Diselenide-based probe for the selective imaging of hypochlorite in living cancer cells†
Abstract
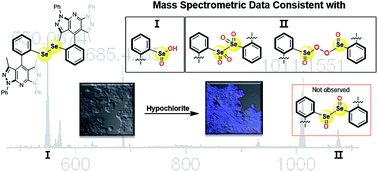
A non-traditional and robust probe skeleton was derivatized for chemosensing applications to investigate a potential novel mode of hypochlorite detection. The BDPP-DSe probe gave a ∼180-fold “turn-on” response to hypochlorite. Confocal fluorescence imaging demonstrated detection of hypochlorite in living cells and cell membrane permeability.
- This article is part of the themed collection: Selenium & Tellurium chemistry at the beginning of the 3rd millennium: a celebration of ICCST

 Please wait while we load your content...
Please wait while we load your content...