Three iodocuprate hybrids symmetrically modulated by positional isomers and the chiral conformation of N-benzyl-methylpyridinium†
Pengfei Hao,
Yanrong Qiao,
Tanlai Yu,
Junju Shen,
Fang Liu and
Yunlong Fu*
School of Chemistry & Material Science, Shanxi Normal University, Linfen 041004, P. R. China. E-mail: yunlongfu@sxnu.edu.cn; Fax: +86 357 2053716; Tel: +86 357 2053716
First published on 17th May 2016
Abstract
Three iodocuprates, [N-Bz-2-MePy]2[Cu5I7] (1), [N-Bz-3-MePy]2[Cu5I7] (2), and [N-Bz-4-MePy][Cu4I5] (3) (N-Bz-2-MePy+ = N-benzyl-2-methylpyridinium, N-Bz-3-MePy+ = N-benzyl-3-methylpyridinium, N-Bz-4-MePy+ = N-benzyl-4-methylpyridinium) were synthesized and structurally characterized, using benzylated methylpyridinium as a potential conformationally chiral structure-directing agent (SDA). The N-Bz-2-MePy+ cations in 1 are featured in coexisting chiral and achiral conformations, forming a left-handed helical supramolecular chain, while N-Bz-3-MePy+ cations in 2 take on two pairs of mirror-image conformations, aggregating into a racemic supramolecular (10,3)-b net. Comparatively, N-Bz-4-MePy+ cations in 3 possess a couple of mirror-image conformations, forming a supramolecular (6,3) net through π–π and C–H⋯π interactions. Accordingly, 1 and 2 crystallize as conglomerate and racemate packing states, respectively, and 3 possesses no chiral features, which shows a regular directing effect of the positional variation of a methyl group on the construction of iodocuprate frameworks. Furthermore, compounds 2 and 3 exhibit interesting low temperature reversible thermochromism.
Introduction
The construction of noncentrosymmetric (NCS) materials is one of the frontier fields of current research owing to their far-ranging applications, such as second-order nonlinear optics (SHG),1 pyroelectricity,2 piezoelectric,3 ferroelectricity,4 and enantioselective sorption, separation, and catalysis.5 Especially, structural directing construction has revealed obvious advantages for noncentrosymmetric organic–inorganic hybrids.6 To guide chiral or acentric inorganic–organic hybrid frameworks, three strategies are usually considered. The first one is based on the occasionally spontaneous chiral resolution of inorganic moieties directed by achiral SDAs, such as noncentrosymmetric (NH4)2Te2WO8 and chiral [CN3H6][Sn4P3O12] charge balanced by the asymmetric units of TeO4, SnO3 and WO6,7 [Ti3P6O27]·5[NH3CH2CH2NH3]·2[H3O] and chiral (NH4)2[Zn(SO4)2] by low symmetrical linkages of a primary building unit (PBU).8 The second one is the occasionally spontaneous chiral resolution of racemic SDAs, such as tri-chelated metal complexes9 and racemic organic amines,10 and consequential directing effect. The last one is based on the direct usage of enantiopure organic SDAs, for example, chiral metallophosphates directed by chiral organic amines,11 iodates, borates and selenites resulting from chiral or acentric metal complexes.12 Although the enantiopure organic SDA route has made significant progress, the high economic expense and relative scarcity of appropriate reagents limits the application. Meanwhile, compared with the first strategy which is largely dependent on “luck”, the second one is likely to be a reasonable choice due to its abundance of available compounds. It is noteworthy that except for the common tri-chelated metal complexes9 and racemic organic amines,10 organic conformational chirality as a common phenomenon, is rarely used in directing the construction of noncentrosymmetric organic–inorganic hybrids.13Moreover, a supramolecular template based on versatile weak interactions exhibits flexible structural matching ability for porous materials14,15 and inorganic–organic hybrids materials,16–18 due to its flexible modulation on spatial and electronic configuration. Meanwhile, halometallates possess structural diversity, hyperpolarizable abilities and sensitivity to the subtle variants of SDAs. Therefore, a combination of supramolecular templates and halometallates should be an excellent system to further understand the construction of chiral or acentric structures with multifunctional properties.
As an extension of our recently reported two NCS iodometallate hybrids13a directed by conformationally chiral benzylpyridinium, further investigations were conducted using positional isomeric methyl-pyridine via in situ benzylation to inspect their structural directing effects on the construction of NCS iodocuprate hybrids. Herein, we report three iodocuprate hybrids, [N-Bz-2-MePy]2[Cu5I7] (1), [N-Bz-3-MePy]2[Cu5I7] (2), and [N-Bz-4-MePy][Cu4I5] (3) directed by conformationally chiral N-Bz-MePy+ cations and their supramolecular aggregates.
Experimental
Materials and methods
All chemicals and solvents were commercially available and used without further purification. X-ray powder diffraction (XRPD) patterns were collected on a Rigaku Ultima IV–185 diffractometer. Elemental analyses (C, H, N) were carried out on a Perkin-Elmer 240 instrument. Inductively coupled plasma (ICP) analyses were performed on a Perkin-Elmer 2400 Elemental Analyzer. FT-IR spectra were performed from KBr pellets in the range of 4000–400 cm−1 on a Nicolet 5DX spectrometer. UV-vis diffuse reflectance spectra were recorded on a Varian Cary 5000 UV-vis spectrophotometer at room temperature. Thermogravimetric analysis-differential scanning calorimetry-mass spectral (TG-DSC-MS) data were collected in an argon atmosphere (flow rate 100 cm3 min−1, thermal ramp 10 °C min−1, temperature range 25–710 °C) using a Setaram SETSYS TGA coupled with a hidden HPR20 QIC R&D mass spectrometer. Photoluminescence analysis was performed on an Edinburgh FLS920 luminescence spectrometer.Preparation of [N-Bz-2-MePy]2[Cu5I7] (1)
A mixture of 2-methylpyridine (99.0 wt%, 0.061 g, 0.65 mmol), CuI (99 wt%, 0.570 g, 3.0 mmol), acetonitrile (99 wt%, 4.0 mL) was heated with concentrated HI (0.37 mL, 1.95 mmol, 45%), and benzyl alcohol (98 wt%, 1.0 mL) in a 15 mL Teflon-lined stainless steel vessel for 2 days at 110 °C. The mother liquor was stored at room temperature for 1 day to afford 1 as pale-yellow rod crystals. The yield was 40% based on Cu. Chemical analysis indicates the content of Cu and I as 20.14 and 56.38 wt% (calculated: 20.18 and 56.42 wt%), giving the Cu/I molar ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7. Anal. calcd for C26H28N2Cu5I7: C, 19.83; H, 1.79; N, 1.78%. Found: C, 19.88; H, 1.78; N, 1.75%. IR (KBr, cm−1): ν 3046(w), 2928(w), 2856(w), 1636(s), 1516(w), 1440(m), 1295(w), 1164(w), 1031(w), 736(m), 690(m), 546(w).
7. Anal. calcd for C26H28N2Cu5I7: C, 19.83; H, 1.79; N, 1.78%. Found: C, 19.88; H, 1.78; N, 1.75%. IR (KBr, cm−1): ν 3046(w), 2928(w), 2856(w), 1636(s), 1516(w), 1440(m), 1295(w), 1164(w), 1031(w), 736(m), 690(m), 546(w).
Preparation of [N-Bz-3-MePy]2[Cu5I7] (2)
A mixture of 3-methylpyridine (98.0 wt%, 0.061 g, 0.65 mmol), CuI (99 wt%, 0.380 g, 2.0 mmol), acetone (99 wt%, 3.0 mL) was heated with concentrated HI (0.37 mL, 1.95 mmol, 45%), and benzyl alcohol (98 wt%, 4.0 mL) in a 15 mL Teflon-lined stainless steel vessel for 3 days at 110 °C. The mother liquor was stored at room temperature for 1 day to afford 2 as orange plate crystals. The yield was 32% based on Cu. Chemical analysis indicates the content of Cu and I as 20.23 and 56.36 wt% (calculated: 20.18 and 56.42 wt%), giving the Cu/I molar ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7. Anal. calcd for C26H28N2Cu5I7: C, 19.83; H, 1.79; N, 1.78%. Found: C, 19.80; H, 1.75; N, 1.76%. IR (KBr, cm−1): ν 3050(w), 2926(w), 2851(w), 1624(s), 1505(w), 1453(m), 1381(w), 1151(w), 1042(w), 737(m), 690(m), 571(w).
7. Anal. calcd for C26H28N2Cu5I7: C, 19.83; H, 1.79; N, 1.78%. Found: C, 19.80; H, 1.75; N, 1.76%. IR (KBr, cm−1): ν 3050(w), 2926(w), 2851(w), 1624(s), 1505(w), 1453(m), 1381(w), 1151(w), 1042(w), 737(m), 690(m), 571(w).
Preparation of [N-Bz-4-MePy][Cu4I5] (3)
The procedure was analogous to the synthesis of compound 2, except that 4-methylpyridine (98.5 wt%, 0.061 g, 0.65 mmol) was replaced by 3-methylpyridine and 3.0 mmol of CuI was used. Orange block crystals of 3 were achieved in 49% yield (based on Cu). Chemical analysis indicates the content of Cu and I as 23.73 and 59.08 wt% (calculated: 23.69 and 59.14 wt%), giving the Cu/I molar ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5. Anal. calcd for C13H14NCu4I5: C, 14.55; H, 1.32; N, 1.31%. Found: C, 14.57; H, 1.30; N, 1.33%. IR (KBr, cm−1): ν 3043(w), 2930(w), 2854(w), 1637(s), 1512(w), 1466(m), 1295(w), 1151(m), 1038(w), 803(m), 713(m), 598(w).
5. Anal. calcd for C13H14NCu4I5: C, 14.55; H, 1.32; N, 1.31%. Found: C, 14.57; H, 1.30; N, 1.33%. IR (KBr, cm−1): ν 3043(w), 2930(w), 2854(w), 1637(s), 1512(w), 1466(m), 1295(w), 1151(m), 1038(w), 803(m), 713(m), 598(w).
X-Ray crystallography
Single crystals of 1, 2, and 3 were mounted with glue on a glass fiber and crystal data were recorded at 293 K on the Oxford Gemini diffractometer using graphite monochromated Mo-Kα (λ = 0.71073 Å). An empirical absorption correction with spherical harmonics was carried out by the SCALE3 ABSPACK scaling algorithm.19 All three structures were solved by direct method and refined by full-matrix least-squares techniques on F2 performed with the SHELXTL-97 program.20 All non-hydrogen atoms were treated anisotropically. A summary of the crystal data is described in detail in Table S1.† Selected bond lengths and angles are listed in Table S2 of the ESI.†Results and discussion
IR spectral aspects
The IR spectra of 1, 2 and 3 (Fig. S1†) show bands at around 2928 cm−1 and 2856 cm−1, which are assigned to the C–H stretching vibration of saturated hydrocarbons (methyl and methylene groups), while the bands at around 3046 cm−1 are ascribed to the C–H stretching vibration of the unsaturated hydrocarbon (pyridyl and benzene group). One set of peaks at around 1636, 1516, 1440, 1295, 1164 and 1031 cm−1 further confirm the existence of a pyridyl or benzene group.21Description of structures
Positional isomerism influence on the chiral conformations, cationic supramolecular aggregates and packing structures of cuprous hybrids
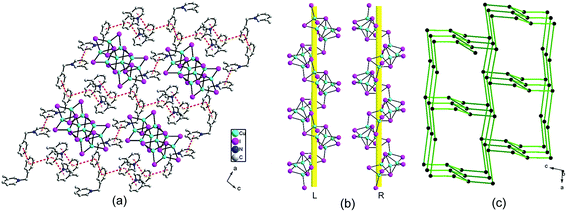
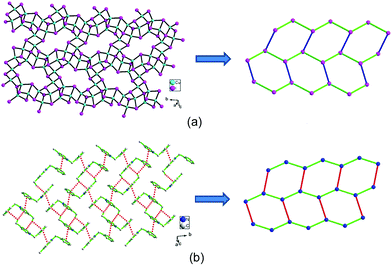
The hierarchical structure comparison of the compounds 1–3 is shown in Table 1. For 1, coexistence of the P-1 chiral and achiral conformations of N-Bz-2-MePy+, consequential 1-D supramolecular left-handed helical chain and corresponding left-handed helical [Cu5I7]n anionic chain imply spontaneous resolution and the asymmetric transfer from N-Bz-2-MePy+ cations to the iodocuprate framework. For 2, comparatively, generation of the two couples of mirror-image N-Bz-3-MePy+, consequential racemic supramolecular (10,3)-b net and left- and right-handed helical [Cu5I7]n anionic chains exhibit local chiral feature and an overall racemic packing arrangement. Interestingly, the N-Bz-4-MePy+ cations in 3 take on a couple of mirror-image conformation, while the 2-D supramolecular layer and anionic [Cu4I5]n layer with (6,3) topology show chiral disappearance and structural correlation at the polymeric level. Therefore, the positional variation of methyl groups in N-Bz-MePy+ exert remarkable regular effects on the conformational manifestation of N-Bz-MePy+ at the molecular level (Fig. S9–S11†), consequential supramolecular aggregating forms and construction of iodocuprate frameworks, exhibiting interesting hierarchical asymmetric transfer in the iodocuprate hybrids and flexible charge matching of iodocuprates, which are attributed to the versatile weak interactions. Additionally, a similar directing effect of the basic organic motif of N-Bz-MePy+ can be witnessed by the occurrence of incomplete cubane-like [Cu3I4] units in all three iodocuprate hybrids.| 1 | 2 | 3 | |
|---|---|---|---|
| Organic SDAs | N-Bz-2-MePy+ | N-Bz-3-MePy+ | N-Bz-4-MePy+ |
| Conformational profiles | M-1, P′-1 | M-2, M′-2, P-2, P′-2 | M-3, P-3 |
| Supramolecular aggregates | Left-handed helical supramolecular chains in 1 | 3-Connected supramolecular (10,3)-b network in 2 | 3-Connected 2-D supramolecular (6,3) network in 3 |
| SBU for inorganic moiety | Cu5I9 building block | Cu5I9 building block | Cu4I9 building block |
| Inorganic framework | Left-handed helical chains in 1 | Left- and right-handed helical chains in 2 | 3-Connected 2-D layer with (6,3) network in 3 |
| Packing state | Conglomerate | Racemate | — |
TG-DSC-MS studies
TG-DSC-MS studies were carried out for compounds 1, 2 and 3 in argon atmosphere and some selected data are shown in Table S4.† All three compounds exhibit multi-step weight loss as shown in Fig. S5.† The TGA curves show that 1, 2 and 3 are stable up to about 210 °C, 230 °C and 245 °C, respectively. The weight losses of 37.8% (210–620 °C in 1), 38.1% (230–630 °C in 2), and 28.2% (245–645 °C in 3) are due to the loss of [N-Bz-2-MePy]I (calc. 39.5%), [N-Bz-3-MePy]I (calc. 39.5%), [N-Bz-4-MePy]I (calc. 29.0%), respectively. This pattern of decomposition is supported by the mass spectrometry (Fig. S6†) (the loss of benzyl group in the temperature range 245–580 °C for 1, 230–585 °C for 2, and 230–575 °C for 3, respectively, and the loss of methylpyridinium in the temperature range 282–620 °C for 1, 275–630 °C for 2, and 295–660 °C for 3, respectively). The different decomposition temperature of the organic moieties in 1, 2 and 3 may be ascribed to the difference of the weak interactions of cations.29 CuI is formed after the loss of organic moieties, which is supported by strong endothermic peaks in the DSC curves (Fig. S5†). The weight losses in the range of 620–710 °C for 1, 630–710 °C for 2, and 640–710 °C for 3 are attributed to the decomposition of CuI, respectively.Optical absorption spectra and thermochromic property
The experimental X-ray powder diffraction pattern of 1, 2 and 3 is in agreement with the simulated one (Fig. S7†), showing that the as-synthesized products are a single phase. The UV-vis diffuse reflectance spectra of 1, 2 and 3 were measured at room temperature (Fig. 4). The optical absorption spectra were calculated from the diffuse-reflectance data (R) using the Kubelka–Munk function (F(R) = (1 − R)2/2R). The band gaps (2.67 eV for 1, 2.56 eV for 2 and 2.64 eV for 3) were obtained using a straightforward extrapolation method,30 implying a semiconductive nature (Fig. S8†) and remarkable red shift relative to that of CuI (2.95 eV). The remarkable red shift of the excitonic absorption for 1, 2 and 3 should be attributed to the existence of intermolecular charge transfer (CT) from the N-benzyl-methylpyridinium cations to iodocuprate anions.31 Compared with 1, the apparent red-shift of 2 is probably attributed to the relatively stronger electron-accepting ability of [N-Bz-3-MePy]+ with respect to [N-Bz-2-MePy]+. Furthermore, although the electron-accepting ability of [N-Bz-4-MePy]+ is weaker than that of [N-Bz-2-MePy]+, the red-shift of 3 with respect to 1 is probably due to the stronger donating ability of anionic layer in 3 relative to anionic chain in 1.It is noteworthy that compounds 2 and 3 exhibit interesting low temperature reversible thermochromism. As shown in Fig. 5, compounds 2 and 3 undergo a color change from brown-yellow (room temperature, 293 K) to pale-yellow (liquid nitrogen, 77 K). The color changed rapidly after the crystals were immersed in liquid nitrogen and then gradually recovered the initial color as the temperature increased to 293 K. Based on our previous work,32 the thermochromic property of 2 and 3 could be attributed to the cold-induced decrease of intermolecular CT population.
 | ||
| Fig. 5 Thermochromic behavior of compounds 2 and 3: digital photographs of crystals taken at room temperature (293 K) and at the temperature of liquid nitrogen (77 K). | ||
Photoluminescence property
All three compounds 1, 2 and 3 exhibit photoluminescence at room temperature in the solid state as shown in Fig. S12.† It is revealed that a high-energy emission and a low-energy emission were found in 1, 2 and 3. The high-energy emission appears at 399 nm for 1, 398 nm for 2, and 397 nm for 3. The compounds 1–3 show bright luminescence of yellow (λmax = 550 nm), orange (λmax = 615 nm), and yellow (λmax = 570 nm), respectively. The photoluminescence properties and mechanisms of compounds 1, 2 and 3 are similar to those of iodocuprates.33 The emission bands are probably ascribed to a triplet cluster-centered excited state,33d which has integrated iodide-to-metal charge transfer and metal-centered transfer character.Conclusions
In summary, the successful synthesis and structural analysis of three novel iodocuprate frameworks exhibit an excellent structural directing effect of the conformationally chiral N-Bz-MePy+. Meanwhile, the slight change of organic SDAs and consequential supramolecular aggregates can lead to the formation of completely different structures, exhibiting the subtle template effect of methyl substituent on the formation of iodocuprate hybrids. The result provides an effective strategy for the rational construction of organic–inorganic hybrids through conformationally chiral template and versatile weak interactions.Acknowledgements
This work is supported by the National Natural Science Foundation of China (no. 21171110) and the Research Fund for the Doctoral Program of Higher Education of China (no. 20131404110001).References
- (a) E. Cariati, R. Macchi, D. Roberto, R. Ugo, S. Galli, N. Casati, P. Macchi, A. Sironi, L. Bogani, A. Caneschi and D. Gatteschi, J. Am. Chem. Soc., 2007, 129, 9410 CrossRef CAS PubMed; (b) A. M. Guloy, Z. J. Tang, P. B. Miranda and V. I. Srdanov, Adv. Mater., 2001, 13, 833 CrossRef CAS.
- (a) H. Y. Chang, S. H. Kim, P. S. Halasyamani and K. M. Ok, J. Am. Chem. Soc., 2009, 131, 2426 CrossRef CAS PubMed; (b) H. Y. Chang, S. H. Kim, K. M. Ok and P. S. Halasyamani, Chem. Mater., 2009, 21, 1654 CrossRef CAS.
- M. H. Choi, S. H. Kim, H. Y. Chang, P. S. Halasyamani and K. M. Ok, Inorg. Chem., 2009, 48, 8376 CrossRef CAS PubMed.
- (a) H. R. Zhao, D. P. Li, X. M. Ren, Y. Song and W. Q. Jin, J. Am. Chem. Soc., 2010, 132, 18 CrossRef CAS PubMed; (b) Z. Guo, R. Cao, X. Wang, H. Li, W. Yuan, G. Wang, H. Wu and J. Li, J. Am. Chem. Soc., 2009, 131, 6894 CrossRef CAS PubMed; (c) G. Xu, Y. Li, W. W. Zhou, G. J. Wang, X. F. Long, L. Z. Cai, M. S. Wang, G. C. Guo, J. S. Huang, G. Batorb and R. Jakubas, J. Mater. Chem., 2009, 19, 2179 RSC; (d) Y. Tang, Z. Sun, C. Ji, L. Li, S. Zhang, T. Chen and J. Luo, Cryst. Growth Des., 2015, 15, 457 CrossRef CAS.
- (a) L. Q. Ma, C. Abney and W. B. Lin, Chem. Soc. Rev., 2009, 38, 1248 RSC; (b) M. M. J. Treacy and J. M. Newsam, Nature, 1988, 332, 249 CrossRef CAS.
- (a) O. R. Evans and W. Lin, Acc. Chem. Res., 2002, 35, 511 CrossRef CAS PubMed; (b) S.-J. Hwu, M. Ulutagay-Kartin, J. A. Clayhold, R. Mackay, T. A. Wardojo, C. J. O’Connor and M. Kraweic, J. Am. Chem. Soc., 2002, 124, 12404 CrossRef CAS PubMed; (c) M. E. Welk, A. J. Norquist, F. P. Arnold, C. L. Stern and K. R. Poeppelmeier, Inorg. Chem., 2002, 41, 5119 CrossRef CAS PubMed; (d) P. A. Maggard, C. L. Stern and K. R. Poeppelmeier, J. Am. Chem. Soc., 2001, 123, 7742 CrossRef CAS PubMed; (e) C. J. Kepert, T. J. Prior and M. J. Rosseinsky, J. Am. Chem. Soc., 2000, 122, 5158 CrossRef CAS.
- (a) J. H. Kim, J. Baek and P. S. Halasyamani, Chem. Mater., 2007, 19, 5637 CrossRef CAS; (b) S. Ayyappan, X. Bu, A. K. Cheetham and C. N. R. Rao, Chem. Mater., 1998, 10, 3308 CrossRef CAS.
- (a) D. W. Guo, S. Y. Yao, G. Zhang and Y. Q. Tian, CrystEngComm, 2010, 12, 2989 RSC; (b) Y. H. Guo, Z. Shi, J. H. Yu, J. D. Wang, Y. L. Liu, N. Bai and W. Q. Pang, Chem. Mater., 2001, 13, 203 CrossRef CAS.
- (a) X. Zhang, Z.-X. Lei, W. Luo, W.-Q. Mu, X. Zhang, Q.-Y. Zhu and J. Dai, Inorg. Chem., 2011, 50, 10872 CrossRef CAS PubMed; (b) G. Liu, X. Jiang, M. Wu, G. Wang, G.-C. Guo and J. Huang, Inorg. Chem., 2011, 50, 5740 CrossRef CAS PubMed; (c) J. Zhao, J. Liang, J. Chen, Y. Pan, Y. Zhang and D. Jia, Inorg. Chem., 2011, 50, 2288 CrossRef CAS PubMed; (d) K. A. McGee and K. R. Mann, J. Am. Chem. Soc., 2009, 131, 1896 CrossRef CAS PubMed; (e) P. Li, X. M. Lu, B. Liu, S. Wang and X. Wang, Inorg. Chem., 2007, 46, 5823 CrossRef CAS PubMed; (f) P. Chen, J. Li, F. Duan, J. Yu, R. Xu and R. P. Sharma, Inorg. Chem., 2007, 46, 6683 CrossRef CAS PubMed; (g) M. Seitz, A. Kaiser, S. Stempfhuber, M. Zabel and O. Reiser, Inorg. Chem., 2005, 44, 4630 CrossRef CAS PubMed; (h) K. Kano and H. Hasegawa, J. Am. Chem. Soc., 2001, 123, 10616 CrossRef CAS PubMed; (i) Y. Wang, J. Yu, M. Guo and R. Xu, Angew. Chem., Int. Ed., 2003, 42, 4089 CrossRef CAS PubMed; (j) C. Tang, F. Wang, J. Lu, D. Jia, W. Jiang and Y. Zhang, Inorg. Chem., 2014, 53, 9267 CrossRef CAS PubMed.
- (a) Z. Dong, L. Zhao, Z. Liang, P. Chen, Y. Yan, J. Li, J. Yu and R. Xu, Dalton Trans., 2010, 39, 5439 RSC; (b) W. J. Liu, K. Yao, W. F. Shangguan and J. Yuan, J. Mater. Chem., 2009, 19, 8803 RSC; (c) L. Zhao, J. Y. Li, P. Chen, Z. J. Dong, J. H. Yu and R. R. Xu, CrystEngComm, 2008, 10, 497 RSC; (d) L. Chen and X. H. Bu, Chem. Mater., 2006, 18, 1857 CrossRef CAS; (e) J. Fan, C. Slebodnick, R. Angel and B. E. Hanson, Inorg. Chem., 2005, 44, 552 CrossRef CAS PubMed; (f) L.-H. Cao, Y.-L. Wei, Y. Yang, H. Xu, S.-Q. Zang, H.-W. Hou and T. C. W. Mak, Cryst. Growth Des., 2014, 14, 1827 CrossRef CAS.
- (a) C. H. Lin and S. L. Wang, Inorg. Chem., 2001, 40, 2918 CrossRef CAS PubMed; (b) H. M. Lin and K. H. Lii, Inorg. Chem., 1998, 37, 4220 CrossRef CAS PubMed.
- (a) C. F. Sun, C. L. Hu, X. Xu, B. P. Yang and J. G. Mao, J. Am. Chem. Soc., 2011, 133, 5561 CrossRef CAS PubMed; (b) B. P. Yang, C. L. Hu, X. Xu, C. F. Sun, J. H. Zhang and J. G. Mao, Chem. Mater., 2010, 22, 1545 CrossRef CAS; (c) F. Kong, C. F. Sun, B. P. Yang and J. G. Mao, Struct. Bonding, 2012, 144, 43 CrossRef CAS; (d) B. P. Yang, C. L. Hu, X. Xu, C. Huang and J. G. Mao, Inorg. Chem., 2013, 52, 5378 CrossRef CAS PubMed; (e) H. X. Zhang, J. Zhang, S. T. Zheng, G. M. Wang and G. Y. Yang, Inorg. Chem., 2004, 43, 6148 CrossRef CAS PubMed; (f) C. Rong, Z. Yu, Q. Wang, S. T. Zheng, C. Y. Pan, F. Deng and G. Y. Yang, Inorg. Chem., 2009, 48, 3650 CrossRef CAS PubMed; (g) S. T. Zheng, C. Y. Pan, G. M. Wang and G. Y. Yang, Angew. Chem., Int. Ed., 2005, 44, 6881 CrossRef PubMed; (h) J. Zhou, W. H. Fang, C. Rong and G. Y. Yang, Chem.–Eur. J., 2010, 16, 4852 CrossRef CAS PubMed; (i) D. J. Williams, J. S. Kruger, A. F. McLeroy, A. P. Wilkinson and J. C. Hanson, Chem. Mater., 1999, 11, 2241 CrossRef CAS; (j) S. M. Stalder and A. P. Wilkinson, Chem. Mater., 1997, 9, 2168 CrossRef CAS.
- (a) Y. R. Qiao, P. F. Hao and Y. L. Fu, Inorg. Chem., 2015, 54, 8705 CrossRef CAS PubMed; (b) H. Zhang, P. Lin, X. Shan, L. Han and S. Du, CrystEngComm, 2014, 16, 1245 RSC.
- (a) J. Y. Li, A. Corma and J. H. Yu, Chem. Soc. Rev., 2015, 44, 7022 RSC; (b) Z. P. Wang, J. H. Yu and R. R. Xu, Chem. Soc. Rev., 2012, 41, 1729 RSC; (c) J. H. Yu and R. R. Xu, Acc. Chem. Res., 2010, 43, 1195 CrossRef CAS PubMed; (d) E. R. Parnham and R. E. Morris, Acc. Chem. Res., 2007, 40, 1005 CrossRef CAS PubMed; (e) S. H. Feng and R. R. Xu, Acc. Chem. Res., 2001, 34, 239 CrossRef CAS PubMed.
- (a) Z. Jiang, W. Lu, Z. Li, K. H. Ho, X. Li, X. Jiao and D. Chen, J. Mater. Chem. A, 2014, 2, 8603 RSC; (b) C. Masquelier and L. Croguennec, Chem. Rev., 2013, 113, 6552 CrossRef CAS PubMed; (c) Y. D. Liu, J. Goebl and Y. D. Yin, Chem. Soc. Rev., 2013, 42, 2610 RSC; (d) N. D. Petkovich and A. Stein, Chem. Soc. Rev., 2013, 42, 3721 RSC; (e) C. N. R. Rao, J. N. Behera and M. Dan, Chem. Soc. Rev., 2006, 35, 375 RSC; (f) P. Y. Feng, X. H. Bu and N. F. Zheng, Acc. Chem. Res., 2005, 38, 293 CrossRef CAS PubMed.
- (a) J. B. Willems and M. Köckerling, Chem. Commun., 2001, 1380 RSC; (b) T. Akutagawa, A. Hashimoto, S. Nishihara, T. Hasegawa and T. Nakamura, J. Phys. Chem. B, 2003, 107, 66 CrossRef CAS; (c) C. R. Li, S. L. Li and X. M. Zhang, Cryst. Growth Des., 2009, 9, 1702 CrossRef CAS; (d) T. Chatterjee, M. Sarma and S. K. Das, Cryst. Growth Des., 2010, 10, 3149 CrossRef CAS; (e) T. Endo, T. Akutagawa, S. Noroa and T. Nakamura, Dalton Trans., 2011, 40, 1491 RSC; (f) X. X. Huang, L. G. Qiu, W. Zhang, Y. P. Yuan, X. Jiang, A. J. Xie, Y. H. Shen and J. F. Zhu, CrystEngComm, 2012, 14, 1613 RSC; (g) N. Hoshino, Y. Yoshii, M. Aonuma, K. Kubo, T. Nakamura and T. Akutagawa, Inorg. Chem., 2012, 51, 12968 CrossRef CAS PubMed.
- (a) Z. Liu, K. Kubo, L. Lin, N. Hoshino, S. Noro, T. Akutagawac and T. Nakamura, Dalton Trans., 2013, 42, 2930 RSC; (b) T. Endo, K. Kubo, M. Yoshitake, S. Noro and N. Hoshino, Cryst. Growth Des., 2015, 15, 1186 CrossRef CAS; (c) Z. J. Zhang and M. J. Zaworotko, Chem. Soc. Rev., 2014, 43, 5444 RSC; (d) Z. J. Zhang, S. C. Xiang, G. C. Guo, G. Xu, M. S. Wang, J. P. Zou, S. P. Guo and J. S. Huang, Angew. Chem., Int. Ed., 2008, 47, 4149 CrossRef CAS PubMed; (e) L. M. Wu, X. T. Wu and L. Chen, Coord. Chem. Rev., 2009, 253, 2787 CrossRef CAS.
- (a) L. Gomez-Hortigüela, F. Lopez-Arbeloa, F. Corà and J. Perez- Pariente, J. Am. Chem. Soc., 2008, 130, 13274 CrossRef PubMed; (b) Q. H. Pan, J. Y. Li, K. E. Christensen, C. Bonneau, X. Y. Ren, L. Shi, J. L. Sun, X. D. Zou, G. H. Li, J. H. Yu and R. R. Xu, Angew. Chem., Int. Ed., 2008, 47, 7868 CrossRef CAS PubMed; (c) T. L. Yu, J. J. Shen, Y. B. Fu and Y. L. Fu, CrystEngComm, 2014, 16, 5280 RSC; (d) J. Heine, Dalton Trans., 2015, 44, 10069 RSC; (e) R. M. Franco, A. Cantín, M. Moliner and A. Corma, Chem. Mater., 2014, 26, 4346 CrossRef; (f) R. M. Franco, A. Cantín, A. V. Moya, M. Moliner and A. Corma, Chem. Mater., 2015, 27, 2981 CrossRef.
- CrysAlisPro, version 1.171.33.56, Oxford Diffraction Ltd., Oxfordshire, UK, 2010 Search PubMed.
- (a) G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112 CrossRef CAS PubMed; (b) G. M. Sheldrick, SHELXS-97, A Program for X-ray Crystal Structure Solution, and SHELXL-97, A Program for X-ray Structure Refinement, Göttingen University, Germany, 1997 Search PubMed.
- H. Khili, N. Chaari, M. Fliyou, A. Koumina and S. Chaabouni, Polyhedron, 2012, 36, 30 CrossRef CAS.
- H.-H. Li, Z.-R. Chen, Y. Liu, K.-N. Ding, J.-Q. Li, C.-C. Huang and L.-Q. Guo, J. Cluster Sci., 2007, 18, 817 CrossRef CAS.
- (a) S. Mishra, E. Jeanneau, G. Ledoux and S. Daniele, Inorg. Chem., 2014, 53, 11721 CrossRef CAS PubMed; (b) S. Mishra, E. Jeanneau, G. Ledouxc and S. Daniele, Dalton Trans., 2009, 4954 RSC.
- K. Takahashi, Y. Mazaki and K. Kobayashi, Chem. Commun., 1996, 2275 RSC.
- W. J. Gee and S. R. Batten, Cryst. Growth Des., 2013, 13, 2335 CAS.
- O. V. Dolomanov, A. J. Blake, N. R. Champness and M. Schröder, J. Appl. Crystallogr., 2003, 36, 1283 CrossRef CAS.
- L. Öhrström and K. Larsson, Kristallografiya, 2004, 347 Search PubMed.
- (a) Q. Y. Li and Y. L. Fu, CrystEngComm, 2009, 11, 1515 RSC; (b) S. Mishra, E. Jeanneau, G. Ledouxc and S. Daniele, CrystEngComm, 2012, 14, 3894 RSC; (c) T. R. ttgers and W. S. Sheldrick, J. Solid State Chem., 2000, 152, 271 CrossRef.
- (a) W. L. Zhang, Z. P. Huang, C. J. Ma, Y. X. Zai, Q. Yang and Y. Y. Niu, Inorg. Chim. Acta, 2015, 425, 52 CrossRef CAS; (b) Y. Z. Qiao, W. Z. Fu, J. M. Yue, X. C. Liu, Y. Y Niu and H. W. Hou, CrystEngComm, 2012, 14, 3241 RSC.
- (a) G. Kotiim, Reflectance Spectroscopy, Springer-Verlag, New York, 1969 Search PubMed; (b) O. Schevciw and W. B. White, Mater. Res. Bull., 1983, 18, 1059 CrossRef CAS.
- (a) H. Chan, Y. Chen, M. Dai, C. Ning, H. F. Wang, Z. G. Ren, Z. J. Huang, C. Y. Ni and J. P. Lang, CrystEngComm, 2012, 14, 466 RSC; (b) Y. Chen, Z. Yang, C. X. Guo, C. Y. Ni, H. X. Li, Z. G. Rena and J. P. Lang, CrystEngComm, 2011, 13, 243 RSC.
- (a) J. J. Shen, C. F. Zhang, T. L. Yu, L. An and Y. L. Fu, Cryst. Growth Des., 2014, 14, 6337 CrossRef CAS; (b) T. L. Yu, L. An, L. Zhang, J. J. Shen, Y. B. Fu and Y. L. Fu, Cryst. Growth Des., 2014, 14, 3875 CrossRef CAS.
- (a) L. Maini, P. P. Mazzeo, F. Farinella, V. Fattori and D. Braga, Faraday Discuss., 2014, 170, 93 RSC; (b) L. Zhang, B. Li and Z. Su, Langmuir, 2009, 25, 2068 CrossRef CAS PubMed; (c) K. Umakoshi, T. Kojima, K. Saito, S. Akatsu, M. Onishi, S. Ishizaka, N. Kitamura, Y. Nakao, S. Sakaki and Y. Ozawa, Inorg. Chem., 2008, 47, 5033 CrossRef CAS PubMed; (d) S.-L. Li and X.-M. Zhang, Inorg. Chem., 2014, 53, 8376 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Infrared Spectroscopy (Fig. S1), the asymmetric unit diagrams (Fig. S2 for 1, Fig. S3 for 2 and Fig. S4 for 3), TG and DSC analyses (Fig. S5), MS for compounds 1, 2 and 3 (Fig. S6), X-ray powder diffraction (XRPD) patterns (Fig. S7), optical absorption spectra at room temperature (Fig. S8), chiral conformations of 1, 2 and 3 (Fig. S9–S11), photoluminescence emissions and excitations of 1, 2 and 3 (Fig. S12), a summary of the crystal data (Table S1), selected bond lengths (Å) and angles (°) for 1, 2 and 3 (Table S2), parameters of the chiral conformations in the compounds 1–3 (Table S3), and selected TG-DTA-MS data for compounds 1–3 (Table S4). CCDC 1449176–1449178. For ESI and crystallographic data in CIF or other electronic formats see DOI: 10.1039/c6ra08692f |
| This journal is © The Royal Society of Chemistry 2016 |