Bioinformatical analysis and preliminary study of the role of lipase in lipid metabolism in Mucor circinelloides†
Abstract
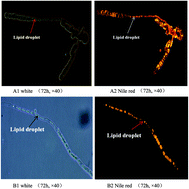
The filamentous fungus Mucor circinelloides has been widely used as a model organism to investigate the mechanisms of lipid accumulation. Although a lot of work has been done to analyze and explore many of the essential enzymes/genes related to lipid accumulation in M. circinelloides, the function of lipase in this fungus has not been studied at all. In this study, we report some important characteristics of all 30 lipases, and especially 4 lipases Lip6, Lip10, Lip19 and Lip24 with functional analyses in Mucor circinelloides CBS 277.49 based on the genome sequences. Transcriptional analyses revealed that the Lip6 or Lip10 gene expression increased significantly during 24 h to 72 h of lipid accumulation while Lip19 and Lip24 genes were down-regulated along with the increase in lipid accumulation. Over-expression of either lipase Lip6 or Lip10 led to a slight increase in cell dry weight, but no significant effect on lipid accumulation. More interestingly over-expression of Lip6 resulted in a 17% increase in C18:3 content of the lipids. However, over-expression of lipase Lip19 and Lip24 decreased the cell dry weight by 9.08–18.8% and lipid content by 37.86–41.42%, respectively. The fatty acid profiles of strains with Lip19 and Lip24 over-expression were also significantly changed as compared to control. Analysis of lipase genes revealed that the sequences of Lip6 and Lip10 not only contained the typical lipase motif (G/A) XSXG, but also the consensus sequence motif HXXXXD (a typical of acyltransferase motif). These results suggest that Lip6 or Lip10 may control acyltransferase activity in Mucor circienlloides and play a role in fatty acid reconstruction of TAG, while Lip19 or Lip24 may work as a TAG lipase involved in TAG metabolism/degradation.

 Please wait while we load your content...
Please wait while we load your content...