 2008108988 A1 20080912, PCT Int. Appl., 2008; (b) I. L. Knox and R. B. Rogers, US Pat., 4, 775,762, 1988; (c) B. Japelj, S. Recnik, P. Cebasek, B. Stanovnik and J. Svete, J. Heterocycl. Chem., 2005, 42, 1167 CrossRef CAS; (d) S. Ito, A. Satoh, Y. Nagatomi, Y. Hirata, G. Suzuki, T. Kimura, A. Satow, S. Maehara, H. Hikichi, M. Hata, H. Kawamoto and H. Ohta, Bioorg. Med. Chem., 2008, 16, 9817 CrossRef CAS PubMed; (e) J. Roppe, N. D. Smith, D. H. Huang, L. Tehrani, B. W. Wang, J. Anderson, J. Brodkin, J. Chung, X. H. Jiang, C. King, B. Munoz, M. A. Varney, P. Prasit and N. D. P. Cosford, J. Med. Chem., 2004, 47, 4645 CrossRef CAS PubMed; (f) A. R. Ellanki, A. Islam, V. S. Rama, R. P. Pulipati, D. Rambabu, G. R. Krishna, C. M. Reddy, K. Mukkanti, G. R. Vanaja, A. M. Kalle, K. S. Kumar and M. Pal, Bioorg. Med. Chem. Lett., 2012, 22, 3455 CrossRef CAS PubMed; (g) S. V. Chapyshev and A. V. Chernyak, Synthesis, 2012, 3158 CrossRef CAS; (h) T. Merckx, P. Verwilst and W. Dehaen, Tetrahedron Lett., 2013, 54, 4237 CrossRef CAS; (i) Y. Okumura, Y. Maya, Y. Shoyama and T. Onishi, Patent WO2012176587 A1, 2012; (j) F. Li, Y. J. Park, J.-M. Hah and J.-S. Ryu, Bioorg. Med. Chem. Lett., 2013, 23, 1083 CrossRef CAS PubMed; (k) C. HirthDietrich, P. Sandner, J.-P. Stasch, M. Hahn and M. Follmann, Eur. Pat. Appl., 2013, 2594270 Search PubMed; (l) F. Li, Y. J. Park, J.-M. Hah and J.-S. Ryu, Bioorg. Med. Chem. Lett., 2013, 23, 1083 CrossRef CAS PubMed.
- (a) Y. J. Li, J. C. Huffman and A. H. Flood, Chem. Commun., 2007, 2692 RSC; (b) S. J. Gu, H. Xu, N. Zhang and W. Z. Chen, Chem.–Asian J., 2010, 5, 1677 CrossRef CAS PubMed; (c) I. Stengel, A. Mishra, N. Pootrakulchote, S.-J. Moon, S. M. Zakeeruddin, M. Graetzel and P. Baeuerle, J. Mater. Chem., 2011, 21, 3726 RSC; (d) A. Bolje and J. Kosmrlj, Org. Lett., 2013, 15, 5084 CrossRef CAS PubMed; (e) J. Sudarat, K. Teerachanan, P. Thongkam, S. Kiatisevi, T. Khamnaen, P. Phiriyawirut, S. Charoenchaidet, T. Sooksimuang, P. Kongsaeree and P. Sangtrirutnugul, J. Organomet. Chem., 2014, 750, 35 CrossRef; (f) R. Sun, H. Wang, J. Hu, J. Zhao and H. Zhang, Org. Biomol. Chem., 2014, 12, 5954 RSC . and references cited therein.
- (a) D. Alberico, M. E. Scott and M. Lautens, Chem. Rev., 2007, 107, 174 CrossRef CAS PubMed; (b) L.-C. Campeau, D. R. Stuart and K. Fagnou, Aldrichimica Acta, 2007, 40, 35 CAS; (c) D. R. Stuart and K. Fagnou, Science, 2007, 316, 1172 CrossRef CAS PubMed; (d) F. Kakiuchi and N. Chatani, Adv. Synth. Catal., 2003, 354, 1077 CrossRef; (e) J. A. Lablinger and J. E. Bercaw, Nature, 2002, 417, 507 CrossRef PubMed; (f) V. Ritleng, C. Sirlin and M. Pfeffer, Chem. Rev., 2002, 102, 1731 CrossRef CAS PubMed; (g) M. Miura and M. Nomura, Top. Curr. Chem., 2002, 219, 211 CrossRef CAS; (h) Y. Fujiwara and J. Chengguo, Pure Appl. Chem., 2001, 73, 319 CrossRef CAS; (i) G. Dyker, Angew. Chem., Int. Ed., 1999, 38, 1698 CrossRef; (j) A. Shilov and G. Shulpin, Chem. Rev., 1997, 97, 2879 CrossRef CAS PubMed.
- (a) R. F. Jordan and D. F. Taylor, J. Am. Chem. Soc., 1989, 111, 778 CrossRef CAS; (b) E. J. Moore, W. Pretzer, T. J. O'Connell, J. Harris, L. LaBounty, L. Chou and S. S. Grimmer, J. Am. Chem. Soc., 1992, 114, 5888 CrossRef CAS; (c) T. Fukuyama, N. Chatani, J. Tatsumi, F. Kakiuchi and S. Murai, J. Am. Chem. Soc., 1998, 120, 11522 CrossRef CAS; (d) M. Murakami and S. Hori, J. Am. Chem. Soc., 2003, 125, 4720 CrossRef CAS PubMed.
- (a) L. Lutz Ackermann and S. Fenner, Chem. Commun., 2011, 47, 430 RSC and references cited therein; (b) F. Gosselin, S.-J. Savage, N. Blaquiere and S. T. Staben, Org. Lett., 2014, 14, 862 CrossRef PubMed.
- K. S. Kanyiva, Y. Nakao and T. Hiyama, Angew. Chem., Int. Ed., 2007, 46, 8872 CrossRef CAS PubMed.
- For direct arylation of azine N-oxides, see: (a) L.-C. Campeau, S. Rousseaux and K. Fagnou, J. Am. Chem. Soc., 2005, 127, 18020 CrossRef CAS PubMed; (b) L.-C. Campeau, D. R. Stuart, J.-P. Leclerc, M. Bertrand-Laperle, E. Villemure, H.-Y. Sun, S. Lasserre, N. Guimond, M. Lecavallier and K. Fagnou, J. Am. Chem. Soc., 2009, 131, 3291 CrossRef CAS PubMed; (c) L.-C. Campeau, D. J. Schipper and K. Fagnou, J. Am. Chem. Soc., 2008, 130, 3266 CrossRef CAS PubMed; (d) D. J. Schipper, L.-C. Campeau and K. Fagnou, Tetrahedron, 2009, 65, 3155 CrossRef CAS; For direct arylation of N-iminopyridiniumylides, see: (e) A. Larivee, J. J. Mousseau and A. B. Charette, J. Am. Chem. Soc., 2008, 130, 52 CrossRef CAS PubMed; (f) D. Alberico, M. E. Scott and M. Lautens, Chem. Rev., 2007, 107, 174 CrossRef CAS PubMed.
- (a) A. Padwa and P. D. Stull, Tetrahedron Lett., 1987, 28, 5407 CrossRef CAS; (b) R. M. Savizky and D. J. Austin, Rhodium(II)-Catalyzed 1,3-Dipolar Cycloaddition Reactions, in Modern Rhodium-Catalyzed Organic Reactions, ed. P. A. Evans, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG, 2005, DOI:10.1002/3527604693.ch19; (c) Q.-Q. Cheng, Y. Qian, P. Y. Zavalij and M. P. Doyle, Org. Lett., 2015, 17, 3568 CrossRef CAS PubMed.
- see recent review: H. M. L. Davies and J. S. Alford, Chem. Soc. Rev., 2014, 43, 5151 RSC.
- H. P. Kokatla, P. F. Thomson, S. Bae, V. R. Doddi and M. K. Lashman, J. Org. Chem., 2011, 76, 7842 CrossRef CAS PubMed.
- K. Wang, X. Bi, S. Xing, P. Liao, Z. Fang, X. Meng, Q. Zhang, Q. Liu and Y. Ji, Green Chem., 2011, 13, 562 RSC.
- J. M. Keith, J. Org. Chem., 2010, 75, 2722 CrossRef CAS PubMed.
- V. Balasubramanian, Chem. Rev., 1966, 66, 567 CrossRef.
- (a) A. R. Katritzky, T. Kurtz, S. Zhang, M. Voronkov and P. J. Steel, Heterocycles, 2001, 55, 1703 CrossRef CAS; (b) H. Law, I. Baussanne, J. M. G. Fernandez and J. Defaye, Carbohydr. Res., 2003, 338, 451 CrossRef CAS PubMed.
- W. K. C. Lo, G. S. Huff, J. R. Cubanski, A. D. W. Kennedy, C. J. McAdam, K. C. Gordon and D. Crowley, Inorg. Chem., 2015, 54, 1572 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1475885 and 1475932. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra10452e |
| This journal is © The Royal Society of Chemistry 2016 |