Quickly FeCl3-catalyzed highly chemo- and stereo-selective [3 + 2] dipolar cycloaddition of aziridines with isothiocyanates†
Lingfeng Gao,
Kai Fu and
Gengxiu Zheng*
School of Chemistry and Chemical Engineering, University of Jinan, Jinan 250022, Shandong, P. R. China. E-mail: chm_zhenggx@ujn.edu.cn; Tel: +86 53182765841
First published on 27th April 2016
Abstract
A FeCl3-catalyzed [3 + 2] cycloaddition reaction of aziridine with isothiocyanate was developed. The cycloaddition reaction was completed in two minutes with high chemoselectivity, stereoselectivity. 15 (tosylthiazolidin-2-ylidene)anilines were obtained with moderate to excellent yields.
[3 + 2] cycloaddition reactions of nitrones, azides, and related 1,3-dipolar species have emerged as powerful tools for the construction of cyclic compounds with great utility in organic synthesis, chemical biology, and materials science.1 Huisgen has conducted pioneering studies for [3 + 2] dipolar cycloaddition reactions, provided detailed explanations for the mechanism, and also predicted the structures of several unknown 1,3-dipoles at the same time.2 After that, a variety of dipoles and unsaturated compounds were applied in [3 + 2] cycloadditions.2 Recently, [3 + 2] dipolar cycloadditions of aziridines as 1,3-dipolar species with unsaturated bonds such as C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C,3a,5 C
C,3a,5 C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C,3e C
C,3e C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O,3f C
O,3f C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N,3j C
N,3j C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N3k and N
N3k and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se3l have been applied to the synthesis of many nitrogen-containing biologically active molecules and functional materials.3 However, highly chemoselective and stereoselective [3 + 2] cycloaddition reaction of aziridines with a molecule bearing two unsaturated bonds via C–N cleavage of aziridines remains a challenge.4,7
Se3l have been applied to the synthesis of many nitrogen-containing biologically active molecules and functional materials.3 However, highly chemoselective and stereoselective [3 + 2] cycloaddition reaction of aziridines with a molecule bearing two unsaturated bonds via C–N cleavage of aziridines remains a challenge.4,7
Wender and Ding's works,3a and our recent research results5 suggested that Lewis acid-catalyzed aziridine ring-opening is highly efficient to generate a zwitterionic intermediate. Encouraged by our previous work, the stable dipolar ion generated from the ring-opening of aziridine, with some reagents containing two unsaturated bonds, should be also translated to a new structural cycloaddition product.
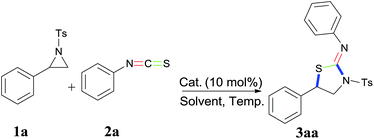
Herein, we report a novel [3 + 2] dipolar cycloaddition reaction of aziridine with isothiocyanate catalyzed by FeCl3 (Table 1). Notably, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bond selectively participates in cycloaddition rather than C
S bond selectively participates in cycloaddition rather than C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond of the isothiocyanate, and X-ray analysis demonstrated the structure of cycloaddition products (Z)-N-phenyl-5-(p-tolyl)-3-tosylthiazolidin-2-imine (3ba) was a stereospecific five-membered ring, the tosyl group and aniline group were in the trans position of the five-member ring (Fig. 1). A series of multifunctional (tosylthiazolidin-2-ylidene)anilines were obtained via one step [3 + 2] dipolar cycloaddition.
N bond of the isothiocyanate, and X-ray analysis demonstrated the structure of cycloaddition products (Z)-N-phenyl-5-(p-tolyl)-3-tosylthiazolidin-2-imine (3ba) was a stereospecific five-membered ring, the tosyl group and aniline group were in the trans position of the five-member ring (Fig. 1). A series of multifunctional (tosylthiazolidin-2-ylidene)anilines were obtained via one step [3 + 2] dipolar cycloaddition.
| Entry | Catalyst | Solvent | T/°C | t/min | Yield/% |
|---|---|---|---|---|---|
| a Reaction conditions: aziridine 1a (136 mg, 0.5 mmol), isothiocyanatobenzene 2a (1 mmol), catalyst (0.05 mmol), solvent (1.0 mL).b Yield of isolated product.c 0.1 g 4 Å molecular sieve. | |||||
| 1 | FeCl3 | CH3NO2 | rt | 2 | 60 |
| 2 | FeCl2·4H2O | CH3NO2 | rt | 2 | Trace |
| 3 | Fe(OTf)3 | CH3NO2 | rt | 1 | 41 |
| 4 | Fe(NO3)3·9H2O | CH3NO2 | rt | 30 | Trace |
| 5 | Fe2(SO4)3·5H2O | CH3NO2 | rt | 30 | Trace |
| 6 | Fe(acac)3 | CH3NO2 | rt | 30 | 0 |
| 7 | Zn(OTf)2 | CH3NO2 | rt | 60 | 45 |
| 8 | Cu(OTf)2 | CH3NO2 | rt | 120 | 38 |
| 9 | Zr(OTf)4 | CH3NO2 | rt | 30 | 25 |
| 10 | CuI | CH3NO2 | rt | 30 | Trace |
| 11 | FeCl3 | CH2Cl2 | rt | 120 | 43 |
| 12 | FeCl3 | (CH2)2Cl2 | rt | 2 | 52 |
| 13 | FeCl3 | CH3NO2 | 0 | 2 | 68 |
| 14 | FeCl3 | CH3NO2 | −20 | 2 | 73 |
| 15 | FeCl3 | CH3NO2 | −20 | 2 | 80c |
| 16 | FeCl3 + Ph3P | CH3NO2 | −20 | 30 | 0 |
| 17 | Binol phosphonic acid | CH3NO2 | rt | 30 | 0 |
In our initial studies, 2-phenyl-1-tosylaziridine (1a) and isothiocyanatobenzene (2a) were chosen the model substrates in the catalysis of FeCl3 in CH3NO2 as the solvent at room temperature for two minutes, affording (Z)-N-(5-phenyl-3-tosylthiazolidin-2-ylidene)aniline (3aa) in 60% yield. When different Lewis acids including iron, copper, zinc, and zirconium salts were examined, FeCl3 gave the highest yield for this cycloaddition reaction (Table 1, entries 1–9). Next, dichloromethane, dichloroethane, nitromethane5 were chosen as solvent in the iron–catalyst reaction, and nitromethane was found to be the best solvent for the reaction. To our delight, lowering temperature to −20 °C increased the reaction yield up to 73% (Table 1, entries 14).6 In addition, adding 4 Å molecular sieve can inhibit hydrolysis of aziridine, the yield increased to 80% (Table 1, entries 15). FeCl3 co-ordinated with Ph3P did not catalyzed the reaction at all (Table 1, entries 16). Furthermore, Brønsted acid binol phosphonic acid was also not effective under the same reaction conditions (Table 1, entry 17).‡
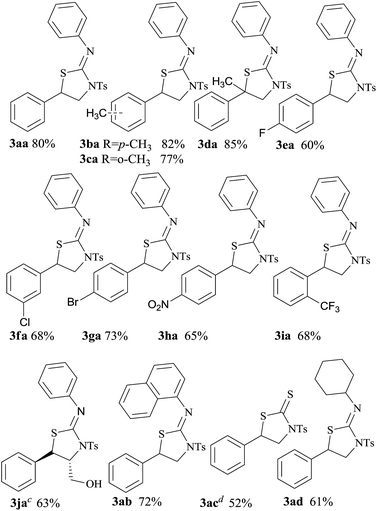
After the optimization of the reaction conditions, we examined the generality of the transformation. Firstly, we focused on the reactivity of different aromatic substituent aziridines 1 (Table 2). A variety of aromatic substituted aziridines with electron-donating or electron-withdrawing groups were employed as the reaction substrates, which afforded the corresponding products 3 in moderate to excellent yields in two minutes. Electron-donating substituents favored the reaction for electron-donating substituents high-efficiency stabilizing benzyl cation, so the corresponding (tosylthiazolidin-2-ylidene)anilines were obtained in excellent yields (Table 2, 3ba, 3ca, 3fa and 3ga). On the other hand, electron-withdrawing substituted aziridines were used, the cyclization reaction still worked well, but with a slightly low yield (Table 2, 3ea, 3ha, 3ia). When 1,1-disubstituted aziridine was used in the selective cycloaddition, the reaction still worked very well, no obvious steric effect was found, 2-methyl-2-phenyl-1-tosylaziridine could get 85% yield (Table 2, 3da). Another (Z)-3-phenyl-1-tosylaziridin-2-yl methanol also afforded 63% yield and kept the trans-structure (Table 2, 3ja). In another part of isothiocyanate, we predicted different isothiocyanate to give similar cycloaddition products. Aromatic 1-isothiocyanatonaphthalene and nonaromatic isothiocyanatocyclohexane were chosen the substrates, but aliphatic substituted isothiocyanate afforded lower yield (Table 2, 3ab and 3ac). Carbon disulfide could participate in the selective [3 + 2] cycloaddition, however, only giving a moderate yield (Table 2, 3ad).
The results led us to address the question, could FeCl3 catalyze cycloaddition between aliphatic–aziridine and isothiocyanate? With the questions, we synthesized an aliphatic aziridine 7-tosyl-7-azabicyclo[4.1.0]heptane (1k), greatly, we obtained the corresponding cycloaddition products 3ka, only with slightly lower yield and longer reaction time (Table 3). Exciting, carbon disulfide and 7-tosyl-7-azabicyclo[4.1.0]heptane converted to 3-tosylhexahydrobenzo[d]thiazole-2(3H)-thione with 47% yield (Table 3, 3kb). Furthermore, we also got similar cycloaddition product 3kc from 1k and isothiocyanatocyclohexane (Table 3, 3kc).
In the reaction process, the ring-opening of aziridine is the most important stage. The ring-opening of aziridine is high-efficiency in the catalyst of Lewis acid to generate a stable dipolar ion, because tosyl group could stable nitrogenanion and phenyl could stable carbocation. We examined the reaction activity of aziridine without the tosyl group [Scheme 1, eqn (1)], as expected, no desired product were detected when unactivated aziridines 1l or 1m were employed in the reaction with isothiocyanatobenzene.
In consideration of the low yields of some substrates and some reports, we designed the reaction aziridine 1a with FeCl3 [Scheme 1, eqn (2)], as expected, N-(2-chloro-2-phenylethyl)-4-methylbenzenesulfonamide 4a was isolated in 16% yield.6 Under the above control experimental results and reports, we proposed a plausible mechanism in Scheme 2. Aziridine 1a was catalyzed ring opening by FeCl3 to form a zwitterionic intermediate A, which is stabilized by tosyl group and phenyl group (Scheme 2). The carbon of isothiocyanatobenzene was attacked by the nitrogen anion of the 1,3-dipolar ion, and then FeCl3 combined with sulphur to generate intermediate B. Last, sulphur attacked intramolecular carbocation to generate FeCl3 to catalyze the next reaction circulation.
 | ||
| Scheme 2 A plausible mechanism for the selective [3 + 2] cycloaddition of aziridine and isothiocyanatobenzene. | ||
Conclusions
In conclusion, we have developed a new chemo- and stereo-selective [3 + 2] dipolar cycloaddition of aziridine with isothiocyanate catalysed by cheap Lewis acid FeCl3. A series of new heterocyclic (tosylthiazolidin-2-ylidene)anilines were synthesized from readily available aziridines and isothiocyanates under mild conditions in two minutes. Simple FeCl3 as catalyst renders the protocol be suitable for large-scale synthesis.Acknowledgements
We are grateful to the University of Science and Technology of China and the great support from the School of Chemistry and Chemical Engineering, University of Jinan.Notes and references
- (a) 1,3-Dipolar Cycloaddition Chemistry, ed. A. Padwa, Wiley: New York, 1984, vol. 1, pp. 1–176 Search PubMed; (b) K. V. Gothelf and K. A. Jørgensen, Chem. Rev., 1998, 98, 863 CrossRef CAS PubMed; (c) H. C. Kolb, M. G. Finn and B. K. Sharpless, Angew. Chem., Int. Ed., 2001, 40, 2004 CrossRef CAS; (d) V. V. Rostovtsev, L. G Green, V. V Folkin and B. K. Sharpless, Angew. Chem., Int. Ed., 2002, 41, 2596 CrossRef CAS; (e) W. Zeng, G. Y. Chen, Y. G. Zhou and Y. X. Li, J. Am. Chem. Soc., 2007, 129, 750 CrossRef CAS PubMed; (f) D. J. St. Cyr and B. A. Arndtsen, J. Am. Chem. Soc., 2007, 129, 12366 CrossRef CAS PubMed; (g) W. Z. Gao, X. M. Zhang and M. Raghunath, Org. Lett., 2005, 7, 4241 CrossRef CAS PubMed; (h) Z. J. Liu, F. Shi, P. D. G. Martinez, C. Raminelli and R. C. Larock, J. Org. Chem., 2008, 73, 219 CrossRef CAS PubMed; (i) V. Aucagne and D. A. Leigh, Org. Lett., 2006, 8, 4505 CrossRef CAS PubMed; (j) Z. Yuan, G. C. Kuang, R. J. Clark and L. Zhu, Org. Lett., 2012, 14, 2590 CrossRef CAS PubMed; (k) S. A. Ingale and F. Seela, J. Org. Chem., 2013, 78, 3394 CrossRef CAS PubMed; (l) S. Arumugam and V. V. Popik, J. Org. Chem., 2014, 79, 2702 CrossRef CAS PubMed.
- (a) H. Rolf, Angew. Chem., Int. Ed., 1963, 2, 565 CrossRef; (b) H. Rolf, Angew. Chem., Int. Ed., 1963, 2, 633 CrossRef; (c) L. M. Stanley and M. P. Sibi, Chem. Rev., 2008, 108, 2887 CrossRef CAS PubMed; (d) V. Nair and T. D. Suja, Tetrahedron, 2007, 63, 12247 CrossRef CAS; (e) H. Pellissier, Tetrahedron, 2007, 63, 3235 CrossRef CAS; (f) G. Molteni, Heterocycles, 2006, 68, 2177 CrossRef CAS; (g) A. E. Koumbis and J. K. Gallos, Curr. Org. Chem., 2003, 7, 397 CrossRef; (h) A. E. Koumbis and J. K. Gallos, Curr. Org. Chem., 2003, 7, 585 CrossRef CAS; (i) A. E. Koumbis and J. K. Gallos, Curr. Org. Chem., 2003, 7, 771 CrossRef CAS; (j) Synthetic Applications of 1,3-Dipolar Cycloaddition Chemistry toward Heterocycles and Natural Products, ed. A. Padwa, W. H. Pearson, Wiley, New York, 2002 Search PubMed; (k) S. Karlsson and H. E. Högberg, Org. Prep. Proced. Int., 2001, 33, 105 CrossRef; (l) U. K. Nadir and N. Basu, J. Org. Chem., 1995, 60, 1458 CrossRef CAS.
- (a) P. A. Wender and D. Strand, J. Am. Chem. Soc., 2009, 131, 7528 CrossRef CAS PubMed; (b) I. Ungureanu, K. Philippe and A. Mann, Angew. Chem., Int. Ed., 2000, 39, 4615 CrossRef CAS; (c) I. Ungureanu, C. Bologa, S. Chayer and A. Mann, Tetrahedron Lett., 1999, 40, 5315 CrossRef CAS; (d) P. Tascedda and E. Dũnach, Chem. Commun., 2000, 449 RSC; (e) M. Brichacek, D. E. Lee and J. T. Njardarson, Org. Lett., 2008, 10, 5023 CrossRef CAS PubMed; (f) C. H. Ding, L. X. Dai and X. L. Hou, Tetrahedron, 2005, 61, 9586 CrossRef CAS; (g) M. K. Ghorai and K. Ghosh, Tetrahedron Lett., 2007, 48, 3191 CrossRef CAS; (h) D. D. Chen, X. L. Hou and L. X. Dai, J. Org. Chem., 2008, 73, 5578 CrossRef CAS PubMed; (i) J. S. Yadav, B. V. S. Reddy, T. Pandurangam and U. V. S. Reddy, Chem. Lett., 2008, 37, 824 CrossRef CAS; (j) J. Wu, X. Y. Sun and H. G. Xia, Tetrahedron Lett., 2006, 47, 1509 CrossRef CAS; (k) X. Y. Sun, W. Sun, R. H. Fan and J. Wu, Adv. Synth. Catal., 2007, 349, 2151 CrossRef CAS; (l) S. Matsukawa and K. Tsukamoto, Org. Biomol. Chem., 2009, 7, 3792 RSC; (m) M. Sengoden and T. Punniyamurthy, Angew. Chem., Int. Ed., 2013, 52, 572 CrossRef CAS PubMed; (n) H. C. Guo, Q. H. Xu and O. Kwon, J. Am. Chem. Soc., 2009, 131, 6318 CrossRef CAS PubMed; (o) M. Sayyad, Y. Nanaji and M. K. Ghorai, J. Org. Chem., 2015, 80, 12659 CrossRef CAS PubMed.
- (a) M. Sengoden and T. Punniyamurthy, Angew. Chem., Int. Ed., 2013, 52, 572 CrossRef CAS PubMed; (b) K. M. Partridge, I. A. Guzei and T. P. Yoon, Angew. Chem., Int. Ed., 2010, 49, 930 CrossRef CAS PubMed; (c) J. Y. Wu, Z. B. Luo, L. X. Dai and X. L. Hou, J. Org. Chem., 2008, 73, 9137 CrossRef CAS PubMed; (d) U. K. Nadir and N. Basu, J. Org. Chem., 1995, 60, 1458 CrossRef CAS.
- J. M. Fan, L. F. Gao and Z. Y. Wang, Chem. Commun., 2009, 5021 RSC.
- (a) C. R. Brindaban, L. Adsk and S. Banerjee, Can. J. Chem., 2007, 85, 366 CrossRef; (b) D. Biswanath, K. Maddeboina and L. Keetha, J. Chem. Res., 2007, 2, 82 Search PubMed; (c) G. Shikha, B. Alakesh, Prasad, B. A. Bhanu and V. K. Singh, J. Org. Chem., 2007, 72, 2133 CrossRef PubMed; (d) D. Biswanath, K. Maddeboina and V. Katta, Chem. Lett., 2007, 36, 82 CrossRef; (e) K. G. Manas, D. Kalpataru, K. Amit and K. Ghosh, Tetrahedron Lett., 2005, 46, 4103 CrossRef; (f) J. S. Yadav, R. B. V. Subba and K. G. Mahesh, Synlett, 2001, 9, 1417 CrossRef; (g) G. Sabitha, R. S. Babu, M. Rajkumar, C. S. Reddy and J. S. Yadav, Tetrahedron Lett., 2001, 42, 3955 CrossRef CAS.
- (a) D. A. Evans, M. M. faul and M. T. Bilodeau, J. Am. Chem. Soc., 1994, 116, 2742 CrossRef CAS; (b) D. A. Evans, M. M. faul and M. T. Bilodeau, J. Org. Chem., 1991, 56, 6745 Search PubMed; (c) T. Ando, D. Kano, S. Minakata, I. Ryu and M. Komatsu, Tetrahedron, 1998, 54, 13485 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: General Information, further optimizing for the reaction conditions, synthesis of substrates, characterization data for the products and crystallographic data. CCDC 110418. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra04923k |
‡ Representative procedure for selective [3 + 2] dipolar cycloaddition of aziridine with isothiocyanate: FeCl3 (8 mg, 0.05 mmol) and aziridine 1a (137.0 mg, 0.5 mmol), 1 mL CH3NO2, 0.1 g 4 Å molecular sieve were loaded into a 10 mL oven-dried flask and the system put under vacuum and charged with N2 three times. The system was cooled to −20 °C and isothiocyanatobenzene 2a (127.0 mg, 1.0 mmol) was added. The resulting mixture was maintained at −20 °C for another 2 min, then the reaction was quenched with 0.5 mL H2O and the solvent was evaporated under reduced pressure. The residue was purified by column chromatography on silica gel using PE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc EtOAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Et3N (20 Et3N (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.03, v/v/v) as eluent to give (tosylthiazolidin-2-ylidene)aniline 3aa as a white solid (163.0 mg, 80%). 0.03, v/v/v) as eluent to give (tosylthiazolidin-2-ylidene)aniline 3aa as a white solid (163.0 mg, 80%). |
| This journal is © The Royal Society of Chemistry 2016 |