Fe3+-induced bioinspired chitosan hydrogels for the sustained and controlled release of doxorubicin†
Abstract
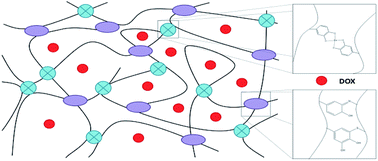
In this study, a novel Fe3+-induced bioinspired chitosan hydrogel was developed to easily deliver the anticancer drug, doxorubicin (DOX). Catechol–chitosan conjugates (CCS) and an N-acetyl cysteine–chitosan conjugate (NACCS) were synthesized and used to prepare the hydrogels. The addition of NACCS accelerated the gelation rate and improved the mechanical strength of the hydrogels, due to the Michael addition reaction between NACCS and the oxidation product of CCS. This study demonstrated that the Fe3+-induced CCS–NACCS hydrogel was a dual covalent-coordination crosslinking system under acidic conditions. Release curves for DOX were evaluated at different pH values, and the release kinetics and mechanism were also investigated. The CCS–NACCS hydrogel showed no obvious toxicity and the DOX released from the hydrogel could effectively inhibit the proliferation of several kinds of tumor cells.

 Please wait while we load your content...
Please wait while we load your content...