Electrodeposition-based electrochromic devices with reversible three-state optical transformation by using titanium dioxide nanoparticle modified FTO electrode†
Tao Ye,
Yong Xiang*,
Hong Ji,
Congjin Hu and
Gang Wu*
School of Energy Science and Engineering, University of Electronic Science and Technology of China, 2006 Xiyuan Ave, West High-Tech Zone, Chengdu, Sichuan 611731, China. E-mail: xiang@uestc.edu.cn; wugang2012@uestc.edu.cn
First published on 11th March 2016
Abstract
Reversible electrodeposition-based electrochromic devices are highly promising for extensive applications owing to their facile and low-cost fabrication. Herein, a novel electrodeposition-based electrochromic device with reversible three-state optical transformation, i.e. transparent, mirror, and black, was fabricated by introducing a fluorine-doped tin oxide (FTO) electrode modified with commonly available and inexpensive titanium dioxide (TiO2) nanoparticles. Typically, the fabricating strategy mainly involved three procedures, namely obtaining a stable dispersion of TiO2 nanoparticles by milling, achieving a surface modification of the FTO electrode with TiO2 nanoparticles by spin-coating and sintering, and assembling the device by sandwiching gel electrolyte between the modified FTO electrode and a flat FTO electrode. By applying different voltages for a short while, this transparent smart device can be immediately switched to black (+2.5 V/20 s) or mirror (−2.5 V/20 s) state by depositing Ag on the surface of the modified or unmodified FTO electrode respectively. There is below 1% transmittance in the black state and over 80% reflectance in the mirror state for the device. By changing the surface structure of the TiO2 modified FTO electrode, the optical properties of the device in different states can be controlled effectively. Moreover, the optical transformation exhibited good stability over 1500 cycles of testing.
1. Introduction
Chromism mainly depends on the change of electron states in the molecule, especially the π electrons and d electrons. In theory, once various stimuli change the electron states of materials, the chromism phenomenon will occur.1 Typically, when voltage is applied as an external stimulus to some materials, their optical properties chiefly involving transmittance and reflectance can be reversibly modulated, that is, so-called electrochromism.2,3 Since Deb prepared the first WO3-based electrochromic device in 1969,4 studies of electrochromism have been paid great attention owing to their extensive potential applications, e.g. smart windows,5,6 rear-view mirrors,7 electrochromic displays,8–10 cathode ray tubes with variable transmittance,11 and security.12–14 Up to now, electrochromic materials mainly fall within the following categories: transition metal oxides,15–17 conjugated conducting polymers,18,19 viologens,20,21 metal coordination complexes,22,23 hybrid electrochromic materials,24,25 and electrodeposition-based electrochromic materials.26,27,32–35For the electrodeposition-based electrochromic materials, one of the most important electrochromic categories, electrochromic devices show a simple sandwich-type structure consisting of the electrolyte, electrodeposition components, and flat electrodes. When voltage is applied to the electrodes, the metal element dissolved in the electrolyte will be deposited on the electrode surface. On the contrary, the deposited metal will be dissolved again into the electrolyte upon removing the voltage. In this way, the transmittance and reflectance of the device can be changed reversibly. Bi/Cu alloy as one of most studied electrodeposition systems was reported first by John P. Ziegler, and a black and white pixel contrast ratio of 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was achieved.28 In this system, the redox reaction between Cu2+ and metallic bismuth can facilitate the reversible conversion between Bi and Bi3+; meanwhile, the Cu may also increase the efficiency of the electrochemical reaction and accelerate the deposition and dissolution of metallic bismuth.29–31 Based on the above researches, the Ag/Cu system was developed recently as a novel reversible electrodeposition system, and it has been used to fabricate electrochromic devices.32–35 For instance, Shingo Araki et al. achieved a reversible black state through depositing Ag onto an indium tin oxide (ITO) nanoparticle modified ITO electrode.33 Furthermore, by controlling the growth of Ag grains under different voltages, the devices showed multiple color states.34 In addition, by integrating a thiol modified ITO electrode and ionic liquids, Chihyun Park et al. obtained switchable silver mirrors with long memory effects.35
1 was achieved.28 In this system, the redox reaction between Cu2+ and metallic bismuth can facilitate the reversible conversion between Bi and Bi3+; meanwhile, the Cu may also increase the efficiency of the electrochemical reaction and accelerate the deposition and dissolution of metallic bismuth.29–31 Based on the above researches, the Ag/Cu system was developed recently as a novel reversible electrodeposition system, and it has been used to fabricate electrochromic devices.32–35 For instance, Shingo Araki et al. achieved a reversible black state through depositing Ag onto an indium tin oxide (ITO) nanoparticle modified ITO electrode.33 Furthermore, by controlling the growth of Ag grains under different voltages, the devices showed multiple color states.34 In addition, by integrating a thiol modified ITO electrode and ionic liquids, Chihyun Park et al. obtained switchable silver mirrors with long memory effects.35
In this study, an electrodeposition-based electrochromic device with a reversible three-state optical transformation was fabricated by a facile and low-cost strategy which involved sandwiching gel electrolyte containing Ag/Cu deposition materials between a flat FTO electrode and a TiO2 nanoparticle modified FTO electrode. Upon applying a suitable voltage, this smart device can change optical state from transparent to black or mirror by depositing Ag on the surface of the modified or unmodified FTO electrode. The optical properties of the device in different states can be controlled effectively by changing the surface structure of the TiO2 modified FTO electrode. Moreover, the optical transformation exhibited good stability over 1500 cycles of testing.
2. Experimental
2.1. Materials
We used FTO glass electrodes 2.5 × 3 cm in size (10 Ω sq−1, Wuhan Lattice Solar Energy Technology Co. Ltd.), TiO2 nanopowders with three different average diameters (i.e. 5–10 nm, 40 nm, 100 nm, ≥99.8%, Aladdin Co. Ltd.), dimethyl sulfoxide (DMSO, ≥99.8%, J&K Chemical Co. Ltd.), tetra-n-butylammonium bromide (TBABr, 99%, J&K Chemical Co. Ltd.), silver nitrate (AgNO3, ≥99.8%, Guangdong Guanghua Sci-Tech Co. Ltd.), copper chloride (CuCl2, ≥99.0%, KeLong Chemical Co. Ltd.), poly(vinyl butyral) (PVB, Sekisui Chemical Co. Ltd.), ethyl cellulose (≥99.5%, Hanzhou Lanbo Industrial Co. Ltd.), lauric acid (≥99.8%, KeLong Chemical Co. Ltd.), terpineol (≥98.0%, KeLong Chemical Co. Ltd.), ethyl alcohol (≥99.7%, KeLong Chemical Co. Ltd.) and Teflon sheet, all of which, except for the FTO glass electrodes and Teflon sheet, were obtained from commercial sources and used directly without further purification. Teflon sheet with a thickness of 0.5 mm was cut to 2.5 × 2.5 cm with a 2 × 2 cm hole. The FTO glass electrodes and Teflon sheet were cleaned with ethanol and de-ionized water several times before use.2.2. Preparation of TiO2 nanoparticle dispersion and gel electrolyte
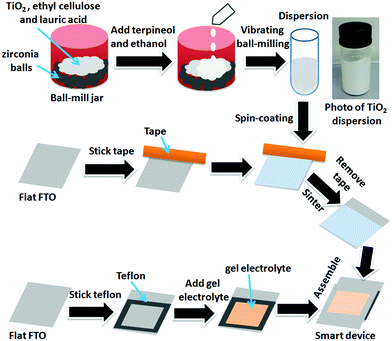
A typical method to prepare the TiO2 nanoparticle dispersion is as follows: TiO2 nanopowders (2.5 g), ethyl cellulose (0.75 g) and lauric acid (0.25 g) were placed in a ball-mill jar, and then mixed with terpineol (16 mL) and ethyl alcohol (10 mL). The mass ratio of TiO2, ethyl cellulose, lauric acid, and terpineol was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.3
0.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1
0.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.36 After 50 min milling, a slurry was obtained in the ball-mill jar. Finally, the TiO2 nanoparticle dispersion was prepared by diluting the slurry with ethyl alcohol. In this experiment, the terpineol and ethyl cellulose acted as the adhesive, and lauric acid as surfactant, and the ethanol was used to adjust the concentration of the slurry.
6.36 After 50 min milling, a slurry was obtained in the ball-mill jar. Finally, the TiO2 nanoparticle dispersion was prepared by diluting the slurry with ethyl alcohol. In this experiment, the terpineol and ethyl cellulose acted as the adhesive, and lauric acid as surfactant, and the ethanol was used to adjust the concentration of the slurry.
A typical method to prepare the gel electrolyte is as follows: TBABr (806 mg, 2.5 mmol), silver nitrate (85 mg, 0.5 mmol), and copper chloride (13 mg, 0.1 mmol) were dissolved in 10 mL of DMSO. After that, 1.32 g (10 wt%) PVB was added to the solution to obtain the gel electrolyte.
2.3. Modification of the FTO electrode by TiO2 nanoparticles and fabrication of the devices
Ethyl alcohol (10 mL) as a diluent was added to the TiO2 nanoparticle dispersion (5 mL), and then the dispersion was ultrasonically vibrated for 20 min. After that, 0.25 mL of the above mentioned dispersion was spin-coated onto the surface of the FTO electrode twice (500 rpm, 5 s; 1500 rpm, 15 s). Then, the spin-coated samples were sintered for 30 min at 500 °C to get the TiO2 nanoparticle modified FTO electrode.The devices were assembled by sandwiching the gel electrolyte between a flat FTO electrode and a TiO2 nanoparticle modified FTO electrode. In this procedure, a 2.5 × 2.5 cm Teflon sheet with a 2 × 2 cm hole was used as spacer, and the gel electrolyte was contained in the hole.
2.4. Characterization
The morphology of the samples was observed with a field-emission scanning electron microscope (S-3400, Hitachi). The X-ray diffraction (XRD) patterns were acquired on a diffractometer (DX-1000, Dandong Haoyun Instrument Co. Ltd.) with Cu Kα (λ = 1.542 Å) radiation at 40 kV and 25 mA. The components of deposited films were detected with energy dispersive X-ray spectroscopy (EDXS, Oxford INCA). An electrochemical workstation (CHI660D, CHI) was used for the electrochemical tests, and a UV-Vis spectrophotometer (Cary 5000, Agilent) was used to record the optical properties.3. Results and discussion
3.1. Electrochemical analysis
Fig. 1 shows the 3-electrode cyclic voltammograms (CV) of the DMSO solutions with different electrolyte components at a fixed concentration (the working electrode is the flat FTO electrode). For TBABr and TBABr/CuCl2 systems, only a very weak peak current can be detected. But for TBABr/AgNO3 and TBABr/AgNO3/CuCl2 systems a strong peak current was observed, reflecting a redox reaction. For the TBABr/AgNO3/CuCl2 ternary system, the cathode current occurred at about −0.8 V and reached a peak at −1.65 V, which resulted from the electrochemical reductions of Ag+ and Cu2+.For the voltage sweep from −2 V in the positive direction, an anodic current was observed at −0.5 V and reached a maximum at +0.6 V, indicating that the electrodeposited Ag and Cu were oxidized. Indeed, the EDXS composition analysis for the films electrodeposited on the modified and unmodified FTO electrodes supported this result and showed that Ag with over 20 atom% was obviously dominant compared with the trace Cu. In addition, compared with the TBABr/AgNO3 system, the oxidation current in the TBABr/AgNO3/CuCl2 system was stronger, and the voltage at the peak current was higher. Accordingly, it can be supposed that Cu2+ can greatly improve the electrochemical reaction efficiency of Ag and accelerate the metal deposition and dissolution similar to the Bi/Cu system.29–31 There was an additional oxidation current at around +0.9 V attributed to the oxidation of Cu+ to Cu2+.
Fig. S1† shows CV curves for the FTO electrodes modified with different sizes of TiO2 nanoparticles (5–10 nm, 40 nm, 100 nm) in DMSO gel electrolyte (100 mM TBABr, 5 mM CuCl2, and 5 mM AgNO3). It can be seen that the CV curves of the modified FTO electrodes are different from the CV curve of unmodified FTO electrode in the same electrolyte, indicating that the introduction of TiO2 will influence the electrochemical reaction partly. However, all of the CV curves for the different sized TiO2 nanoparticle modified FTO electrodes are similar.
3.2. Preparation of a voltage-controlled smart device with reversible three-state optical transformation
The strategy for preparing the smart device mainly involved three procedures as shown in Fig. 2. Firstly, a stable dispersion of TiO2 nanoparticles was obtained by milling. Secondly, this dispersion was spin-coated onto the conducting surface of a clean FTO electrode, and then sintering was used to achieve surface modification of the FTO with TiO2 nanoparticles. Finally, the smart device was assembled by sandwiching gel electrolyte between the modified FTO electrode and a flat FTO electrode.Fig. S2† shows the X-ray powder diffraction (XRD) patterns of TiO2 film, sintered TiO2 film, fresh TiO2 nanopowders, and a flat FTO electrode. It can be seen that the diffraction peaks obtained for all of these materials appear at the same positions (TiO2 anatase, JCPDS 21-1217), indicating that the TiO2 remained in the anatase structural form throughout the whole procedure for modifying FTO. Moreover, the diffraction peaks of tin oxide (SnO2, JCPDS 46-1088) on the FTO electrode surface were not found in the XRD pattern of the TiO2 modified FTO electrode, which indicates that TiO2 nanoparticles completely covered the FTO electrode surface.
As shown in Fig. 3, the smart device can achieve a reversible three-state optical transformation from mirror, through transparent, to black by application of different voltages. When a negative voltage (−2.5 V) was applied to the unmodified flat FTO electrode, the mirror state was obtained, and the reflection of the yellow pen was obviously visible in the device (Fig. 3a). On the contrary, when a positive voltage (2.5 V) was applied to the unmodified flat FTO electrode, the black state was triggered, and of course the reflection of the yellow pen was not seen in the device (Fig. 3c). Without the application of voltage, the device was transparent, and the white paper as a background could be seen through the device (Fig. 3b). An essential mechanism is Ag electrodeposition and dissolution according to previous reports.29–31 However, the surface modification of the flat FTO with TiO2 nanoparticles is extremely important to realize the black optical state due to the formation of the rough Ag deposition layer which resulted in absorption and/or multiple scattering of light.33
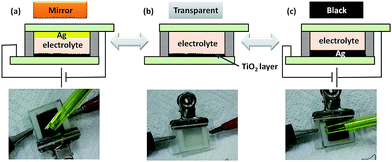 | ||
| Fig. 3 Photographs and the schematic mechanism of the voltage-controlled smart device with different optical states. (a) Mirror (−2.5 V), (b) transparent (0 V), (c) black (+2.5 V). | ||
3.3. Adjustable optical properties of the smart device by changing surface structure of the modified FTO
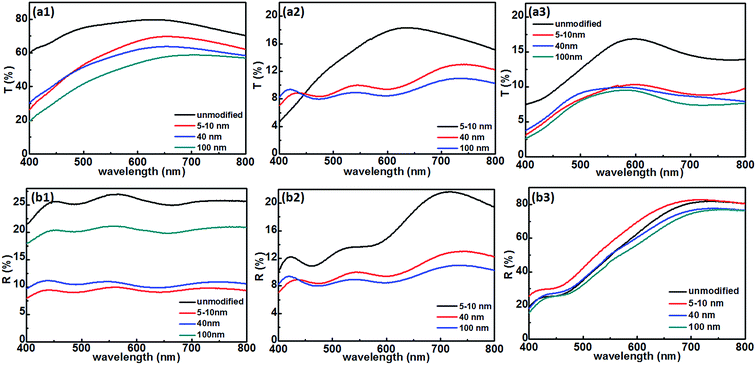
As mentioned above, the surface modification of the flat FTO played an important role for the realization of the black optical state. Hence, the optical properties of the smart device are expected to be altered by changing the surface structure of the modified FTO. In this connection, three dispersions containing different sized TiO2 nanoparticles (5–10 nm, 40 nm, and 100 nm) at the same concentration were prepared in the same way and respectively used to modify the surface of the flat FTO electrode. Apparently, all of the smart devices with different sized TiO2 particle modified FTO electrodes presented the voltage-controlled three optical states (transparent, mirror, and black) very well (Fig. 4). However, the light transmission capacity of the smart devices in the transparent state was weakened with increasing size of TiO2 particles.The transmission and reflection spectra of the smart devices with different sized TiO2 modified FTO electrodes are shown in Fig. 5. For the transmission spectra, the negative pole was connected to the flat FTO electrode (−2.5 V/20 s) and the TiO2 nanoparticle modified FTO electrode (2.5 V/20 s), respectively, to get the mirror and the black state. For the reflection spectra, the negative pole was connected similarly, and the same voltage was provided for 90 s to separately get the mirror and the black states. As shown in Fig. 5a1, modification of the flat FTO electrode by using TiO2 nanoparticles can slightly affect the light transmission capacity of devices, and the transmittance was gradually reduced with increasing size of TiO2 nanoparticles. Similarly, the TiO2 nanoparticle modified devices showed a weakened reflectivity, and the reflectance was gradually increased with the increase of the TiO2 nanoparticle size (Fig. 5b1). Moreover, when the modified devices were in the black state, the transmittance and reflectance were uniformly low and decreased to below 11% with increasing the size of the TiO2 nanoparticles to 100 nm (Fig. 5a2 and b2). With regard to the mirror state (Fig. 5a3 and b3), the devices showed high reflectance and low transmittance. However, the influences of TiO2 size on transmittance and reflectance were quite limited owing to the deposition of Ag on the surface of the unmodified FTO electrode.
In order to reveal the relationship between the optical properties of the smart device and the microstructure of the modified flat FTO with different sizes of TiO2 nanoparticles, field emission scanning electron microscopy (FESEM) was applied. As shown in the FESEM images Fig. 6b2, b3, c2, c3, d2, and d3, the modified flat FTO electrodes in the transparent state before Ag deposition (Fig. 6b1–d1) were obviously more rough than the unmodified flat FTO electrode (Fig. 6a2 and a3), and the roughness increased with the increase of TiO2 nanoparticle size. The rough surface of the modified flat FTO electrodes should be attributed to the aggregation and agglomeration of TiO2 nanoparticles during the spin-coating and sintering, respectively. Moreover, when the deposition of Ag on the surfaces of the different flat FTO electrodes to obtain the mirror or black state was completed (Fig. 6a4–d4), the FESEM images of the unmodified and 5–10 nm TiO2 modified FTO electrodes observably presented a less smooth surface than before the Ag deposition (Fig. 6a5, a6, b5, and b6), while the surfaces of 40 and 100 nm TiO2 modified flat FTO electrodes were still very rough due to the presence of big aggregated and agglomerated TiO2 nanoparticles (Fig. 6c5, c6, d5, and d6). The Ag deposited layer was thick and compact. Typically, a side view FESEM image of the 5–10 nm TiO2 particle modified FTO electrode with a TiO2 layer of about 400 nm and a 20 nm SnO2 layer (Fig. S3a†) after Ag deposition shows that the thickness of the compact Ag layer was approximately 4 μm (Fig. S3b†).
Considering the results of the transmission and reflection spectra, it can be concluded that the TiO2 nanoparticles applied to modify the flat FTO electrodes mainly contributed to the black optical state of the devices, and the black optical state was enhanced with increasing the sizes of TiO2 nanoparticles owing to the rougher surface structure of the modified FTO electrode with greater size of TiO2 nanoparticles.
3.4. Study of switching time and cycle stability of the smart device
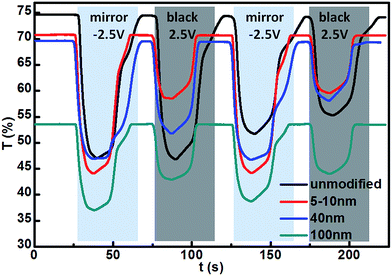
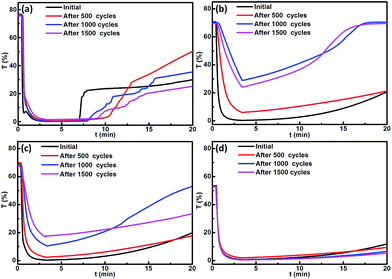
The switching times of different optical states for the unmodified and modified devices were evaluated by measuring transmittance changes at 700 nm during 2-electrode CV tests. As shown in Fig. 7, the time spent in the mirror state for the modified devices was shorter than that for the unmodified device, which seems to indicate that the modified devices can switch more quickly from mirror to transparent state than the unmodified device. Also, the switching times for the different size TiO2 nanoparticle modified devices were different, and there should be the shortest switching time for the 5–10 nm TiO2 nanoparticle modified device. However, the time in the black state for the different size TiO2 nanoparticle modified devices showed no obvious difference. Notably, during the second CV cycle, the tendency for transmittance variation for the modified devices was almost consistent with that of the first CV cycle, while an obvious decay of the transmittance variation for the unmodified device was observed, probably reflecting that the stability of the modified devices was better than the unmodified device during the CV cycles. This was solidly supported by the longer CV cycles (Fig. S4†).By repeatedly applying sequential voltages to the devices, the cycle stability was further investigated as shown in Fig. 8. For all of the devices, at the beginning of the first test cycle the transmittance at 700 nm was sharply reduced and it remained at approximately 0% while the 2.5 V constant voltage was implemented, reflecting that the conversion of optical state from transparent to black was complete. However, after the 2.5 V voltage was removed, the transmittance of the modified devices gradually increased with time (Fig. 8b–d), and the 100 nm TiO2 nanoparticle modified device presented the least increasing trend, indicating that spontaneous restoration to the original transparent state was occurring, which was more difficult for the greater size TiO2 nanoparticle modified devices. For the unmodified device (Fig. 8a), after removing the 2.5 V voltage, an abrupt increase in the transmittance was observed, which could be attributed to a peeling off of the Ag film deposited on the flat FTO electrode. Subsequently, the sequential voltages were cyclically applied to the devices. Through 500 cycles, the changing trend of transmittance for the devices modified with either 40 or 100 nm TiO2 was quite consistent with that of the initial devices (Fig. 8c and d), while the transmittance curve of the 5–10 nm TiO2 modified device was obviously offset from the initial curve (Fig. 8b). With increasing numbers of cycles up to 1500, the deviation was more prominent for small size TiO2 modified devices, while the transmittance curve of the 100 nm TiO2 modified device almost overlapped with the initial curve. Accordingly, it is inferred that the cycle stability of the modified devices was enhanced by increasing the size of TiO2 nanoparticles.
4. Conclusion
In summary, we have successfully developed a simple and low-cost three-step approach for the fabrication of a novel electrodeposition-based electrochromic device with reversible transparent, mirror, and black three-state optical transformation by introducing an FTO electrode modified with common and inexpensive TiO2 nanoparticles. It is significant for the potential applications that transmittance in the black state and reflectance in mirror state for this device were below 1% and over 80% respectively. Moreover, the optical properties of the device in different states can be adjusted easily by manipulating the surface structure of the TiO2 modified FTO electrode, and the cycle stability of the device is considerable, even for over 1500 cycles of testing.Acknowledgements
The authors appreciate the financial support from National Natural Science Foundation of China (No. 51472044 and No. 51503026) and Fundamental Research Funds for the Central Universities (No. ZYGX2014J090).Notes and references
- V. K. Thakur, G. Ding, J. Ma, P. S. Lee and X. Lu, Adv. Mater., 2012, 24, 4071 CrossRef CAS PubMed
.
- C. G. Granqvist, Thin Solid Films, 2014, 564, 1 CrossRef CAS
.
- R. J. Mortimer, Annu. Rev. Mater. Res., 2011, 41, 241 CrossRef CAS
.
- S. K. Deb, Appl. Opt., 1969, 8, 192 CrossRef PubMed
.
- C. Bechinger, S. Ferrere, A. Zaban, J. Sprague and B. A. Gregg, Nature, 1996, 383, 608 CrossRef CAS
.
- A. Kraft and M. Rottmann, Sol. Energy Mater. Sol. Cells, 2009, 93, 2088 CrossRef CAS
.
- D. Krause and V. Paquet, Electro-Opt. Syst. Des., 1977, 9, 54 CAS
.
- B. S. Cho, W. J. Kwon, S.-J. Choi, P. Kim, S.-A. Park, J. Kim, S. J. Son, R. Xiao, S.-H. Kim and S. B. Lee, Adv. Mater., 2005, 17, 171 CrossRef
.
- U. Bach, D. Corr, D. Lupo, F. Pichot and M. Ryan, Adv. Mater., 2002, 14, 845 CrossRef CAS
.
- F. C. Krebs, Nat. Mater., 2008, 7, 766 CrossRef CAS PubMed
.
- G. C. Vries, Electrochim. Acta, 1999, 44, 3185 CrossRef
.
- P. M. S. Monk, C. Turner and S. P. Akhtar, Electrochim. Acta, 1999, 44, 4817 CrossRef CAS
.
- B. Baloukas, J. M. Lamarre and L. Martinu, Appl. Opt., 2011, 50, C41 CrossRef PubMed
.
- B. Baloukas and L. Martinu, SVC Bulletin Fall., 2013, 36.
- H. Li, G. Shi, H. Wang, Q. Zhang and Y. Li, J. Mater. Chem. A, 2014, 2, 11305 CAS
.
- E. O. Zayim, I. Turhan, F. Z. Tepehan and N. Ozer, Sol. Energy Mater. Sol. Cells, 2008, 92, 164 CrossRef
.
- M. Dhanasankar, K. K. Purushothaman and G. Muralidharan, Solid State Sci., 2010, 12, 246 CrossRef CAS
.
- W. Lu, A. G. Fadeev, B. Qi, E. Smela, B. R. Mattes, J. Ding, G. M. Spinks, J. Mazurkiewicz, D. Zhou, G. G. Wallace, D. R. MacFarlane, S. A. Forsyth and M. Forsyth, Science, 2002, 297, 983 CrossRef CAS PubMed
.
- M. Icli, M. Pamuk, F. Algi, A. M. Onal and A. Cihaner, Chem. Mater., 2010, 22, 4034 CrossRef CAS
.
- T. Kakibe and H. Ohno, J. Mater. Chem., 2009, 19, 4960 RSC
.
- X. W. Sun and J. X. Wang, Nano Lett., 2008, 8, 1884 CrossRef CAS PubMed
.
- Q. Zeng, A. McNally, T. E. Keyes and R. J. Forster, Electrochem. Commun., 2008, 10, 466 CrossRef CAS
.
- G. de la Torre, C. G. Claessens and T. Torres, Chem. Commun., 2007, 2000 RSC
.
- H. Ling, J. Lu, S. Phua, H. Liu, L. Liu, Y. Huang, D. Mandler, P. S. Lee and X. Lu, J. Mater. Chem. A, 2014, 2, 2708 CAS
.
- J. Zhang, J. Tu, D. Zhang, Y. Qiao, X. Xia, X. Wang and C. Gua, J. Mater. Chem., 2011, 21, 17316 RSC
.
- M. Nakashima, T. Ebine, M. Shishikura, K. Hoshino, K. Kawai and K. Hatsusaka, ACS Appl. Mater. Interfaces, 2010, 2, 1471 CAS
.
- A. Imamura, M. Kimura, T. Kon, S. Sunohara and N. Kobayashi, Sol. Energy Mater. Sol. Cells, 2009, 93, 2079 CrossRef CAS
.
- J. P. Ziegler, Sol. Energy Mater. Sol. Cells, 1999, 56, 477 CrossRef CAS
.
- C. O. Avellaneda, M. A. Napolitano, E. K. Kaibara and L. O. S. Bulhoes, Electrochim. Acta, 2005, 50, 1317 CrossRef CAS
.
- S. I. Torresi and I. A. Carlos, J. Electroanal. Chem., 1996, 414, 11 CrossRef
.
- L. H. Mascaro, E. K. Kaibara and L. O. S. Bulhoes, J. Electrochem. Soc., 1997, 144, 273 CrossRef
.
- M. R. S. Oliveira, D. A. A. Mello, E. A. Ponzio and S. C. de Oliveira, Electrochim. Acta, 2010, 55, 3756 CrossRef CAS
.
- S. Araki, K. Nakamura, K. Kobayashi, A. Tsuboi and N. Kobayashi, Adv. Mater., 2012, 24, OP122 CAS
.
- A. Tsuboi, K. Nakamura and N. Kobayashi, Adv. Mater., 2013, 25, 3197 CrossRef CAS PubMed
.
- C. Park, S. Seo, H. Shin, B. D. Sarwade, J. Na and E. Kim, Chem. Sci., 2015, 6, 596 RSC
.
- H.-S. Kim, C.-R. Lee, J.-H. Im, K.-B. Lee, T. Moehl, A. Marchioro, S.-J. Moon, R. H. Baker, J.-H. Yum, J. E. Moser, M. Graetzel and N.-G. Park, Sci. Rep., 2012, 2, 591 Search PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available: CV, XRD patterns, FESEM images, and transmittance change graphs. See DOI: 10.1039/c6ra03315f |
| This journal is © The Royal Society of Chemistry 2016 |