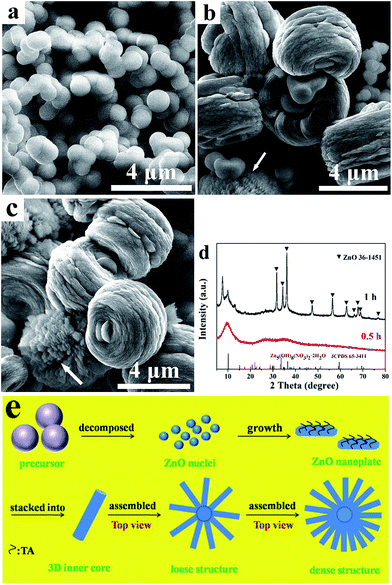
Formation of nanoplate-based clew-like ZnO mesocrystals and their photocatalysis application
Xueling Songa,
Dehua Dongb and
Ping Yang*a
aSchool of Material Science and Engineering, University of Jinan, Jinan, 250022, P. R. China. E-mail: mse_yangp@ujn.edu.cn; Fax: +86-531-87974453; Tel: +86-531-89736225
bFuels and Energy Technology Institute, Curtin University, GPO Box U1987, Perth WA6845, Australia
First published on 20th May 2016
Abstract
Clew-like ZnO mesocrystals comprised of ZnO nanoplates were created through a hydrothermal procedure in an aqueous solution using tartaric acid as the structure-directing additive. A detailed formation process of the nanoplate-based clew-like ZnO mesocrystal was investigated by varying reaction conditions especially the concentration of hexamethylenetetramine (HMT) and reaction time. It was found that the concentration of HMT played a significant role in the size distribution of ZnO products and the pileup degree of the nanoplates. Combined with the results obtained from the systematic time-dependent experiments, the formation of the nanoplate-based ZnO mesocrystal was mainly ascribed to the synergistic effect of the dipole moment generated among nanoplates and the electric field derived from a pre-formed inner core. The photocatalytic activity of samples synthesized using different HMT concentrations was also investigated. The results demonstrated that the adsorption percentage in the dark and degradation efficiency under UV-light irradiation were gradually improved with increasing the concentration of HMT. This was mainly ascribed to the increased specific surface area which promoted the migration of photon-generated carriers between the photocatalysts and rhodamine B (RhB) molecules.
Introduction
Nanomaterials have attracted wide research interest owing to their novel physicochemical properties and specific collective properties derived from nanoparticle ensembles.1,2 In particular, the study of mesocrystals has received a great deal of attention since the discovery of a series of ordered nanoparticle superstructures reported by Cölfen and Antonietti in 2005.3 Thus, it is still a challenge to build highly ordered superstructures with unique geometry and size-dependent characteristics by the self-assembly of subunits.4,5 As is well known, the properties of the materials such as different morphology, size and microstructure could dramatically influence their properties and applications. Therefore, it is meaningful to investigate mesocrystals considering their size range and ordered structures which endow them with an extensive range of applications, such as in photocatalysis, catalysis and other fields.2,6Up to now, many research groups have fabricated various mesocrystals and discussed the corresponding formation mechanism with the emergence of mesocrystals.7–12 For instance, Song and co-workers prepared CaCO3 mesocrystals with high versatility using poly(4-styrenesulfonate-co-maleic acid) as the crystal modifier.13 Hong and co-workers synthesized nanoporous rutile TiO2 mesocrystals with wulff-shaped and nanorod-like morphologies employing sodium dodecyl benzene sulfonate as the surfactant.14 Deng and co-workers successfully fabricated Cu2O nanowire mesocrystals in the presence of o-anisidin and the Cu2O mesocrystals-based composite, reduced graphene oxide–Cu2O, exhibited enhanced sensitivity toward NO2 gas due to their high specific surface area and improved conductivity.15 The formation mechanism of the mesocrystal has also received much attention. Differing from the classical crystallization, the nonclassical crystallization provides a pathway for investigating the formation of ordered superstructures. However, the driving mechanism of mesocrystals is still demanded to investigate.16,17 Compared with the previous reported 3D mesocrystals constituted of nanoparticles or nanowires, the synthesis of mesocrystals consisted of densely stacked 2D nanoplates with high specific surface area and excellent properties is necessary to explore.
As a common semiconductor material, ZnO nanocrystals and microcrystals have obtained wide attention due to excellent photoelectrical properties as well as diverse morphology, such as nanorods, rhombic sheets and nanoplate-based 3D hierarchical structure.18–20 The formation mechanism was mainly illustrated by the classical crystallization process. Similarly, researchers have paid much attention to ZnO mesocrystals owing to their unique morphology and crystallization process. For example, Liu and coworkers first reported a novel apple-like nanoplatelet-based ZnO mesocrystals prepared by a low temperature hydrothermal method at 95 °C.11 Meanwhile, the formation mechanism of the as-obtained mesocrystal was systematically discussed and a dipole-field-driven mechanism was proposed.11 Ye and coworkers have fabricated ZnO bundled mesocrystals by a low-temperature, solution-phase approach in the presence of poly(vinyl pyrrolidone) (PVP) as a crystal growth modifier.21 Besides, ZnO mesocrystals with different morphologies were also successfully synthesized by applying different structure-directing agents, such as tetrabutyl ammonium hydroxide (TBAH), gelatin and others.22,23 However, despite the prepared various ZnO mesocrystal assembled by different subunits, it is highly desirable to synthesize ZnO mesocrystals in large scale by a simple method and to investigate their properties. Especially, no report focused on the preparation of clew-like ZnO mesocrystal microspheres. Such morphology may be advantages in tuning the performance.
Inspired by these previous reports, we prepared a novel clew-like ZnO mesocrystal microspheres comprised of densely stacked nanoplates in the presence of tartaric acid (TA) as the structure-directing additive. It was found that the size and the morphology of ZnO mesocrystal can be adjusted by varying different reaction conditions, such as reaction temperature and the concentration of raw materials. Furthermore, the formation mechanism of the ZnO mesocrystal was also discussed on the basis of the nonclassical crystallization crystal growth. In detail, the formation of such ZnO mesocrystal was depended on synergistic effect of the dipole moment generated among nanoplates and the electric field derived from the inner core. Moreover, ZnO mesocrystals prepared using 0.1 M hexamethylenetetramine (HMT) concentration exhibited the highest normalized kinetic constant compared with others. This was mainly ascribed to high specific surface area.
Experimental
Materials
All reagents were of analytical grade and used as received without further purification. Zinc nitrate hexahydrate (Zn(NO3)2·6H2O) and rhodamine B (RhB) were purchased from Shanghai Guoyao Chemical Reagent Co. Ltd., (Shanghai, China). HMT and TA were applied by Tianjin Guangcheng Chemical Reagent Company (Tianjin, China). Anhydrous ethanol (CH3CH2OH) was of analytical grade and purchased from Tianjin Fuyu Chemical Industry Co. Ltd., (Tianjin, China). Deionized (DI) water with a resistance of 18.2 MΩ cm was supplied by a Milli-Q synthesis system and used for all experiments.Synthetic procedure of ZnO mesocrystal microspheres
ZnO samples were prepared by a one-step hydrothermal method. In a typical synthetic procedure, Zn(NO3)2 (2.5 mmol), HMT (2.5 mmol) and TA (0.5 mmol) were dissolved in 50 mL of H2O with vigorously stirring to form a clear solution. Subsequently, the above solution was then transferred to a 100 mL of Teflon-lined steel autoclave and hydrothermally treated at 150 °C for 6 h. After cooling to room temperature naturally, the white precipitate was centrifuged and washed sequentially with H2O and CH3CH2OH three times each, before drying at 60 °C for 10 h in an oven. Finally, the white products were collected for subsequent characterization. In addition, to investigate the optimal experimental parameters for the synthesis of ZnO mesocrystal microspheres, a series of experiments were carried out by altering the hydrothermal temperature and the concentration of Zn(NO3)2, TA and HMT, respectively. Furthermore, to analyse the formation mechanism of the typical sample, a series of samples were prepared by varying reaction time. Detailed preparation parameters were summarized in Table 1.| Sample | Zn(NO3)2·6H2O/M | HMT/M | TA/M | Reaction temperature/°C | Reaction time/h |
|---|---|---|---|---|---|
| S1 | 0.05 | 0.05 | 0.01 | 150 | 6 |
| S2 | 0.05 | 0.05 | 0 | 150 | 6 |
| S3 | 0.05 | 0.05 | 0.008 | 150 | 6 |
| S4 | 0.05 | 0.05 | 0.015 | 150 | 6 |
| S5 | 0.05 | 0.05 | 0.01 | 200 | 6 |
| S6 | 0.02 | 0.05 | 0.01 | 150 | 6 |
| S7 | 0.05 | 0.075 | 0.01 | 150 | 6 |
| S8 | 0.05 | 0.1 | 0.01 | 150 | 6 |
| S9 | 0.05 | 0.15 | 0.01 | 150 | 6 |
| S10 | 0.05 | 0.05 | 0.01 | 150 | 0.5 |
| S11 | 0.05 | 0.05 | 0.01 | 150 | 1 |
Characterization
The crystal structure of the as-synthesized ZnO samples was examined using a powder X-ray diffraction (XRD) with a D8 Advance Bruker X-ray diffractometer using a Cu Kα target in a 2θ range of 20° to 80°. The morphologies and sizes of products were characterized via field-emission scanning electron microscopy (SEM, FEI, QUANTA FEG 250). The high-resolution TEM (HRTEM) image was recorded using a JEM-2100 under an acceleration voltage of 200 kV to investigate interplanar distances of ZnO. The Brunauer–Emmett–Teller (BET) specific surface area and the Barrett–Joyner–Halenda (BJH) pore size distribution of different samples were performed on a multi-function adsorption instrument (MFA-140 of Beijing Builder Company) at liquid nitrogen temperature of 77 K. Before the measurements, all samples were degassed under vacuum at 110 °C for 3 h. Thereinto, the BET specific surface areas were evaluated on the basis of nitrogen adsorption isotherms. The BJH pore size distribution was obtained from desorption data. The absorption spectra of RhB solution were performed on a Hitachi U-4100 spectrophotometer with a quartz cell.Photocatalytic degradation measurement
The photocatalytic degradation activities of different ZnO samples were determined using RhB as a target dye pollutant under a UV light (12 W). Firstly, 20 mL of a 5 mg L−1 RhB aqueous solution was prepared by dissolving RhB in H2O. 10 mg of as-prepared ZnO photocatalyst was then dispersed in the above RhB solution. The mixture was sonicated for 3 min and subsequently placed in the dark for 30 min with continuous stirring to reach the adsorption–desorption equilibrium between photocatalysts and RhB. Afterwards, the mixed solution was exposed to the UV light. At an interval of 1 h, 2 mL of the suspension was withdrawn and immediately centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 3 min to remove the photocatalyst. The concentration of RhB was monitored by recording variations of the maximum absorbance at 553 nm using the Hitachi U-4100 spectrometer.
000 rpm for 3 min to remove the photocatalyst. The concentration of RhB was monitored by recording variations of the maximum absorbance at 553 nm using the Hitachi U-4100 spectrometer.
Results and discussion
SEM images of the solid sample produced from the reaction between Zn(NO3)2 and HMT in the presence of TA shown in Fig. 1a–d represent the obtained sample (S1) with clew-like structures. A low-magnification SEM image in Fig. 1a suggests that the products with rough surface are made up of large-scale clew-shaped microspheres with the diameter of 4.0–8.6 μm (average size of 6.3 μm) and shows typical mesocrystal character. The SEM images in Fig. 1 show that nearly every microsphere has a hole (the white arrows). The result from the high-magnification SEM image shown in Fig. 1b clearly shows that each mesocrystal is comprised of abundant densely packed nanoplates which are aligned perpendicularly to the microsphere pointing to the same center. Further observation indicates that the shape of these mesocrystal microspheres is not fully spherical exhibiting an almost hexagonal structure with rounded corners. In addition, the existence of holes is further observed. Fig. 1c shows that the nanoplates as building block with the average thickness of ca. 30.8 nm arrange regularly on the surface of the as-synthesized mesocrystal. The inner architecture can be confirmed by a crushed microsphere, and the corresponding SEM image is showed in Fig. 1d. A clear boundary, which separated the inner nonradial structure from the outer radial structure, was witnessed. Furthermore, it can be found that the outer radial structure was arranged pointing to the inner axis, indicating that stacking manner of the outer nanoplates was closely related to the inner axis. The HRTEM image of ZnO nanoplates which are the building block of the above-mentioned mesocrystal microspheres is depicted in Fig. 1e. From the image, the measured lattice fringes of 0.26 nm correspond to the (002) crystal plane of hexagonal ZnO phase. Although HMT could hydrolyze to form HMT-4H+ cationic which can absorbed onto the O2−-terminated (000−1) plane, the main role of HMT is to act as hydroxide anion precursor in the process of the hydrothermal reaction. And the result from the HRTEM image indicates that the growth of nanoplates is blocked along the [0001] direction due to the effect of carboxyl groups in TA. Furthermore, it can be clearly noticed that some defect regions (marked by circles) are existed, which is the assembly characteristic of mesocrystals. The results strongly indicate that the formation mechanism of the microsphere is a nonclassical process instead of an ion-by-ion crystallization one.24,25 The corresponding XRD pattern depicted in Fig. 1f exhibits that all diffraction peaks of the clew-like mesocrystal (S1) are exclusively indexed to a wurtzite structured ZnO (JCPDS file no. 36-1451). Additionally, no impurity peaks are detected in this XRD pattern, implying that the high purity ZnO products. Moreover, it is noted that the diffraction peaks are sharp and have high intensity, indicating the as-prepared ZnO products are highly crystallized.It is well known that the reaction conditions are important in determining the formation of mesocrystals.26,27 Thereinto, the concentration of TA has significant effect on the formation ZnO mesocrystals as reported in previous literature.27 Hence, the relevant controlled experiments were carried out via changing the TA concentration. The morphologies of ZnO samples were characterized by SEM as shown in Fig. 2. Irregularly flower-like ZnO comprised by ZnO rods with hexagonal structure was obtained in the absence of TA additive as shown in Fig. 2a and b. With decreasing the TA concentration to 0.008 M from 0.1 M (Fig. 2c and d), aggregations of ZnO mesocrystals are obtained differing from well-dispersed mesocrystals as products shown in Fig. 1 and 4. To investigate the effect of increased TA concentration, the product was synthesized when the concentration of TA was increased to 0.015 M. As depicted in Fig. 2e and f, a lot of irregular particles with small amount of microspheres are observed. The result demonstrates that the concentration of TA has to be about 0.01 M to gain ZnO mesocrystals with fine dispersibility. The above result confirms that the concentration of TA plays a crucial role in the formation of clew-like ZnO mesocrystals due to that the negatively charged carboxyl groups in TA molecules are easily anchored on the Zn2+-terminated (0001) plane. When the concentration of TA was low, repulsive force among mesocrystals was weak. Thus, the formed individual ZnO mesocrystals aggregated driving by reduction of the surface free energy. Nevertheless, it is unfavourable to prepare well-dispersed ZnO mesocrystals by applying high concentration of TA. The possible reason is that the surface of precursors which will be mentioned in Fig. 5 is covered with large amount of TA molecules, leading to that the precursor is difficult to decompose. Driven by minimum surface energy, a small percentage of ZnO particles generated by decomposition of the precursor could aggregate quickly to form microspheres.
 | ||
| Fig. 2 SEM images of different ZnO products prepared under different concentration of TA: (a and b) in the absence of TA (S2), (c and d) with 0.008 M TA (S3) and (e and f) with 0.015 M TA (S4). | ||
 | ||
| Fig. 4 SEM images of ZnO mesocrystals prepared at different concentration of HMT. (a and b) 0.075 M, S7, (c and d) 0.1 M, S8, and (e and f) 0.15 M, S9. | ||
Moreover, to investigate the influence of other reaction conditions, ZnO samples with different morphologies were synthesized as shown in Fig. 3. It is worthwhile to mention that the hydrothermal temperature has a great influence on the morphology of samples.14 When the reaction system is conducted at a high temperature (200 °C), hamburger-like and football-like ZnO appear which has been labeled in Fig. 3a besides clew-like microspheres. These results indicated relatively low temperature was favorable for the formation of clew-like ZnO mesocrystals with uniform particle size distribution. This was probably due to high nucleation and growth rate at high temperature leading to inhomogeneous morphology. Interestingly, nanoplate-based ZnO microspheres with average diameter of 8.85 μm are obtained with decreasing the concentration of Zn(NO3)2 to 0.02 M on the basis of the typical experiment (Fig. 3b). Further observation on the inset of Fig. 3b indicates that the thickness of these nanoplate subunits is about 76.9 nm on average, which is 2.5 times larger than that of S1 (about 30.8 nm) shown in Fig. 1. This was attributed that the shortage of Zn(NO3)2 resulted in slow growth which was beneficial to generate ZnO thick nanoplates and self-assembled products with large dimension. The results suggested that the concentration of Zn(NO3)2 had to be adjusted to 0.05 M to create nanoplate-based clew-like ZnO mesocrystals with relatively uniform size distribution.
To get insight into the effect of the concentration of HMT, controlled experiments were carried out through simply varying HMT concentrations from 0.075 to 0.15 M with other parameters kept constant compared with the typical experiment. The SEM image in Fig. 4a indicates that the diameter of the clew-like ZnO structure is in the range of 3.17 to 4.63 μm with increasing the concentration of HMT to 0.075 M. Some ZnO products have large defect on their surface which suggests the sample are not fully developed (Fig. 4a) and thin nanoplates are also well aligned forming the clew-like ZnO (Fig. 4b). When the HMT concentration is increased to 0.1 M, the SEM image shows that the products are consisted of clew-like microspheres with 2.34–5.62 μm in diameter (Fig. 4c). Interestingly, some microspheres with large size exhibited no holes on their surface and small ones still have typical holes characteristic. Meanwhile, it can be observed that a few of the products has a more distinct hexagonal shape (Fig. 4d) compared with other ZnO samples. Actually, clew-shaped, nanoplate-based ZnO mesocrystals with uneven size distribution (2.31–7.69 μm in diameter) shown in Fig. 4e were prepared when 0.15 M of HMT was used. Furthermore, large irregular aggregates were also obtained. The phenomenon that the typical holes structure only existed in these ZnO mesocrystals with small size was consistent with the result gained from Fig. 4c. The result implies that HMT concentrations play a significant role in the size and structure of ZnO products.
In order to gain a complete understanding for the growth process of the clew-like ZnO mesocrystal, a systematic time-dependent morphology evolution study was performed at 150 °C for different hydrothermal time. In general, much important information can be provided via such time-resolved experiments for elucidating the underlying formation mechanism. In Fig. 5a, it can be observed that a large number of precursor particles displaying smooth surface with the diameter in the range of 0.9 to 1.2 μm are obtained at the primary stage of the crystallization process (i.e. nucleation stage of mesocrystal) due to fast nucleation caused by the reaction of Zn(NO3)2 and HMT. The corresponding XRD pattern suggests that the composition of S10 can be referred to Zn5(OH)8(NO3)2·2H2O by comparison with the standard pattern from JCPDS card no. 64-3411, while the broadness of the diffraction peaks implies the weak crystallinity (the red line in Fig. 5d). When the reaction time is reached 1 h, clew-like microstructures appear and coexist with a small quantity of particles in S11 (Fig. 5b and c). Further observation found that the size of particles was in a wide range of 385–769 nm, which suggested that the product shown in Fig. 5a was gradually decomposed with time. Meanwhile, some structures consisted of loosely packed nanoplates can be observed (marked by white arrows). Hereby, we speculate that the clew-like microspheres with loose structure could turn into the structures constituted of densely packed ZnO nanoplates represented in Fig. 1 with prolonging reaction time. As depicted in Fig. 5b, it is apparent that a core has been formed and nanoplates have aligned perpendicularly to the surface of the clew-like microsphere because of the influence of the core-based electric field. The result obtained from the XRD pattern of S11 (the black line in Fig. 5d) was further confirmed that the formation of a partial crystalline ZnO structure because of the decomposition of the precursor.
On the basis of the aforementioned experimental results and analysis, a plausible growth mechanism of the clew-like ZnO microspheres was proposed and the corresponding schematic model was depicted in Fig. 5e. In initial stage, ZnO nuclei were formed by the decomposition of Zn5(OH)8(NO3)2·2H2O precursors. Because the TA molecules had negatively charged carboxyl groups, they could easily adsorb onto Zn2+-terminated (0001) plane in the ZnO crystal, which was similar to previous literature.11,21,23 Therefore, this might block crystal growth along c-axis resulting in the generation of ZnO nanoplates, which has been observed in Fig. 1e. The adsorption of TA molecules can result in the aggregation of nanoplates for charge neutralization and plane stabilization.28 Driving by a strong dipole–dipole interaction between the nanoplates,29 they are inclined to stack together along the c-axis to build a 3D inner core structure. Afterwards, the effects of the electric field generated by the inner core and the minimum surface energy drive nanoplates to assemble to form clew-like ZnO microspheres with loosely packed structures. By increasing the reaction time, the dipole moment generated by the core is gradually equal to that of nanoplates with the growth of ZnO crystals. At the same time, to minimize the exposed high energy planes of nanoplates, the stack way of such subunits would alter to dense structure from loose structure. Finally, clew-like nanoplatelet-based ZnO mesocrystal microspheres are formed. From the result obtained from Fig. 2, it was observed that flower-like structure consisted of rods was generated in the absence of TA. For hexagonal ZnO crystal, it has both polar and nonpolar faces. Compared to nonpolar faces, the polar faces are thermodynamically unstable. Hence, in the hydrothermal reaction without TA, the Zn2+-terminated (0001) plane grows faster than other plans. Moreover, the generated HMT-4H+ cationic by the hydrolysis of HMT can absorb onto the O2−-terminated (000−1) plane to inhibit the growth of the (000−1) plane. Thus, the rod-like structure is easily to create. Finally, the formed ZnO rods are inclined to aggregate to produce flower-like ZnO. According to the analysis for Fig. 4, it was found that the amount of HMT plays a certain effect on the morphology of final ZnO mesocrystal. As previous reports, it usually been accepted that HMT can serve as a the nucleation-control reagent in most of the HMT-assisted hydrothermal systems.30,31 This is because HMT, as a weak base, can decompose to form protonated ammonia which in turn reacts with water to release OH− ions at elevated temperature. It has been reported that the overall reaction of HMT in the solution may be simply formulated as follows:32
| (CH2)6N4 + 6H2O ↔ 4NH3 + 6HCHO | (1) |
| NH3 + H2O ↔ NH3·H2O ↔ NH4+ + OH− | (2) |
With increasing the concentration of HMT, the above equilibrium will be inclined to the right side of those reactions, which is in favor of producing more OH− anions. With introducing more HMT into this system, the more ZnO nucleus was formed by the decomposition of Zn5(OH)8(NO3)2·2H2O precursors. Thus, non-uniform ZnO microspheres formed at high HMT concentration is attributed to the influence of abundant nuclei plus the shortage of raw materials. Moreover, the observation that the holes on ZnO mesocrystal microspheres with large size vanish by attaching nanoplates near the hole implies that the crystallization process of clew-like ZnO mesocrystals, differing from the conventional “ion-by-ion” crystal growth is non-classical crystallization since the existence of the long-range electric field that has been verified by Kniep and co-workers.33
Fig. 6 depicts the N2 adsorption–desorption isotherms and the corresponding BJH pore size distribution curves (insets) of S1 and S7–S9, respectively. All samples displayed typical IV isotherms with a hysteresis loop which is characteristic of a mesoporous structure.34 The BET surface areas and the pore size of such samples were summarized in Table 2. It is apparent that the specific surface area of ZnO mesocrystals increases monotonically with increasing concentration of HMT. Specifically, the measured surface areas of S1, S7, S8 and S9 are 11.0, 16.3, 21.5 and 23.5 m2 g−1, respectively. As observed in the BJH pore size distribution curves (insets of Fig. 6), all samples show the size of pores in the range of 5–20 nm. The average pore diameters of S1, S7, S8 and S9 are 8.2, 7.6, 6.2 and 5.2 nm, respectively, which should be mainly contributed from the pileup of the nanoplates. It could be concluded that the pore diameter could gradually decrease with the increasing of the HMT concentration. It is ascribed to that ZnO mesocrystals obtained at high HMT concentration are consisted of densely packed ZnO nanoplates, which is attributed to the high OH− concentration released from HMT (Fig. 4).
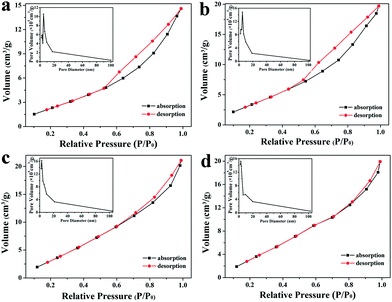 | ||
| Fig. 6 BET adsorption/desorption isotherms and the corresponding BJH pore size distribution curves for different ZnO samples: (a) S1, (b) S7, (c) S8 and (d) S9. | ||
| S1 | S7 | S8 | S9 | |
|---|---|---|---|---|
| Specific surface area (m2 g−1) | 11.0 | 16.3 | 21.5 | 23.5 |
| Average pore diameter (nm) | 8.2 | 7.6 | 6.2 | 5.2 |
In general, the ZnO nanocrystals and microcrystals are excellent photocatalyst for degradation of contaminants.35–37 Moreover, organic dye RhB is a widely used cationic xanthene dye in the textile industry, which has gave rise to severe environmental pollution. Hence, considering the unique mesocrystal structure, the photocatalytic performance of the as-synthesized ZnO mesocrystal microspheres was investigated with RhB as a model pollutant. As depicted in Fig. 7a, a series of absorption spectra of the RhB aqueous solution were characterized by the Hitachi U-4100 spectrophotometer in the presence of S9 under UV light irradiation for different durations. It is found that the intensities of the maximum absorption peaks of RhB at 553 nm diminish gradually with prolonging irradiation time, which indicates the decreasing of RhB concentration. Meanwhile, the color of the aqueous solution almost disappeared when the time was reached to 5 h, signifying the decomposition of the chromophoric structure in RhB. Fig. 7b shows the comparison of the degradation efficiencies in the presence of different ZnO photocatalysts with the irradiation time of 5 h. The degradation efficiency of ZnO was here defined as C/C0, where C0 and C were the concentration after the adsorption–desorption equilibrium in the dark and the temporal concentration of RhB at different irradiation times, respectively. Therefore, a lower value of C/C0 demonstrated a higher photodegradation degree of RhB. The degradation efficiency of ZnO photocatalysts under UV light irradiation for 5 h followed the order of S9 (95.64%) > S8 (94.51%) > S7 (87.19%) > S1 (40.38%). In contrast, S9 and S8 prepared with higher HMT concentration showed higher photocatalytic activity exposed to UV light in comparison with other samples. In our previous work, the degradation efficiency of the hierarchical flower-like ZnO is only about 29.37% after UV light irradiation for 3 h.20 By comparison, the photocatalytic efficiency of S8 and S9 is improved owing to their unique clew-like morphology. However, the efficiency of S8 and S9 is relatively lower than sheet-like and butterfly-like ZnO nanostructures,20,35 which is probably attributed to their lower surface area. To obtain a better understanding about the photocatalytic efficiency, the pseudo-first order kinetics analysis of the various ZnO photocatalysts toward RhB degradation is discussed as follows. As shown in Fig. 7c, the photocatalytic reaction followed the pseudo-first order kinetics on the basis of the values of correlation coefficient (R2, inset of Fig. 7d). As shown in Fig. 7d, it is found that the kinetic constants of as-synthesized ZnO samples followed the order of S9 (0.6215 h−1) > S8 (0.5724 h−1) > S7 (0.4115 h−1) > S1 (0.1046 h−1). Furthermore, the kinetic constants normalized to the measured BET surface area of different samples followed the order of S8 (26.6 h−1 m2 mg−1) > S9 (26.4 h−1 m2 mg−1) > S7 (25.2 h−1 m2 mg−1) > S1 (9.51 h−1 m2 mg−1). The kinetic constants for S8 and S9 were about three times than that of S1 due to their higher surface area. These results showed that the photodegradation efficiency of the as-prepared photocatalysts was closely related to their specific surface area. Generally, the difference in photodegradation ability could be associated with the adsorption capacity of photocatalysts toward RhB dye. The adsorption percentage of different ZnO samples in the dark for 30 min followed the order of S8 (8.14%) > S9 (7.24%) > S7 (6.67%) > S1 (5.70%). It was well known that the lower absorption percentage implied the lower probability of photon-generated carriers transfer between the photocatalysts and the RhB molecules.
As reported in previous study, the degradation efficiency of a photocatalyst was ascribed to its microstructure, such as crystallinity degree, BET specific surface area, and others.35 Generally, a high surface area results in the good adsorption and photocatalytic performance. Therefore, the higher specific surface area of S8 and S9 is the main reason for explaining the enhanced photocatalytic activity. The relevant photocatalytic formula reactions have been reported in previous study.36,37 In our study, a series of reactions are showed in the following eqn (3)–(6) when the as-synthesized ZnO mesocrystal are exposed to a UV light. Under the light irradiation, the ZnO material is excited to produce separated electrons (e−) and holes (h+). Afterwards, the photogenerated electrons in conduction band (CB) are easily trapped by the dissolve oxygen to yield O2˙− radicals. And the photogenerated holes in valence band (VB) are reacted with H2O molecules to produce OH˙ radicals. Finally, the obtained radicals with high activity can degrade RhB into H2O and CO2 small molecules.
| ZnO + hν → ZnO(hvb+ + ecb−) | (3) |
| O2(absorbed) + ecb− → O2˙− | (4) |
| hvb+ + H2O/OH− → OH˙ | (5) |
| O2˙− + OH˙ + RhB(dye) → CO2 + H2O | (6) |
Conclusions
In summary, unique clew-shaped nanoplate-based ZnO mesocrystals have been successfully fabricated by a hydrothermal method in the case of TA as the structure-directing additive through a nonclassical crystallization process. The formation of ZnO mesocrystals was influenced by the dipole moment generated among nanoplates and the electric field derived from a pre-formed inner core. It revealed that several reaction conditions including the reaction temperature, the concentration of TA, Zn(NO3)2 and HMT played significant roles in the size and morphology of ZnO mesocrystals. The photocatalytic property of ZnO samples with different size and pileup degree toward RhB was further explored. The result indicated that the larger specific surface area of products was, the higher photocatalytic activity was.Acknowledgements
This work was supported by the program for Taishan Scholars, the projects from National Natural Science Foundation of China (Grant no. 51572109, 51501071, 51302106, 51402123, and 51402124).References
- D. V. Talapin, J. S. Lee, M. V. Kovalenko and E. V. Shevchenko, Chem. Rev., 2010, 110, 389 CrossRef CAS PubMed.
- J. Ye, W. Liu, J. Cai, S. Chen, X. Zhao, H. Zhou and L. Qi, J. Am. Chem. Soc., 2011, 133, 933 CrossRef CAS PubMed.
- H. Cölfen and M. Antonietti, Angew. Chem., Int. Ed., 2005, 44, 5576 CrossRef PubMed.
- M. J. Bierman, Y. K. A. Lau, A. V. Kvit, A. L. Schmitt and S. Jin, Science, 2008, 320, 1060 CrossRef CAS PubMed.
- M. Grzelczak, J. Vermant, E. M. Furst and L. M. Liz-Marzán, ACS Nano, 2010, 4, 3591 CrossRef CAS PubMed.
- R. Q. Song and H. Cölfen, Adv. Mater., 2010, 22, 1301 CrossRef CAS PubMed.
- J. Zhan, H. P. Lin and C. Y. Mou, Adv. Mater., 2003, 15, 621 CrossRef CAS.
- X. Geng, L. Liu, J. Jiang and S. H. Yu, Cryst. Growth Des., 2010, 10, 3448 CAS.
- M. Adachi, Y. Murata, J. Takao, J. Jiu, M. Sakamoto and F. Wang, J. Am. Chem. Soc., 2004, 126, 14943 CrossRef CAS PubMed.
- M. S. Mo, S. H. Lim, Y. W. Mai, R. K. Zheng and S. P. Ringer, Adv. Mater., 2008, 20, 339 CrossRef CAS.
- Z. Liu, X. D. Wen, X. L. Wu, Y. J. Gao, H. T. Chen, J. Zhu and P. K. Chu, J. Am. Chem. Soc., 2009, 131, 9405 CrossRef CAS PubMed.
- B. Hu, L. H. Wu, S. J. Liu, H. B. Yao, H. Y. Shi, G. P. Li and S. H. Yu, Chem. Commun., 2010, 46, 2277 RSC.
- R. Q. Song, H. Cölfen, A. W. Xu, J. Hartmann and M. Antonietti, ACS Nano, 2009, 3, 1966 CrossRef CAS PubMed.
- Z. Hong, M. Wei, T. Lan and G. Cao, Nano Energy, 2012, 1, 466 CrossRef CAS.
- S. Deng, V. Tjoa, H. M. Fan, H. R. Tan, D. C. Sayle, M. Olivo, S. Mhaisalkar, J. Wei and C. H. Sow, J. Am. Chem. Soc., 2012, 134, 4905 CrossRef CAS PubMed.
- Z. R. Tian, J. A. Voigt, J. Liu, B. Mckenzie and M. J. Mcdermott, J. Am. Chem. Soc., 2002, 124, 12954 CrossRef CAS PubMed.
- Z. Quan and J. Fang, Nano Today, 2010, 5, 390 CrossRef CAS.
- X. Dong, P. Yang and R. Shi, Mater. Lett., 2014, 135, 96 CrossRef CAS.
- X. Dong, P. Yang, J. Wang and B. Huang, ChemPlusChem, 2014, 79, 1681 CrossRef CAS.
- X. Song, Y. Liu, Y. Zheng, K. Ding, S. Nie and P. Yang, Phys. Chem. Chem. Phys., 2016, 18, 4577 RSC.
- F. Ye, Y. Peng, G. Y. Chen, B. Deng and A. W. Xu, J. Phys. Chem. C, 2009, 113, 10407 CAS.
- Z. Li, A. Geßner, J. P. Richters, J. Kalden, T. Voss, C. Kübel and A. Taubert, Adv. Mater., 2008, 20, 1279 CrossRef CAS.
- Y. H. Tseng, H. Y. Lin, M. H. Liu, Y. F. Chen and C. Y. Mou, J. Phys. Chem. C, 2009, 113, 18053 CAS.
- L. Zhou and P. O'Brien, Small, 2008, 4, 1566 CrossRef CAS PubMed.
- S. Sarkar, K. P. McGowan, S. Kuppuswamy, I. Ghiviriga, K. A. Abboud and A. S. Veige, J. Am. Chem. Soc., 2012, 134, 4905 CrossRef PubMed.
- L. Zhou, D. Smyth-Boyle and P. O'Brien, J. Am. Chem. Soc., 2008, 130, 1309 CrossRef CAS PubMed.
- Y. Yang, Y. Yang, H. Wu and S. Guo, CrystEngComm, 2013, 15, 2608 RSC.
- L. Jia, W. Cai, H. Wang and H. Zeng, Cryst. Growth Des., 2008, 8, 4367 CAS.
- Z. Tang, N. A. Kotov and M. Giersig, Science, 2002, 297, 237 CrossRef CAS PubMed.
- B. Wen, Y. Huang and J. J. Boland, J. Phys. Chem. C, 2008, 112, 106 CAS.
- J. Zhang, S. Wang, M. Xu, Y. Wang, B. Zhu, S. Zhang, W. Huang and S. Wu, Cryst. Growth Des., 2009, 9, 3532 CAS.
- W. Guo, T. Liu, L. Huang, H. Zhang, Q. Zhou and W. Zeng, Physica E, 2011, 44, 680 CrossRef CAS.
- P. Simon, D. Zahn, H. Lichte and R. Kniep, Angew. Chem., 2006, 118, 1945 CrossRef.
- S. Zhang, W. Xu, M. Zeng, J. Li, J. Li, J. Xu and X. Wang, J. Mater. Chem. A, 2013, 1, 11691 CAS.
- Y. Hong, C. Tian, B. Jiang, A. Wu, Q. Zhang, G. Tian and H. Fu, J. Mater. Chem. A, 2013, 1, 5700 CAS.
- S. Ma, J. Xue, Y. Zhou and Z. Zhang, J. Mater. Chem. A, 2014, 2, 7272 CAS.
- M. T. Qamar, M. Aslam, I. M. Ismail, N. Salah and A. Hameed, ACS Appl. Mater. Interfaces, 2015, 7, 8757 CAS.
| This journal is © The Royal Society of Chemistry 2016 |