Application of spent mushroom (Lentinula edodes) substrate and acclimated sewage sludge on the bioremediation of polycyclic aromatic hydrocarbon polluted soil
Can Wang,
Dong Yu,
Wenjin Shi,
Kai Jiao,
Bin Wu and
Heng Xu*
Key Laboratory of Bio-resources and Eco-environment (Ministry of Education), College of Life Sciences, Sichuan University, Chengdu, Sichuan 610064, China. E-mail: xuheng64@sina.com
First published on 4th April 2016
Abstract
Two widespread agro-industrial wastes, spent mushroom substrate of Lentinus edodes (SMS) and acclimated sewage sludge (ASS), were investigated for the remediation of multi-polycyclic aromatic hydrocarbon (multi-PAH) polluted soil. Results clearly showed that multi-PAH contaminated soil was not amendable to natural attenuation. In contrast, application of sterilized ASS and SMS significantly stimulated resident microbes in the soil and consequently enhanced the removal efficiency of PAHs. Active ASS and SMS exerted a better effect than sterilized ones. ASS improved total bacterial abundance better, and it had a better degradation effect on low molecular weight PAHs than SMS, while SMS improved the fungal population better and had a better effect on the degradation of heavy molecular weight PAHs by its ligninolytic enzymes. The combination of ASS–SMS exerts the best PAH removal and soil improvement effect with degradation rates beyond 94% in all PAHs. The present study strongly supports SAS and SMS as amendments for the remediation or the improvement of environmental soil which could also open a new approach for recycling agro-industry waste.
1 Introduction
Polycyclic aromatic hydrocarbons (PAHs) are persistent organic pollutants which could be toxic, carcinogenic and mutagenic to an organism. With industry development and human activity, PAHs have become ubiquitous contaminants in the environment and the remediation of them has gained a lot of attention.1 There are many alternatives to remediate PAH contaminated sites including in situ or ex situ techniques which rely on either physicochemical or biological approaches.2 However, the leading process used for successful elimination of PAHs from the environment is bioremediation based on microbial transformation and degradation, which is generally achieved via biostimulation or bioaugmentation depending on soil conditions and microbial community structure.3,4 Biostimulation on indigenous microbial communities of PAH-impacted soil can be achieved through the addition of organic matter, which was found to be successful.5 The bioremediation of PAH polluted sites could also be based on the addition of exogenous microorganisms, which is referred to as bioaugmentation.3Lignin-degrading fungi are known to produce extracellular lignin-modifying enzymes with low substrate specificity including laccase and Mn-peroxidase (MnP) which enable them to degrade a wide range of organic pollutants like PAH.6 Mushroom production is the biggest solid-state-fermentation industry in the world with global production greater than six million tons.7 At least 30 million tons of spent mushroom substrate (SMS) is produced annually which is challenging for disposal management.8 Lentinus edodes (L. edodes) is the world’s second largest cultivated mushroom and a lignin-degrading fungus which has been found to be an efficient producer of both laccase and MnP.9,10 High levels of residual nutrients and enzymes left in SMS make it an appropriate and low cost material for the remediation of PAH contaminated soil.11 The utilization of SMS on soil remediation also provides an applicable approach for disposal management.
Urbanization and industrialization have dramatically increased the volume of sewage sludge produced by wastewater treatment plants throughout the world. In China, approximately 30 million tons of sewage sludge was generated in 2010 and only 44% of the sewage sludge was used in the agriculture sector (this compares with: 71% in UK, 54% in Germany, 54% in Spain, 65% in France, and 60–65% in the USA).12 Application of sewage sludge to agricultural land delivers well recognized benefits in terms of nutrient addition, soil organic matter increase, soil structure improvement and consequently crop yield growth.13 In addition, sewage sludge may enhance the bioremediation of hydrocarbon-polluted soils by introducing microorganisms and nutrients that compensate for scant indigenous microbial activity.14 However, its remediation effect on PAH contaminated soil was not very good, which restricted its potential use on soil remediation.14 But some studies suggested that the acclimation of activated sewage sludge by particular organics could contribute to pollutant tolerance and degradation ability of microorganisms in sludge, and then enhance its utilization value.15,16 Reports indicated that a consortium acclimated by some PAHs could also be used to degrade other PAHs because of their similar molecular formation and degradation mechanism.17,18 In this study, acclimated sewage sludge (ASS) by simulated wastewater contaminated with naphthalene (Nap) and pyrene (Pyr) was used to investigate its potential for soil remediation. Nap and Pyr were chosen for acclimation because they are typical and representative PAHs in the present study.
This research provides direct evidence about the feasibility of SMS and ASS on the remediation of PAH polluted soil in view of being environmentally and profitably sound. Furthermore, this necessarily implies a variety of manipulations of the wastes prior to their application, to gain more insight into their ability to act as a supplier of exogenous complex microbiota and organic matter.
In this research, obtained ASS was applied in PAH contaminated soil with SMS for the investigation of their effects on soil remediation. For the investigation of their bioaugmentation and biostimulation effect, active material or sterilized material were both introduced. Application of ASS and SMS alone or combined were both implemented to seek the best remediation effect. The PAH dissipation rate, soil pH, ligninolytic enzymes, soil microorganism quantity, and soil respiration after remediation were determined. In addition, soil ecotoxicity represented by total hydrolase activity (THA), dehydrogenase activity (DHA), soil urease activity, soil germinability and reduction of carcinogenic risk assessment (RCRA) were also analyzed.
2 Methods and materials
2.1 Amendment preparation
SMS of L. edodes was obtained from a mushroom cultivation base in Shuangliu Chengdu, China consisting of 1% gypsum, 20% wheat brain, and 79% saw dust. Before application, SMS was smashed into pieces about 0.2–0.5 cm in size. Sewage sludge obtained from Chengdu First Sewage Treatment Plant (Chengdu, China) was acclimated by simulated wastewater containing 500 mg L−1 sodium acetate, 500 g L−1 ammonium chloride, 1000 mg L−1 sodium chloride, 300 mg L−1 potassium dihydrogen phosphate, and 300 mg L−1 dipotassium phosphate. Pyr and Nap dissolved in acetone were added, their dosage was gradually increased and finally maintained at 170 mg L−1. Wastewater was changed every 3 days and the acclimation process lasted for 4 weeks before application. After acclimation, ASS was centrifuged and the sediment was preserved at 0 °C before application.2.2 Soil preparation
A soil sample originally free of PAHs used for the study was collected from a paddy field in Shuangliu Chengdu, Sichuan province, China. The soil sample was air dried and sieved by a 2 mm sieve to remove foreign substances. The soil was spiked with Pyr, Nap, phenanthrene (Phe), anthracene (AnT), and fluoranthene (FluA) at 200 mg kg−1 by dissolving in acetone and eventually classified to heavily contaminated soil according to the class of soil contamination by Maliszewska-Kordybach et al.19 All treatments contained the same amount of acetone and the remediation process was conducted after acetone evaporation. The initial concentration of PAHs in soil and their toxic equivalency factor (TEF) values are presented in Table 1. Physicochemical characteristics of the soil and amendments are presented in Table 2.| PAHs | Abbreviation | Rings | Concentration (mg kg−1) | TEF valuea | Carcinogenic classificationb |
|---|---|---|---|---|---|
| a TEF: toxic equivalence factor.b IARC 2010: 1 carcinogenic, 2A probable carcinogenic, 2B possible carcinogenic, 3 not classifiable as carcinogenic. | |||||
| Naphthalene | Nap | 2 | 200 | 0.001 | 3 |
| Phenanthrene | Phe | 3 | 200 | 0.001 | 3 |
| Anthracene | AnT | 3 | 200 | 0.01 | 3 |
| Fluoranthene | FluA | 4 | 200 | 0.001 | 3 |
| Pyrene | Pyr | 4 | 200 | 0.001 | 3 |
| Parameter | SMS | ASS | Soil |
|---|---|---|---|
| OM (%) | 90.0 ± 0.5 | 40.1 ± 0.3 | 16.8 ± 1.31 |
| Initial pH | 4.3 ± 0.11 | 6.7 ± 0.2 | 8.7 ± 0.2 |
| C (%) | 49.8 ± 0.6 | 43.5 ± 1.2 | 0.7 ± 0.01 |
| N (%) | 0.59 ± 0.01 | 3.5 ± 0.16 | 0.03 ± 0.001 |
| P (%) | 0.61 ± 0.02 | 2.4 ± 0.11 | 0.14 ± 0.015 |
| K (%) | 0.22 ± 0.01 | 1.6 ± 0.07 | 0.42 ± 0.02 |
| Humidity (%) | 59.6 ± 0.6 | 96.4 ± 1.1 | 5.3 ± 0.1 |
| Bacterial counts (log10 CFU g−1) | 8.66 ± 0.14 | 9.21 ± 0.22 | 5.82 ± 0.32 |
| Fungal counts (log10 CFU g−1) | 7.21 ± 0.19 | 5.24 ± 0.14 | 3.88 ± 0.15 |
2.3 Remediation process
The remediation process was carried out in plastic pots with 200 g (dry mass) of contaminated soil each. The soil moisture content was adjusted to 70% of their water holding capacity prior to incubation by adding ultrapure water. The soil moisture content was determined by a gravimetric method. 100 g of the soil sample was dried in an oven at 105 °C for 12 h and the water content was calculated by subtracting the oven dry weight from the initial field soil weight.20 Water holding capacity (WHC) was measured by soaking the soil for 12 h in a plastic cylinder (diameter = 35 mm, height = 40 mm) with a 0.3 mm nylon mesh in the bottom. After the soil was drained for 1 h, the soil was emptied into a container and WHC was determined for the gravimetric water content.21Treatments were set as shown in Table 3. Treatment 1 (T1) was set to investigate the effect of natural attenuation, and T2 was set in comparison with T1 to reveal the role of the original microorganism in the soil. T3 and T4 were set to analyze the bioaugmentation and biostimulation effect of ASS. T5 and T6 were set for the same reason. T7 and T8 were set to investigate the combination remediation effect of ASS and SMS. SMS or ASS was homogeneously mixed with soil artificially. Each treatment was carried out in triplicate and incubated for 42 days under stable condition in a greenhouse with the temperature controlled between 25 and 30 °C. Ultrapure water was sprayed everyday to maintain soil moisture.
| Treatment | Soil (g) | SMS (%) | Sterilized SMS (%) | ASS (%) | Sterilized ASS (%) |
|---|---|---|---|---|---|
| 1 | 200 | — | — | — | — |
| 2 | 200 (sterilized) | — | — | — | — |
| 3 | 200 | — | — | 10 | — |
| 4 | 200 | — | — | — | 10 |
| 5 | 200 | 25 | — | — | — |
| 6 | 200 | — | 25 | — | — |
| 7 | 200 | 25 | — | 10 | — |
| 8 | 200 | — | 25 | — | 10 |
2.4 pH and ligninolytic enzyme analysis
The pH of soil was determined in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 water soluble extract (w/v).22 5 g of soil was extracted with 50 mL of distilled water in a 100 mL Erlenmeyer flask, and the pH value was determined by a pH meter (PB-10) after standing for 10 min.
10 water soluble extract (w/v).22 5 g of soil was extracted with 50 mL of distilled water in a 100 mL Erlenmeyer flask, and the pH value was determined by a pH meter (PB-10) after standing for 10 min.
Ligninolytic enzymes were extracted from the soil samples (3 g) at 5 °C for 1 h with 30 mL of 0.1 M acetate buffer (pH 5.0), containing 0.005 M CaCl2, 0.05% Tween 80, and 3% insoluble polyvinylpolypyrrolidone.23 Then, the aqueous suspension was centrifuged (6000 × g, 30 min) and the supernatant was assayed for ligninolytic enzyme activity.
Laccase activity was determined through spectrophotometry with 2,2-azino-di-[3-ethyl-benzothiazoline-sulphonate] (ABTS) as the substrate at 420 nm.24 The assay mixture (1 mL) contained 880 μL of 100 mM sodium acetate buffer (pH 5.0), 100 μL of ABTS stock, and 20 μL of appropriately diluted crude enzyme. The enzyme activity was calculated using the molar extinction coefficient of oxidized ABTS (E420 = 3.6 × 104 M−1 cm−1). One unit of enzyme activity was defined as the amount of enzyme which produced 1 μmol of product per minute.
MnP activity was assayed by oxidation of Mn2+ and formation of a Mn3+–tartrate complex (ε 2386.5 mM−1 cm−1) from 0.1 mM MnSO4 in 0.1 mM sodium tartrate buffer at pH 5 with 0.1 mM H2O2. One unit of enzyme activity was defined as the amount of the enzyme which can produce 1 μM Mn3+ from the oxidation of Mn2+ per minute.25
2.5 PAH removal and microbe count in soil
Total extraction of PAHs was performed according to Garcia-Delgado et al. and analyzed by Gas Chromatography Mass Spectrometry (GC-MS) was performed according to De Luca et al.26,27 Acetone/hexane (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was used as the extract solvent. Three replicates were performed. 5 g of the sample plus 25 mL of the solvent were shaken for 2 h at 200 rpm. The solution was left for 30 min before decanting. Then solutions were filtered through 0.45 μm nylon syringe filters (Whatman International, Maidstone, UK). One mL of solution was dried by N2 flow. The residue was dissolved in 1 mL of acetonitrile solvent. The GC-MS unit (Thermo-Quest 8000 top with an electronic control of pressure) was equipped with a splitless injector (injector temperature: 260 °C; splitting time: 1 min; flow: 100 cm3 min−1). A carrier pressure of 130 kPa at 90 °C in the head of the column was selected. The sample was injected into the column (a CP SIL 8DB, 50 m × 0.25 mm fused silica, low-bleed column; Chrompack) held at 90 °C for 1 min and programmed from 90 to 180 °C at 5 °C min−1, and from 180 to 280 °C at 15 °C min−1. Helium was the carrier gas and the flow rate was 1 cm3 min−1. The mass spectrometer was a Thermo-Quest Voyager quadrupole working in the electron ionization mode (70 eV). The temperatures of the transfer line and ionization source were 260 and 200 °C, respectively. Mass spectra were scanned from 100 to 350 m/z ratio (acquisition speed: 2 scan per s). Each sample was analyzed twice. The PAH degradation rate was calculated as follows:
1) was used as the extract solvent. Three replicates were performed. 5 g of the sample plus 25 mL of the solvent were shaken for 2 h at 200 rpm. The solution was left for 30 min before decanting. Then solutions were filtered through 0.45 μm nylon syringe filters (Whatman International, Maidstone, UK). One mL of solution was dried by N2 flow. The residue was dissolved in 1 mL of acetonitrile solvent. The GC-MS unit (Thermo-Quest 8000 top with an electronic control of pressure) was equipped with a splitless injector (injector temperature: 260 °C; splitting time: 1 min; flow: 100 cm3 min−1). A carrier pressure of 130 kPa at 90 °C in the head of the column was selected. The sample was injected into the column (a CP SIL 8DB, 50 m × 0.25 mm fused silica, low-bleed column; Chrompack) held at 90 °C for 1 min and programmed from 90 to 180 °C at 5 °C min−1, and from 180 to 280 °C at 15 °C min−1. Helium was the carrier gas and the flow rate was 1 cm3 min−1. The mass spectrometer was a Thermo-Quest Voyager quadrupole working in the electron ionization mode (70 eV). The temperatures of the transfer line and ionization source were 260 and 200 °C, respectively. Mass spectra were scanned from 100 to 350 m/z ratio (acquisition speed: 2 scan per s). Each sample was analyzed twice. The PAH degradation rate was calculated as follows:
 | (1) |
Microbe counts in the soil were made on an agar medium, the total number of colony forming units (CFUs) of microbe were counted. Aqueous extracts of a 5 g soil sample were serially diluted in sterilized distilled water. Three appropriate portions (1 mL) of serial dilutions were all repeated twice, spread in Petri dishes, then mixed well with plate count agar (tryptone: 5.0 g, yeast extract: 2.5 g, glucose: 1.0 g, agar: 15.0 g, distill water: 1000 mL, pH: 7.0 ± 0.2, sterilization: 121 °C for 30 min) for bacteria counting, and also mixed well with potato dextrose agar (potato: 200 g, glucose: 20.0 g, agar: 15.0 g, distill water: 1000 mL, sterilization: 115 °C for 30 min) for fungal counting. Bacteria were counted after incubation at 28 °C for 3–5 days in the dark, fungi were counted after incubation at 30 °C for 3–5 days in the dark.
2.6 Biochemical determination and soil ecotoxicity estimation
Soil total microbial activity and eco-toxicological assessment of treatments were carried out by two soil health parameters: THA by hydrolysis of fluorescein diacetate (FDA), which was presented as the content of fluorescein in dry soil (mg g−1 24 h−1); and DHA with 2,3,5-triphenyltetrazolium chloride, which was presented as the content of triphenylformazan in dry soil (mg g−1 24 h−1).28,29 Soil urease activity was presented as the content of amino nitrogen in dry soil (mg g−1 24 h−1).30Soil respiration was determined according to Park et al.31 10 g of soil (amended with 0.05 g glucose) with water content adjusted to 50–60% of its WHC was incubated for 24 h at 28 °C in a hermetically sealed flask and the produced CO2 was measured by titration. Soil respiration was expressed as μg CO2 per g soil per hour.
Soil germinability test was conducted as described by Garcia-Delgado et al.32 25 Lactuca sativa seeds were incubated for 3 days in the dark at 28 °C in 90 mm Petri dishes containing Whatman no. 42 filters soaked with 2.0 mL of acetone![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) hexane extracts obtained during PAH extraction from treatments at 42 days of incubation per triplicate. After addition of PAH extracts, the filters were kept at room temperature for 12 h to allow solvent evaporation. Then, 2.0 mL of distilled water was added on a daily basis. A test conducted with the presence of distilled water was also run for three times as a control. Germinated seeds were counted. The percentage of germinability was calculated from the following equation:
hexane extracts obtained during PAH extraction from treatments at 42 days of incubation per triplicate. After addition of PAH extracts, the filters were kept at room temperature for 12 h to allow solvent evaporation. Then, 2.0 mL of distilled water was added on a daily basis. A test conducted with the presence of distilled water was also run for three times as a control. Germinated seeds were counted. The percentage of germinability was calculated from the following equation:
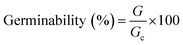 | (2) |
RCRA of the PAH present in the soil was based on the Nisbet and LaGoy toxic equivalency factors (TEFs) of PAH for environmental exposure and was determined as follows:33
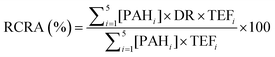 | (3) |
2.7 Data analysis
Mean and standard deviation values of three replicates were calculated in the present experiment. Statistical analyses P < 0.05 were performed using SPSS 21.0. All figures were produced using Origin V8.5 software.3 Results and discussion
3.1 pH and ligninolytic enzyme analysis
The pH values of the treatments at 42 day (d) are shown in Fig. 1. Ranged from high to low, the order of pH in the treatments was: T1, T2, T3, T4, T6, T5, T8, T7. However, no significant difference was found among pH values of the treatments. T7 presented the lowest pH value (8.08 ± 0.51), but only 8.39% lower than T1 (8.82 ± 0.16). The differentiation of amendments in each treatment caused the differentiation of the pH values. Treatments amended with SMS (T5, 6, 7, 8) presented lower pH than treatments without amendment or amended with ASS. This is mainly due to the lower pH of SMS (pH at 4.3 ± 0.11) than ASS (pH at 6.7 ± 0.2) and organic acid secreted by living fungi in SMS during the experiment.34 The longest survival time and strong vitality of fungi verified by its high laccase activity could be one main contributor of the lowest pH in T7, the acidity of ASS and SMS could be another.The activities of laccase and MnP in the treatments were determined by their acceleration ability in the early oxidation steps of PAHs and as an indicator of fungal activity (Fig. 2). MnP was detected in all the treatments except T1 and T2, but only lasted in T5 and T7 until the end of the incubation. T5 and T7 maintained a relatively high MnP level before 35 d but decreased with time; at 42 d low MnP activities were detected in T5 and T7, and MnP activity in T5 was higher than T7. MnP activities in T3 and T4 were low before 21 d and they were not detected at 28 d. T6 and T8, which were amended with sterilized SMS, presented increasing MnP activities at an early stage but decreased dramatically and vanished at the end. Laccase was only detected in T5–T8, which were amended with SMS. T5 presented the highest laccase activity but declined dramatically with time. T7 showed lower laccase activity than T5 at 7 d. Laccase activities of both of them declined with time but T7 declined much slower and presented the highest laccase activity at 42 d. No laccase activity was detected in T6 and very low laccase activity was detected in T8 at 7 d. During incubation, they presented an increasing followed by a decreasing trend and T6 maintained a higher laccase level than T8 at 42 d.
 | ||
| Fig. 2 Ligninolytic enzymes (MnP and laccase) activity in treatments with different amendments during incubation. Error bars represent the standard deviation of three sampled pots. | ||
It was noteworthy that no treatment developed filamentous fungi except T5 and T7 during incubation. However, fungi colonization in T5 and T7 was not completed, the hyphae lasted a few days and were not observed later because of their poor colonization. Different colonization depth and survival time of L. edodes mycelia could be the main reason for various expression levels of laccase and MnP. Higher activities of MnP and laccase in treatments amended with SMS were secreted by living L. edodes mycelia in SMS, but declined with time because of the decrease of the L. edodes population due to poor colonization, nutrition exhaustion and competition with indigenous microorganisms. This coincides with a report by Garcia-Delgado et al., where shrinking laccase and, to a much lower extent, MnP activity were only detected in the bioaugmented microcosms amended with SMS.29 Enzyme activities of treatments with sterilized SMS showed an increasing followed by a decreasing pattern during incubation. Another study by Garcia-Delgado et al. presented similar results, where no ligninolytic enzyme was found in natural attenuation treatments and a decrease after an increase in ligninolytic enzyme activity was presented in treatments with sterilized SMS.35 Despite of microbe depletion by sterilization, tremendous nutrition was introduced by sterilized SMS which stimulated the development of indigenous fungi in T4, T6 and T8. Hence, the increase of enzyme activities. The decrease later may also be due to the exhaustion of nutrition and competition of other microorganisms.36 Laccase and MnP are important parameters which reflect biological viability of SMS during incubation where remediation purposes are concerned. Long lasting and comparatively high levels of laccase and MnP in T5 and T7 made these treatments effective in the management of organic pollutant contaminated soils.
3.2 Removal of PAHs
Degradation rates of PAHs during incubation are presented in Table 4. At 42 d, degradation rates for all PAHs were beyond 90% in all treatments except T1 and T2. T7 presented the best PAH removal efficiency. Degradation rates for all PAHs in T7 were beyond 94%, and its degradation rate for Nap was 100%. The most degradable PAHs were those with low molecular weight (Nap and Phe). PAHs with higher molecular weight (Pyr and FluA) presented lower degradation rates. Degradation rates for PAHs with higher molecular weight were higher in T5 than T3, indicating higher degradation efficiency for high molecular weight PAHs of SMS on account of higher ligninolytic enzymes level (Fig. 2). Research showed that fungi had an advantage in biodegrading heavy molecular weight PAHs compared with bacteria due to their ability to produce ligninolytic enzymes.37 While for PAHs with lower molecular weight, the degradation rate was higher in T3, which may be due to its higher bacteria quantity. It has been reported that bacteria metabolism could be the main contributor in the passivation of low molecular weight PAHs.29| PAH degradation rate (%) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nap | Ant | Phe | FluA | Pyr | |||||||||||
| 7 d | 21 d | 42 d | 7 d | 21 d | 42 d | 7 d | 21 d | 42 d | 7 d | 21 d | 42 d | 7 d | 21 d | 42 d | |
| a Different lowercase letters indicate significant differences between degradation rates of the same PAH in treatments at the same sampling time (P < 0.05). Different uppercase letters denote significant differences of degradation rates of the same PAH in the same treatment at a different sampling time (7 d, 21 d and 42 d) (P < 0.05). | |||||||||||||||
| T1 | 42.9A,b | 65.2B,b | 83.2C,b | 40.1A,b | 58.7B,b | 78.4C,a | 40.8A,b | 61.0B,b | 79.5C,b | 34.1A,b | 54.7B,b | 78.1C,a | 27.2A,b | 50.0B,b | 75.1C,a |
| T2 | 33.9A,a | 56.9B,a | 78.6C,a | 29.7A,a | 50.2B,a | 77.2C,a | 31.9A,a | 53.0B,a | 73.1C,a | 22.0A,a | 40.6B,a | 76.0C,a | 18.1A,a | 39.7B,a | 74.0C,a |
| T3 | 72.2A,e | 89.3B,de | 99.2C,d | 66.2A,ef | 86.2B,de | 93.6C,de | 68.4A,e | 87.8B,ef | 98.9C,d | 57.3A,e | 83.1B,e | 92.8C,bc | 47.7A,e | 81.5B,de | 92.0C,c |
| T4 | 63.2A,c | 84.3B,c | 97.4C,cd | 60.0A,d | 81.4B,cd | 90.3C,c | 60.8A,cd | 82.4B,d | 97.4C,cd | 51.8A,ad | 76.8B,cd | 91.2C,b | 42.8A,cd | 71.1B,c | 90.4C,c |
| T5 | 73.5A,e | 91.2B,e | 98.7C,cd | 67.4A,f | 83.9B,d | 92.0C,cd | 69.4A,ef | 85.0B,de | 97.2C,cd | 62.2A,f | 84.4B,e | 93.2C,bc | 55.0A,f | 82.8B,e | 92.7C,c |
| T6 | 60.8A,c | 82.5B,c | 94.3C,c | 56.0A,c | 77.6B,c | 86.8C,b | 57.9A,c | 77.0B,c | 92.2C,c | 49.7A,c | 73.8B,c | 91.2C,b | 39.5A,c | 71.2B,c | 85.0C,b |
| T7 | 73.0A,e | 91.5B,e | 100.0C,d | 69.6A,f | 89.7B,e | 95.0B,e | 71.6A,f | 91.0B,f | 99.2C,d | 68.2A,g | 86.6B,e | 95.5C,c | 62.4A,g | 85.6B,e | 94.2C,c |
| T8 | 66.9A,d | 86.3B,cd | 98.2C,cd | 62.7A,de | 83.8B,d | 91.2C,cd | 62.1A,d | 81.6B,d | 96.6C,cd | 54.0A,de | 79.0B,d | 91.6C,b | 44.1A,de | 76.8B,d | 91.2C,c |
Fig. 3 presents residual PAH concentrations in treatments during incubation. Residual PAHs in treatments decreased with time and the reduction quickly slowed followed by a slow trend. At 42 d, all the treatments reached the standard of non-contaminated soil except T1 and T2.19 T2 showed the highest PAH remnants (282.39 ± 17.23 mg kg−1) followed by T1 (211.38 ± 16.45 mg kg−1), while T7 presented the lowest residual PAHs (32.01 ± 5.63 mg kg−1, 32.3% lower than T3 which was next to the lowest). No significant difference was found between total residual PAH concentrations of T3 and T5 (47.3 ± 6.12 and 52.3 ± 11.7 mg kg−1 at 42 d, respectively), however, for a different PAH, different residual concentrations were presented as described in the previous paragraph.
The fates of PAHs in contaminated soil mainly include leaching, biodegradation, plant uptake, volatilization, photo-degradation and other biotic or abiotic losses.38 Apparently, natural attenuation of PAH contaminated soil was infeasible, and the combined use of SMS and ASS showed the best remediation effect in the present study. Biostimulation of indigenous microorganisms by organic matter and bioaugmentation by living microorganisms in amendment could be the main cause of PAH removal in soil.39 In this study, the better PAH removal efficiency of treatments with active material than treatments with sterilized material indicated the main position of bioaugmentation in the remediation process other than biostimulation. This was contrary to the study about comparative assessment of industrial polluted soil bioremediation through biostimulation and bioaugmentation by S. Lladó et al., in which a lignocellulosic substrate (biostimulation approach) had a better effect on PAH degradation than fungal bioaugmentation with white-rot fungi (bioaugmentation approach) because related fungi were hampered by active native soil microbiota.40 But it was supported by a study of Garcia-Delgado et al. about soil bioremediation by spent mushroom substrate, where bioaugmentation through SMS had a better effect on PAH passivation than biostimulation through sterilized SMS.35 It was also in agreement with the study about remediation of diesel contaminated soil by Taccari et al., where bioaugmentation through inoculation of adapted microbial consortium had a better effect on diesel degradation than biostimulation through compost addition.41 Mycoremediation potential of fungi has been well investigated in the operation of soil with a single pollutant or contaminated by multi-pollutants. For example, Wu et al. and Zhou et al. have sufficiently investigated the mycoremediation potential of Coprinus comatus and Clitocybe maxima etc. on soil contaminated with heavy metal and organic pollutants. In this respect, possible mycoaugmentation approaches might fail due to promoted growth of indigenous fungal and bacterial populations in practical operation. A lab-scale assessment of interactions between indigenous microbiota and the selected allochthonous species is needed when it comes to the implementation of bioaugmentation.
3.3 Estimation of microbes in soil
The bacterial and fungal population during incubation are presented in Fig. 4A and B, respectively. Treatments with amendment (T3–T8) showed a much higher microbe population than control (T1 and T2). T7 showed the highest but with a decreasing microbe population during incubation. T5 followed a similar pattern with T7. T3 followed this pattern only with the bacteria population. The fungal population of T3 did not change dramatically like T4. The bacterial population of T4, T6 and T8 presented an increasing trend first but decreased later, and the fungi population of T6 and T8 followed a similar pattern. At 42 d, the difference in the microbe population for treatments with active material and sterilized material reduced, such as T7 and T8. T2 developed the lowest microbe population but a significant microbe growth was detected during incubation. However, no significant microbe growth was found in T1. At 42 d, no significant difference in the microbe population was found between T2 and T1.T1 and T2 were set as controls to assess the effect of natural attenuation. A microbe increase in T2 (which had been sterilized) was due to microorganism colonization from the ambient environment, while a slight microbe growth in T1 may be evidence of soil detoxicity. The highest microbe population in T7 was predictable because of the inherent microbial population of SMS and ASS. Significant time dependent changes in the fungal population in T6 and T8 indicated that with sterilized SMS, fungal growth was stimulated, which was in agreement with several former studies which showed that the application of sterilized lignocellulose waste exerted a positive effect on the resident fungi in contaminated soils.42–44 This could be explained by its valuable nutrition supplement for resident fungi in sterilized SMS corroborated by the long term large amount of fungal colonization in treatments amended with SMS. A high fungal population of T5 and T7 despite the slight decrease with time also indicated favourable colonization of fungi in soil led by an abundant fungal population and nutriment in SMS. According to a study by Ribas et al., L. edodes tend to have greater tolerance to mycelium fragmentation when performed with sample homogenization than other fungus species like A. subrufescens.45 Similar results were found in the estimation of the bacterial population in T3, T5 and T7. T3, which was amended with ASS, showed a high bacterial population but a comparatively low fungal population, indicating a great amount of bacteria but a small number of fungus colonization during the acclimation of ASS and it was in agreement with Table 1. It can been seen that the microbial population in treatments corresponded with PAH residue (Fig. 3). Treatments with a higher microbial population had higher PAH degradation and lower PAH residue which verified the role of microorganisms in soil remediation.
3.4 Biochemical determination and soil eco-toxicity estimation
DHA is considered an indicator of oxidative metabolism and microbiological activity in soils and it has been used as a possible index of detoxification in remediation microcosms as previously suggested for hydrocarbon polluted soils.46,47 The DHA increase in T4, T6 and T8 could be due to stimulation of aboriginal microorganisms by organic matter in amendments, while increased DHA in T1 and T2 could be due to a PAH toxicity decrease because of indigenous microbe metabolism and microbe colonization from the ambient environment, respectively. A high DHA in T3, T5 and T7 could be due to colonization and metabolism of a high microbe density in amendments. With nutrient consumption, microbiological activity went down but was maintained at a relatively high level, indicating that soil microbiota retained a high functional activity despite a prolonged incubation time which may be caused by decreased PAH toxicity and this corresponds with a former report.29
THA, another soil eco-toxicological parameter, which has been found to be significantly correlated with microbial biomass and could be used as an alternative estimate of the content of the size of soil microflora, is a good indicator of total microbial activity.28,48 It has been reported that the addition of organic matter could lead to a significant increase in microbial biomass and microbial activity of the soil because of a stimulation effect and decline later because of nutrient consumption.49 This short term change was in agreement with THA of T3, T5 and T7 which increased drastically at 21 d but decreased at 42 d. As for treatments without amendments or amended with sterilized material, increased THA was found which shared a lot with DHA. The similarity between THA and DHA of treatments confirmed the functional activity of nutritious material and the abundant microbes in amendments (ASS and SMS) on contaminated soils.
Urease is one of the most commonly assayed soil enzymes because its great influence on the transformation and fate of important fertilizers and could be an indicator of soil fertility.30 Pollutants could exert complex effects on soil urease activity by direct activation of them on enzymatic molecules and competition with enzymes which eventually lead to the variation of enzymatic activity.50 It has been reported that soil urease activity was sensitive to soil toxicity and decreased heavily with the existence of PAHs.51 Higher urease activity in treatments with amendments indicated the improvement of soil fertility and the decrease of soil toxicity. Higher urease activity in T3 than T5 indicated higher fertility in ASS compared to SMS. This may be because of the greater amount of available organic matter in ASS than SMS which is mainly made from sawdust and wheat bran. Some sources like SMS might supply enough nutrients to restore the microbial population, but become unavailable because of their low solubility. Knowledge of the bioavailability of nutrients is necessary in the planning of an efficient bioremediation strategy.52 The above results implied that ASS showed a greater contribution to urease activity than SMS and was more suitable for the improvement of soil fertility in the soil remediation process.
Higher CO2 production in T3–T8 than that in the control provided conclusive evidence for the potential character of PAH degradation and microbial metabolic increase in soil after remediation. The highest CO2 production in T7, and higher CO2 production in T3 than that in T5 was in agreement with their bacterial population. It can be deduced that the bacterial population posed a much greater contribution on soil respiration since bacteria was the majority in most treatments. The decrease of CO2 production in treatments amended with active material indicated the decrease of the microbial population and metabolism due to nutrient consumption.29,53 Increase of CO2 production was found in treatments amended with sterilized amendments or sterilized before. For T2, colonization and development of ambient microorganisms could be the main reason. For others, it was because of the biostimulation effect of amendments on indigenous microorganisms.
Higher germinability in treatments with amendments indicated an improvement of soil quality and decrease of soil toxicity. Higher germinability of treatments amended with active materials (T7, T3, T5) reconfirmed their better functional effect on soil detoxicity. Higher germinability rate of T3 than that of T5 could be ascribed to the higher content of utilizable nutrition for microbes in ASS than in SMS which is mainly constituted by sawdust and wheat bran. Higher germinability in T1 and T2 than in the initial soil indicated that natural attenuation could contribute to soil detoxicity because of the adaptation and metabolism of indigenous microorganisms, but this contribution was relatively small.35,36
RCRA in T1 and T2 verified the existence of natural attenuation and its weak effect. The close RCRA of T3 and T5 indicated that ASS and SMS exerted a similar detoxicity effect on PAH polluted soil. In this case, although a higher degradation of Phe and FluA in T5 was observed compared with T3, the total residual PAH concentration in T5 was higher (Fig. 3), leading to higher RCRA in T3. Better degradation of AnT (exhibited the highest TEF value) in T3 could be the participating part. By this token, sterilized or not, ASS exerted a greater contribution for RCRA than SMS in this study, and combined application of SMS and ASS was the optimal choice with regards to RCRA.
4 Conclusions
The present work identified the potential use of L. edodes SMS and ASS for the remediation of multi-PAH polluted soil. Although exclusive use of active or sterilized SMS and ASS can exert a certain remediation and soil improvement effect, their combined application presented a better effect on the microbial community, PAH passivation, soil improvement and toxicity reduction etc. Removal rates of all PAHs in the ASS-SMS combined treatment was beyond 94% and the total PAH residue was only 32.01 ± 5.63 mg kg−1 after 42 d. Remarkably, ASS exerted a better effect on the bacterial increment and degradation of low molecular weight PAHs, while SMS exerted a better effect on heavy molecular weight PAHs. The wide spatiotemporal and low cost availability of SMS and ASS, along with their confirmed efficacy in PAH biodegradation, make their application technically feasible for environmental remediation purposes and opens a perspective for relevant agro-industry waste management.Acknowledgements
This study was financially supported by the National High Technology Research and Development Program of China (No. 2013AA06A210), and the NSFC (No. J1103518). The authors also wish to thank Professor Guanglei Cheng and Dong Yu from Sichuan University for their technical assistance.References
- H. Hamdi, L. Manusadžianas, I. Aoyama and N. Jedidi, Chemosphere, 2006, 65, 1153–1162 CrossRef CAS PubMed.
- S. Gan, E. V. Lau and H. K. Ng, J. Hazard. Mater., 2009, 172, 532–549 CrossRef CAS PubMed.
- A. Haritash and C. Kaushik, J. Hazard. Mater., 2009, 169, 1–15 CrossRef CAS PubMed.
- M. Tyagi, M. M. R. da Fonseca and C. C. C. R. de Carvalho, Biodegradation, 2011, 22, 231–241 CrossRef CAS PubMed.
- S. Covino, K. Svobodova, M. Cvancarova, A. D’Annibale, M. Petruccioli, F. Federici, Z. Kresinova, E. Galli and T. Cajthaml, Chemosphere, 2010, 79, 855–864 CrossRef CAS PubMed.
- A. Majcherczyk, C. Johannes and A. Huttermann, Enzyme Microb. Technol., 1998, 22, 335–341 CrossRef CAS.
- K. T. Semple, B. J. Reid and T. R. Fermor, Environ. Pollut., 2001, 112, 269–283 CrossRef CAS PubMed.
- W. S. Kwak, S. H. Jung and Y. I. Kim, Bioresour. Technol., 2008, 99, 2947–2955 CrossRef CAS PubMed.
- P. S. Bisen, R. K. Baghel, B. S. Sanodiya, G. S. Thakur and G. B. K. S. Prasad, Curr. Med. Chem., 2010, 17, 2419–2430 CrossRef CAS PubMed.
- S. Covino, K. Svobodová, Z. Křesinová, M. Petruccioli, F. Federici, A. D’Annibale, M. Čvančarová and T. Cajthaml, Bioresour. Technol., 2010, 101, 3004–3012 CrossRef CAS PubMed.
- K. T. Semple, B. J. Reid and T. Fermor, Environ. Pollut., 2001, 112, 269–283 CrossRef CAS PubMed.
- S. Khan, N. Wang, B. J. Reid, A. Freddo and C. Cai, Environ. Pollut., 2013, 175, 64–68 CrossRef CAS PubMed.
- R. El-Motaium and M. A. El-Seoud, Nutr. Cycling Agroecosyst., 2007, 78, 133–142 CrossRef.
- S. Khan, W. Ning, B. J. Reid, A. Freddo and C. Chao, Environ. Pollut., 2013, 175, 64–68 CrossRef CAS PubMed.
- L. Wang, J. Zhao and Y. Li, J. Chem. Technol. Biotechnol., 2014, 89, 391–400 CrossRef CAS.
- S. F. Yang, C. F. Lin, C. J. Wu, K. K. Ng, A. Y. C. Lin and P. K. A. Hong, Water Res., 2012, 46, 1301–1308 CrossRef CAS PubMed.
- C. L. Guo, H. W. Zhou, Y. S. Wong and N. F. Y. Tam, Mar. Pollut. Bull., 2005, 51, 1054–1061 CrossRef CAS PubMed.
- S. Y. Yuan, S. H. Wei and B. V. Chang, Chemosphere, 2000, 41, 1463–1468 CrossRef CAS PubMed.
- B. Maliszewska-Kordybach, Appl. Geochem., 1996, 11, 121–127 CrossRef.
- I. A. Lunt, S. S. Hubbard and Y. Rubin, J. Hydrol., 2005, 307, 254–269 CrossRef CAS.
- U. Ilstedt, A. Nordgren and A. Malmer, Soil Biol. Biochem., 2000, 32, 1591–1599 CrossRef CAS.
- M. A. Sanchez-Monedero, A. Roig, C. Paredes and M. P. Bernal, Bioresour. Technol., 2001, 78, 301–308 CrossRef CAS PubMed.
- A. D’Annibale, F. Rosetto, V. Leonardi, F. Federici and M. Petruccioli, Appl. Environ. Microbiol., 2006, 72, 28–36 CrossRef PubMed.
- K. Murugesan, Y.-M. Kim, J.-R. Jeon and Y.-S. Chang, J. Hazard. Mater., 2009, 168, 523–529 CrossRef CAS PubMed.
- M. J. Lopez, M. D. Vargas-Garcia, F. Suarez-Estrella, N. N. Nichols, B. S. Dien and J. Moreno, Enzyme Microb. Technol., 2007, 40, 794–800 CrossRef CAS.
- C. Garcia-Delgado, F. Yunta and E. Eymar, Commun. Soil Sci. Plant Anal., 2013, 44, 817–825 CrossRef CAS.
- G. De Luca, A. Furesi, R. Leardi, G. Micera, A. Panzanelli, P. C. Piu and G. Sanna, Mar. Chem., 2004, 86, 15–32 CrossRef CAS.
- G. Adam and H. Duncan, Soil Biol. Biochem., 2001, 33, 943–951 CrossRef CAS.
- C. Garcia-Delgado, A. D’Annibale, L. Pesciaroli, F. Yunta, S. Crognale, M. Petruccioli and E. Eymar, Sci. Total Environ., 2015, 508, 20–28 CrossRef CAS PubMed.
- H. Guo, J. Yao, M. Cai, Y. Qian, Y. Guo, H. H. Richnow, R. E. Blake, S. Doni and B. Ceccanti, Chemosphere, 2012, 87, 1273–1280 CrossRef CAS PubMed.
- J. H. Park, G. K. Choppala, N. S. Bolan, J. W. Chung and T. Chuasavathi, Plant Soil, 2011, 348, 439–451 CrossRef CAS.
- C. Garcia-Delgado, I. Alfaro-Barta and E. Eymar, J. Hazard. Mater., 2015, 285, 259–266 CrossRef CAS PubMed.
- I. C. Nisbet and P. K. LaGoy, Regul. Toxicol. Pharmacol., 1992, 16, 290–300 CrossRef CAS PubMed.
- P. Valentao, G. Lopes, M. Valente, P. Barbosa, P. B. Andrade, B. M. Silva, P. Baptista and R. M. Seabra, J. Agric. Food Chem., 2005, 53, 3626–3630 CrossRef CAS PubMed.
- C. Garcia-Delgado, F. Yunta and E. Eymar, J. Hazard. Mater., 2015, 300, 281–288 CrossRef CAS PubMed.
- L. Ruberto, S. C. Vazquez and W. P. Mac Cormack, Int. Biodeterior. Biodegrad., 2003, 52, 115–125 CrossRef CAS.
- X. Li, X. Lin, J. Zhang, Y. Wu, R. Yin, Y. Feng and Y. Wang, Curr. Microbiol., 2010, 60, 336–342 CrossRef CAS PubMed.
- X. Sheng-You, C. Ying-Xu, L. Kuang-Fei, C. Xin-Cai, L. Qi, L. Feng and W. Zhao-Wei, Pedosphere, 2009, 19, 265–272 CrossRef.
- A. I. Pelaez, I. Lores, A. Sotres, C. Mendez-Garcia, C. Fernandez-Velarde, J. A. Santos, J. L. R. Gallego and J. Sanchez, Environ. Pollut., 2013, 181, 190–199 CrossRef CAS PubMed.
- S. Lladó, S. Covino, A. Solanas, M. Viñas, M. Petruccioli and A. D’annibale, J. Hazard. Mater., 2013, 248, 407–414 CrossRef PubMed.
- M. Taccari, V. Milanovic, F. Comitini, C. Casucci and M. Ciani, Int. Biodeterior. Biodegrad., 2012, 66, 39–46 CrossRef CAS.
- E. Federici, M. A. Giubilei, T. Cajthaml, M. Petruccioli and A. D’Annibale, J. Hazard. Mater., 2011, 186, 1263–1270 CrossRef CAS PubMed.
- E. Federici, M. Giubilei, G. Santi, G. Zanaroli, A. Negroni, F. Fava, M. Petruccioli and A. D’Annibale, Microb. Cell Fact., 2012, 11, 1–35 CrossRef PubMed.
- S. Lladó, N. Jiménez, M. Viñas and A. M. Solanas, Biodegradation, 2009, 20, 593–601 CrossRef PubMed.
- L. Ribas, M. De Mendonça, C. Camelini and C. Soares, Bioresour. Technol., 2009, 100, 4750–4757 CrossRef CAS PubMed.
- K. Velmourougane, M. Venugopalan, T. Bhattacharyya, D. Sarkar, D. Pal, A. Sahu, S. Ray, K. Nair, J. Prasad and R. Singh, Geoderma, 2013, 197, 186–192 CrossRef.
- J. Dawson, E. Godsiffe, I. Thompson, T. K. Ralebitso-Senior, K. Killham and G. Paton, Soil Biol. Biochem., 2007, 39, 164–177 CrossRef CAS.
- T. E. Schumacher, A. Eynard and R. Chintala, Environ. Sci. Pollut. Res., 2015, 22, 4759–4762 CrossRef CAS PubMed.
- A. Pérez-Piqueres, V. Edel-Hermann, C. Alabouvette and C. Steinberg, Soil Biol. Biochem., 2006, 38, 460–470 CrossRef.
- C. L. Yang, T. H. Sun, W. X. He, Q. X. Zhou and S. Chen, J. Environ. Sci., 2007, 19, 210–216 CrossRef CAS.
- G. Shen, Y. Lu and J. Hong, Ecotoxicol. Environ. Saf., 2006, 63, 474–480 CrossRef CAS PubMed.
- F. M. Bento, F. A. Camargo, B. C. Okeke and W. T. Frankenberger, Bioresour. Technol., 2005, 96, 1049–1055 CrossRef CAS PubMed.
- S. Covino, M. Cvancarova, M. Muzikar, K. Svobodova, A. D’annibale, M. Petruccioli, F. Federici, Z. Kresinova and T. Cajthaml, J. Hazard. Mater., 2010, 183, 669–676 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |






