Nickel-oxide multiwall carbon-nanotube/reduced graphene oxide a ternary composite for enzyme-free glucose sensing†
Abstract
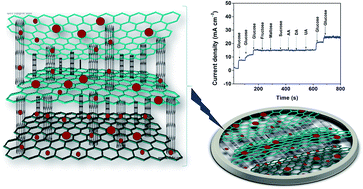
We report a solvent-free method of preparation for a NiO–carbon nanotube/graphene ternary composite using nickel formate as a green precursor via a thermal decomposition method. In this ternary composite, NiO with an average particle size of 7 nm is regularly decorated on the surfaces of conductive carbon matrix networks such as MWCNTs and reduced graphene oxide (rGO). Here rGO serves as an ideal support for the uniform distribution of NiO nanoparticles and also functions as an efficient transducer material, whereas, MWCNTs act as a spacer between rGO, which enhances the electrical conductivity and accessibility of the active reaction sites for direct glucose oxidation. The electrochemical performances were evaluated by cyclic voltammetry and amperometric techniques. Under the optimal conditions, the 20 wt% NiO–MWCNT/rGO/GCE exhibits a sensitivity of 4223.3 μA cm−2 mM−1 and a detection limit of 0.92 μM over a linear glucose concentration range up to 19 mM. Furthermore, the constructed sensor is effectively employed to detect glucose in real human blood serum samples with adequate results. The modified 20 wt% NiO–MWCNT/rGO/GCE also shows a high sensitivity, greater selectivity, excellent reproducibility and long-term stability.

 Please wait while we load your content...
Please wait while we load your content...