Covalent conjugation of cysteine-engineered scFv to PEGylated magnetic nanoprobes for immunotargeting of breast cancer cells†
Christophe Alric a,
Nicolas Aubreyb,
Émilie Allard-Vanniera,
Anne di Tommasob,
Thibaut Blondya,
Isabelle Dimier-Poissonb,
Igor Chourpaa and
Katel Hervé-Aubert*a
a,
Nicolas Aubreyb,
Émilie Allard-Vanniera,
Anne di Tommasob,
Thibaut Blondya,
Isabelle Dimier-Poissonb,
Igor Chourpaa and
Katel Hervé-Aubert*a
aUniversité François Rabelais de Tours, EA6295 ‘Nanomédicaments et Nanosondes’, F 37200 Tours, France. E-mail: katel.herve@univ-tours.fr; Tel: +33 247 36 71 57
bUniversité François Rabelais de Tours, UMR1282 INRA ‘Infectiologie et Santé Publique’, F 37000 Tours, France
First published on 7th April 2016
Abstract
In the present study, we describe the synthesis and characterization of new generation of cancer-targeting magnetic nanoprobes: superparamagnetic iron oxide nanoparticles (SPIONs) coated with polyethylene glycol (PEG) shell functionalized with recombinant anti-HER2 single chain fragment variable (scFv) of Trastuzumab antibody. An anti-HER2 scFv with terminal cysteine (scFv 4D5-Cys) has been rationally engineered in order to favor its orientation- and site-directed covalent conjugation to the polymeric surface of PEGylated SPIONs. Optimization of scFv and nanoparticles production allowed to obtain well-characterized SPIONs-PEG–scFv nanoparticles carrying ∼7 fragments per nanoparticle, having a hydrodynamic diameter of ca. 86 nm and nearly neutral surface. The nanoprobes-scFv capability to recognize the HER2 protein has been confirmed by enzyme-linked immunosorbent assay (ELISA). Compared to non-targeted PEGylated SPIONs, the SPIONs–PEG–scFv nanoprobes showed an enhanced binding to HER2-overexpressing cells (SK-BR3) in vitro as it was shown by immunofluorescence. Finally, ICP-AES measurements shown that in 1 hour the uptake of SPIONs–PEG–scFv in HER2-overexpressing cells is 2.1 times greater than non-targeted PEGylated SPIONs. Therefore, both due to their physico-chemical characteristics and the immunotargeting of HER2-positive breast cancer cells, the SPIONs–PEG–scFv appear as promising nanoplatforms for future applications in theranostic treatment of cancers.
Introduction
Breast cancer is today the most frequent cancer in women (∼25% of all cancers). According to the International Agency for Research on Cancer (IARC), 1.67 million new cases of breast cancer were detected in 2012 in the world, and the number of deaths is estimated at 0.52 million. Currently, standard diagnostic procedures of breast tumors involve clinical examination, ultrasound and mammography. The main drawback of the latter is low efficiency in numerous cases like in younger women exploration and patient exposure to X-rays. As an adjunct and/or an alternative to the above methods, breast cancer diagnosis approaches are being developed using imaging methods such as magnetic resonance imaging (MRI). MRI is non-invasive and non-ionizing and provides a reliable evaluation of any lesion within the breast with a spatial resolution of hundreds of μm. Whereas the sensitivity of breast MRI is usually high, specificity depends on many factors and requires an MRI-guided breast biopsy.1 The breast imaging is performed in both asymptomatic and symptomatic women (in patients with known malignancies, including those that are initially treated medically with neoadjuvant chemotherapy). Therefore, imaging is helpful to detect breast cancer in its early stages, in the assessment of response to treatment and the evaluation of residual disease and thus increases the prognosis and survival of breast cancer patients.1 In order to improve detection of cancer cells, both sensitivity and specificity of breast MRI can be enhanced by the use of targeted magnetic nanoprobes. Due to their magnetic properties and their ability to be functionalized with several types of targeting ligand, SPIONs are potent platforms for targeted MRI nanoprobes2 or cancer therapy by hyperthermia or magnetically triggered drug delivery.3HER2 protein, which is composed of tyrosine kinase receptors involved in regulation of proliferation and survival of epithelial cells, is overexpressed in 25–30% of breast cancers and is associated to aggressiveness and poor prognosis.4 Thus, development of MRI nanoprobes specifically targeting HER2-overexpressing cancer cells is a promising perspective for breast cancer theranosis (diagnosis and therapy).
To be employed in such applications, SPIONs need to gather some fundamental properties, like the ability to interact with HER2-overexpressing cancer cells and adequate physico-chemical properties known to allow systemic injection.
This paper is focused on elaboration and in vitro studies of a novel type of targeted nanoprobes. As their final goal was to be intravenously injected, properties governing the in vivo behavior of nanoparticles were used as rules to guide the design of these nanoprobes.
To be administrated systemically, nanoprobes have to satisfy several requirements, like colloidal stability at physiological conditions and stealthiness against immune system. To avoid the rapid removal of the nanoparticles (NPs) from blood circulation, their surface can be coated with neutral hydrophilic polymers, PEG being the most used.5,6 Aqueous suspensions of PEGylated nanoparticles are stable against aggregation due to steric hindrance of the hydrophilic polymer chains.7 Moreover, the half-life of NPs injected in blood is also prolonged due to the fact that neutral PEG prevents the plasma proteins adsorption to the particle surface8 (opsonization) and consequently the recognition by the mononuclear phagocyte system (MPS).9 Moreover, the use of adequate reactive PEG chains allows further functionalization of nanoparticles with targeting moiety.10 Surface charge (zeta potential) and hydrodynamic diameter (DH) of nanoparticles are key parameters for their biodistribution as they govern both their elimination and their tumor accumulation. For example, nanoparticles with a neutral or a weakly negatively charged surface have reduced clearance from the blood by the MPS.11 PEGylated nanoparticles smaller than 100 nm have reduced plasma protein adsorption on their surface and reduced hepatic filtration,12 whereas nanoparticles with hydrodynamic diameter lower than 200 nm accumulate progressively in tumors due to the so-called enhanced permeability and retention (EPR) effect.13 Thus, the hydrodynamic diameter of nanoprobes should be lower than 100 nm to both reduce fast clearance from the blood and to allow passive tumor accumulation by EPR effect. Finally, to reach their target (cancer cells), the nanoprobes should be able not only to progressively accumulate within diseased zones but also to interact specifically with targeted cells. This combination of passive and active targeting is an essential condition to increase the accumulation of nanosystems in tumors.
Conjugation of whole antibodies to nanoparticles is a classical way to specifically target disease cells.14 MRI nanoprobes decorated with whole HER2-targeting antibodies have been recently described as being significantly accumulated in HER2-positive cancers.2,15 However, the conjugation of whole antibody to nanoparticles induces several drawbacks that limit the efficiency of cancer targeting, such as poor control of functionalization geometry (which can cause the loss of the biological activity of the antibody16) or rapid elimination of nanoparticles via Fc-mediated clearance.17 Moreover, the conjugation of whole antibodies on the surface of nanoparticles can induce an important increase of their surface charge and/or hydrodynamic diameter18,19 and thus decrease their colloidal stability and stealthiness.
To circumvent these limitations, one more recently proposed strategy consists in functionalizing the nanoprobes with an antibody fragment, such as single chain Fragment variable (scFv).17,20 The scFv fragments are composed of variable parts of heavy (VH) and light (VL) chains joined together with a flexible peptide linker. The scFv is the smallest functional antigen-binding domain of an antibody and has following advantages over whole antibody: (i) lower molecular weight (∼27 vs. 150 kDa) allows to keep nanoparticles small, stable and stealthy; (ii) absence of the Fc constant domain reduces the NPs immunogenicity; (iii) ability to be engineered to display desired amino acids favors the fragment purification/detection and its conjugation to the NPs; (iv) biotechnology protocols allow the scFv production to be relatively easy and low cost.
The conjugation of scFv at the surface of nanoparticles generally implies carbodiimide chemistry,20,21 which is known to give random orientation of the bound fragment.22 Recently, several works emphasized the need to perform directional-oriented conjugation of antibodies or antibody fragments on the surface of nanoparticles to preserve their antigen binding capacity.16,23 To achieve this, different strategies can be employed, such as exploiting the histidine tag of scFv24 or creating disulfide bridges between scFv and nanoparticles.25 Besides, thanks to the possibility of engineering the scFv structure, a C-terminal cysteine can be introduced within its sequence to allow the bonding of the scFv on the surface of maleimide functionalized nanoparticles.26
In this work, we describe synthesis, characterization and in vitro immunotargeting assay of new generation of magnetic nanoprobes functionalized with anti-HER2 scFv. The interest/novelty of this new generation compared to the previously reported nanoprobes rests upon the fact that our nanoparticles satisfy all the above described requirements necessary for efficient and specific targeting of HER2-positive tumor cells: (i) the orientation- and site-directed covalent conjugation of specifically designed scFv on the polymeric coating of the nanoparticles to provide an optimal immunotargeting with a moderate number of antibody fragments per NP; (ii) the moderate number of antibody fragments per NP to preserve both neutral surface and small size compatible with long circulation in blood and with EPR effect; (iii) the sample purity guaranteed against presence of free antibody fragments to avoid non-specific interactions in vivo. These nanosystems are composed of SPIONs core covalently coated with a layer of PEG which is functionalized with recombinant scFv of Trastuzumab antibody that specifically recognizes breast and ovarian cancer cells.4 The anti-HER2 antibody fragment selected for this study (scFv 4D5-Cys) was first engineered and produced in bacterial cultures before being covalently linked to PEGylated nanoparticles with orientation- and site-directed conjugation. The physico-chemical properties of the nanoparticles hereafter called SPIONs–PEG–scFv were determined by numerous analytical methods including atomic absorption and photon correlation spectroscopies. The presence of scFv fragments was analyzed both qualitatively and quantitatively by means of optical spectroscopy (fluorescence and UV-visible). The ability of the scFv-nanoprobes to interact with both HER2 protein and HER2-overexpressing cells has been assessed using ELISA and immunofluorescence methods.
Materials and methods
Chemicals
Anhydrous dimethyl sulfoxide (DMSO, 99.9%), EDTA disodium salt and DTT were purchased from Sigma Aldrich (Saint-Quentin-Fallavier, France). α-Maleinimidohexanoic-ω-NHS PEG, Mw 5000 Da, (NHS–PEG–Mal) was obtained from Rapp Polymere (Tübingen, Germany). Tris(2-carboxyethyl)phosphine hydrochloride (TCEP) was purchased from Thermo Fisher/Pierce. All other reagents were of analytical grade. In all the experiments, water was previously deionized (18 MΩ cm). Dialysis tubing (cellulose ester, molecular weight cut off 1000 kDa) was obtained from Spectrum Labs (France).Protein expression, purification and detection
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 × g, 4 °C, 10 min). Swiss Institute of Bioinformatics software (ProtParam tool) was used to determine the theoretical molecular weight (27
000 × g, 4 °C, 10 min). Swiss Institute of Bioinformatics software (ProtParam tool) was used to determine the theoretical molecular weight (27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 303.1 Da) and the extinction coefficient (ε = 1835 L g−1 cm−1).29 The purification process was checked using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) on homogeneous 12% gel with Coomassie brilliant blue staining under reducing (with DTT) or non-reducing (without DTT) conditions, and by size-exclusion chromatography with an ÄKTApurifier FPLC system equipped with a prepacked Superdex 200 10/300 GL column (GE Healthcare). The column was loaded with 15 μg of the sample of scFv 4D5-Cys to be analyzed. The protein was eluted with PBS pH 7.4 at a flow rate of 0.5 mL min−1 and detected with a UV detector at 280 nm.
303.1 Da) and the extinction coefficient (ε = 1835 L g−1 cm−1).29 The purification process was checked using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) on homogeneous 12% gel with Coomassie brilliant blue staining under reducing (with DTT) or non-reducing (without DTT) conditions, and by size-exclusion chromatography with an ÄKTApurifier FPLC system equipped with a prepacked Superdex 200 10/300 GL column (GE Healthcare). The column was loaded with 15 μg of the sample of scFv 4D5-Cys to be analyzed. The protein was eluted with PBS pH 7.4 at a flow rate of 0.5 mL min−1 and detected with a UV detector at 280 nm.Nanoparticles preparation
To obtain scFv-functionalized PEGylated SPIONs, silanized SPIONs were first PEGylated before to be functionalized with scFv 4D5-Cys. Each type of nanoparticles were characterized by SAA, DLS, zeta potential measurements and fluorescence spectroscopy.Analytical methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v). The total iron concentration of the cell samples was determined by ICP-AES measurements (Ultima spectrometer, Jobin Yvon Horiba).
1 v/v). The total iron concentration of the cell samples was determined by ICP-AES measurements (Ultima spectrometer, Jobin Yvon Horiba).Results and discussion
Production of the scFv 4D5-Cys
The published amino acid sequence of the humanized monoclonal antibody variant h4D5v8 (ref. 27) (Trastuzumab) was used as a template to design the nucleotide sequence of the synthetic gene encoding 4D5-Cys. This gene was composed of sequences encoding for VH and VL of h4D5v8 joined together with a short peptide linker (G4S)3 and a flag composed of a hexahistidine tag, a GS spacer and a terminal cysteine at the C-terminus.26 The plasmid pSW1-scFv 4D5-Cys was cloned into the HB2151 E. coli strain, and the recombinant protein was expressed and exported to the bacterial periplasm by its leader sequence PelB.33 Soluble proteins were extracted from the periplasm of induced bacteria by osmotic shock, and the purification of scFv 4D5-Cys fragments was achieved by affinity chromatography on a protein L-agarose gel column.28,34 Protein L affinity chromatography is a fast and easy method allowing a highly specific purification of scFvs and their recovery with high purity.35 Finally, yields of 2.5 mg L−1 of bacterial culture of soluble affinity-purified scFv 4D5-Cys were obtained.Fig. 1 presents construction, identification and functionality assessment of the scFv 4D5-Cys. The schematic representation and 3D molecular model are shown in Fig. 1(A and B). The C-terminus of scFv contains six histidine residues followed by a GS spacer and a terminal cysteine. The insertion of six histidine residues (hexahistidine tag) to a protein is a commonly used method for the detection and/or purification of recombinant proteins. Here, purification was performed using a high specific protein L-based resin and thus the hexahistidine tag was only used for scFv detection. The cysteine insertion at the C-terminus was performed in order to promote oriented conjugation of scFv on maleimide functionalized PEGylated SPIONs while preserving its antigen-binding ability.26,36 Indeed, when scFv is grafted on the nanoparticle surface, its paratope should be favorably exposed to the solvent and must remained freely accessible to antigens (HER2) to allow their specific recognition.23 However, the classical carbodiimide chemistry bioconjugation involves the attachment of scFv through primary amines (from lysine residues) and results in the creation of amide bonds at several sites of scFv structure. This can lead to a random orientation of antibody fragments at the nanoparticles surface, what can cause a decrease of its biological activity. In order to obtain a site specific-oriented conjugation, it is useful to selectively attach scFv by a unique grafting site which can be placed as desired. Such a conjugation strategy can be implemented using the reaction between thiol (e.g. cysteine residues from proteins) and maleimide groups that is known to be efficient and selective at neutral pH and gives a stable thioether bond.37 Thus, a single unpaired cysteine residue should be introduced as far away as possible from the scFv paratope to act as a unique bonding site. Previous works have shown that such a condition is met when the cysteine residue is located within the C-terminus flag, at the opposite of the antigen-binding site.26,36 Furthermore, the position of the cysteine within the amino acid flag sequence seemed to have an influence on its reactivity.36 Based on these considerations, we placed the unpaired cysteine residue most distal to the antigen binding regions, namely at the end of the flag, as shown in Fig. 1(A and B) and ESI S1.†
Analysis of purified scFv 4D5-Cys by size-exclusion chromatography showed two elution peaks, at volume of ∼15.7 mL and ∼17 mL, corresponding respectively to dimeric and monomeric forms of the protein (Fig. 1D). This result is concordant with the analysis of purified scFv 4D5-Cys by SDS-PAGE under non-reducing conditions (absence of DTT) that showed the presence of two bands (MW ∼54 kDa and MW ∼27 kDa). As expected, under reducing conditions (presence of DTT), only the ∼27 kDa (monomeric form) subsisted (Fig. 1C). From these observations, it can be deduced that: (i) when the protein was secreted into the oxidizing environment of the bacterial periplasm, a significant amount of dimeric form of scFvs was formed via disulfide bridges between free cysteine moieties of scFvs; (ii) reducing conditions allowed to break all those dimers. This behavior is similar to that observed for engineered scFvs having a free terminal cysteine and produced under similar conditions.35,36 As the covalently formed dimers of scFvs are not reactive toward maleimide groups, it was necessary to reduce the intermolecular disulfide bridges and thus to restore the reactivity of the terminal cysteines. Indeed, scFv dimers were easily reduced by (2-carboxyethyl)phosphine (TCEP)26 before their conjugation on the surface of nanoparticles, to obtain the monomeric form of scFv with preserved functionality (ESI Fig. S2(A and B)†).
Compared to the initial amino acids sequence of the antibody variant h4D5v8 VH and VL, the sequence of scFv 4D5-Cys we produced was slightly different: a linker was added between VH and VL and the N-terminal sequence was modified to introduce a histidine tag and a terminal cysteine residue. In order to verify the immunoreactivity of the purified scFv 4D5-Cys, we performed an indirect ELISA assay using HER2 protein as a target. Increasing concentrations of the HER2 protein were put as a coating in well plates and scFv 4D5-Cys was then added at the constant concentration of 10 μg mL−1. Thanks to the high affinity of protein L to light chains of the scFv,30 the addition to the wells of protein L-peroxydase in presence of a chromatic substrate (TMB) caused coloration only if the scFv was present. The obtained results showed that, for the same quantity of the scFv added, the content of the fragment retained in wells increased proportionally to the HER2 protein content (Fig. 1E), thus confirming the protein binding was specific.
Synthesis and characterization of aqueous suspensions of PEGylated SPIONs
Aqueous suspensions of PEGylated SPIONs were prepared by covalent coupling of polyethylene glycol (PEG) chains to the surface of SPIONs pre-coated with aminated trialkoxysilane (Fig. 2A). Silane coating of SPIONs was obtained due to covalent Si–O bonds formed between silane molecules and the surface hydroxyls groups of SPIONs.7 According to DLS data (intensity distribution), the silanized SPIONs dispersed in acidic aqueous medium had an average hydrodynamic diameter (DH) of about 68 nm and a polydispersity index of ∼0.15. Compared to 8–10 nm dimension of the SPIONs cores observed with transmission electron microscopy (ESI Fig. S3A†), the average DH values of the silane-coated NPs in aqueous colloids are usually higher because they take into account both solvation layers around NPs and possible SPIONs combination in small clusters.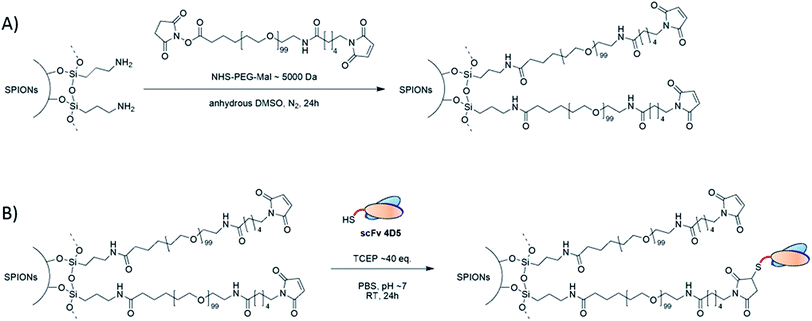 | ||
| Fig. 2 Synthesis scheme of (A) PEGylated SPIONs (SPIONs–PEG–Mal) and (B) scFv functionalized PEGylated SPIONs (SPIONs–PEG–scFv). | ||
According to zeta potential measurements, the isoelectric point of silanized SPIONs was observed at pH value of 10, very close to the pKa value of amino groups. These results are concomitant with the presence of amino groups at the SPIONs surface and thus confirm the effective silanization of SPIONs.7 We used the primary amino groups of the silanized SPIONs to covalently link N-hydroxysuccinimidyl (NHS) ester terminal groups of the polyethylene glycol chains (Fig. 2A).7 For further covalent functionalization of the PEGylated SPIONs with cysteine-derived scFv (4D5-Cys), we used a thiol–maleimide reaction (Fig. 2B) which is known to give stable site-specific and oriented covalent bonding.38 Therefore, we used heterobifunctional PEG chains (hereafter called NHS–PEG–Mal) that possess an amine reactive NHS-ester group at one terminus and a maleimide group at the other terminus (Fig. 2A).
The NHS–PEG–Mal chains were grafted onto silanized SPIONs by formation of amide bonds between NHS-ester groups of PEG chains and primary amino groups of silane, as shown in Fig. 2A. This reaction led to the formation of PEG–maleimide-coated iron oxide nanoparticles (SPIONs–PEG–Mal). It should be noticed that all steps of the preparation protocol of SPIONs–PEG–Mal were designed to minimize maleimide groups hydrolysis, which depends on pH and temperature.37,39 The SPIONs–PEG–Mal were then extensively purified by dialysis against water during 48 hours at 4 °C and were used as soon as prepared.
The iron concentration in the suspensions was 150–300 mg L−1 as determined by atomic absorption spectroscopy. According to the dynamic light scattering (DLS) data, the PEG–Mal coating of SPIONs increased the average hydrodynamic diameter (DH) of NPs from ∼68 nm to ∼82 nm (Fig. 3A) while the size of iron oxide cores determined by TEM remained unchanged (ESI Fig. S3B†). This increase is consistent with PEG coating and is similar to previous studies for this type of PEG chains.7 As expected, the hydrodynamic diameter of the SPIONs–PEG–Mal remained stable over a broad pH range 3–12 (Fig. 3A). The effective coating of silanized SPIONs with neutral PEG was also confirmed by their zeta potential: it was very low (<|10| mV) and was nearly insensitive to pH change from 3 to 12. These results suggest that the neutral PEG shell protects SPIONs from aggregation by steric repulsion mechanism. The fact that zeta potential became almost neutral could be explained by amine groups consumption and their replacement by neutral maleimide groups at the surface of NPs during the PEGylation process. Similar result was shown in a recent work where the zeta-potential of aminodextran coated iron oxide nanoparticles functionalized with a NHS-derived maleimide linker was found to be almost neutral.40
The infrared absorption spectrum of SPIONs–PEG–Mal (Fig. 3B) shows characteristic bands of PEG at 2680–2660 cm−1 (a) attributed to C–H stretching mode from methylene groups from PEG, and at 1100 cm−1 and 1060 cm−1 (b), from two C–O–C stretching modes, respectively asymmetric and symmetric. The presence of a band attributed to carbonyl groups of amide linkage (at 1650 cm−1) confirms the covalent bonding of NHS–PEG–Mal on the surface of silanized SPIONs. Moreover, the presence of maleimide function is attested by the presence of characteristic IR bands from imide carbonyl groups at 1740 cm−1 (asymmetric) and 1705 cm−1 (symmetric).41 Therefore, both the interpretation of colloidal properties seen in DLS and zetametry and the direct analysis of chemical composition by IR spectroscopy confirmed the effective synthesis of SPIONs–PEG–Mal with desired specifications: DH < 100 nm and nearly neutral surface within a large pH range around physiological value of 7.4.
Preparation of PEGylated SPIONs functionalized by scFv 4D5-Cys
As explained above, the oriented coupling of our scFv 4D5-Cys on the surface of PEGylated SPIONs was performed via the formation of stable covalent thioether bonds between terminal cysteine residues of the antibody fragment and maleimide terminal groups of the polymer chains (Fig. 2B). The bioconjugation reaction was performed in mild conditions (in PBS, at room temperature and pH 7.0) to both keep the selectivity of the coupling reaction and to preserve the scFv immunoreactivity.Purification is a critical step in production of bioconjugated nanoparticles for in vivo biomedical applications where there is a need to ensure reproducibility of synthesis, quality assurance, predictable behavior in intended use.42 Indeed, the bioconjugated nanoparticles purity should be carefully controlled to verify the absence of free biomolecules that could interfere with the immunoreactivity of the nanoparticles. We first tried to purify SPIONs–PEG–scFv by centrifugation, but these technique failed to fully separate SPIONs–PEG–scFv from free scFv. Indeed, a significant part of nanoparticles remained in suspension even after several centrifugation cycles of 1 h at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 × g or 30
000 × g or 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 × g. A higher centrifugation speed could cause a degradation of the nanoparticles surface. To preserve the integrity of scFvs grafted on nanoparticles surface, we then chose to purify SPIONs–PEG–scFv nanoparticles with mild purification method, such as size exclusion chromatography (SEC). SEC has already been used to separate scFv-functionalized nanoparticles from unconjugated scFvs.20,43 In our very first experiments, SPIONs–PEG–scFv nanoparticles were purified by SEC using an ÄKTApurifier FPLC system equipped with a prepacked Superdex 75 column. Surprisingly, although the elution peaks of SPIONs–PEG–scFv and free scFv were well resolved, purity assessment by SDS-PAGE of scFv-functionalized NPs has shown the minor presence of unconjugated scFv (ESI Fig. S4†). This minor presence could be due to some residual scFv while the electrophoresis-induced degradation of the SPIONs–PEG–scFv could not be excluded.
000 × g. A higher centrifugation speed could cause a degradation of the nanoparticles surface. To preserve the integrity of scFvs grafted on nanoparticles surface, we then chose to purify SPIONs–PEG–scFv nanoparticles with mild purification method, such as size exclusion chromatography (SEC). SEC has already been used to separate scFv-functionalized nanoparticles from unconjugated scFvs.20,43 In our very first experiments, SPIONs–PEG–scFv nanoparticles were purified by SEC using an ÄKTApurifier FPLC system equipped with a prepacked Superdex 75 column. Surprisingly, although the elution peaks of SPIONs–PEG–scFv and free scFv were well resolved, purity assessment by SDS-PAGE of scFv-functionalized NPs has shown the minor presence of unconjugated scFv (ESI Fig. S4†). This minor presence could be due to some residual scFv while the electrophoresis-induced degradation of the SPIONs–PEG–scFv could not be excluded.
The presence of traces of residual scFv could be explained by a decrease of scFv stability (formation of aggregates inseparable from nanoparticles) during the bioconjugation process and/or by the entrapment of scFv into the PEG shell of nanoparticles. On the other hand, for NPs conjugated with another type of scFv purified under the same conditions, the elution fractions containing purified SPIONs–PEG–scFv did not show the presence of unconjugated scFv (data not shown). Although this result is not currently fully understood, it highlights the need to carefully choose the NPs purification method and to control the purification result. Nonetheless, the purification step is not always described in literature, although it is critical to avoid overestimation of the number of biomolecules grafted on the surface of nanoparticles.
Optimization of the purification process of SPIONs–PEG–scFv had been performed and dialysis against PBS using 1000 kDa pores sizes membranes appeared efficient to eliminate unconjugated scFv. After purification by dialysis, SPIONs–PEG–scFv and SPIONs–PEG–Mal were obtained at iron concentration in the range 50–180 mg L−1. Purity of final SPIONs–PEG–scFv was assessed by controlling the absence of free scFv using SDS-PAGE with Coomassie brilliant blue staining (Fig. 4A). Although it was present before dialysis, the free scFv was no more observed after purification. The presence of the scFv 4D5-Cys bound to the surface of SPIONs–PEG–scFv was confirmed by detection of the intrinsic scFv fluorescence in UV (ex/em 260/340 nm, Fig. 4B) assigned to the aromatic amino acids (mainly tryptophan and tyrosine).
The quantity of scFv covalently bound to SPIONs–PEG–scFv was determined using Bradford assay32 (ESI Fig. S5†) which is widely exploited for the determination of total protein. This assay is based on the strong red shift (from 465 to 595 nm) of the absorption maximum of Coomassie G-250 dye when it binds to proteins. This approach seem to be suited for SPIONs-grafted protein quantification as the absorbance of SPIONs at 595 nm is very weak.44 The obtained results showed that a suspension of SPIONs–PEG–scFv at 55 mg L−1 of iron contained ∼7.2 μg mL−1 of scFv 4D5-Cys. That represents ∼130 μg of scFv per mg of iron, i.e. more than 43 μg of whole antibody per mg of iron reported by Shanehsazzadeh et al.18 From this value, one obtains that the final SPIONs–PEG–scFv carry ∼7 scFv molecules per nanoparticle, which is close to 5–6 scFv per nanoparticle obtained by Mazzucchelli et al.25
Amount of antibodies/antibody fragments per mg of iron (and thus the number of antibody per nanoparticles) depends on several factors, such as type of grafted biomolecules, chemical conjugation protocol or quantification method. In literature, the number of antibodies or antibody fragments conjugated per nanoparticle is highly variable: from 1.8 whole antibody to ∼28 scFv per nanoparticle for iron oxide nanoparticles with DH about 50–60 nm or ∼10 Fab per nanoparticles with DH about 30 nm.20,40,45 According to literature, even a weak content of antibody or scFv (∼2 per nanoparticle) can be sufficient to allow specific in vitro detection of antigen-overexpressing cells.45,46 Moreover, covalent bonding of ∼6 to 25 scFv molecules per nanoliposomes (DH ∼ 77 nm) did not significantly increase their in vitro binding efficiency to cells overexpressing targeted antigen.36
An other very important result of the present study was that the covalent conjugation of ∼7 scFv molecules per nanoparticle did not change significantly the hydrodynamic diameter of the PEGylated nanoparticles (see the DLS data in Fig. 4A). Indeed, in PBS pH 7.4 at 37 °C, SPIONs–PEG–Mal showed a hydrodynamic diameter (DH) of 80.7 ± 1.3 nm (PdI = 0.124) while that of SPIONs–PEG–scFv was of 86.2 ± 2.8 nm (PdI = 0.233). The NPs size increase of ca. 5.5 nm was comparable to the DH of free scFv in solution (typically ∼5 nm)47 and can be attributed to the presence of scFv molecules at the polymeric surface of functionalized nanoparticles. As DH increased only moderately, it can be stated that the bioconjugation process did not alter the colloidal stability of nanoparticles. Contrary to some previous works where DH highly increased (and even over 100 nm) after bioconjugation with whole antibodies18,19 or with scFvs,48,49 in our study, the hydrodynamic diameter of our scFv-capped NPs was significantly below 100 nm. Nanoparticles with DH < 100 nm are intended to have a prolonged half-life in blood after their systemic administration and are able of intratumoral accumulation due to the EPR effect.50 It had been verified by TEM that covalent attachment of scFv at the surface of SPIONs–PEG–Mal did not change morphology or size of iron oxide cores (8–10 nm, ESI Fig. S3C†). Organic shell was not observable in TEM due to its low electronic density.43
The conjugation of whole antibodies/scFv to the polymeric surface of nanoparticles can often lead (when stated) to relatively strong (>10 mV) change of their zeta potential.51,52 In our study, zeta potential went from −4.3 ± 0.6 mV to −5.4 ± 0.8 mV. Thus, after bioconjugation, zeta potential of SPIONs–PEG–scFv remained almost neutral, which is also a very important result for their further in vivo applications. Stability of zeta potential after NPs conjugation with antibody fragment (Fab or scFv) had already been described, but no explanation was given.40,49 In our case, theoretical isoelectric point of scFv 4D5-Cys is about 8.75, so scFv would be slightly positively charged in PBS pH 7.4. The fact that zeta potential of SPIONs–PEG–scFv remained stable after scFv bonding could be explained by the relatively low number of conjugated scFvs and by hiding of positively charged scFv amino acids.
To summarize, the covalent conjugation of scFv 4D5-Cys at the surface of PEGylated SPIONs had only a weak impact on the size and surface charge of the nanoparticles. At physiologic pH, their hydrodynamic diameter was below 90 nm and their zeta potential was almost neutral. These properties are known to favor prolonged retention time in the blood after in vivo injection: the nanoparticles should avoid opsonization and rapid elimination from the body and would have a better chance to be progressively accumulated in tumors due to their small size favoring the EPR effect usually considered as passive targeting.11,12 The active targeting of cancer cells by SPIONs–PEG–scFv would be insured if the bound scFv remain functional.
The functionality of scFv 4D5-Cys carried by SPIONs–PEG–scFv has been assessed by indirect enzyme linked immunosorbent assay (ELISA) in which scFv-functionalized PEGylated nanoparticles played a role of a primary antibody that detects the antigen (HER2 protein). HER2 coating at increasing protein concentrations was first introduced in wells and colloidal suspensions of SPIONs–PEG–Mal or SPIONs–PEG–scFv were then added at the same iron concentration in each well. The presence of scFv at the nanoparticles surface both allows antigen recognition and nanoparticles detection by the specific conjugate protein L-peroxydase. Results shown that whereas SPIONs–PEG–Mal were not retained, SPIONs–PEG–scFv were held in wells. For SPIONs–PEG–scFv, the signal increased with HER2 concentration, what confirmed a specific recognition (Fig. 5A). As we have previously shown that no free scFv 4D5-Cys were present in SPIONs–PEG–scFv colloidal suspensions, this result confirmed the immunoreactivity of the nanoparticles toward HER2 protein.
The immunoreactivity of the SPIONs–PEG–scFv was additionally confirmed by immunofluorescence labelling of HER2-positive breast cancer cells SK-BR3 (Fig. 5(B and C)). The cells were first fixed in order to block nanoparticle endocytosis and to be able to isolate membrane interactions between HER2 receptor and SPIONs–PEG–scFv. The cells were first incubated in the presence of SPIONs–PEG–Mal or SPIONs–PEG–scFv, then with a primary anti-histidine antibody followed by a secondary fluorescent antibody. The SK-BR3 cells incubated with SPIONs–PEG–scFv and not the cells treated with SPIONs–PEG–mal showed a significant fluorescence on cellular membranes where HER2 protein is overexpressed. As expected for fixed cells, no internalization was observed. Thus, functionality of scFvs i.e. their ability to specifically recognize the antigen was preserved. This suggests that the scFv molecules attached on the NPs surface were not denatured and were favorably exposed to the exterior.
Lastly, we quantified the uptake of SPIONs–PEG–Mal and SPIONs–PEG–scFv on HER2-overexpressing BT-474 breast cancer cells in vitro. The cells incubated for 1 hour with suspensions of the two types of NPs, were washed and the cellular iron concentration was determined by ICP-AES. The ICP-AES results showed that, the uptake of SPIONs–PEG–scFv was 2.1 times greater than that of SPIONs–PEG–Mal (ESI Fig. S6†). So, the presence of scFv fragment on the nanoprobes surface increased significantly their uptake by HER2-positive breast cancer cells.
Conclusion
The rational design and the protocols developed in this work enabled the successful synthesis and purification of PEGylated iron oxide nanoparticles functionalized with scFv fragment of antibody Trastuzumab specific to breast cancer. First, a monomeric anti-HER2 scFv 4D5-Cys with a C-terminal cysteine residue has been engineered, produced with a high yield, and purified using a highly specific affinity chromatography method. Its functionality have been confirmed by indirect ELISA assay. Then, the oriented site-specific covalent binding of the scFv on the surface of PEGylated SPIONs (∼7 scFv molecules per nanoparticle) was performed in mild conditions. The SPIONs–PEG–scFv dispersed in aqueous suspensions had a hydrodynamic diameter below 90 nm and a nearly neutral surface. We confirmed that covalent conjugation of scFv 4D5-Cys at the surface of PEGylated SPIONs did not hinder its antigen binding ability: the NPs were found to be immunoreactive in vitro toward the HER2 protein and HER2-protein overexpressing cells. Moreover, the presence of scFv on the surface of nanoprobes allowed to significantly increase their uptake by HER2-positive cancer cells. Finally, as these nanoparticles possessed adequate properties for further in vivo use, they have an interesting potential to be used as targeted nanosystems in HER2-positive breast cancer theranosis, namely for MR imaging of HER2-positive cancer cells and for anticancer agents delivery. The in vivo experiments with these nanosystems are now in progress in our lab.As HER2 protein is overexpressed not only in breast cancers but also in ovarian or gastric cancers and gliomas,53 our SPIONs–PEG–scFv 4D5-Cys could be used to detect those HER2-positive tumors, too. Moreover, thanks to their modular character, SPIONs–PEG–Mal can be functionalized with scFvs molecules designed to specifically target other cancers types, seeing that a terminal cysteine residue is added to scFv using biotechnology protocols.
Acknowledgements
We are thankful to the “Région Centre-Val de Loire” (NCIS project) and “Ligue Nationale contre le Cancer” for their financial support and especially to the local comities 28, 37, 53, 79 and 56. We thank Agnès Pallier (CBM, UPR CNRS 4301, Orléans) for technical assistance in the determination of iron content in cells by ICP-AES.References
- R. M. Mann, C. K. Kuhl, K. Kinkel and C. Boetes, Eur. J. Radiol., 2008, 18, 1307–1318 CrossRef CAS PubMed.
- Y. W. Jun, J. H. Lee and J. Cheon, Angew. Chem., Int. Ed., 2008, 47, 5122–5135 CrossRef CAS PubMed.
- A. J. Cole, V. C. Yang and A. E. David, Trends Biotechnol., 2011, 29, 323–332 CrossRef CAS PubMed.
- M. Colombo, F. Corsi, D. Foschi, E. Mazzantini, S. Mazzucchelli, C. Morasso, E. Occhipinti, L. Polito, D. Prosperi, S. Ronchi and P. Verderio, Pharmacol. Res., 2010, 62, 150–165 CrossRef CAS PubMed.
- D. E. Owens and N. a. Peppas, Int. J. Pharm., 2006, 307, 93–102 CrossRef CAS PubMed.
- J. Xie, C. Xu, N. Kohler, Y. Hou and S. Sun, Adv. Mater., 2007, 19, 3163–3166 CrossRef CAS.
- K. Hervé, L. Douziech-Eyrolles, E. Munnier, S. Cohen-Jonathan, M. Soucé, H. Marchais, P. Limelette, F. Warmont, M. L. Saboungi, P. Dubois and I. Chourpa, Nanotechnology, 2008, 19, 465608 CrossRef PubMed.
- J. V Jokerst, T. Lobovkina, R. N. Zare and S. S. Gambhir, Nanomedicine, 2011, 6, 715–728 CrossRef PubMed.
- E. Allard-Vannier, S. Cohen-Jonathan, J. Gautier, K. Hervé-Aubert, E. Munnier, M. Soucé, P. Legras, C. Passirani and I. Chourpa, Eur. J. Pharm. Biopharm., 2012, 81, 498–505 CrossRef CAS PubMed.
- K. Kaaki, K. Hervé-Aubert, M. Chiper, A. Shkilnyy, M. Soucé, R. Benoit, A. Paillard, P. Dubois, M. Saboungi and I. Chourpa, Langmuir, 2012, 28, 1496–1505 CrossRef CAS PubMed.
- X. Duan and Y. Li, Small, 2013, 9, 1521–1532 CrossRef CAS PubMed.
- F. Alexis, E. Pridgen, L. K. Molnar and O. C. Farokhzad, Mol. Pharm., 2008, 5, 505–515 CrossRef CAS PubMed.
- C. M. Dawidczyk, C. Kim, J. H. Park, L. M. Russell, K. H. Lee, M. G. Pomper and P. C. Searson, J. Controlled Release, 2014, 187, 133–144 CrossRef CAS PubMed.
- F. Fay and C. J. Scott, Immunotherapy, 2011, 3, 381–394 CrossRef CAS PubMed.
- H. Yang, C. W. Park, M. Woo, M. Il Kim, Y. M. Jo, H. G. Park and J.-D. Kim, Biomacromolecules, 2010, 11, 2866–2872 CrossRef CAS PubMed.
- J. Montenegro, V. Grazu, A. Sukhanova, S. Agarwal, J. M. de la Fuente, I. Nabiev, A. Greiner and W. J. Parak, Adv. Drug Delivery Rev., 2013, 65, 677–688 CrossRef CAS PubMed.
- R. E. Kontermann, Curr. Opin. Mol. Ther., 2006, 8, 39–45 CAS.
- S. Shanehsazzadeh, C. Gruettner, A. Lahooti, M. Mahmoudi, B. J. Allen, M. Ghavami, F. J. Daha and M. A. Oghabian, Contrast Media Mol. Imaging, 2015, 10, 225–236 CrossRef CAS PubMed.
- J. Conde, C. Bao, D. Cui, P. V. Baptista and F. Tian, J. Controlled Release, 2014, 183, 87–93 CrossRef CAS PubMed.
- K. L. Vigor, P. G. Kyrtatos, S. Minogue, K. T. Al-Jamal, H. Kogelberg, B. Tolner, K. Kostarelos, R. H. Begent, Q. A. Pankhurst, M. F. Lythgoe and K. A. Chester, Biomaterials, 2010, 31, 1307–1315 CrossRef CAS PubMed.
- X. Qian, X.-H. Peng, D. O. Ansari, Q. Yin-Goen, G. Z. Chen, D. M. Shin, L. Yang, A. N. Young, M. D. Wang and S. Nie, Nat. Biotechnol., 2008, 26, 83–90 CrossRef CAS PubMed.
- J. Li and C. K. Ng, in Antibody-Mediated Drug Delivery Systems, ed. Y. Pathak and S. Benita, John Wiley & Sons, Inc., Hoboken, NJ, USA, 2012, pp. 191–207 Search PubMed.
- S. Avvakumova, M. Colombo, P. Tortora and D. Prosperi, Trends Biotechnol., 2014, 32, 11–20 CrossRef CAS PubMed.
- L. Yang, H. Mao, Y. A. Wang, Z. Cao, X. Peng, X. Wang, H. Duan, C. Ni, Q. Yuan, G. Adams, M. Q. Smith, W. C. Wood, X. Gao and S. Nie, Small, 2008, 5, 235–243 CrossRef PubMed.
- S. Mazzucchelli, S. Sommaruga, M. O'Donnell, P. Galeffi, P. Tortora, D. Prosperi and M. Colombo, Biomater. Sci., 2013, 1, 728–735 RSC.
- S. K. E. Messerschmidt, A. Kolbe, D. Müller, M. Knoll, J. Pleiss and R. E. Kontermann, Bioconjugate Chem., 2008, 19, 362–369 CrossRef CAS PubMed.
- P. Carter, L. Presta, C. M. Gorman, J. B. Ridgway, D. Henner, W. L. Wong, A. M. Rowland, C. Kotts, M. E. Carver and H. M. Shepard, Proc. Natl. Acad. Sci. U. S. A., 1992, 89, 4285–4289 CrossRef CAS.
- N. Aubrey, C. Devaux, P.-Y. Sizaret, H. Rochat, M. Goyffon and P. Billiald, Cell. Mol. Life Sci., 2003, 60, 617–628 CrossRef CAS PubMed.
- E. Gasteiger, A. Gattiker, C. Hoogland, I. Ivanyi, R. D. Appel and A. Bairoch, Nucleic Acids Res., 2003, 31, 3784–3788 CrossRef CAS PubMed.
- B. H. K. Nilson, A. Solomon, L. Björck and B. Åkerström, J. Biol. Chem., 1992, 267, 2234–2239 CAS.
- R. Massart, IEEE Trans. Magn., 1981, 17, 1247–1248 CrossRef.
- M. M. Bradford, Anal. Biochem., 1976, 72, 248–254 CrossRef CAS PubMed.
- S. P. Lei, H. C. Lin, S. S. Wang and G. Wilcox, J. Bacteriol., 1987, 169, 4379–4383 CAS.
- B. H. K. Nilson, L. Lögdberg, W. Kastern, L. Björck and B. Akerström, J. Immunol. Methods, 1993, 164, 33–40 CrossRef CAS PubMed.
- D. Das, T. M. Allen and M. R. Suresh, Protein Expression Purif., 2005, 39, 199–208 CrossRef CAS PubMed.
- T. Völkel, P. Hölig, T. Merdan, R. Müller and R. E. Kontermann, Biochim. Biophys. Acta, Biomembr., 2004, 1663, 158–166 CrossRef PubMed.
- G. G. Hermanson, Bioconjugate Techniques, Academic Press, London, 3rd edn, 2013 Search PubMed.
- J. L. Zimmermann, T. Nicolaus, G. Neuert and K. Blank, Nat. Protoc., 2010, 5, 975–985 CrossRef CAS PubMed.
- G. Shen, M. F. G. Anand and R. Levicky, Nucleic Acids Res., 2004, 32, 5973–5980 CrossRef CAS PubMed.
- C. N. Dong, J. a. Tate, W. C. Kett, J. Batra, E. Demidenko, L. D. Lewis, P. J. Hoopes, T. U. Gerngross and K. E. Griswold, PLoS One, 2015, 10, e0115636 Search PubMed.
- M. Milne, P. Gobbo, N. McVicar, R. Bartha, M. S. Workentin and R. H. E. Hudson, J. Mater. Chem. B, 2013, 1, 5628–5635 RSC.
- K. E. Sapsford, K. M. Tyner, B. J. Dair, J. R. Deschamps and I. L. Medintz, Anal. Chem., 2011, 83, 4453–4488 CrossRef CAS PubMed.
- D. K. Kirui, D. A. Rey and C. A. Batt, Nanotechnology, 2010, 21, 105105–105114 CrossRef PubMed.
- G. Rimkus, S. Bremer-Streck, C. Grüttner, W. A. Kaiser and I. Hilger, Contrast Media Mol. Imaging, 2011, 6, 119–125 CrossRef CAS PubMed.
- S. Mazzucchelli, M. Colombo, C. De Palma, A. Salvadè, P. Verderio, M. D. Coghi, E. Clementi, P. Tortora, F. Corsi and D. Prosperi, ACS Nano, 2010, 4, 5693–5702 CrossRef CAS PubMed.
- M. Colombo, S. Mazzucchelli, V. Collico, S. Avvakumova, L. Pandolfi, F. Corsi, F. Porta and D. Prosperi, Angew. Chem., Int. Ed., 2012, 51, 9272–9275 CrossRef CAS PubMed.
- S. Kubetzko, C. A. Sarkar and A. Plückthun, Mol. Pharmacol., 2005, 68, 1439–1454 CrossRef CAS PubMed.
- S. Mazzucchelli, M. Colombo, P. Verderio, E. Rozek, F. Andreata, E. Galbiati, P. Tortora, F. Corsi and D. Prosperi, Angew. Chem., Int. Ed., 2013, 52, 3121–3125 CrossRef CAS PubMed.
- X.-H. Peng, Y. Wang, D. Huang, Y. Wang, H. J. Shin, Z. Chen, M. B. Spewak, H. Mao, X. Wang, Y. Wang, Z. G. Chen, S. Nie and D. M. Shin, ACS Nano, 2011, 5, 9480–9493 CrossRef CAS PubMed.
- H. Arami, A. Khandhar, D. Liggitt and K. M. Krishnan, Chem. Soc. Rev., 2015, 44, 8576–8607 RSC.
- S. Richard, M. Boucher, A. Herbet, Y. Lalatonne, S. Mériaux, D. Boquet and L. Motte, J. Mater. Chem. B, 2015, 3, 2939–2942 RSC.
- H. Xu, Z. P. Aguilar, L. Yang, M. Kuang, H. Duan, Y. Xiong, H. Wei and A. Wang, Biomaterials, 2011, 32, 9758–9765 CrossRef CAS PubMed.
- L. Accardi and P. Di Bonito, Curr. Med. Chem., 2010, 17, 1730–1755 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Amino acids sequence of scFv 4D5-Cys, control of the influence of scFv 4D5-Cys reduction on immunoreactivity, TEM images of nanoprobes, quality control of SPIONs–PEG–scFv purified by size exclusion chromatography, quantification of scFv concentration in a colloidal suspensions of SPIONs–PEG–scFv by the Bradford assay and ICP-AES quantification of nanoparticles uptaken by HER2-overexpressing BT-474 cancer cells. See DOI: 10.1039/c6ra06076e |
| This journal is © The Royal Society of Chemistry 2016 |




