Dielectric investigation of the Sr3WO6 double perovskite at RF/microwave frequencies
Abstract
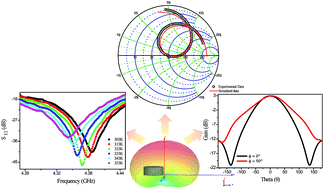
This work reports the dielectric properties of Sr3WO6 (SWO) double perovskite at radio frequencies as a function of the temperature and a study in microwaves range to evaluate the material as a novel dielectric resonator. Thermo-activated charge transfer process for SWO ceramic was observed and two resistor–capacitor associations were fitted for the Nyquist diagram. SWO presented negative values of temperature coefficient of capacitance (TCC) and the activation energies of dielectric relaxation processes were measured by conductivity (1.35 eV), imaginary modulus (1.38 eV) and imaginary impedance (1.36 eV). For the microwave range, SWO shows high dielectric permittivity (13.57) and dielectric loss (0.0281). The dielectric resonator antenna (DRA) fabricated from SWO ceramic presented an operation frequency in 4.1 GHz and return loss below −40 dB. It was possible to evaluate the performance of the antenna from numerical simulation. From these results, the SWO based DRA shows good properties to be used as a novel microwave dielectric resonator.


 Please wait while we load your content...
Please wait while we load your content...