Preparation of MgO-coated nano CaO using adsorption phase reaction technique for CO2 sorption
Abstract
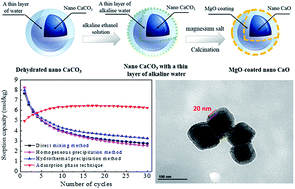
This paper describes the preparation and evaluation of a novel MgO-coated nano CaO for cyclic CO2 sorption by an adsorption phase reaction technique. The influences of the MgO sources, MgO content and carbonation temperature on cyclic CO2 sorption capacity and durability of the as-prepared sorbent were investigated in detail by thermogravimetric analysis (TGA). A forming mechanism of MgO-coated nano CaO sorbent using adsorption phase reaction technique was put forward. The results showed that the thickness of the coating layer could be modified by varying MgO content, and the optimal MgO content was 12 wt%. A durable CO2 sorption without decay could be obtained when carbonation reaction was taken under 650 °C to 700 °C due to the occurrence of self-reactivation phenomenon during the cycles. The sorption capacity of MgO-coated nano CaO prepared via adsorption phase reaction technique showed no decay after 30 cycles, implying better cyclic CO2 sorption durability than that of MgO modified sorbents using mixing or precipitation methods. This is attributed to the MgO coating structure prevented the sintering of nano CaCO3 during multiple carbonation–calcination cycles.

 Please wait while we load your content...
Please wait while we load your content...