Highly efficient As(V)/Sb(V) removal by magnetic sludge composite: synthesis, characterization, equilibrium, and mechanism studies†
Li Wanga,
Jing-mei Wanga,
Ren Zhanga,
Xin-gang Liua,
Guo-xin Songa,
Xiao-feng Chena,
Yi Wang*ab and
Ji-lie Kongb
aCenter of Analysis and Measurement, Fudan University, Shanghai 200433, China. E-mail: wangyifd@fudan.edu.cn; Fax: +86-21-65643010; Tel: +86-21-65643010
bDepartment of Chemistry, Innovative Center of Chemistry for Energy Materials, Fudan University, Shanghai 200433, China
First published on 25th April 2016
Abstract
The development of inexpensive and effective adsorbents for the highly efficient removal of arsenic (As) and antimony (Sb) from water is of great importance for the environment and human health. In the present study, magnetic bio-sludge (MS) containing activated sludge and magnetite (Fe3O4) nanoparticles (diameter: 2–25 nm) was investigated as an adsorbent. MS was synthesized by a facile co-precipitation method and characterized using various physicochemical methods. MS featured a macroporous structure with a surface area of 78 m2 g−1 and a pore volume of 0.53 cm3 g−1. MS could be readily and easily separated from water using an external magnetic field. Importantly, MS could be applied for the removal of As(V) and Sb(V) contaminants from water. It exhibited high stability, ultrafast adsorption/separation rates, and a large adsorption capacity. Moreover, it achieved near-complete removal of As(V) and Sb(V) in 10 μg L−1 solutions. Its high adsorption performance was almost unaffected by the presence of ion interferents in solution. Thus, the present study demonstrates the potential of MS as an adsorbent for the removal of As(V) and Sb(V) from groundwater to meet environmental and public health standards.
1. Introduction
Rapid industrialization has led to increased groundwater contamination by heavy metals (e.g., As, Sb, Hg, Cd), which are harmful to the environment and human health.1,2 Among heavy metal contaminants, arsenic (As) is the most toxic and, often occurs together with antimony (Sb).3 Of the various species of As/Sb, As(V) and Sb(V) oxyanions are predominant and stable in oxic systems, possibly transforming to more toxic species under certain conditions, thereby presenting a more serious threat.4 The World Health Organization (WHO) has set thresholds for safe drinking water of 10 μg L−1 (10 ppb) for As and 5 μg L−1 (5 ppb) for Sb.5,6 Globally, and especially in South and Southeast Asia, tens of millions of people are at risk of exposure to these contaminants in groundwater.7 Therefore, it is important to remove them from water. Various technologies, including ion exchange,8 oxidation,9 electro-chemical precipitation,10 membrane separation,11 and adsorption,12 have been used to remove As and Sb from aqueous media. Among the proposed techniques, magnetic iron oxide nanomaterials have attracted much attention because of their effective adsorption performance and convenient separation capacity. As/Sb-contaminated water primarily originated from acid mine drainage.13 Iron oxide nanomaterials with small particle sizes and abundant adsorption sites are often unstable (i.e., they can dissolve or oxidize), and are difficult to be applied in practical treatment systems. By comparison, large nanoparticles (NPs) lack sufficient sites for As/Sb adsorption.14,15 Traditional carbon or silica nanomaterials, such as carbon nanotubes,16 graphene,17 mesoporous carbon,18 and macroporous silica,19 have been widely used as substrates to stabilize magnetic NPs for As/Sb removal. However, the rigid substrates, easy aggregation and complex preparation procedures are often involved in the application of these nanomaterials, which consequently cannot achieve good removal efficiency. Furthermore, magnetic carbon- or silica-based nanomaterials are expensive, thus limiting their application in the purification of contaminated water.20 Therefore, more effective and cheaper magnetic adsorbents are required.It is well known that activated sludge is abundant and inexpensive. It can easily be obtained from wastewater treatment plants and has many microorganisms on its surface. It is commonly used to treat organic or eutrophicated wastewater, which often has a high chemical oxygen demand or a high total nitrogen and phosphorus content.21,22 Recently, although activated sludge has often been used as a precursor for environmental applications.23–25 most of these strategies focus on the employment of activated sludge to drive activated carbons with high surface area,26–28 which thus fall into the category of inorganic rigid substrate with above-mentioned defects. Owing to the flexible structure, large surface area, low thermal conductance, and abundant surface sites for anchoring of the nanoparticles, direct application of activated sludge in water treatment will be more energy-saving and promising. Especially, combing magnetic iron oxide NPs (M) with activated sludge (S) to produce a novel adsorbent i.e., magnetic sludge (MS) for removing most toxic As and Sb from water, will be much significant. In this novel adsorbent, this magnetic bio-sludge can act as a substrate to disperse and stabilize the magnetic iron oxide NPs with fully exposed adsorption sites and increased surface area. Negative charged sludge can strongly adsorb positively charged iron species, subsequently producing more active sites for the complete removal of As and Sb. The microorganisms on the sludge may act as a buffering agent against acid dissolution of the active NPs. Furthermore, MS can be easily dispersed in water and readily separated using a magnetic field because of the uniform distribution of magnetic NPs on the hydrophilic sludge.
Based on the environment friendly properties, natural resource availability, and low cost of raw materials, the aims of the present study are to (1) prepare and synthesize MS via a facile co-precipitation method, (2) characterize and identify the morphology, structure, composition, and stability of MS using various physicochemical methods, (3) investigate and compare the removal performance of As(V)/Sb(V) by MS by examining adsorption isotherms, kinetics, and mechanisms, and the effect of influencing factors, and (4) elucidate the potential of this novel composite material in quick separation and purification of drinking water.
2. Experimental
2.1 Chemical reagents
FeCl3·6H2O, FeCl2·4H2O, NH3·H2O (30%), and H4N2·H2O (50%) were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). Na2HAsO4·7H2O and KSb(OH)6 were purchased from Sigma-Aldrich (St. Louis, MO, USA). Activated sludge was obtained from Quyang wastewater treatment plant (Yangpu District, Shanghai, China).2.2 Synthesis of MS
MS was synthesized by a facile co-precipitation method. Typically, 0.7 g of dry activated sludge, which was obtained by washing the wet sludge and subsequent freeze-drying, was first dispersed in 250 mL of aqueous FeCl3 (10 mmol L−1) and FeCl2 (5 mmol L−1). After stirring for 10 min, the solution was adjusted to pH 10 with ammonia. Then, 5 mL of hydrazine hydrate was added, and the mixture was stirred for 5 h at 90 °C, generating MS as a black solid. The latter product was washed with water and magnetically separated from the washing solution. The washing process was repeated multiple times to remove any unreacted chemicals. For the comparison, pristine Fe3O4 NPs were also synthesized using the same procedure, however, in the absence of sludge. While calcined MS was obtained by heating MS at 550 °C for 5 h under N2 atmosphere.2.3 Characterization
X-ray diffraction (XRD) patterns were recorded on a PANalyticalX'pert PRO diffractometer (PANalytical, Almelo, The Netherlands) using Cu Kα radiation at 40 kV and 60 mA. X-ray photoelectron spectroscopy (XPS) was performed on a RBD upgraded PHI-5000C ESCA system (PerkinElmer, Waltham, MA, USA) using Al Kα radiation (hν = 1486.6 eV) as the X-ray source for excitation. Morphologic observation was performed using scanning electron microscopy (SEM) (Zeiss Ultra 55, Germany). Transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM), energy-dispersive X-ray spectroscopy (EDS), and electron diffraction patterns were obtained on a JEM-2010 (Jeol, Tokyo, Japan) operating at an acceleration voltage of 200 kV. Magnetic hysteresis curves, zero-field-cooled and field-cooled magnetization curves were obtained on a MPMS (SQUID) vibrating sample magnetometer (VSM) system (Quantum Design Corp., San Diego, CA, USA). Raman spectra were obtained on a Labram-1B (Dilor, France) confocal Raman microscope using a 533 nm wavelength incident laser light. Fourier transform infrared (FTIR) spectroscopy, employing KBr pellets was conducted on a Nicolet-470 FTIR spectrometer (Nicole, USA) in the spectral range of 400–4000 cm−1. Thermogravimetric analysis (TG) was performed on a Pyris Diamond TG/differential thermal analysis (DTA) thermogravimetric analyzer (PerkinElmer, USA). Nitrogen adsorption–desorption isotherms were measured using a Tristar 3000 (Micromeritics, USA). Inductively coupled plasma atomic emission spectrometry (ICP-AES) was performed on Optima 8000 (PerkinElmer, USA), the zero point charge, pHzpc of MS was determined according to the method presented therein.29 Photographs were taken using a digital camera (Panasonic DMC-FX2, Japan).2.4 Adsorption studies
Na2HAsO4·7H2O and KSb(OH)6 were used as sources of As(V) and Sb(V), respectively. Stock solutions of As(V) and Sb(V) at a concentration of 1000 mg L−1 were prepared by dissolving the powders in deionized water and diluting to the required concentrations for subsequent tests. The solution pH was adjusted by adding 0.1 mol L−1 NaOH or HCl. To study the effect of pH, various pHs from 0.9 to 11.5 were used. Typically, 1.4 g L−1 of sorbent was mixed with 10 mg L−1 of As(V) or Sb(V), followed by shaking (200 rpm) at 25 °C for 5 h. For comparison, the adsorption performance of pristine Fe3O4 NPs and activated sludge was also examined at pH 2.6, using a sorbent dosage of 0.7 g L−1 and initial target ions concentrations of 0.1 and 5 mg L−1. Various concentrations of As(V)/Sb(V) (10, 50, 100, and 500 μg L−1) were selected to investigate the adsorption capacity of MS at pH 2.6 at 25 °C, and a sorbent dosage of 0.7 g L−1. The effect of co-existing ions was investigated using four common anions (Cl−, NO3−, SO42−, PO43−) with concentrations ranging from 1 to 50 mmol L−1 at pH 2.6 at 25 °C. The supernatants were sampled as required, subjected to magnetic separation or centrifugation (Feige TGL-16GB, China), and ion concentrations were determined by ICP-AES.3. Results and discussion
3.1 Synthesis, structure and properties of MS
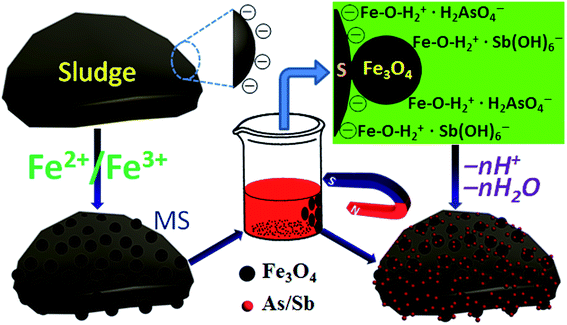
The synthesis of the MS and removal procedure of As(V)/Sb(V) are illustrated in Scheme 1. A simple co-precipitation method was used to prepare MS. The adsorption performance of MS was compared with that of activated sludge (without NPs) and magnetic iron oxide NPs (without activated sludge). | ||
| Fig. 1 (a) XRD patterns of sludge (S), pristine magnetite nanoparticles (M), and magnetic sludge (MS). (b) Survey, (c) C 1s, and (d) Fe 2p XPS spectra of MS. | ||
FTIR analysis confirmed the presence of hydrated hydroxyls (3410 cm−1). Additionally, the results revealed that MS was a composite of magnetic Fe3O4 NPs and activated sludge, as evidenced by Fe–O (563 cm−1) stretching vibrations, corresponding to magnetite, and C–H (asymmetric: 2923 cm−1, symmetric: 2850 cm−1), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (1642 cm−1), and C–O (1036 cm−1) stretching vibrations, corresponding to the microorganism in the sludge (Fig. S1b†).34
C (1642 cm−1), and C–O (1036 cm−1) stretching vibrations, corresponding to the microorganism in the sludge (Fig. S1b†).34
The XPS spectrum of MS in Fig. 1b displayed four typical peaks at C 1s, N 1s, O 1s, and Fe 2p binding energies. The deconvoluted C 1s spectrum (Fig. 1c) revealed the presence of functional groups that originated from the microorganism, namely C–C at 284.7 eV, C–O/C–N at 286.4 eV and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O at 288.8 eV.35 The deconvoluted Fe 2p spectrum (Fig. 1d) showed typical peaks of magnetite Fe3O4, namely Fe 2p3/2 at 711.5 eV and Fe 2p1/2 at 724.9 eV; however, peaks relating to other iron (oxide impurities) were absent.17,36
O at 288.8 eV.35 The deconvoluted Fe 2p spectrum (Fig. 1d) showed typical peaks of magnetite Fe3O4, namely Fe 2p3/2 at 711.5 eV and Fe 2p1/2 at 724.9 eV; however, peaks relating to other iron (oxide impurities) were absent.17,36
The incorporation of magnetic Fe3O4 into the layers of sludge in MS was further supported by the properties of calcined MS using Raman spectroscopy analysis. As shown in the Raman spectra in Fig. S3a,† in addition to the characteristic magnetite peaks at 338, 496, and 655 cm−1, the calcined MS displayed signals similar to those relating to the symmetry A1g mode and E2g mode of defected graphene at 1330 cm−1 (D-band) and 1580 cm−1 (G-band),37 respectively. These signals revealed the formation of a Fe3O4@C composite by graphitization of the microorganisms on the sludge. Moreover, the SEM, TEM, and HRTEM images (Fig. S3b and c†) showed that the highly crystalline iron oxide NPs were dispersed within the mesocellular structure of the carbon matrix, which is a typical feature of magnetite NPs on the surface of activated sludge after calcination.30 The presence of microorganism on the activated sludge and highly dispersed Fe3O4 NPs determined the surface area (78 m2 g−1), pore volume (0.53 cm3 g−1), and macropore diameter (44 nm) of MS. These results were obtained from the N2 adsorption–desorption type III isotherm and the pore size distribution curve shown in Fig. S4a.†
3.2 Adsorption studies
 | ||
| Fig. 4 Removal efficiency of As(V) and Sb(V) by MS as a function of pH and corresponding loss of Fe from MS. (T: 25 °C, C0: 10 mg L−1, contact time: 5 h, rotation speed: 200 rpm). | ||
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qt − (k1/2.303)t, qt − (k1/2.303)t,
| (1) |
| t/qt = 1/(k2qe2) + t/qe, | (2) |
As deduced from Table 2, the adsorption kinetics of MS at different initial concentrations of As(V) and Sb(V) was best described by the pseudo-second-order model. Moreover, negative correlations between the rate constants (k2) and the initial concentrations of As(V) and Sb(V) confirmed that the adsorption process was mass transfer-dependent, especially at the high initial ions concentrations. Although the half-adsorption time (t1/2) increased with increases in the initial As(V) and Sb(V) concentration, overall, it was low (i.e., 4.1 min for As(V) adsorption and 2.6 min for Sb(V) adsorption) at the highest concentration tested (i.e., 20 mg L−1). It is expected that this property and the fast magnetic separation (10 s) ability will greatly reduce the time required for As(V) and Sb(V) removal in practical applications.
| MS | C0 (mg L−1) | qe,exp (mg g−1) | Pseudo-first-order | Pseudo-second-order | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| qe,calc (mg g−1) | k1 (min−1) | r2 | qe,calc (mg g−1) | k2 (g mg−1 min−1) | h (mg g−1 min−1) | t1/2 (min) | r2 | |||
| As(V) | 5 | 6.786 | 0.701 | 0.081 | 0.832 | 6.803 | 0.274 | 12.7 | 0.54 | 1.000 |
| 10 | 12.13 | 3.418 | 0.088 | 0.919 | 12.30 | 0.042 | 6.4 | 1.9 | 0.999 | |
| 20 | 15.1 | 5.366 | 0.051 | 0.869 | 15.38 | 0.016 | 3.8 | 4.1 | 1.000 | |
| Sb(V) | 5 | 6.758 | 0.715 | 0.078 | 0.838 | 6.805 | 0.273 | 12.6 | 0.54 | 1.000 |
| 10 | 13.09 | 2.601 | 0.078 | 0.956 | 13.20 | 0.063 | 11.0 | 1.2 | 1.000 | |
| 20 | 21.65 | 5.568 | 0.051 | 0.826 | 22.22 | 0.017 | 8.4 | 2.6 | 1.000 | |
| Ce/qe = Ce/qm + 1/(bqm), | (3) |
The Freundlich isotherm model, which accounts for multilayered adsorption on heterogeneous surfaces, is described as follows:42
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = 1/n qe = 1/n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce + ln Ce + ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kF kF
| (4) |
Both isotherms models, especially the Langmuir model (R2 > 0.990), were good fits for the adsorption data of MS obtained at different initial ions concentrations and temperatures. The associated adsorption parameters are shown in Table 3. As observed, the maximum monolayer uptake amount of Sb(V) calculated from the Langmuir fit was more than two times that of As(V) at all tested temperatures. The monolayer uptake of both As(V) and Sb(V) increased with increasing temperatures. The dependence of the adsorption uptake on temperature and concentration suggests that the process is endothermic and mass transfer-dependent. Therefore, the low thermal conductance (for heat preservation) and porous framework (for mass transfer) of activated sludge in MS are important operating features of the adsorbent toward As(V) and Sb(V) removal. Thus, the adsorption of the ions can be controlled by adjusting the experimental parameters.
| MS | T (°C) | Langmuir model | Freundlich model | ||||
|---|---|---|---|---|---|---|---|
| qm (mg g−1) | b (L mg−1) | r2 | kF (mg g−1) | n | r2 | ||
| As(V) | 5 | 13.7 | 1.03 | 0.996 | 6.44 | 4.15 | 0.960 |
| 25 | 18.5 | 1.10 | 0.994 | 8.31 | 3.80 | 0.929 | |
| 50 | 21.3 | 1.38 | 0.993 | 10.8 | 4.43 | 0.976 | |
| Sb(V) | 5 | 26.3 | 0.97 | 0.996 | 9.74 | 2.78 | 0.970 |
| 25 | 35.7 | 1.00 | 0.990 | 12.9 | 2.95 | 0.952 | |
| 50 | 43.5 | 1.64 | 0.990 | 18.7 | 2.50 | 0.910 | |
3.3 Mechanism of removal of As(V) and Sb(V)
The removal of As(V) and Sb(V) by MS could be explained by a charge-mediated enhanced adsorption process followed by dehydration (Scheme 1). As shown in Fig. S8,† the pHzpc of MS is 7. As(V) and Sb(V) can hydrolyze into different species, e.g., H2AsO4−, HAsO42−, AsO43−, and Sb(OH)6−, at different pHs (Fig. 8). At pH values greater than pHzpc, MS is negatively charged and thus would repel As(V) and Sb(V) anions, thereby leading to a reduced adsorption amount. As pH decreases, the surface of MS is gradually protonated, leading to a positively charged surface. As(V) and Sb(V) adsorption thus increases because of electrostatic attraction to the positively charged MS surface. However, in a strong acid environment (e.g., pH < 1.7), some Fe species are expected to be discharged from MS, and the active sites on MS affinity to the As(V) and Sb(V) anions decreases, consequently leading to a reduced removal efficiency. Therefore, pH 2.6 was determined as the optimum pH for As(V) and Sb(V) removal. At this pH, Fe species in MS are protonated (e.g.,![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Fe–OH2+). Thus, H2AsO4− and Sb(OH)6− species are dominant and can be attracted by positively charged
Fe–OH2+). Thus, H2AsO4− and Sb(OH)6− species are dominant and can be attracted by positively charged ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Fe–OH2+, forming outer-sphere complexes (i.e.,
Fe–OH2+, forming outer-sphere complexes (i.e., ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Fe–OH2+·H2AsO4− and
Fe–OH2+·H2AsO4− and ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Fe–OH2+·Sb(OH)6−) (Scheme 1). Dehydration then occurs, resulting in formation of inner-sphere complexes (i.e.,
Fe–OH2+·Sb(OH)6−) (Scheme 1). Dehydration then occurs, resulting in formation of inner-sphere complexes (i.e., ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Fe–O–HAsO3− and
Fe–O–HAsO3− and ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Fe–O–Sb(OH)5−) with stronger chemical bonds.46,47 The non-dependency of inner-sphere complex formation on ionic strength supported the lack of interfering effects from anions (Cl−, NO3−, and SO42−). Moreover, sludge can attract many positively charged Fe species during the synthesis of MS, which would enhance the adsorption of negatively charged target ions by MS further. Therefore, MS was determined as an excellent adsorbent for the highly efficient removal of As(V) and Sb(V) removal (Scheme 1).
Fe–O–Sb(OH)5−) with stronger chemical bonds.46,47 The non-dependency of inner-sphere complex formation on ionic strength supported the lack of interfering effects from anions (Cl−, NO3−, and SO42−). Moreover, sludge can attract many positively charged Fe species during the synthesis of MS, which would enhance the adsorption of negatively charged target ions by MS further. Therefore, MS was determined as an excellent adsorbent for the highly efficient removal of As(V) and Sb(V) removal (Scheme 1).
4. Conclusions
A novel magnetic sludge composite material for the efficient and inexpensive removal of As(V)/Sb(V) was successfully prepared by a facile co-precipitation method. Combining highly crystalline and dispersed magnetite NPs (diameter: 2–25 nm) with the microorganisms of activated sludge produced a superparamagnetic sludge that could be quickly separated from solution using an external magnetic field. The sludge had a macroporous structure with a surface area of 78 m2 g−1 and a pore volume of 0.53 cm3 g−1. MS featured a low thermal conductance and abundant exposed adsorption sites, and the NPs on the surface were tightly packed. Such characteristics were responsible for the high stability of MS in acid, fast separation (within 10 s) of MS from solution, and large adsorption capacity of MS. Furthermore, the adsorption performance of MS was almost unaffected by interfering effects from the presence of other ions in solution. Specifically, MS achieved near-complete removal of As(V)/Sb(V) at 10 μg L−1. It is believed that the presently developed magnetic sludge can be effectively applied in existing techniques for removing As(V)/Sb(V) from contaminated water to meet water quality standards for the environment and human health.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 21303022 and 21175029) and the Natural Science Foundation of Shanghai City of China (Grant No. 13ZR1451400).References
- L. Rodríguez-Lado, G. Sun, M. Berg, Q. Zhang, H. Xue, Q. Zheng and J. Ca, Science, 2013, 341, 866–868 CrossRef PubMed.
- M. L. Polizzotto, B. D. Kocar, S. G. Benner, M. Sampson and S. Fendorf, Nature, 2008, 454, 505–508 CrossRef CAS PubMed.
- T. Wen, X. Wu, X. Tan, X. Wang and A. Xu, ACS Appl. Mater. Interfaces, 2013, 5, 3304–3311 CAS.
- B. Dousova, F. Buzek, L. Herzogova, V. Machovic and M. Lhotka, Eur. J. Soil Sci., 2015, 66, 74–82 CrossRef CAS.
- M. Zhang, Bioresour. Technol., 2013, 130, 457–462 CrossRef CAS PubMed.
- G. Ungureanu, S. Santos, R. Boaventura and C. Botelho, J. Environ. Manage., 2015, 151, 326–342 CrossRef CAS PubMed.
- Y. Wang, G. Morin, G. Ona-Nguema and G. E. Brown Jr, Environ. Sci. Technol., 2014, 48, 14282–14290 CrossRef CAS PubMed.
- T. M. Clancy, K. F. Hayes and L. Raskin, Environ. Sci. Technol., 2013, 47, 10799–10812 CrossRef CAS PubMed.
- L. Yu, X. Peng, F. Ni, J. Li, D. Wang and Z. Luan, J. Hazard. Mater., 2013, 246, 10–17 CrossRef PubMed.
- J. F. Rivera, C. Bucher, E. Saint-Aman, B. L. Rivas, M. del Carmen Aguirre, J. Sanchez, I. Pignot-Paintrand and J. C. Moutet, Appl. Catal., B, 2013, 129, 130–136 CrossRef CAS.
- H. R. Pant, H. J. Kim, M. K. Joshi, B. Pant, C. H. Park, J. I. Kim, K. Hui and C. S. Kim, J. Hazard. Mater., 2014, 264, 25–33 CrossRef CAS PubMed.
- L. Li, G. Zhou, Z. Weng, X. Y. Shan, F. Li and H. M. Cheng, Carbon, 2014, 67, 500–507 CrossRef CAS.
- J. Liu, X. Huang, J. Liu, W. Wang, W. Zhang, F. Dong and K. Hudson-Edwards, Mineral. Mag., 2014, 78, 73–89 CrossRef CAS.
- J. Yang, H. Zhang, M. Yu, I. Emmanuelawati, J. Zou, Z. Yuan and C. Yu, Adv. Funct. Mater., 2014, 24, 1354–1363 CrossRef CAS.
- S. Zhang, X. Y. Li and J. P. Chen, Carbon, 2010, 48, 60–67 CrossRef CAS.
- J. Ma, Z. Zhu, B. Chen, M. Yang, H. Zhou, C. Li, F. Yu and J. Chen, J. Mater. Chem. A, 2013, 1, 4662–4666 CAS.
- V. Chandra, J. Park, Y. Chun, J. W. Lee, I. C. Hwang and K. S. Kim, ACS Nano, 2010, 4, 3979–3986 CrossRef CAS PubMed.
- Z. Wu, W. Li, P. A. Webley and D. Zhao, Adv. Mater., 2012, 24, 485–491 CrossRef CAS PubMed.
- W. Ma, F. Meng, Z. Cheng, G. Xin and S. Duan, Bioresour. Technol., 2015, 356–359 CrossRef CAS PubMed.
- S. Zhang, M. Zeng, J. Li, J. Li, J. Xu and X. Wang, J. Mater. Chem. A, 2014, 2, 4391–4397 CAS.
- M. F. Zuthi, Bioresour. Technol., 2013, 139, 363–374 CrossRef CAS PubMed.
- M. F. Dignac, P. Ginestet, D. Rybacki, A. Bruchet, V. Urbain and P. Scribe, Water Res., 2000, 34, 4185–4194 CrossRef CAS.
- A. Bagreev, S. Bashkova, D. C. Locke and T. J. Bandosz, Environ. Sci. Technol., 2001, 35, 1537–1543 CrossRef CAS PubMed.
- C. C. Liu, M. Kuang-Wang and Y. S. Li, Ind. Eng. Chem. Res., 2005, 44, 1438–1445 CrossRef CAS.
- L. Gu, N. W. Zhu, D. F. Zhang, Z. Y. Lou, H. P. Yuan and P. Zhou, Bioresour. Technol., 2013, 136, 719–724 CrossRef CAS PubMed.
- L. Gu, N. W. Zhu, H. Q. Guo, S. Q. Huang, Z. Y. Lou and H. P. Yuan, J. Hazard. Mater., 2013, 246–247, 145–153 CrossRef CAS PubMed.
- P. Devi and A. K. Saroha, Bioresour. Technol., 2014, 169, 525–531 CrossRef CAS PubMed.
- P. Devi and A. K. Saroha, Chem. Eng. J., 2015, 271, 195–203 CrossRef CAS.
- S. Yadav, V. Srivastava, S. Banerjee, C. H. Weng and Y. C. Sharma, Catena, 2013, 100, 120–127 CrossRef CAS.
- D. Ye, L. Wang, R. Zhang, B. Liu, Y. Wang and J. Kong, J. Mater. Chem. A, 2015, 3, 15171–15176 CAS.
- W. Wei, S. Yang, H. Zhou, I. Lieberwirth, X. Feng and K. Müllen, Adv. Mater., 2013, 25, 2909–2914 CrossRef CAS PubMed.
- Y. Wang, R. Huang, G. Liang, Z. Zhang, P. Zhang, S. Yu and J. Kong, Small, 2014, 10, 109–116 CrossRef CAS PubMed.
- U. Holzwarth and N. Gibson, Nat. Nanotechnol., 2011, 6, 534 CrossRef CAS PubMed.
- K. P. Kepp and P. Dasmeh, J. Phys. Chem. B, 2013, 117, 3755–3770 CrossRef CAS PubMed.
- D. Ye, R. Zhang, Y. Fu, J. Bu, Y. Wang, B. Liu and J. Kong, Electrochim. Acta, 2015, 160, 306–312 CrossRef CAS.
- W. Chen, S. Li, C. Chen and L. Yan, Adv. Mater., 2011, 23, 5679–5683 CrossRef CAS PubMed.
- H. Jin, H. Huang, Y. He, X. Feng, S. Wang, L. Dai and J. Wang, J. Am. Chem. Soc., 2015, 137, 7588–7591 CrossRef CAS PubMed.
- Y. Liu, T. C. Hughes, B. W. Muir, L. J. Waddington, T. R. Gengenbach, C. D. Easton, T. M. Hinton, B. A. Moffat, X. Hao and J. Qiu, Biomaterials, 2014, 35, 378–386 CrossRef CAS PubMed.
- S. Lagergren, K. Sven. Vetenskapsakad. Handl., 1898, 24, 1–39 Search PubMed.
- Y. S. Ho and G. Mckay, Process Biochem., 1999, 34, 451–465 CrossRef CAS.
- I. Langmuir, J. Am. Chem. Soc., 1918, 40, 1361–1403 CrossRef CAS.
- H. Freundlich, J. Phys. Chem., 1906, 57, 385–470 CAS.
- S. Lin, D. Lu and Z. Liu, Chem. Eng. J., 2012, 211, 46–52 CrossRef.
- X. Li, X. Dou and J. Li, J. Environ. Sci., 2012, 24, 1197–1203 CrossRef CAS.
- J. Xi, M. He and C. Lin, Microchem. J., 2011, 97, 85–91 CrossRef CAS.
- M. Aryal, M. Ziagova and M. Liakopoulou-Kyriakides, Chem. Eng. J., 2010, 162, 178–185 CrossRef CAS.
- L. Wang, C. L. Wan, Y. Zhang, D. J. Lee, X. Liu, X. F. Chen and J. H. Tay, J. Hazard. Mater., 2015, 284, 43–49 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra06208c |
| This journal is © The Royal Society of Chemistry 2016 |