Docetaxel-conjugated monomethoxy-poly(ethylene glycol)-b-poly(lactide) (mPEG-PLA) polymeric micelles to enhance the therapeutic efficacy in oral squamous cell carcinoma
Abstract
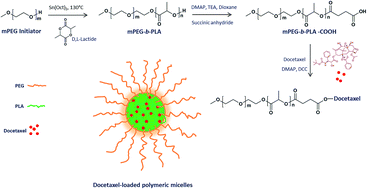
In this study, docetaxel (DTX) was successfully conjugated to the monomethoxy-poly(ethylene glycol)-b-poly(lactide) (mPEG-PLA) polymer block via an ester linkage (DTX-PM). The polymer–drug conjugate resulted in the formation of nanosized micelles with a clear spherical shape. This size range is suitable for passive targeting via an EPR effect in cancer drug delivery. The DTX-PM exhibited a sustained release of the drug over 160 h with slightly accelerated release at acidic conditions. A cytotoxicity assay clearly revealed a time-dependent anticancer effect of DTX-PM in the squamous cancer cells. At 24 h incubation, the free drug exhibited a potent anticancer effect in the cancer cells while DTX-PM exhibited a superior anticancer effect by the end of 48 h incubation. The higher cytotoxic potential of DTX-PM suggests potential therapeutic advantages for cancer treatments. The annexin-V/PI based apoptosis assay further confirmed the anticancer potential of DTX-PM. The DTX-PM induced significantly higher cancer cell apoptosis in HSC-3 cancer cells. Importantly, DTX-PM significantly controlled the tumor progression in HSC-3 cancer cells bearing a tumor xenograft. Consistently, DTX-PM treated mice however showed a severe necrosis of the cancer cells in H&E staining and showed remarkably higher TUNEL positive apoptotic cells. Overall, a polymer–drug conjugate (DTX-PM) is a promising strategy to improve the therapeutic efficacy in oral squamous cell carcinoma.

 Please wait while we load your content...
Please wait while we load your content...