Experimental determination and analysis of gold nanorod settlement by differential centrifugal sedimentation†
Ruimin Wang,
Yinglu Ji,
Xiaochun Wu,
Renxiao Liu,
Lan Chen* and
Guanglu Ge*
CAS Key Laboratory of Standardization and Measurement for Nanotechnology, CAS Center for Excellence in Nanoscience, National Center for Nanoscience and Technology, Beijing, 100190, China. E-mail: chenlan@nanoctr.cn; gegl@nanoctr.cn
First published on 26th April 2016
Abstract
Direct measurement of the sedimentation coefficients of gold nanorods with a controlled shape factor was performed using differential centrifugal sedimentation (DCS). The results serve as a testing ground for various existing models describing non-spherical nanoparticles moving in a centrifugal field based on sphericity and force analysis, and point to further refinement.
Sorting polydispersed colloidal nanoparticles into fractions utilizing their differential movement dictated by their size and shape has made significant progress in recent years.1–4 Among various methods for this purpose, centrifugation-based separation has gained popularity due to its convenient operation and post-treatment. It is also thought to be an efficient approach to maximize the yield of the monodisperse samples.5,6 Good separation performance using density gradient centrifugation, either in aqueous or organic media, has been reported.7,8–11 More recently, Sun et al. reported the separation of one-dimensional nanostructures self-assembled by gold nanoparticles using ultracentrifugation.12 To support these efforts and design more quantitative strategy of purification in terms of selectivity and efficiency, better understanding of nanoparticle sedimentation in a centrifugal field is highly desired.
The challenges in analysing non-spherical nanoparticle sedimentation stem from the orientation-dependent drag force under centrifugal field. For example, inconsistent observations on sedimentation have been reported by many groups when they tried to separate and purify colloidal gold nanorods (GNRs). Murphy et al. have reported that long GNRs settled preferentially down to bottom because of their larger mass when they used high aspect ratio cylindrical GNRs.13 Similar phenomenon has been observed in Sun's work where ultrashort single-walled carbon nanotube mixture was separated by centrifugation, which is consistent with Murphy's theoretical assumption.14 A conclusion has been drawn based on these experimental results that the mass of GNRs is the key factor for GNRs settlement. However, Srinivasarao et al. have observed that the GNRs settled faster with shorter length and wider diameter than those with comparable volume or mass but larger aspect ratio.15 Similar results have also been seen when separating gold nanoparticles (GNPs) from GNRs using traditional centrifugation techniques,6,16 where the sedimentation of GNRs depends more on their diameter than their mass or aspect ratio.15
Several groups have laid the foundation towards a comprehensive model for analysing nanorod sedimentation under centrifugation. Sharma et al.15 first put forward a model for the GNRs sedimentation coefficient. However, as later pointed out,17 predicting sedimentation velocity of the nanorods with given size and aspect ratio using their equations is difficult since the calculation involves solving several indeterminate equations. He et al. later modelled GNRs as Brownian ellipsoidal particles and derived an orientation-averaged sedimentation coefficient along the vertical direction of apparent movement,17 but it is lack of experimental data to verify and support his theory. The suitability of the model to real nanorod samples has yet to be confirmed. Moreover, the experimental validation of these works was mostly based on preparative separation using benchtop centrifuges, which could not provide precisely-measured sedimentation coefficients. Therefore, the relationship among the measurable settlement time, fluid properties including viscosity and density, and the feature size, e.g. aspect ratio and diameter of the GNRs need to be established and refined by additional experimental measurement.
Directly measuring the sedimentation coefficients of nanoparticles using DCS simplifies the procedure and avoids solving complicated Lamn equation in analytical ultracentrifugation (AUC). The magnitude of the sedimentation coefficient for gold nanoparticle (nanorods) is about 103 to 104 S which is out of the upper limit of AUC (∼102 S) for precise measurements. We carried out differential centrifugal sedimentation (DCS) in a disc centrifuge configuration, which is equipped with a high speed centrifugation motor (up to 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm) and an optical detector with a detectable size limit down to 10 nm (CPS Instruments, Inc.).19 DCS has usually been used to analyse particle size and density, where the size resolution of 0.2 nm has been achieved in ligand-passivated Au clusters.19 Recently, Dawson et al. have employed DCS to study the biomolecular–nanoparticle interaction by comparing the difference of the stokes diameter of GNPs obtained before and after the formation of the protein corona.20 These cases have demonstrated that CPS is an efficient technique to obtain time-space dependent parameters such as size and density. Furthermore, our studies indicate that the technique can be used to measure time-dependent parameter directly e.g. sedimentation coefficient assisted by the use of reference materials.
000 rpm) and an optical detector with a detectable size limit down to 10 nm (CPS Instruments, Inc.).19 DCS has usually been used to analyse particle size and density, where the size resolution of 0.2 nm has been achieved in ligand-passivated Au clusters.19 Recently, Dawson et al. have employed DCS to study the biomolecular–nanoparticle interaction by comparing the difference of the stokes diameter of GNPs obtained before and after the formation of the protein corona.20 These cases have demonstrated that CPS is an efficient technique to obtain time-space dependent parameters such as size and density. Furthermore, our studies indicate that the technique can be used to measure time-dependent parameter directly e.g. sedimentation coefficient assisted by the use of reference materials.
Gold nanorods with either equal volume or diameter were synthesized by a seeds-mediated method.18 The UV-visible spectra of the GNRs used in this study are shown in Fig. 1, where Fig. 1a is for GNRs with equal mass/volume, and Fig. 1b is for GNRs with equal diameter. In Fig. 1b, an obvious red-shift of the longitudinal surface plasmon resonance (SPR) peak from 600 to 750 nm is observed while the transverse SPR peak keep unchanged. The aspect ratio of the GNRs calculated using the wavelength of their longitudinal SPR peak λLSPR according to eqn (1) are listed in Table S1.†21
 | (1) |
As stated above, it is relatively straightforward to obtain sedimentation coefficient in DCS. According its definition (s = v/ω2r), since v = dr/dt, s can be rewritten as s = dr/ω2rdt or d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) r/ω2dt. After integration, s becomes ln(RD/R0)/ω2t, where t is the settling time of the particles moving from the starting point R0 to the detector position RD as shown in Scheme 1. ln(RD/R0) is difficult to be experimentally determined but it can be obtained by the use of the reference materials (RM), e.g. monodisperse SiO2 nanoparticles with a certified diameter of 164 nm, and with s calculated from Stokes law.
r/ω2dt. After integration, s becomes ln(RD/R0)/ω2t, where t is the settling time of the particles moving from the starting point R0 to the detector position RD as shown in Scheme 1. ln(RD/R0) is difficult to be experimentally determined but it can be obtained by the use of the reference materials (RM), e.g. monodisperse SiO2 nanoparticles with a certified diameter of 164 nm, and with s calculated from Stokes law.
For spherical particles, the settling velocity can be calculated by Stokes law based on the drag-centrifugal force balance and expressed as
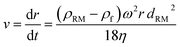 | (2) |
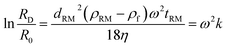 | (3) |
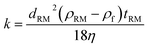 while all other parameters such as dRM, ρRM, ρf, ω and η are known under the experimental conditions. Therefore, the sedimentation coefficient (sm) of nanorods can be obtained using known ln(RD/R0) by:
while all other parameters such as dRM, ρRM, ρf, ω and η are known under the experimental conditions. Therefore, the sedimentation coefficient (sm) of nanorods can be obtained using known ln(RD/R0) by:
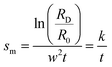 | (4) |
In this work, special attention will be paid on the influence of two factors i.e. mass and aspect ratio of the GNRs on their sedimentation behaviour.
A series of gold nanorods with either equal volume or equal diameter but different aspect ratio have been interrogated using DCS. As shown in Fig. 2a, the settling time of GNRs with equal volume increases gradually with the increase of their aspect ratio. It suggests that nearly-spherical GNRs (with smaller aspect ratio) move faster than their rod-like counterparts. The corresponding sedimentation coefficient (sm) is displayed in Fig. 2c, where sm decreases with the increase of the aspect ratio. On the other hand, as shown in Fig. 2b, for GNRs with equal diameter and different length (aspect ratio), their settling time decreases with the increase of the aspect ratio. As a consequence, the measured sedimentation coefficient increases with the increase of the aspect ratio as shown in Fig. 2d. That is consistent with the usual observation that particles would sediment faster with larger mass or smaller drag area (longitudinally paralleling area).
 | ||
| Fig. 2 The relationship between the aspect ratio and the settling time (a and b) and the measured sedimentation coefficient (c and d) for the GNRs with either equal volume or diameter. | ||
The sedimentation coefficient for a given sample with monodisperse size and known density is generally considered to be a constant under fixed fluid viscosity, density and rotation speed. For GNRs, when the centrifugal force Fc, buoyant force Fb and drag force Fd acting on the particle are transiently balanced, eqn (5) applies, where V is the volume of the particle, ρp and ρf is the density of the particle and fluid, ω is the revolution speed of the disc, r is the distance from the rotation axis to the location of the particle.
 | (5) |
A few assumptions are made here for the nanorod samples used in this study. First, the influence of the ligand on the surface of the GNRs on the sedimentation is ignored, where the aspect ratio of a nanorod is expressed as β = L/dc. Second, the ends of the nanorod are considered as two hemispheres on top of one cylinder with same diameter (dc) as shown in Scheme 1 and, therefore, its volume can be written as 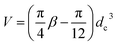 . Finally, the motion of the GNRs follows the swirling direction of the fluid inside the chamber, i.e. a spiral movement of the nanorods whose longitudinal direction is aligned along the tangent of the disc-concentric circle at any time.
. Finally, the motion of the GNRs follows the swirling direction of the fluid inside the chamber, i.e. a spiral movement of the nanorods whose longitudinal direction is aligned along the tangent of the disc-concentric circle at any time.
Various empirical formula based on particle shape factors (ratio between particle surface area and that of equivalent volume) exist in the literature which are quite successful in fitting experimental sedimentation velocities for different particle shapes, including prolate ellipsoidal, cylinders, prisms, spheroids, double conical, and chains of particles. For the drag force in eqn (5)  ,
,  is the projected area of the nanorods in the settlement direction. Drag coefficient CD can be correlated with the Reynolds number, Re, the overall sphericity, the lengthwise sphericity and the transverse sphericity, following Leith22 and Sommerfeld et al.'s formula.23 Note Sommerfeld et al.23 modified the Leith expression (eqn (6)) using lengthwise sphericity (eqn (7)) and better predicted the drag coefficient of spherical particles24 and cylindrical particles25 with the consideration of the particle orientation.
is the projected area of the nanorods in the settlement direction. Drag coefficient CD can be correlated with the Reynolds number, Re, the overall sphericity, the lengthwise sphericity and the transverse sphericity, following Leith22 and Sommerfeld et al.'s formula.23 Note Sommerfeld et al.23 modified the Leith expression (eqn (6)) using lengthwise sphericity (eqn (7)) and better predicted the drag coefficient of spherical particles24 and cylindrical particles25 with the consideration of the particle orientation.
 | (6) |
 | (7) |
For nanorods considered in this study, the sphericity Φ is expressed as the ratio between the surface area  of the volume equivalent sphere with a diameter of
of the volume equivalent sphere with a diameter of  and that (SNR) of the considered nanorod in eqn (8). The crosswise sphericity Φ⊥ is defined as the ratio between the cross-sectional area (Ae) and the laterally projected cross-sectional area (S⊥) as shown in eqn (9). The lengthwise sphericity Φ‖ is the ratio between the cross-sectional area
and that (SNR) of the considered nanorod in eqn (8). The crosswise sphericity Φ⊥ is defined as the ratio between the cross-sectional area (Ae) and the laterally projected cross-sectional area (S⊥) as shown in eqn (9). The lengthwise sphericity Φ‖ is the ratio between the cross-sectional area  of the volume equivalent sphere and the difference between half the surface area and the longitudinally projected cross-sectional area (S‖) of the considered particle as proposed by Sommerfeld and shown in eqn (10). In order to examine the effect of the orientation-based sphericity and drag area on the sedimentation coefficient, several orientations from paralleling to vertical are used to calculate the s. The experimental results show that the calculated s value is closest to the measured one when the GNR is perpendicular to its displacement direction. The Reynolds number Re is expressed as
of the volume equivalent sphere and the difference between half the surface area and the longitudinally projected cross-sectional area (S‖) of the considered particle as proposed by Sommerfeld and shown in eqn (10). In order to examine the effect of the orientation-based sphericity and drag area on the sedimentation coefficient, several orientations from paralleling to vertical are used to calculate the s. The experimental results show that the calculated s value is closest to the measured one when the GNR is perpendicular to its displacement direction. The Reynolds number Re is expressed as  , where η is the averaged viscosity of the fluid at the temperature of measurement.
, where η is the averaged viscosity of the fluid at the temperature of measurement.
 | (8) |
 | (9) |
 | (10) |
The sedimentation coefficients calculated from different literature models (for calculation details see ESI S3†) are summarized in Fig. 3 and compared with our experimental data. For both 3a and 3b, calculated results using Leith's formula (overall and crosswise sphericity) are consistent with experiment in terms of slope, but differ the most in absolute values. Sommerfeld's modified formula appears to provide values closer to the experiments, implying the lengthwise sphericity does give better description for nanorods in this case. For GNRs with equal volume, He's formula provides a close result in values but trend in a reverse way.17 For GNRs with equal diameter, He's work fits experimental results quite well. In short, He's model is ideal for nanorods with equal diameter but not for nanorods with equal volume (mass), which indicates this model more sensitive to the mass rather than shape factor, i.e. the nanorods with ellipsoidal or cylindrical shape are not much different in the calculated s but quite different with different mass. However, Sommerfeld's model provides a better description on the influence of the shape factor of non-spherical particles on their settlement behaviour based on sphericity analysis, but it only applies to the particles in either macro- or micro-scale.26,27 Therefore, new development and improvement is required to calculate the particles sedimentation coefficient in nanoscale based on this model. Moreover, the direct measurement on the sedimentation coefficient of the nanorods has been achieved for the first time, which provides a comparable ground for further theoretical modelling and quantitative analysis.
 | ||
| Fig. 3 Sedimentation coefficient of the GNRs with equal volume (a) and equal diameter (b) determined by different model (see ESI†). | ||
There are several factors that may cause the difference between the measured and calculated values (1) measurement error in nanorods dimensions; (2) the effect of the ligand layer nanorod surface, which increases the cross-sectional area perpendicular to movement direction, and lower the effective density of the particles; (3) the deterioration of the pre-filled gradient fluid caused by the temperature fluctuation and the inter-layer sucrose diffusion, which transfers to inconsistency between reference material calibration and actual measurement on nanorods. To improve the accuracy and precision for both the measurement and theoretical description, analyzing these influencing factors will be the main task in future.
Conclusions
In this work, using high speed DCS and high quality gold nanorods samples synthesized with either equal volume or diameter, the influence of the size and aspect ratio on the settlement behaviour of the gold nanorods has been systemically investigated. The experimental results show that the settling time becomes longer with the increase of the aspect ratio for the GNRs with equal volume (mass) and the decrease of the length (aspect ratio) for those with equal diameter. Experimentally, it is the first attempt to measure the sedimentation coefficient of non-spherical particles directly and the results provide a comparable base to evaluate the s value predicted by other models. Insightful trends have been observed suggesting this is a reliable way to measure and predict the sedimentation behaviour of the rod-like particles moved under a vertically mounted centrifugal field, i.e. CPS disc centrifugation. Further study on analysing influencing factors and determining the sedimentation coefficient of other non-spherical particles is underway.Acknowledgements
This work is supported by the Strategic Priority Research Program of the Chinese Academy of Sciences, Grant No. XDA09040400.Notes and references
- C. Hu and Y. Chen, Chem. Eng. J., 2015, 271, 128–134 CrossRef CAS.
- J. Walter, T. Thajudeen, S. Suss, D. Segets and W. Peukert, Nanoscale, 2015, 7, 6574–6587 RSC.
- S. K. Balasubramanian, L. M. Yang, L. Y. L. Yung, C. N. Ong, W. Y. Ong and L. E. Yu, Biomaterials, 2010, 31, 9023–9030 CrossRef CAS PubMed.
- K. Park, H. Koerner and R. A. Vaia, Nano Lett., 2010, 10, 1433–1439 CrossRef CAS PubMed.
- V. A. Turek, L. N. Elliott, A. I. I. Tyler, A. Demetriadou, J. Paget, M. P. Cecchini, A. R. Kucernak, A. A. Kornyshev and J. B. Edel, ACS Nano, 2013, 7, 8753–8759 CrossRef CAS PubMed.
- S. Li, Z. Chang, J. Liu, L. Bai, L. Luo and X. Sun, Nano Res., 2011, 4, 723–728 CrossRef CAS.
- S. H. Ko, F. Vargas-Lara, P. N. Patrone, S. M. Stavis, F. W. Starr, J. F. Douglas and J. A. Liddle, Soft Matter, 2014, 10, 7370–7378 RSC.
- X. Sun, S. M. Tabakman, W. S. Seo, L. Zhang, G. Zhang, S. Sherlock, L. Bai and H. Dai, Angew. Chem., Int. Ed., 2009, 48, 939–942 CrossRef CAS PubMed.
- L. Bai, X. J. Ma, J. F. Liu, X. M. Sun, D. Y. Zhao and D. G. Evans, J. Am. Chem. Soc., 2010, 132, 2333–2337 CrossRef CAS PubMed.
- X. M. Sun, D. C. Luo, J. F. Liu and D. G. Evans, ACS Nano, 2010, 4, 3381–3389 CrossRef CAS PubMed.
- O. Akbulut, C. R. Mace, R. V. Martinez, A. A. Kumar, Z. Nie, M. R. Patton and G. M. Whitesides, Nano Lett., 2012, 12, 4060–4064 CrossRef CAS PubMed.
- X. Qi, M. Li, Y. Kuang, C. Wang, Z. Cai, J. Zhang, S. You, M. Yin, P. Wan, L. Luo and X. Sun, Chem.–Eur. J., 2015, 21, 7211–7216 CrossRef CAS PubMed.
- N. R. Jana, L. Gearheart and C. J. Murphy, J. Phys. Chem. B, 2001, 105, 4065–4067 CrossRef CAS.
- X. Sun, S. Zaric, D. Daranciang, K. Welsher, Y. Lu, X. Li and H. Dai, J. Am. Chem. Soc., 2008, 130, 6551–6555 CrossRef CAS PubMed.
- V. Sharma, K. Park and M. Srinivasarao, Proc. Natl. Acad. Sci. U. S. A., 2009, 106, 4981–4985 CrossRef CAS PubMed.
- V. Sharma, K. Park and M. Srinivasarao, Mater. Sci. Eng., R, 2009, 65, 1–38 CrossRef.
- B. Xiong, J. Cheng, Y. Qiao, R. Zhou, Y. He and E. S. Yeung, J. Chromatogr. A, 2011, 1218, 3823–3829 CrossRef CAS PubMed.
- B. Nikoobakht and M. A. El-Sayed, Chem. Mater., 2003, 15, 1957–1962 CrossRef CAS.
- Z. Krpetic, A. M. Davidson, M. Volk, R. Levy, M. Brust and D. L. Cooper, ACS Nano, 2013, 7, 8881–8890 CrossRef CAS PubMed.
- P. M. Kelly, C. Aberg, E. Polo, A. O'Connell, J. Cookman, J. Fallon, Z. Krpetic and K. A. Dawson, Nat. Nanotechnol., 2015, 10, 472–479 CrossRef CAS PubMed.
- S. Link, M. A. El-Sayed and M. B. Mohamed, J. Phys. Chem. B, 2005, 109, 10531–10532 CrossRef CAS.
- D. Leith, Aerosol Sci. Technol., 1987, 6, 153–161 CrossRef CAS.
- A. Holzer and M. Sommerfeld, Powder Technol., 2008, 184, 361–365 CrossRef.
- A. Terfous, A. Hazzab and A. Ghenaim, Powder Technol., 2013, 239, 12–20 CrossRef CAS.
- X. H. Guo, J. Z. Lin and D. M. Nie, Particuology, 2011, 9, 114–120 CrossRef.
- L. W. Rong, Z. Y. Zhou and A. B. Yu, Powder Technol., 2015, 285, 146–156 CrossRef CAS.
- W. Devarrewaere, D. Foque, U. Heimbach, D. Cantre, B. Nicolai, D. Nuyttens and P. Verboven, Environ. Sci. Technol., 2015, 49, 7310–7318 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra07829j |
| This journal is © The Royal Society of Chemistry 2016 |


