A novel amphoteric ion-exchange membrane prepared by the pore-filling technique for vanadium redox flow batteries†
Abstract
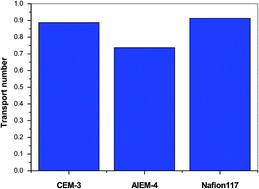
A novel amphoteric ion-exchange membrane (AIEM) was prepared through the pore-filling technique, for vanadium redox flow battery (VRBs) applications. Their physico-chemical properties including vanadium ion permeability and electrochemical properties were investigated as a function of cross-linking degrees. Vanadium ion permeability was effectively suppressed by the quaternary ammonium group-induced Donnan exclusion effect and was only 7.7% of that of Nafion117. Finally, open circuit voltage measurements showed that VRBs assembled with the AIEM maintained voltages >1.3 V for over 43 h, which was superior to the cation exchange membranes (CEMs), studied. In addition, the VRBs with AIEM-4 exhibited higher columbic efficiency (ca. 98%) and energy efficiency (ca. 89%) than those with CEMs. Therefore, it may be concluded that pore-filled AIEMs are more suitable for use in VRBs than pore-filled CEMs. This work provides a convenient method for preparing new types of IEMs for various applications.

 Please wait while we load your content...
Please wait while we load your content...