Improved lateral heat spreading performance for polyvinylidene fluoride composite film comprising silver nanowire in light-emitting diode†
Zhao Li,
Li Zhang,
Rong Qi,
Fan Xie and
Shuhua Qi*
Department of Applied Chemistry, School of Science, Northwestern Polytechnical University, Xi'an 710072, PR China. E-mail: qishuhuanwpu@163.com
First published on 29th March 2016
Abstract
Silver nanowire (AgNW) attracts great attention as a heat dissipation material due to its excellent thermal transfer property which exceeds most traditional heat sink materials. Heat accumulation has a vital risk for light-emitting diodes (LED) devices. In this work, a lateral heat spreader with enhanced thermal conductivity was reported as an additional escaping channel to improve thermal management of LED devices. A lateral heat spreader was prepared based on AgNW/polyvinylidene fluoride (PVDF) composite film which the AgNW was embedded in the PVDF matrix. The through-plane and in-plane thermal conductivity values of 25 vol% AgNW/PVDF composite film are 0.31 W (m K)−1 and 127.6 W (m K)−1 respectively, which far exceed that of pristine PVDF film, and are comparable to those of a commercial copper foil. The surface temperatures of AgNW/PVDF composite film during the transfer processes were captured by a thermal infrared imager and the contours of temperature distribution were simulated. Consequently, the simulated heat transfer is very consistent with actual situations. The data combined with computational simulation indicates that the heat transfers along the direction of AgNW and more Joule heat diffused efficiently along the lateral heat spreader.
1. Introduction
Heat dissipation is a critical issue that needs to be addressed immediately to improve long-term reliability and performances of electronic devices.1–3 The development and research for highly heat conductive materials is essential for the microelectronic industry. An LED, as a semiconductor component, has different energy gaps emitting visible light of different colors. Recent decades have witnessed prevalence of LEDs in general lighting as a substitute for conventional incandescent and fluorescent lamps. The LED is widely used in various aspects due to its various advantages, such as low power consumption, highly efficient light, fast response time, and long lifetime. Higher output performances need a higher driving current in the microelectronic industry. However, LED devices operating at a high driving currents not only produce high output performance, but also aggregate plenty of Joule heating at the junctions of materials, which is harmful to illumination intensity and working life.4 To dissipate heat generated in LED devices, many methods were investigated to enhance heat dissipation.Electronic devices such as mobile phones, tablets, and laptop computers have become more and more familiar in our day-to-day life.5 The miniaturization trend of electronic devices requires components that have high heat dissipation.6 There is no doubt that operating temperature has affected reliability of the electronic devices.7 It is urgent to improve this property of electronic devices because of the demand of high thermal conductivity for traditional materials. The thermal conductivity of polymers is usually approximately 0.2 W (m K)−1,6 making their use as traditional thermal conductivity materials greatly restricted. To overcome these drawbacks, various novel materials of high thermal conductivity such as silver nanowire, Ag, carbon nanotubes,8 graphene,9,10 and metal nanostructures11 are used for thermal transfer media to fill the polymer matrix. These fillers can significantly increase the thermal conductivity of polymers, and simultaneously enhance the electrical conductivity of them.12 Besides high thermal conductivity, there are other excellent properties of these new types of fillers, such as superior mechanical properties, lightweight, excellent chemical properties, etc. These good performances can cater to special demands for lightweight and miniaturized electronic materials.
As we know, high thermal conductive materials can meet the heat transfer demand of a majority of electronic devices. Thus, some ceramic fillers are blended with polymer substrates as thermal conductive fillers including boron nitride,13,14 aluminum nitride,15 silicon carbide,16 and alumina. However, a high volume fraction of ceramic fillers, generally exceeding 60%, is necessary to obtain high thermal conductivity for a composite. Not only that, these fillers seriously affect the mechanical properties and flexibility of a polymer composite for practical applications. Thermal conductivity of a polymer filled with ceramic fillers is lower because of thermal resistance at the interface between the fillers and polymer.17
Metallic nanowires, such as silver nanowires, are very promising candidates for fabricating transparent and flexible films18 for replacement of traditional materials. Silver nanowires are excellent candidates for transparent, flexible, and high electrical and thermal conductivity films because of their high aspect ratio and excellent electrical, optical, magnetic, and thermal properties. More and more researchers have revealed various applications based on AgNW, such as transparent electrodes in thin-film solar cells,19,20 OLEDs,21 touch panels,22 fuel cells,23 and stretchable electronic devices.24,25
Due to polyvinylidene fluoride being a partially fluorinated polymer, it exhibits superior electrochemical and thermal stability and exhibits a great potential for applications in sensors, piezoelectric materials,26 and biomedical fields.27 Considering the excellent properties of AgNW and PVDF, AgNWs were added to PVDF as a novel type of filler. The AgNWs tend to aggregate without other additives to prevent their aggregation because of their nanometer properties.28 Previous research showed that silica nanoparticles can improve thermomechanical properties and other properties. In this work, flexible and high thermal conductivity film was fabricated via thermally assisted evaporation phase separation (TAEPS). The PVDF particles were dissolved in N,N-dimethylformamide (DMF) and formed a PVDF/DMF homogeneous phase solution. The AgNW/PVDF composite film was fabricated via a mixed solution, composed of self-made silver nanowire (AgNW), PVDF/DMF homogeneous phase solution, and silica nanoparticles.
In this letter, a simple wire-bar-coating process is reported as a suitable method for depositing a homogeneous solution,29 which is comprised of silver nanowire and a PVDF/DMF precursor solution. The AgNW/PVDF composite film was obtained from a semitransparent polymer homogeneous solution by intermittent and multiple wire-bar-coating processes on a 6 cm glass or plastic substrate. The film thickness was primarily affected by the solution of polymer, the quality of the bar, the diameter of wound metal wires on the bar, and the coating speed. In this study, the glass was coated in the solution, which was precisely controlled with 50 microliters by pipette each time, and the previous operation was repeated every 60 seconds; this operation was totally repeated 10 times.30 Interestingly, the polymer solution and slide cleaned with deionized water and ethyl alcohol sequentially have a perfect spreadability. The film represents three forms: dried film, drying film, and wetting film in the back of bar movement, which is attributed to the style of intermittent and multiple coating (Fig. 1).
2. Experimental
2.1 Preparation of AgNW and lateral heat spreader
The AgNW was synthesized successfully and used as thermal conductive fills in the fabrication of a lateral heat spreader. The fabricated processes of AgNW were shown in ESI Fig. S1.† The amounts of AgNW/PVDF precursor solution (the processes of fabrication were shown in ESI Fig. S2†) were controlled precisely via pipette and transferred onto the pre-cleaned glass substrate.31 Then, the AgNW/PVDF precursor solution was bar coated with the wire-wound bar (a diameter of 12 mm) and the wire coil (a diameter of 100 μm) at a bar speed of 40 mm s−1, and dried under a vacuum drying oven for 4 h resulting in the AgNW/PVDF composite film being fabricated successfully (Fig. 2).2.2 Characterization
The surface morphologies of AgNW and AgNW/PVDF composite films were characterized by Scanning Electron Microscope (SEM, JSM-6360LV). Structural characterization of the film was performed using X-ray diffraction (XRD) on an X'Pert MPD PRO Advanced diffractometer with Cu Kα radiation (λ = 1.54056) and using FI-TF Spectrum. The thermal diffusion coefficient of the AgNW/PVDF composite film was measured using a laser flash method (LFA447, NETZSCH, Germany) at room temperature and the heat conduction effect of the AgNW/PVDF composite film was characterized by an Infra-Red system (IRS-6) (Thermal Imaging Electrical Technology Co., Ltd., Shanghai).3. Results and discussion
3.1 Microstructure of AgNW
Herein, to obtain the high thermal conductive film based on PVDF and AgNW, we employed the self-made AgNW obtained via a hydrothermal process and PVDF. Every step of the synthesis process is tightly controlled and precisely measured as shown in Fig. S1.† The photograph of AgNW revealed that the average length of silver nanowire approximately reached 15 μm, which was characterized by a Scanning Electron Microscope. The AgNW dispersed uniformly into the PVDF/DMF precursor solution with the help of silica nanoparticles; previous research reported that the silver nanowires tend to aggregate without the silica nanoparticles but the silver nanowires dispersed well if the solution contained silica nanoparticles. Research found that van der Waals attractive interaction between silica nanoparticles and silver nanowires dominates over the depletion-induced interaction between silver nanowires, which greatly improved the dispersion of AgNW in the DMF solution (Fig. 3).323.2 Crystal structure
AgNWs are expected to be ideal filling materials for thermal conductive film considering their high intrinsic thermal conductivity and large ratio.33 AgNW/PVDF composite films at different volume fractions were prepared and their thermal performances were measured systematically. PVDF, as a kind of partially fluorinated polymer, was chosen as a matrix due to its good crystallization and good film-forming properties.Fig. 4 illustrates the X-ray diffraction spectra (XRD) of the pristine PVDF film and 5 vol%, 10 vol%, 15 vol%, 20 vol%, and 25 vol% AgNW/PVDF composite film, respectively. Four diffraction peaks labeled by red rectangles are observed at 38.5°, 44.5°, 65.0°, and 78.1°, which can be assigned to the (111), (200), (220), and (311) reflections of the surface centered cubic structure of metallic silver. The peak value at 20.6° indicated the β phase,34,35 which was labeled by black rectangles in Fig. 4. Moreover, the results also showed that the thermally assisted evaporation phase separation is an effective method for generation of the β phase. Different peaks in the same location also indicated that the AgNW has little influence on the crystallization of PVDF.
 | ||
| Fig. 4 The X-ray diffraction of pristine PVDF film and 25 vol% AgNW/PVDF composite film with different volume fractions. | ||
3.3 Thermo-gravimetric analysis
Thermal stability is an essential property for a lateral heat spreader in electronic devices, being the limiting factor in both processing and applications. The lateral heat spreader based on AgNW/PVDF composite film was measured by TGA with the results shown in Fig. 5(a). The initial decomposition temperature (corresponding to the decomposition of 5 wt%) of pristine PVDF film and 25 vol% AgNW/PVDF composite film was found to be 441 °C and 447 °C, respectively. This indicated that the 25 vol% AgNW/PVDF composite film had a higher thermal decomposition temperature than pristine PVDF film. The weight loss of this stage was attributed to the volatilization of moisture and decomposition of the oligomer. Decomposition of the 25 vol% AgNW/PVDF composite film started at a high temperature because of AgNW, which has high thermal conductivity and could absorb part of the heat. AgNW, which as a thermal filler played an important role in forming the barrier, improved the resistance to thermal degradation and prevented the diffusion of decomposition products from the polymer into the gas phase. Moreover, the nano-silicon dioxide particles, as auxiliary inorganic fillers, were added into the polymer and have good thermal stability. Considering the above factors, we can draw a conclusion that the thermal decomposition temperature of the 25 vol% AgNW/PVDF composite film shifted to higher temperatures. | ||
| Fig. 5 (a) TG curves of pristine PVDF film and 25 vol% AgNW/PVDF composite film in nitrogen; (b) DTG curves of pristine PVDF film and 25 vol% AgNW/PVDF composite film in nitrogen. | ||
The AgNW/PVDF composite film had better thermal stability compared with pristine PVDF film, which was a good explanation of the reason why AgNW/PVDF composite film was used as a lateral heat spreader in electronic devices. As can be seen in Fig. 5(b), for 25 vol% AgNW/PVDF composite film, the maximum weight loss rate was 0.019% min−1, which was far less than that of pristine PVDF film of 0.03% min−1. That indicated that the decomposition of 25 vol% AgNW/PVDF composite film was less drastic than that of pristine PVDF film; therefore, the former had better thermal stability than the latter.
3.4 Thermal conductivity property and analysis of the heat transfer process
In order to illustrate the thermal conductive property of AgNW/PVDF composite film, the thermal diffusion coefficient was measured via a laser flash method. The experiment indicates that the thermal diffusion coefficient increased with an increase of quality percentage of AgNW. Also, we could acquire thermal conductivity according to equations describing thermal diffusion coefficient and thermal conductivity. The equation governing the thermal diffusion coefficient and thermal conductivity36 was written as:where k = thermal conductivity, α is the thermal diffusivity, c = specific heat capacity, and ρ = density. Therefore, thermal conductivity was calculated by this equation. The thermal conductivity and thermal diffusion coefficient did not present a linear relationship due to the slight changing of density of the AgNW/PVDF composite film. The reasons behind this phenomenon are that the specific heat and density of the AgNW/PVDF composite film have slight changes with the fractional increase of silver nanowire.
Using the infrared camera, the heat-transfer processes of pristine PVDF film and AgNW/PVDF composite film in a rectangular area and in a straight line were examined. Compared to the characteristic of heat transfer of pristine PVDF film and the 25 vol% AgNW/PVDF composite film, the performance of heat transfer of the AgNW/PVDF composite (127.6 W m−1 K−1) was significantly better than pristine PVDF film (0.13 W m−1 K−1). We chose straight line as a monitoring area and researched the characteristics of heat transfer of pristine PVDF and 25 vol% AgNW/PVDF composite film. As shown in Fig. 6(e) and (f), the maximum temperature and average temperature were measured every ten seconds. The 25 vol% AgNW/PVDF composite film had a higher temperature gradient than that of pristine PVDF film. According to Fourier's law, thermal conductivity property is proportional to temperature variation ratio. Therefore, thermal conductivity of 25 vol% AgNW/PVDF composite film is superior to that of PVDF film.
It was of interest to monitor the temperature profile on the surface of the thermal test platform. The surface temperature distribution, peak, and average temperature of the lateral heat spreader were examined using IIRS-6. In order to achieve reliable results of measurements, the lateral heat spreader was placed on the thermal test platform and IR intensity images of temperature distribution on the surface of the lateral heat spreader were obtained. The thermal infrared images were captured at the same intervals; these images are shown in Fig. 6(e) and (f), which indicated that the heat tends to transfer along the 25 vol% AgNW/PVDF composite film rather than pristine PVDF film.
In order to vividly explain the lateral heat transfer performance, we set the physical model of the sample. The sample geometry center was set as the origin, perpendicular bisector of sample's length direction and width direction were set as the Y axis, X axis, respectively. The monitoring points were selected every five millimeters in the X axis and Y axis respectively; the details are shown in Fig. 7(a). The 25 vol% AgNW/PVDF composite film was placed on the thermal test platform under constant power. The heat transfer of 25 vol% AgNW/PVDF composite film in a straight line was analyzed and the results indicated that the property of heat transfer had obviously improved.
To rationalize the experimental results and evaluate the heat transfer property, heat simulations of AgNW/PVDF composite film and pristine PVDF film, including transient simulation and steady simulation, were performed using finite simulation element analysis software (ANSYS Icepak) according to the model of natural convection heat transfer, respectively. In transient state simulation, we chose 18 time points on based on the piecewise linear model in 60 seconds simulated 18 temperature distribution contours, and presented a different temperature gradient; these are shown in ESI Fig. S3.† According to Fourier's law of heat conduction, and partial differential equation of heat conduction, when the temperature increased in x direction 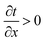 , yet the q < 0, this indicated the heat transferred along the direction of reduction. Conversely, when the
, yet the q < 0, this indicated the heat transferred along the direction of reduction. Conversely, when the 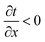 , q > 0, this indicated the heat transferred along the direction of increase.
, q > 0, this indicated the heat transferred along the direction of increase.
According to the transient contours in different times, the 3D color maps on the surface of the AgNW/PVDF composite film (based on three parameters including temperature, position, and time) were obtained via a surface probe (a tool in ANSYS Icepak). The 3D color maps along X direction and Y direction were shown in Fig. 7(c) and (e). When the system reached thermal equilibrium, the contour was shown in Fig. 7(b). As shown in the 3D color maps, we found that the temperature distributed symmetrically on both sides of the origin; the highest temperature was in the heat source center. The temperature of sample surface decreased with the increase of distance from the origin and increased with the increase of heating time. In order to describe vividly the characteristic of symmetric heat transfer, the contours filled with color were established in Fig. 7(f). The contour, which is based on the transient contour in 60 seconds, indicated that the heat flow transferred symmetrically in the X direction and Y direction (Fig. 7(d) and (f)) for the sake of distribution symmetrically on the contour line. A steady state simulation contour obtained in accordance with method of steady state simulation, the temperature of the heat source center reached 47.6 °C and the temperature of the sample boundary was 26.7 °C. Similarly, the temperature decreased with an increase of distance from the origin.
3.5 Lateral heat spreader in LED
The temperature propagation in the LED device is shown in Fig. 8. The experimental results indicate that the heat generated by a LED would spread out along the 25 vol% AgNW/PVDF composite film. Lateral heat transfer performance of AgNW/PVDF composite film is higher than pristine PVDF film. More heat would spread along the heat spreader and be dissipated by two heat sinks at both ends of the LED device due to the excellent through-plane thermal conductivity of the lateral heat spreader concluding 25 vol% AgNW/PVDF composite film. The heat tends to transfer along the film rather than accumulate in surface of the LED device, so the temperature of the LED is low. Heat along the heat spreader is mainly by free electrons of the Ag NW network in the polymer substrate. The thermal conduction is dominated by acoustic phonons; ion-core vibration in a crystal lattice of PVDF. Consequently, the surface temperature curves of the LED indicates that the heat trends to transfer along the heat spreader.4. Conclusions
In conclusion, a lateral heat spreader was prepared comprising an AgNW network in a polyvinylidene fluoride substrate. The thermal conductivity of the lateral heat spreader was greatly improved owing to import of silver nanowires. The lateral heat spreader made of 25 vol% AgNW/PVDF composite film exhibited excellent thermal conductivity of 127.6 W m−1 K−1, which far exceeded the 0.13 W m−1 K−1 of the pristine PVDF film. The lateral heat transfer performance of AgNW/PVDF composite films were examined by a simulation method and was monitored using an infrared camera, respectively. The results indicated that the contours were consistent with the actual results which were reflected from the infrared camera.References
- S. Liu, J. Wang, Y. Lu, D. Huang, C. Huang, W. Hsieh, J. Lee, Y. Tsai, J. Shih and Y.-H. Lee, Reliability Physics Symposium, 2014 IEEE International, 2014, pp. 4A.4.1–4A.4.4 Search PubMed
.
- R. Agaiby, A. G. O'Neill, S. H. Olsen, G. Eneman, P. Verheyen, R. Loo and C. Claeys, IEEE Trans. Electron Devices, 2008, 55, 1568–1573 CrossRef CAS
.
- O. Semenov, A. Vassighi and M. Sachdev, Device Mater. Reliab., IEEE Trans., 2006, 6, 17–27 CrossRef
.
- J. Li, S. Qi, J. Li, M. Zhang and Z. Wang, RSC Adv., 2015, 5, 59398–59402 RSC
.
- J. Li, J. Liang, L. Li, F. Ren, W. Hu, J. Li, S. Qi and Q. Pei, ACS Nano, 2014, 8, 12874–12882 CrossRef CAS PubMed
.
- J. Xia, G. Zhang, L. Deng, H. Yang, R. Sun and C.-P. Wong, RSC Adv., 2015, 5, 19315–19320 RSC
.
- Y. Hwang, M. Kim and J. Kim, RSC Adv., 2014, 4, 17015–17021 RSC
.
- R. C. Tenent, T. M. Barnes, J. D. Bergeson, A. J. Ferguson, B. To, L. M. Gedvilas, M. J. Heben and J. L. Blackburn, Adv. Mater., 2009, 21, 3210–3216 CrossRef CAS
.
- K. Rana, J. Singh and J.-H. Ahn, J. Mater. Chem. C, 2014, 2, 2646–2656 RSC
.
- J. O. Hwang, J. S. Park, D. S. Choi, J. Y. Kim, S. H. Lee, K. E. Lee, Y.-H. Kim, M. H. Song, S. Yoo and S. O. Kim, ACS Nano, 2011, 6, 159–167 CrossRef PubMed
.
- A. R. Rathmell, S. M. Bergin, Y. L. Hua, Z. Y. Li and B. J. Wiley, Adv. Mater., 2010, 22, 3558–3563 CrossRef CAS PubMed
.
- R. Qian, J. Yu, C. Wu, X. Zhai and P. Jiang, RSC Adv., 2013, 3, 17373–17379 RSC
.
- T.-L. Li and S. L.-C. Hsu, J. Phys. Chem. B, 2010, 114, 6825–6829 CrossRef CAS PubMed
.
- W. Yan, Y. Zhang, H. Sun, S. Liu, Z. Chi, X. Chen and J. Xu, J. Mater. Chem. A, 2014, 2, 20958–20965 CAS
.
- A. Agrawal and A. Satapathy, Composites, Part A, 2014, 63, 51–58 CrossRef CAS
.
- J.-P. Cao, X. Zhao, J. Zhao, J.-W. Zha, G.-H. Hu and Z.-M. Dang, ACS Appl. Mater. Interfaces, 2013, 5, 6915–6924 CAS
.
- Y. Shimazaki, F. Hojo and Y. Takezawa, Appl. Phys. Lett., 2008, 92, 133309 CrossRef
.
- L. Hu, H. S. Kim, J.-Y. Lee, P. Peumans and Y. Cui, ACS Nano, 2010, 4, 2955–2963 CrossRef CAS PubMed
.
- M.-G. Kang, H. J. Park, S. H. Ahn, T. Xu and L. J. Guo, IEEE J. Sel. Top. Quantum Electron., 2010, 16, 1807–1820 CrossRef CAS
.
- W. Gaynor, J.-Y. Lee and P. Peumans, ACS Nano, 2009, 4, 30–34 CrossRef PubMed
.
- X. Y. Zeng, Q. K. Zhang, R. M. Yu and C. Z. Lu, Adv. Mater., 2010, 22, 4484–4488 CrossRef CAS PubMed
.
- J. Lee, P. Lee, H. Lee, D. Lee, S. S. Lee and S. H. Ko, Nanoscale, 2012, 4, 6408–6414 RSC
.
- I. Chang, T. Park, J. Lee, H. B. Lee, S. Ji, M. H. Lee, S. H. Ko and S. W. Cha, Int. J. Hydrogen Energy, 2014, 39, 7422–7427 CrossRef CAS
.
- P. Lee, J. Ham, J. Lee, S. Hong, S. Han, Y. D. Suh, S. E. Lee, J. Yeo, S. S. Lee and D. Lee, Adv. Funct. Mater., 2014, 24, 5671–5678 CrossRef CAS
.
- P. Lee, J. Lee, H. Lee, J. Yeo, S. Hong, K. H. Nam, D. Lee, S. S. Lee and S. H. Ko, Adv. Mater., 2012, 24, 3326–3332 CrossRef CAS PubMed
.
- G.-d. Kang and Y.-m. Cao, J. Membr. Sci., 2014, 463, 145–165 CrossRef CAS
.
- B. Chu, X. Zhou, K. Ren, B. Neese, M. Lin, Q. Wang, F. Bauer and Q. Zhang, Science, 2006, 313, 334–336 CrossRef CAS PubMed
.
- S. Nam, H. W. Cho, S. Lim, D. Kim, H. Kim and B. J. Sung, ACS Nano, 2013, 7, 851–856 CrossRef CAS PubMed
.
- D. Khim, H. Han, K. J. Baeg, J. Kim, S. W. Kwak, D. Y. Kim and Y. Y. Noh, Adv. Mater., 2013, 25, 4302–4308 CrossRef CAS PubMed
.
- W. Lee, C. T. Hong, O. H. Kwon, Y. Yoo, Y. H. Kang, J. Y. Lee, S. Y. Cho and K. S. Jang, ACS Appl. Mater. Interfaces, 2015, 7, 6550–6556 CAS
.
- W. J. Lee, W. T. Park, S. Park, S. Sung, Y. Y. Noh and M. H. Yoon, Adv. Mater., 2015, 27, 5043–5048 CrossRef CAS PubMed
.
- S. Nam, H. W. Cho, S. Lim, D. Kim, H. Kim and B. J. Sung, ACS Nano, 2012, 7, 851–856 CrossRef PubMed
.
- S. Wang, Y. Cheng, R. Wang, J. Sun and L. Gao, ACS Appl. Mater. Interfaces, 2014, 6, 6481–6486 CAS
.
- E. H. Abdelhamid, O. D. Jayakumar, V. Kotari, B. P. Mandal, R. Rao, V. M. Naik, R. Naik and A. K. Tyagi, RSC Adv., 2016, 6, 20089–20094 RSC
.
- Z. Li, X. Zhang and G. Li, Phys. Chem. Chem. Phys., 2014, 16, 5475–5479 RSC
.
- Z. Wang, H. Kriegs and S. Wiegand, J. Phys. Chem. B, 2012, 116, 7463–7469 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra04680k |
| This journal is © The Royal Society of Chemistry 2016 |







