p-TsOH-promoted synthesis of (E)-6-phenyl-7-styryl-5,6-dihydrodibenzo[b,h][1,6]naphthyridines via cascade intramolecular aza-Michael addition/Friedlander condensation of 2′-aminochalcones in a SDS/H2O system†
Makthala Ravi‡
a,
Parul Chauhan‡a,
Shikha Singha,
Ruchir Kantb and
Prem. P. Yadav*a
aDivision of Medicinal and Process Chemistry, CSIR-Central Drug Research Institute, Lucknow-226031, India. E-mail: pp_yadav@cdri.res.in; ppy_cdri@yahoo.co.in; Fax: +91-522-2623405; Fax: +91-522-2623938; Tel: +91-522-2612411-18 ext. 4761 Tel: +91-522-2612411-18 ext. 4762
bDivision of Molecular and Structural Biology, CSIR-Central Drug Research Institute, Lucknow-226031, India
First published on 12th May 2016
Abstract
Brønsted acid-promoted cascade synthesis of novel (E)-6-phenyl-7-styryl-5,6-dihydrodibenzo[b,h][1,6]naphthyridines has been achieved via homodimerization of 2′-aminochalcones by employing sodium dodecylsulphate (SDS) as a surfactant in water. Besides water as an environmentally benign reaction medium, the reaction proceeds smoothly with high atom-economy under a sequential one-pot protocol.
Bioactive natural products and natural product-inspired molecules have played a pivotal role in drug discovery research.1 The synthesis of natural and unnatural complex molecules generally involves a large number of synthetic steps using various hazardous organic solvents and reagents, apart from extraction and purification processes in each step. This leads to a large amount of waste that increases the overall cost. Designing cascade sequences to accomplish target-oriented syntheses of structurally complex natural products has remained a major challenge in the art of total synthesis.2
From the standpoint of green and sustainable approaches, the development of synthetic processes for the construction of privileged scaffolds with high atom economy in water rather than organic solvents is eco-friendly and economical.3 In addition, being an ideal green solvent, water can also accelerate organic reactions such as Michael additions, intramolecular cyclizations, rearrangements and nucleophilic substitutions due to its unique reactivity and selectivity at elevated temperature.4 However, the use of aqueous reaction media is often limited due to the poor solubility of organic compounds in water. This can be overcome by the use of surfactants as solubilizing agents via a spontaneous aggregation in water to form reactive micellar system having a hydrophobic core and hydrophilic corona.5
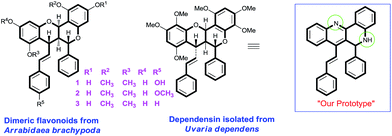
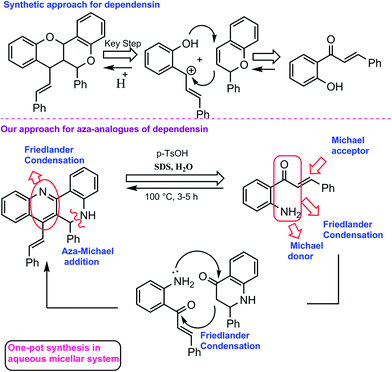
Dependensin, an antiplasmodial natural product isolated from Uvaria dependens and related antiparasitic dimeric flavonoids isolated from Arrabidaea brachypoda are densely functionalized fused benzopyranes bearing four stereocenters and one trans double bond (Scheme 1).6 Additionally, 1,6-naphthyridines have been shown to be potent as HIV-1 integrase inhibitors,7 anticancer8 and antimalarial agents.9 The extended benzenoid core of dibenzo[b,h][1,6]naphthyridine is also an important class of fluorophore and reported as DNA intercalating agents.10 In view of the medicinal and fluorescent properties of 1,6-naphthyridines, the development of sustainable and green methodologies towards the synthesis of dibenzo[b,h][1,6]naphthyridines is highly desirable. Herein, for the first time, a new route for the synthesis of aza-analogues of dimeric flavonoids via Brønsted acid-promoted homodimerization of 2′-aminochalcones in an aqueous micellar (SDS/H2O) system is reported.
As a part of our ongoing efforts towards the development of ecofriendly methods for the synthesis of novel bioactive heterocycles,11 we became interested in natural product inspired synthesis of (E)-6-phenyl-7-styryl-5,6-dihydrodibenzo[b,h][1,6]naphthyridines in a one-pot reaction under aqueous conditions. The reported method for the total synthesis of dependensin involved an acid-promoted ring opening of flavene to a stable benzyl carbocation, which upon further cyclization with another flavene molecule delivered the homodimerized compound (Scheme 2).12 Based on the above analysis and literature reports for the cascade synthesis of dibenzo[b,h][1,6]naphthyridines from a 2′-amino-substituted carbonyl precursor,10,13 we speculated that 2′-aminochalcones may undergo homodimerization via an intramolecular aza-Michael addition in the presence of an acid to give aza-flavanone, which may subsequently react with another 2′-aminochalcone under the same acidic conditions to finally deliver the homodimerized product.
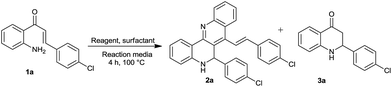
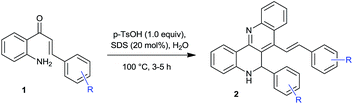
To test this hypothesis initially, the starting material 2′-aminochalcone was synthesized from 2′-aminoacetophenone and benzaldehyde in the presence of base in ethanol.14 The hypothesis was actualized when 2′-aminochalcone 1a was treated with 3 equiv. of p-toluenesulfonic acid (p-TsOH) in EtOH at 80 °C for 4 h. The reaction proceeded well to yield 76% of homodimerized product 2a and 20% of aza-flavanone 3a (Table 1, entry 1). However, 1 equivalent of p-toluenesulfonic acid in EtOH at 80 °C delivered the desired product 2a in 68% yield (Table 1, entry 2). Based on the compatibility of Bronsted acids with aqueous solutions and the feasibility of cyclization reaction in water,15 the reaction was attempted in aqueous medium with 3 equiv. of p-TsOH at 100 °C for 4 h, which afforded homodimerized product 2a in 64% yield (Table 1, entry 3). Delighted with this result, the reaction was performed with 1 equiv. of p-TsOH, which drastically reduced the yield of 2a to 10% (Table 1, entry 4), which may be due to the poor solubility of the substrate. To overcome the solubility issue and the possibility of dehydration reactions in the presence of surfactants,16 the reaction with 20 mol% of sodium dodecyl sulfate (SDS) and 1 equiv. of p-TsOH in water was performed at 100 °C for 4 h. The reaction proceeded smoothly and afforded 78% of homodimerized product 2a and 20% of aza-flavanone 3a (Table 1, entry 5), decreasing the amount of SDS to 10 mol% and p-TsOH to 0.5 equiv. reduced the yield to 55% and 42%, respectively (Table 1, entries 6 and 7). It is noteworthy that the product can be obtained as precipitated solid without any further work up, however, column chromatography was required to achieve analytical purity. Next, the experiment was carried out at room temperature and 60 °C; no product formation was observed under either of these conditions (Table 1, entries 8 and 9). Encouraged by these results, a variety of Brønsted acids and Lewis acids were screened based on their ability to promote the Friedlander condensation17,18 (Table 1, entries 10–16). Among the tested acids TfOH and InCl3 provided homodimerized product 2a in 60% and 67% yields respectively (Table 1, entries 13 and 15). However, p-TsOH produced the best yield of 2a (Table 1, entry 5). Also, various anionic, non-ionic and cationic surfactants (see the ESI,† Table 1, entries 17–21) such as SLS, PF-127, Tween 80, Triton-X100 and TBAI were screened with varying results.
| Entry | Reagent (equiv.) | Surfactant SDS (20 mol%) | Reaction media | Yieldb | |
|---|---|---|---|---|---|
| 2a | 3a | ||||
| a Reaction conditions: 1a (1.0 mmol), reagent (1.0 mmol), surfactant (20 mol%), reaction media (10 mL) at 100 °C.b Isolated yields.c At 80 °C.d 10 mol% of SDS was used.e At room temperature.f At 60 °C. | |||||
| 1c | p-TsOH (3) | — | Ethanol | 76 | 20 |
| 2c | p-TsOH (1) | — | Ethanol | 68 | 18 |
| 3 | p-TsOH (3) | — | Water | 64 | 24 |
| 4 | p-TsOH (1) | — | Water | 10 | 10 |
| 5 | p-TsOH (1) | SDS | Water | 78 | 20 |
| 6d | p-TsOH (1) | SDS | Water | 55 | 18 |
| 7 | p-TsOH (0.5) | SDS | Water | 42 | 24 |
| 8e | p-TsOH (1) | SDS | Water | — | 15 |
| 9f | p-TsOH (1) | SDS | Water | — | 17 |
| 10 | TFA (1) | SDS | Water | 20 | 30 |
| 11 | AcOH (1) | SDS | Water | — | 25 |
| 12 | MeSO3H (1) | SDS | Water | 52 | 35 |
| 13 | TfOH (1) | SDS | Water | 60 | 33 |
| 14 | FeCl3 (1) | SDS | Water | 46 | 20 |
| 15 | InCl3 (1) | SDS | Water | 67 | 25 |
| 16 | I2 (1) | SDS | Water | 15 | 51 |
Among the screened surfactants (see the ESI,† Table 1, entries 17 and 20), only SLS and Triton-X100 provided >70% yield of homodimerized product 2a and other surfactants did not provide a satisfactory result in comparison to SDS (Table 1, entries 17–21 vs. entry 5, see the ESI†). Further solvent optimization with protic solvents such as methanol and isopropanol delivered the homodimerized product 2a in 60% and 30% yields respectively (Table 1, entries 22 and 23, see the ESI†), however, aprotic solvents such as acetonitrile and toluene were ineffective for homodimerization, but produced aza-flavanone 3a exclusively (Table 1, entries 24 and 25, see the ESI†).
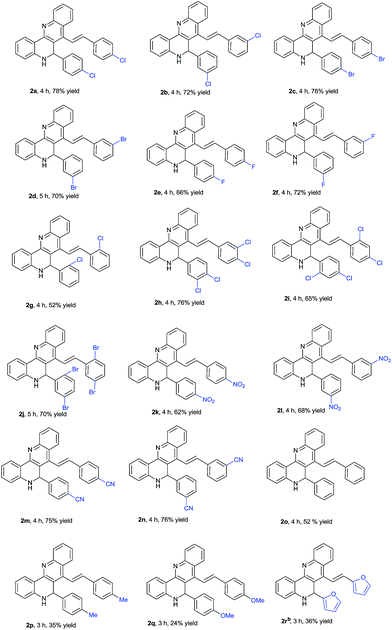
Using optimized reaction conditions, the scope and generality were further investigated. The reactions of 2′-aminochalcones bearing electron withdrawing groups such as chloro, bromo, fluoro, 2,4-dichloro, 3,4-dichloro, 2,5-dibromo, nitro and cyano, afforded the corresponding homodimerized products in moderate to good yields (Table 2, products 2a–2n). 2′-Aminochalcones bearing electron donating groups such as methyl and methoxy on the phenyl ring and furyl substituted 2′-aminochalcone produced homodimerized products, albeit in low yields (Table 2, products 2p–2r). The lower yields in cases with electron donating groups may result from a decrease in the electrophilicity at the carbonyl carbon, which is unfavourable for the Friedlander condensation.
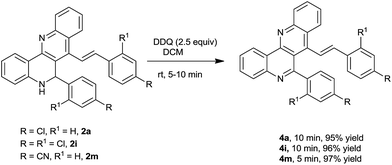
The synthesized (E)-6-phenyl-7-styryl-5,6-dihydrodibenzo[b,h][1,6]naphthyridines were further oxidized to (E)-6-phenyl-7-styryldibenzo[b,h][1,6]naphthyridines in the presence of DDQ in DCM for 5–10 min at room temperature. The reaction proceeded smoothly and provided excellent yields of the corresponding oxidized products (Scheme 3). The structure of oxidized product 4i was unambiguously confirmed by X-ray analysis (see the ESI†).
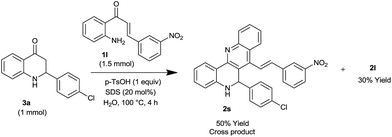
To prove that the exact mechanism involved aza-flavanone formation followed by Friedlander condensation, an experiment was conducted with aza-flavanone (3a) and 2′-aminochalcone 1l under the optimized conditions. The reaction afforded desired Friedlander condensation product 2s in 50% yield along with 30% of the homodimerized product 2l (Scheme 4). The structure of 2s was unambiguously confirmed by X-ray analysis (see the ESI†).
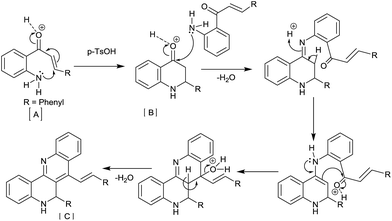
Based on our experimental results and literature reports,17,18 we proposed a plausible mechanism for homodimerization as shown in (Scheme 5). 2′-Aminochalcone [A] undergoes an intramolecular aza-Michael addition in the presence of acid to form aza-flavanone [B], which subsequently reacts with another molecule of 2′-aminochalcone in the presence of acid via a Friedlander condensation to afford homodimerized product [C].
In conclusion, we have developed an efficient and straightforward methodology for the synthesis of (E)-6-phenyl-7-styryl-5,6-dihydrodibenzo[b,h][1,6]naphthyridines in aqueous media by using SDS as a surfactant with good yields. This methodology offered a sequential one-pot synthetic operation with high atom economy, under greener conditions, using water as the medium and no workup with organic solvent. Application of the present methodology towards exploration of unique cascade sequences is under progress.
Acknowledgements
M. R., P. C. and S. S. are thankful to the Council of Scientific and Industrial Research (CSIR), New Delhi, for Fellowship. Mr A. K. Srivastava for providing technical support and SAIF-CDRI, Lucknow for providing spectral and analytical data. This work was supported by CSIR network project “HOPE” (BSC0114). We thank Dr Tejender S. Thakur of the Molecular and Structural Biology Division, CSIR-Central Drug Research Institute (CDRI) for supervising the X-ray data collection and structure determination of our compound reported in this paper. This is CDRI communication No. 9332.Notes and references
- (a) S. Rizzo and H. Waldmann, Chem. Rev., 2014, 114, 4621 CrossRef CAS PubMed; (b) M. S. Butler, A. A. B. Robertson and M. A. Cooper, Nat. Prod. Rep., 2014, 31, 1612 RSC.
- K. C. Nicolaou, D. J. Edmonds and P. G. Bulger, Angew. Chem., Int. Ed., 2006, 45, 7134 CrossRef CAS PubMed.
- (a) C.-J. Li and B. M. Trost, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 13197 CrossRef CAS PubMed; (b) C.-J. Li, Chem. Rev., 2005, 105, 3095 CrossRef CAS PubMed; (c) B. H. Lipshutz and S. Ghorai, Green Chem., 2014, 16, 3660 RSC; (d) A. Chanda and V. V. Fokin, Chem. Rev., 2009, 109, 725 CrossRef CAS PubMed; (e) M. Shekouhy and A. Khalafi-Nezhad, Green Chem., 2015, 17, 4815 RSC; (f) S. Higson, F. Subrizi, T. D. Sheppard and H. C. Hailes, Green Chem., 2016, 18, 1855 RSC.
- (a) S. Shirakawa and S. Kobayashi, Org. Lett., 2007, 9, 311 CrossRef CAS PubMed; (b) F.-Z. Zhang, Y. Tian, G.-X. Li and J. Qu, J. Org. Chem., 2015, 80, 1107 CrossRef CAS PubMed; (c) T. Iwado, K. Hasegawa, T. Sato, M. Okada, K. Sue, H. Iwamura and T. Hiaki, J. Org. Chem., 2013, 78, 1949 CrossRef CAS PubMed; (d) P.-F. Li, H.-L. Wang and J. Qu, J. Org. Chem., 2014, 79, 3955 CrossRef CAS PubMed.
- (a) G. L. Sorella, G. Strukul and A. Scarso, Green Chem., 2015, 17, 644 RSC; (b) T. Dwars, E. Paetzold and G. Oehme, Angew. Chem., Int. Ed., 2005, 44, 7174 CrossRef CAS PubMed; (c) K. Bahrami, M. M. Khodaei and A. Nejati, Green Chem., 2010, 12, 1237 RSC; (d) M. Srivastava, J. Singh, S. B. Singh, K. Tiwari, V. K. Pathak and J. Singh, Green Chem., 2012, 14, 901 RSC; (e) E. Tasca, G. L. Sorella, L. Sperni, G. Strukul and A. Scarso, Green Chem., 2015, 17, 1414 RSC; (f) D. R. M. Arenas, C. A. M. Bonilla and V. V. Kouznetsov, Org. Biomol. Chem., 2013, 11, 3655 RSC; (g) K. Ahlford, J. Lind, L. Maler and H. Adolfsson, Green Chem., 2008, 10, 832 RSC; (h) H. Firouzabadi, N. Iranpoor and A. Garzan, Adv. Synth. Catal., 2005, 347, 1925 CrossRef CAS.
- (a) M. H. H. Nkunya, R. Waibel and H. Achenbach, Phytochemistry, 1993, 34, 853 CrossRef CAS; (b) C. Q. da Rocha, E. F. Queiroz, C. S. Meira, D. R. M. Moreira, M. B. P. Soares, L. Marcourt, W. Vilegas and J.-L. Wolfender, J. Nat. Prod., 2014, 77, 1345 CrossRef PubMed.
- D. J. Hazuda, N. J. Anthony, R. P. Gomez, S. M. Jolly, J. S. Wai, L. Zhuang, T. E. Fisher, M. Embrey, J. P. Guare Jr, M. S. Egbertson, J. P. Vacca, J. R. Huff, P. J. Felock, M. V. Witmer, K. A. Stillmock, R. Danovich, J. Grobler, M. D. Miller, A. S. Espeseth, L. Jin, I.-W. Chen, J. H. Lin, K. Kassahun, J. D. Ellis, B. K. Wong, W. Xu, P. G. Pearson, W. A. Schleif, R. Cortese, E. Emini, V. Summa, M. K. Holloway and S. D. Young, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 11233 CrossRef CAS PubMed.
- E. Kiselev, T. S. Dexheimer, Y. Pommier and M. Cushman, J. Med. Chem., 2010, 53, 8716 CrossRef CAS PubMed.
- S. Zhu, Q. Zhang, C. Gudise, L. Meng, L. Wei, E. Smith and Y. Kong, Bioorg. Med. Chem. Lett., 2007, 17, 6101 CrossRef CAS PubMed.
- K. Okuma, T. Koga, S. Ozaki, Y. Suzuki, K. Horigami, N. Nagahora, K. Shioji, M. Fukuda and M. Deshimaru, Chem. Commun., 2014, 50, 15525 RSC.
- (a) O. P. S. Patel, D. Anand, R. K. Maurya and P. P. Yadav, Green Chem., 2015, 17, 3728 RSC; (b) P. Chauhan, M. Ravi, S. Singh, K. S. R. Raju, V. Bajpai, B. Kumar, M. Wahajuddin and P. P. Yadav, RSC Adv., 2014, 4, 43336 CAS; (c) M. Ravi, P. Chauhan, R. Kant, S. K. Shukla and P. P. Yadav, J. Org. Chem., 2015, 80, 5369 CrossRef CAS PubMed.
- M. Deodhar, D. S. Black and N. Kumar, Tetrahedron, 2007, 63, 5227 CrossRef CAS.
- D. Mao, J. Tang, W. Wang, X. Liu, S. Wu, J. Yu and L. Wang, Org. Biomol. Chem., 2015, 13, 2122 CAS.
- Y. Xia, Z.-Y. Yang, P. Xia, K. F. Bastow, Y. Nakanishi and K.-H. Lee, Bioorg. Med. Chem. Lett., 2000, 10, 699 CrossRef CAS PubMed.
- S. D. Sharma, P. Gogoi and D. Konwar, Green Chem., 2007, 9, 153 RSC; T. Akiyama, Chem. Rev., 2007, 107, 5744 CrossRef CAS PubMed; G.-W. Wang, C.-S. Jia and Y.-W. Dong, Tetrahedron Lett., 2006, 47, 1059 CrossRef.
- (a) K. Manabe, X.-M. Sun and S. Kobayashi, J. Am. Chem. Soc., 2001, 123, 10101 CrossRef CAS PubMed; (b) K. Manabe, S. Iimura, X.-M. Sun and S. Kobayashi, J. Am. Chem. Soc., 2002, 124, 11971 CrossRef CAS PubMed; (c) A. Kumar, M. K. Gupta, M. Kumar and D. Saxena, RSC Adv., 2013, 3, 1673 RSC; (d) A. Kumar, S. Maurya, M. K. Gupta and R. D. Shukla, RSC Adv., 2014, 4, 57953 RSC.
- (a) C.-S. Jia, Z. Zhang, S.-J. Tu and G.-W. Wang, Org. Biomol. Chem., 2006, 4, 104 RSC; (b) T. Brunin, J.-P. Henichart and B. Rigo, Tetrahedron, 2005, 61, 7916 CrossRef CAS; (c) T. Brunin, L. Legentil, J.-P. Henichart and B. Rigo, Tetrahedron, 2006, 62, 3959 CrossRef CAS; (d) G. B. Raolji, S. Garcon, A. E. Greene and A. Kanazawa, Angew. Chem., Int. Ed., 2003, 42, 5059 CrossRef CAS PubMed; (e) S. P. Chavan, K. Pasupathy, M. S. Venkatraman and R. R. Kale, Tetrahedron Lett., 2004, 45, 6879 CrossRef CAS; (f) A. Shaabani, E. Soleimani and Z. Badri, Synth. Commun., 2007, 37, 629 CrossRef CAS.
- (a) S. K. De and R. A. Gibbs, Tetrahedron Lett., 2005, 46, 1647 CrossRef CAS; (b) D. S. Bose and R. K. Kumar, Tetrahedron Lett., 2006, 47, 813 CrossRef CAS; (c) J. Wang, X. Fan, X. Zhang and L. Han, Can. J. Chem., 2004, 82, 1192 CrossRef CAS; (d) J. Wu, H.-G. Xia and K. Gao, Org. Biomol. Chem., 2006, 4, 126 RSC; (e) T. Chanda, R. K. Verma and M. S. Singh, Chem.–Asian J., 2012, 7, 778 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Experimental procedures, compound characterization, X-ray crystal structure data and ORTEP drawing for 4i and 2s; copies of NMR spectra of all the final compounds. For ESI and crystallographic data in CIF. CCDC 1058218 and 1025096. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra04837d |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2016 |