Multi-stacked electrodes employing aluminum coated tissue papers and non-oxidized graphene nanoflakes for high performance lithium–sulfur batteries†
Abstract
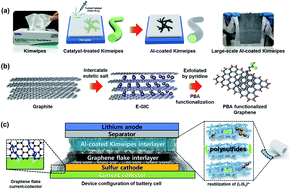
Here, we report a simple approach to Li/S battery cell modification by introducing multi-stacked reactivation layers of 1st-graphene flakes/2nd-Al coated tissue paper (GNFs/Al-coated Kimwipes) between a separator and a sulfur cathode. Our unique chemical solution-based coating technique for an Al thin film on catalytically treated fibrous tissue paper offers a cost-effective sulfur electrode with high electrical conductivity, which is well suited to a scaling up of the sulfur electrode. The cathode with the GNFs/Al-coated Kimwipes not only showed excellent rate capability (497.3 mA h g−1 at 2C), but also delivered a high capacity of 715.9 mA h g−1 up to 100 cycles. It also maintained 669.3 mA h g−1 after 200 cycles at 0.2C with negligible capacity degradation, indicating a good capacity retention of 93.5%. Such superior electrochemical performances should be attributed to the finely designed cell configuration: (i) GNFs on the sulfur electrode as a pseudo-upper current collector that directly suppresses the sulfur dissolution; (ii) porous Al-coated Kimwipes with a high electrical conductivity (∼0.7 Ω □−1) as a main reservoir which reversibly captures and reutilizes the sulfur species. The proposed concept of the sulfur electrode can give an applicable solution for advanced Li/S batteries.


 Please wait while we load your content...
Please wait while we load your content...