Dependence of elastic and optical properties on surface terminated groups in two-dimensional MXene monolayers: a first-principles study
Yuelei Baiab,
Kun Zhou*a,
Narasimalu Srikanthc,
John H. L. Panga,
Xiaodong Heb and
Rongguo Wangb
aSchool of Mechanical and Aerospace Engineering, Nanyang Technological University, 50 Nanyang Avenue, Singapore 639798, Singapore. E-mail: kzhou@ntu.edu.sg; Fax: +65 6792 4062; Tel: +65 6790 5499
bNational Key Laboratory of Science and Technology on Advanced Composites in Special Environments and Center for Composite Materials and Structures, Harbin Institute of Technology, Harbin 150080, P. R. China
cEnergy Research Institute @NTU, Nanyang Technological University, 1 CleanTech Loop, Singapore 637141, Singapore
First published on 5th April 2016
Abstract
Density functional theory is used to investigate the elastic and optical properties as well as the crystal and electronic structures of two-dimensional Ti2CT2 and Ti3C2T2 (T = F, O, and OH) MXene monolayers. It is found that the elastic stiffness, optical response, crystal structure and the electronic structure show strong dependence on the surface terminated groups often formed with MXene during the etching process. The elastic stiffness maintains only with the surface termination of O atoms, but a large degradation is present in the surface terminations of F and OH atoms. The low adsorption and reflectivity in the range from infrared to ultraviolet rays account for the high transmittance of Ti3C2T2 that has been experimentally observed, and it is predicted that Ti2CT2 will have higher optical transmittance in this range. The calculations also demonstrate the presence of the optical bandgap in Ti2CO2, which renders its potential applications in optical and electronic devices.
1. Introduction
Since the discovery of graphene,1,2 studies on two-dimensional (2D) systems have been extensively conducted because they possess properties significantly different from those of bulk materials. In 2011, a new class of 2D transition-metal carbides or nitrides, called MXene,3 was fabricated experimentally by etching the A-group layer from the transition-metal ternary layered compounds with the general formula Mn+1AXn (abbreviated to be MAX), where M represents an early transition metal, A is an A-group element (mostly groups 13 and 14), X is C or N, and n = 1, 2 or 3.4 Notably, in most cases the etching process with acids or alkalis results in the fabricated MXene usually with some surface terminated functional groups, such as F, O, and/or OH.5 Therefore, these terminated MXene species should be referred to as a general formula Mn+1XnTx, where T denotes a type of surface group (F, OH and/or O) and x is the number of terminations. However, recently a significant advance has been made in the synthesis of bare 2D MXene. Bare 2D Mo2C with high crystallinity, very low defect concentration and no surface terminations was successfully grown by chemical vapour deposition (CVD),6 which sheds light on the synthesis of other bare MXenes and opens the door to the understanding of their fundamental physical properties.Originating from the parent MAX phases with different M–X slabs (n),7 each bare MXene monolayer could have three, five or seven atomic layers in thickness. In all cases, the thickness of Mn+1XnTx monolayer is less than 1 nm, while the lateral dimension can reach tens of microns.5 This 2D feature of MXenes inspires some researchers to perform following study because in the previous research other 2D materials8 usually exhibit some interesting and unique properties. Since the potential use as the electrochemical energy storage, such as electrode for batteries, supercapacitors, and hybrid devices was revealed by Gogotsi et al.,9 some theoretical and experimental studies have been extensively performed from pure science to potential applications,5,10 such as crystal structure,11–13 electronic structure,14 elastic properties,15 batteries,16–20 supercapacity,21,22 and so on.5,10
Considering the fact that more than 60 MAX phases have been identified and the solid solution of M, A, and/or X sites could be formed among the formers,4 it is reasonably expected that the MXene family should be quite large, and more MXene phases would be exfoliated from the MAX phases or other ternary compounds in future. However, to date only some selected MXenes were experimentally fabricated, e.g. Ti2C,23 V2C,19 Nb2C,19 Mo2C,24 Ti3C2,3 Ta4C3,23 Nb4C3,25 (Ti0.5Nb0.5)2C,23 (V0.5Cr0.5)3C2,23 Ti3CN.23 Among these MXenes, Ti3C2T2 and Ti2CT2 are by far the most studied because they are the earliest identified,3,23 and exhibit many interesting properties that are applicable to others in the following studies,5,10 such as outstanding intercalation properties with very promising applications, including supercapacity21,22 and metal ion batteries.17,18
In the past decades, the rapid development of computer hardware and computational methods has provided strong tools for the structures and properties of materials at the atomic scale, where the first-principles simulations based on density functional theory (DFT) have shown the powerful ability to predict and understand the mechanisms of phase stability, layered structures and unusual properties of the MAX phases.7,26,27 It is also true in the field of MXenes: some important issues have been clarified or explained using DFT simulations. For instance, DFT simulations play a crucial role in the structures, properties and potential applications of MXenes.5 In fact, the first identified multilayer MXene structure—named terminated Ti3C2T2 layers—was proposed with the support of a DFT modeling,3 which also helps researchers get some more insights into MXenes as metal ion batteries and other applications,13,16,18,20,28 and clarifies why the bare and terminated Ti3C2 MXenes are electronic conductor.14,16
It is well established that the surface termination on 2D materials may drastically change the electronic and accompanying properties. For instance, alternately hydrogenated and hence fully sp3-bonded graphene, so-called graphene, results in a fundamental gap at least up to 5.4 eV.29 The similar case is also present in the recently discovered MXenes: their electronic14 and magnetic30 properties as well as the potential applications for metal ion batteries16–20 and supercapacity21,22 are affected greatly by the surface terminations with different functional groups. However, to date there are few reports concerning the effect of surface terminated groups on the elastic and optical properties of MXenes. Considering the fact that the derived MXenes are usually terminated with some surface functional groups like F, O, and OH, it is necessary and timely to perform a systematic study for the dependence of the elastic and optical properties on terminated groups.
Recently Halim et al.31 reported that about 90% of the light in the infrared-to-visible range was transmitted in the epitaxial Ti3C2Tx film while it exhibited metallic conductivity down to ∼100 K, which indicates that MXenes are promising materials for transparent conductive electrode, sensors and so on. To get some more theoretical insights into this issue, the present work is to investigate the optical properties of two typical MXene Ti3C2 and Ti2C using DFT, emphasizing the effect of surface functional groups. In addition, the elastic stiffness is also studied. The results show that the optical properties and elastic stiffness of Ti3C2T2 MXenes are both highly dependent on the surface terminated groups, and contribute to understanding the experimentally observed high transmittance of Ti3C2T2 MXenes from infrared to ultraviolet rays, and to predicting the higher transmittance in thinner Ti2CT2. The calculations also demonstrate that the optical bandgap is present in Ti2CO2, which renders it as promising candidate materials in applications such as the optical and electronic devices. These results further indicate that the optical and other properties of Ti2CO2 based materials can be tunable by approaches including doping and solid solution.
2. Computational details
All the first-principles calculations are performed using Vienna Ab-initio Simulation Package (VASP) based on density functional theory (DFT) where a plane-wave pseudopotential total energy calculation method is employed.32 The plane-wave cutoff energy is always set as 520 eV, which is tested to ensure the convergence of total energy within 1 meV per atom. Ion-electron interactions are represented by the all-electron projector-augmented wave (PAW) method,33 with 1s1, 2s22p5, 2s22p2, 2s22p4, and 3d24 s2 treated as valence electrons for H, F, O, C, and Ti, respectively. In addition, the semi-core 3s23p6 states are treated as valence for Ti elements. The electronic exchange–correlation energy (xc) is treated according to the Perdue–Burke–Ernzerhof generalized gradient approximations (GGA–PBE)34 and hybrid Heyd–Scuseria–Ernzerhof (HSE06) hybrid functional.35,36 The latter is constructed by replacing 25% short-range part of PBE exchange energy (EPBE,SRx) using a short-range Hartree–Fock exchange term (EHF,SRx), which can be expressed as follows:| EHSExc = 0.25EHF,SRx(μ) + 0.75EPBE,SRx(μ) + EPBE,LRx(μ) + EPBEc(μ) | (1) |
The full geometry optimization including the lattice constants and atomic internal coordinates is conducted using the conjugate gradient algorithm and 12 × 12 × 1 Monkhorst–Pack k-point grid which is tested to ensure the total energy converged within 1 meV per atom, with the convergence criteria set such that the difference in total energy is within 1 × 10−6 eV per cell and the force is less than 0.005 eV Å−1. A vacuum space over 20 Å is added between MXene monolayer and its periodically repeated images along c-axis direction of the periodic structure to avoid any interaction. For the density of states (DOS) and dielectric function of the optimized structure, a much denser k-point mesh of 20 × 20 × 1 is used. For the Brillouin zone integration, in most cases the first-order Methfessel–Paxton and Gaussian smearing with a width of 0.1 eV are employed for metal and semiconductor (Ti2CO2), respectively, where the max entropy term less than 1 meV per atom is achieved. However, the tetrahedron method with Blöchl corrections is employed when calculating DOS.
The elastic tensor is determined from strain–stress relationship by performing six finite distortions of lattice and deriving the lattice constants.37 The elastic tensor is estimated both, for rigid ions, as well, as allowing for relaxation of the ions. The ionic contributions are determined by inverting the ionic Hessian matrix, which is a matrix of the second derivatives of the energy with respect to the atomic positions and here calculated using finite difference, and multiplying with the internal strain tensor.38 The convergence criteria for this part is set such that the difference in total energy is within 1 × 10−6 eV per cell, and the force is less than 0.001 eV Å−1.
The optical properties of a medium can be fully described by the dielectric function, ε(ω) = ε1(ω) + iε2(ω) at all photon energy ω. After the electronic ground state has been available, the dielectric function is estimated in the momentum representation that needs the matrix elements between occupied and unoccupied electronic eigenstates. Specifically, the imaginary part of the dielectric function is determined by a summation over empty states using the following equation:
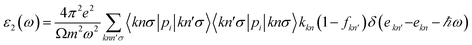 | (2) |
3. Results and discussions
3.1. Configuration and structural properties
The 2D Ti2CT2 and Ti3C2T2 MXene monolayers are constructed appropriately by removing Al atoms from their parent MAX phases, i.e. Ti2AlC and Ti3AlC2.4 With the lowest energy that has been revealed in the previous work,11,14,16 the stable configurations of Ti2CT2 and Ti3C2T2 monolayers are illustrated in Fig. 1, where all the functional groups such as F, O, and OH are located above the hollow site of three neighboring C atoms, and pointed to the Ti atoms in the second Ti atomic layer on both sides of the MXenes.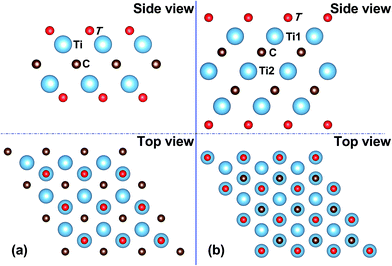 | ||
| Fig. 1 Side and top views of crystal structures of a 2D Mxene monolayer: (a) Ti2CT2 and (b) Ti3C2T2. | ||
The in-plane lattice constant a, atomic distance (bond length) d, and thickness of monolayer L (the distance perpendicular to surface between the topmost and bottommost atomic layers) are listed in Table 1, including other theoretical values for comparison. It can be seen that the present data are well consistent with previous ones.14,15 Comparing with the corresponding MAX (Ti2AlC and Ti3AlC2, Table 1),14 removing Al atoms results in the shrink of a. Moreover, it increases with increasing Ti–C slabs in bare and terminated Ti2C and Ti3C2 MXenes, e.g. 3.0345 Å in Ti2C and 3.0994 Å in Ti3C2. Upon termination, a increases in Ti2CT2, but decreases in Ti3C2T2. In addition, the O terminated Ti2CT2 and Ti3C2T2 have the lowest value of a, while a significantly higher value is present in the F and OH terminated ones.
| a (Å) | L (Å) | dTi(1)–C (Å) | dTi(2)–C (Å) | dTi(1)–T (Å) | Ef (eV) | c11 (GPa) | c44 (GPa) | Comments | |
|---|---|---|---|---|---|---|---|---|---|
| Ti2C | 3.0345 | 2.3046 | 2.0969 | −1.5915 | 609 | 227 | |||
| 3.007 | 2.291 | 636 | CASTEP/Wu-Cohen15 | ||||||
| Ti2CF2 | 3.0595 | 4.7789 | 2.1013 | 2.1647 | −1.4908 | 391 | 136 | ||
| 3.0583 | 4.80 | 210 | 2.16 | VASP/PBE14 | |||||
| Ti2CO2 | 3.0329 | 4.4305 | 2.1850 | 1.9726 | −2.3046 | 607 | 212 | ||
| 3.0329 | 4.45 | 2.19 | 1.98 | VASP/PBE14 | |||||
| Ti2C(OH)2 | 3.0717 | 6.7840 | 2.1141 | 2.1791 | 0.7886 | 285 | 111 | ||
| 3.0712 | 6.79 | 2.12 | 2.18 | VASP/PBE14 | |||||
| Ti3C2 | 3.0994 | 4.6320 | 2.0496 | 2.2216 | −0.4446 | 491 | 206 | ||
| 3.071 | 4.605 | 523 | CASTEP/Wu-Cohen15 | ||||||
| Ti3C2F2 | 3.0777 | 7.2160 | 2.0734 | 2.1936 | 2.1744 | −0.4202 | 436 | 166 | |
| 3.0775 | 7.21 | 2.08 | 2.17 | CASTEP/Wu-Cohen15 | |||||
| Ti3C2O2 | 3.0393 | 6.9592 | 2.1932 | 2.1582 | 1.9755 | −1.3124 | 523 | 191 | |
| Ti3C2(OH)2 | 3.0874 | 9.2170 | 2.0848 | 2.1949 | 2.1904 | 1.5409 | 341 | 137 | |
| Ti2AlC | 3.0687 | VASP/PBE14 | |||||||
| Ti3AlC2 | 3.0816 | VASP/PBE14 |
The terminated MXenes always have higher Ti(1)–C bond length than bare ones, implying that the interaction between terminated group T and surface Ti atoms results in the latter away from the near C atoms. For both Ti2C and Ti3C2, the shortest Ti(1)–T and Ti(2)–C bonds (applicable in Ti3C2), and longest Ti(1)–C bonds are always present in O terminated MXenes, indicating the strong interaction between O termination and surface Ti atoms. To the contrast, the weak interaction could be in the F and OH terminated MXenes that is evidenced in the larger Ti–T bond length. Moreover, for bare and functionalized Ti3C2 except Ti3C2O2, Ti(2)–C bonds are much longer than Ti(1)–C bonds, which is common for their parent MAX phases.7 However, the opposite is present in Ti3C2O2, once again which shows the strong interaction between O termination and surface Ti atoms.
3.2. Electronic structure and chemical bonding
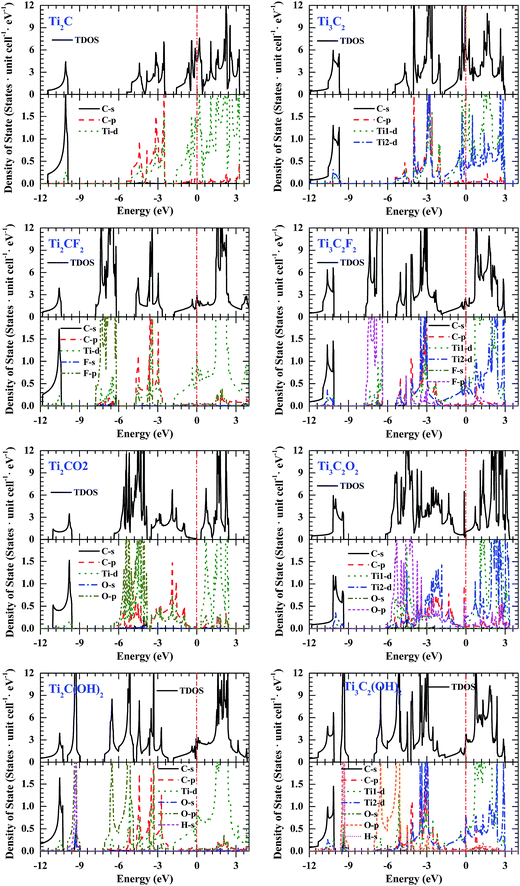
The total density of state (TDOS) of bare and terminated Ti2CT2 and Ti3C2T2 MXene monolayers are shown in Fig. 2. Of most importance, except Ti2CO2 there is a finite DOS value around the Fermi energy Ef in all Ti2CT2 and Ti3C2T2 monolayers although it varies with changing functionalized groups, which indicates that they are all electronic conductors. This is consistent with the previous theoretical14 and experimental results.3,23 It should be noted that most of MAX phases (the parent of MXene) also exhibit the electronic conduction behavior4 because of their metal-like electronic structure.7 It is shown that Ti2CO2 has a narrow bandgap that is formed by decreasing Ef (Table 1) upon O termination, indicating it is semiconductor and could be used for some electronic and optical devices.More insights into the electronic structure and chemical bonding can be gleaned from their partial density of state (PDOS), as shown in Fig. 2. Clearly, the TDOS at Ef are mainly attributed to Ti-d electrons and therefore they are involved in the conduction properties although d electrons are usually considered not to be efficient conductors, as similar with MAX phases.7 The lowest valence bands from −12 eV to −10 eV are formed by the C-s states with a small mixture of Ti-p + d states. The higher valance bands from −5.5 eV to −2.5 eV in bare Ti2C and Ti3C2 monolayer correspond to a strong hybridization of T-d and C-p states (Ti–C bond). However, surface terminations using F, O, or OH group result in a diverge of these bands: the hybridization between p electrons of the terminated atoms and d electrons of surface Ti atoms makes some bands move into lower energy range, which corresponds to the Ti-T bond; the hybridization states between Ti-d and C-p states shift slightly to higher energy range. In the OH terminated MXenes, the peak around −10 eV corresponds to the hybridization between H-s and O-p states with the minor mixture of O-s states (H–O bond). Although the decreasing Ef (Table 1) upon O termination is also present in functionalized Ti3C2 MXenes, no bandgap is observed between two related bands mainly because of the widening of Ti-d states, which is related to the fact that all Ti3C2 MXenes are electronic conductors. In addition, in Ti2CO2 and Ti3C2O2 some hybridization states between Ti-d and O-p electrons with a mixture of minor O-s electrons are present in the much lower energy range around −18.5 eV (not shown in figure), which indicates the strong coupling between surface terminated O atoms and bare MXenes, and contributes to the lowest Ef for Ti2CO2 (−2.3046 eV) and Ti3C2O2 (−1.3124 eV).
3.3. Elastic stiffness
Consistent with the previous work on MXenes by Kurtoglu et al.,15 the elastic constants in-plane cij are calculated using following expression:| cij = cij,cell(c/r) | (3) |
The calculated elastic constants c11 and c44 of Ti2CT2 and Ti3C2T2 monolayers are listed in Table 1. They are well consistent with the previous theoretical values of bare Ti2C and Ti3C2 monolayers by other researchers.15 Of most importance, cij strongly depends on the terminated functional groups: cij of the bare and O terminated MXenes have very high stiffness (over 490 GPa of c11 and around 200 GPa of c44) probably because of the underestimation of the r values and elimination of the A layers that is also evidenced in the shrink of in-plane lattice constant,15 but surface termination using other functional groups results in a decrease of elastic stiffness, where the lowest values are present in OH terminated ones, and the degradation of elastic stiffness is more in Ti2CT2 than Ti3C2T2. Although cij of the bare and O terminated MXenes decreases with increasing Ti–C slabs, an opposite trend is observed in the F and OH terminated ones.
Due to the similar bond length of Ti–C in the terminated MXenes, it is reasonable that the Ti–C bond strength should not change a lot with the termination of functional group. It follows that the key factor for elastic stiffness is the bonding strength between the bare MXenes and terminated groups. As shown in Table 1, the much larger change of Ti–C bond length in the O terminated Ti2CT2 and Ti3C2T2 indicates the strongest coupling between O atoms and bare MXenes, which is also supported by examining PDOS of Ti2CO2 and Ti3C2O2 (Fig. 2). Although the change of Ti–C bond length is comparable in F and OH terminations, the associated increase of layer thickness (Table 1) is much less in the former than later, which results in the lowest elastic stiffness of OH terminated MXene monolayers.
3.4. Dielectric function and optical properties
To investigate the optical properties of MXenes, we have calculated the imaginary part (ε2) of the dielectric function against photon frequency along in-plane and out-plane directions, as displayed in Fig. 3. Of most importance, the optical bandgap is present only in Ti2CO2 because of its semiconductor-like electronic structure in origin (Fig. 2). Notably, the present optical bandgap, around 1 eV, is much higher than the bandgap from DOS in Fig. 2 (around 0.3 eV). The reason is that on the one hand, Ti2CO2 has an indirect bandgap where interband transition needs lager energy because of the different crystal momentum between minimal-energy state in the conduction band and the maximal-energy state of the valence band; one the other hand, PBE usually underestimates the bandgap than the more accurate HSE06 that is used in the calculation of dielectric function.14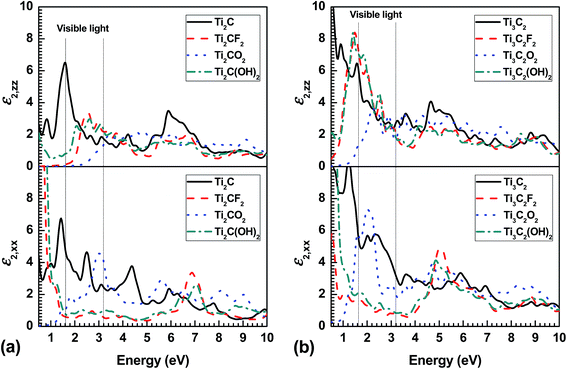 | ||
| Fig. 3 Imaginary part of the in-plane (ε2,xx) and out-plane (ε2,zz) dielectric function as a function of photon frequency for (a) functionalized Ti2CT2 and (b) Ti3C2T2 monolayer. | ||
Similar with electronic structure and elastic constants, ε2 is strongly dependent on the terminated surface groups. And due to the anisotropic crystal structure, the corresponding anisotropic dielectric function is observed that ε2 is different along in-plane (ε2,xx) and out-plane (ε2,zz) directions, especially in the low-energy region. Very interestingly, a optical bandgap is present in the out-plane direction (ε2,zz) even in the metal-like Ti2CF2 and Ti3C2O2, while for Ti2CO2 it is also widen to 2.3 eV, which is attributed to their anisotropic electronic structure and chemical bonding (valence bonds). In addition, ε2 in the F terminated MXenes exhibits similar behavior with OH terminated ones, which originates from their similar electronic structure (TDOS, Fig. 2). Moreover, ε2,xx in the O terminated MXene is very low below around 1 eV, and increases quickly with increasing photon frequency, peaking at a frequency (2–4 eV), while upon other terminations a much higher ε2,xx value is present in the low-frequency range. Notably, ε2,xx is much lower in F and OH terminated MXenes than the others in a narrow range for Ti2CT2 (1.4–6.5 eV) and Ti3C2T2 (1.4–4.5 eV), while it is not true for ε2,zz.
It should be noted that the interband transition that is treated in the DFT simulation is much less important than intraband transition in the low energy (<1 eV) for metal. The latter is the dominant mechanism in low energy of metal and metal-like materials, and should be expressed using empirical Drude term, which can be written as the dielectric function:
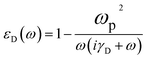 | (4) |
In practice, the experimentally obtained optical properties are usually the adsorption coefficients α and refractivity R. Therefore, the in-plane and out-plane α and R are estimated from the calculated dielectric function as a function of frequency, as illustrated in Fig. 4 and 5, respectively. Considering the anisotropic dielectric function, it is not surprising to obtain anisotropic α and R in the bare and terminated Ti2CT2 and Ti3C2T2. As a semiconductor, Ti2CO2 almost can not adsorb any light below about 1 eV in plane and 2.3 eV out plane, where it reflects only about 12% and 6% light, respectively. The zero adsorption and low refractivity are also present in out-plane direction of Ti2CF2 and Ti3C2O2. In the range from near-infrared to ultraviolet [Ti2CT2 (1.4–6.5 eV) and Ti3C2T2 (1.4–4.5 eV)] including visible light, the in-plane adsorption coefficients αxx is significantly lower in the F and OH terminated MXenes than the bare and O terminated ones, of more importance, with the low refractivity, where the αxx and Rxx both change very slightly with increasing frequency. This indicates that the pure F and OH terminated Ti2CT2 and Ti3C2T2 should exhibit white colour. Moreover, overall Ti3C2T2 has the higher Rxx value than Ti2CT2, which shows that increasing Ti–C slabs or thickness of monolayer can result in the increase of Rxx. It should be noted that due to the metal-like electronic structure, their parent MAX phases always have relative strong adsorption without optical bandgap compared with MXenes,27,39 while R is much higher in the former than the latter especially in the low energy range. In other words, MAX phases are always lighttight, which has been confirmed in previous experiments,40 but in some MXenes like Ti2CO2 the light can be transmitted.
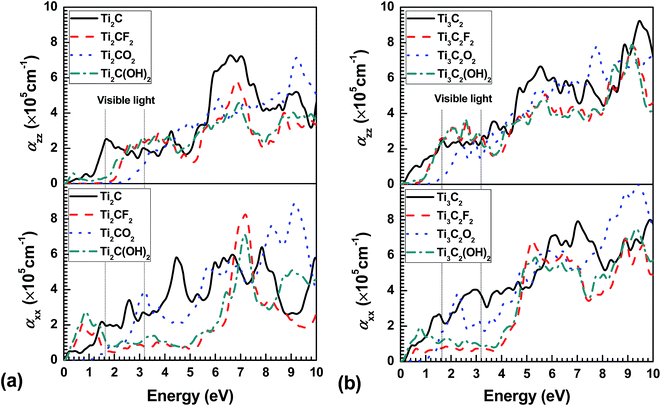 | ||
| Fig. 4 In-plane (αxx) and out-plane (αzz) adsorption coefficient as a function of photon frequency for (a) functionalized Ti2CT2 and (b) Ti3C2T2 monolayer. | ||
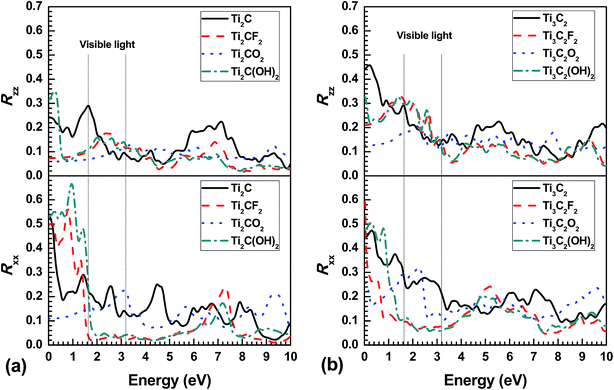 | ||
| Fig. 5 In-plane (Rxx) and out-plane (Rzz) reflectivity as a function of photon frequency for (a) functionalized Ti2CT2 and (b) Ti3C2T2 monolayer. | ||
In the previous work, the recent experimental research on the optical properties of Ti3C2T2 (ref. 31) shows that more than 90% light could be transmitted in a single MXene layer, with the slight change against photon energy. It is well known that light is electromagnetic wave where the electric and magnetic fields of lights are both perpendicular to each other and perpendicular to the direction of energy and wave propagation. This means that the photos react with MXenes in plane when a light shines perpendicularly on it, as in ref. 31. And the recent experimental result41 indicates that MXenes have a surface termination layer that is a mixture of OH and F mainly. Clearly, the low αxx and Rxx of F and OH terminated Ti3C2T2 (Fig. 4 and 5) monolayers are the reason why the high transmittance at over 90% from infrared to ultraviolet rays is observed in the experiment. Moreover, based on the prediction that the much lower Rxx and monolayer thickness are present in F and OH terminated Ti2CT2 (2–4%) than Ti3C2T2 (6–10%) (Fig. 5), it is reasonable to assume that the former should have higher optical transmittance.
Notably, as a semiconductor, Ti2CO2 has very different electronic structure and optical properties, which renders it for applications in the electronic and optical devices. It is inspired that the pure Ti2CO2 would be synthesized and optically characterized in future. In addition, for these potential applications the electronic structure and optical properties of Ti2CO2-based materials can be further tuned by doping or solid solution.
4. Conclusions
The bare and terminated Ti2CT2 and Ti3C2T2 (T = F, O, and OH) MXene monolayers are investigated using first principle calculations, emphasizing the elastic and optical properties with their dependence of surface terminated groups. The surface terminated groups have a significant effect on the crystal structure, electronic structure, chemical bonding, elastic properties, and optical properties. Surface termination results in the shrink in Ti2CT2 and expansion in Ti3C2T2 of the in-plane lattice constant, where the largest change is present in O terminated ones. Correspondingly, the elastic stiffness maintains only in Ti2CO2 and Ti3C2O2 that is also evidenced from their electronic structure: some hybridization states between Ti and O atoms are in a much lower energy range around −18.5 eV, but this is not true with the other terminations. The anisotropic optical properties using hybrid functional HSE06 against photon frequency, including dielectric function, adsorption coefficients and reflectivity, strongly depend on surface terminated groups. The optical bandgap is present only in Ti2CO2, originating from its semiconductor-like electronic structure, which renders its potential applications in optical and electronic devices. Moreover, for these potential applications the optical and other properties of Ti2CO2-based materials can be further tunable by the approaches including doping and solid solution. With the surface termination of F and OH functional groups, the low adsorption and reflectivity from infrared to ultraviolet rays account for the high transmittance of Ti3C2T2 that is observed in experiments, and it is predicted that the thinner Ti2CT2 monolayer should have higher optical transmittance.Acknowledgements
The authors acknowledge financial support from Ministry of Education, Singapore (Academic Research Fund TIER 1 – RG128/14). YB gratefully acknowledges the support from National Natural Science Foundation of China (Grant No. 11302061) and International Postdoctoral Exchange Fellowship Program by the Office of China Postdoctoral Council (Grant No. 20130004).References
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, Y. Zhang, S. V. Dubonos, I. V. Grigorieva and A. A. Firsov, Science, 2004, 306, 666–669 CrossRef CAS PubMed.
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, M. I. Katsnelson, I. V. Grigorieva, S. V. Dubonos and A. A. Firsov, Nature, 2005, 438, 197–200 CrossRef CAS PubMed.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Adv. Mater., 2011, 23, 4248–4253 CrossRef CAS PubMed.
- M. W. Barsoum, Prog. Solid State Chem., 2000, 28, 201–281 CrossRef CAS.
- M. Naguib, V. N. Mochalin, M. W. Barsoum and Y. Gogotsi, Adv. Mater., 2014, 26, 992–1005 CrossRef CAS PubMed.
- C. Xu, L. Wang, Z. Liu, L. Chen, J. Guo, N. Kang, X.-L. Ma, H.-M. Cheng and W. Ren, Nat. Mater., 2015, 14, 1135–1141 CrossRef CAS PubMed.
- Y. L. Bai, X. D. He, R. G. Wang, S. Wang and F. Y. Kong, Comput. Mater. Sci., 2014, 91, 28–37 CrossRef CAS.
- V. Nicolosi, M. Chhowalla, M. G. Kanatzidis, M. S. Strano and J. N. Coleman, Science, 2013, 340, 1420 CrossRef CAS.
- M. R. Lukatskaya, O. Mashtalir, C. E. Ren, Y. Dall'Agnese, P. Rozier, P. L. Taberna, M. Naguib, P. Simon, M. W. Barsoum and Y. Gogotsi, Science, 2013, 341, 1502–1505 CrossRef CAS PubMed.
- J. C. Lei, X. Zhang and Z. Zhou, Front. Phys China, 2015, 10, 276–286 Search PubMed.
- A. N. Enyashin and A. L. Ivanovskii, Comput. Theor. Chem., 2012, 989, 27–32 CrossRef CAS.
- C. Y. Shi, M. Beidaghi, M. Naguib, O. Mashtalir, Y. Gogotsi and S. J. L. Billinge, Phys. Rev. Lett., 2014, 112, 5 Search PubMed.
- X. F. Wang, X. Shen, Y. R. Gao, Z. X. Wang, R. C. Yu and L. Q. Chen, J. Am. Chem. Soc., 2015, 137, 2715–2721 CrossRef CAS PubMed.
- Y. Xie and P. R. C. Kent, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 87 Search PubMed.
- M. Kurtoglu, M. Naguib, Y. Gogotsi and M. W. Barsoum, MRS Commun., 2012, 2, 133–137 CrossRef CAS.
- Q. Tang, Z. Zhou and P. W. Shen, J. Am. Chem. Soc., 2012, 134, 16909–16916 CrossRef CAS PubMed.
- O. Mashtalir, M. Naguib, V. N. Mochalin, Y. Dall'Agnese, M. Heon, M. W. Barsoum and Y. Gogotsi, Nat. Commun., 2013, 4 Search PubMed.
- Y. Xie, M. Naguib, V. N. Mochalin, M. W. Barsoum, Y. Gogotsi, X. Q. Yu, K. W. Nam, X. Q. Yang, A. I. Kolesnikov and P. R. C. Kent, J. Am. Chem. Soc., 2014, 136, 6385–6394 CrossRef CAS PubMed.
- M. Naguib, J. Halim, J. Lu, K. M. Cook, L. Hultman, Y. Gogotsi and M. W. Barsoum, J. Am. Chem. Soc., 2013, 135, 15966–15969 CrossRef CAS PubMed.
- C. Eames and M. S. Islam, J. Am. Chem. Soc., 2014, 136, 16270–16276 CrossRef CAS PubMed.
- M. Ghidiu, M. R. Lukatskaya, M. Q. Zhao, Y. Gogotsi and M. W. Barsoum, Nature, 2014, 516, 78–U171 CAS.
- Y. Dall'Agnese, M. R. Lukatskaya, K. M. Cook, P. L. Taberna, Y. Gogotsi and P. Simon, Electrochem. Commun., 2014, 48, 118–122 CrossRef.
- M. Naguib, O. Mashtalir, J. Carle, V. Presser, J. Lu, L. Hultman, Y. Gogotsi and M. W. Barsoum, ACS Nano, 2012, 6, 1322–1331 CrossRef CAS PubMed.
- R. Meshkian, L. A. Naslund, J. Halim, J. Lu, M. W. Barsoum and J. Rosen, Scr. Mater., 2015, 108, 147–150 CrossRef CAS.
- M. Ghidiu, M. Naguib, C. Shi, O. Mashtalir, L. M. Pan, B. Zhang, J. Yang, Y. Gogotsi, S. J. L. Billinge and M. W. Barsoum, Chem. Commun., 2014, 50, 9517–9520 RSC.
- X. D. He, Y. L. Bai, C. C. Zhu and M. W. Barsoum, Acta Mater., 2011, 59, 5523–5533 CrossRef CAS.
- X. D. He, Y. L. Bai, Y. Chen, C. C. Zhu, M. W. Li and M. W. Barsoum, J. Am. Ceram. Soc., 2011, 94, 3907–3914 CrossRef CAS.
- Q. K. Hu, D. D. Sun, Q. H. Wu, H. Y. Wang, L. B. Wang, B. Z. Liu, A. G. Zhou and J. L. He, J. Phys. Chem. A, 2013, 117, 14253–14260 CrossRef CAS PubMed.
- P. Cudazzo, C. Attaccalite, I. V. Tokatly and A. Rubio, Phys. Rev. Lett., 2010, 104, 4 CrossRef PubMed.
- M. Khazaei, M. Arai, T. Sasaki, C. Y. Chung, N. S. Venkataramanan, M. Estili, Y. Sakka and Y. Kawazoe, Adv. Funct. Mater., 2013, 23, 2185–2192 CrossRef CAS.
- J. Halim, M. R. Lukatskaya, K. M. Cook, J. Lu, C. R. Smith, L. A. Naslund, S. J. May, L. Hultman, Y. Gogotsi, P. Eklund and M. W. Barsoum, Chem. Mater., 2014, 26, 2374–2381 CrossRef CAS PubMed.
- G. Kresse and J. Furthmuller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS.
- P. E. Blochl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- J. Heyd, G. E. Scuseria and M. Ernzerhof, J. Chem. Phys., 2003, 118, 8207–8215 CrossRef CAS.
- J. Paier, M. Marsman, K. Hummer, G. Kresse, I. C. Gerber and J. G. Angyan, J. Chem. Phys., 2006, 124, 13 CrossRef PubMed.
- Y. Le Page and P. Saxe, Phys. Rev. B: Condens. Matter Mater. Phys., 2002, 65, 14 CrossRef.
- X. Wu, D. Vanderbilt and D. R. Hamann, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 72, 13 Search PubMed.
- S. Li, R. Ahuja, M. W. Barsoum, P. Jena and B. Johansson, Appl. Phys. Lett., 2008, 92, 221907 CrossRef.
- M. W. Barsoum, Electronic, Optical, and Magnetic Properties, in MAX Phases, Wiley-VCH Verlag GmbH & Co. KGaA, 2013, pp. 155–185 Search PubMed.
- K. J. Harris, M. Bugnet, M. Naguib, M. W. Barsoum and G. R. Goward, J. Phys. Chem. C, 2015, 119, 13713–13720 CAS.
| This journal is © The Royal Society of Chemistry 2016 |