Control of magneto-transport characteristics of Co-doped ZnO by electron beam irradiation
Seunghun Leea,
Ji Hun Parkb,
Won-Kyung Kimb,
Hyung Dal Parkc,
Byung Cheol Leec,
Chikako Moriyoshid,
Yoshihiro Kuroiwad,
Geun Woo Leee,
Yong Chan Cho*e and
Se-Young Jeong*b
aDepartment of Materials Science and Engineering, University of Maryland, College Park, MD 20742, USA
bDepartment of Cogno-Mechatronics Engineering, Pusan National University, Miryang, 627-706, Republic of Korea. E-mail: syjeong@pusan.ac.kr; Fax: +82-55-353-1314; Tel: +82-55-350-5273
cRadiation Instrumentation Research Division, Korea Atomic Energy Research Institute (KAERI), Daejeon, 305-353, Republic of Korea
dDepartment of Physical Science, Hiroshima University, Higashihiroshima 739-8526, Japan
eFrontier in Extreme Physics, Korea Research Institute of Standards and Science, Daejeon 305-340, Republic of Korea. E-mail: yongchanjo@gmail.com
First published on 20th April 2016
Abstract
Electron beam irradiation can be used to modify the physical properties of materials, but its effects have never been studied in Co-doped ZnO (ZnCoO), a promising magnetic semiconductor material. Here, we demonstrate that electron beam irradiation enables modification of the magneto-transport properties of ZnCoO. The electron beam irradiation cannot affect the electrical and magnetic properties of ZnCoO significantly, due to the insulating nature of pristine ZnCoO showing paramagnetic behavior. However, intentional hydrogen doping increases carrier concentration and induces ferromagnetism in ZnCoO, which allows ZnCoO to be affected by electron beam irradiation. As a consequence of s–d exchange interaction, hydrogen-doped ZnCoO shows positive magnetoresistance and anomalous Hall conductivity. Electron beam irradiation reduced the carrier concentration in hydrogen-doped ZnCoO by removing shallow donor-type hydrogen and consequently increasing s–d exchange interaction, which resulted in an increase in positive magnetoresistance and a decrease in the anomalous Hall conductivity. These findings demonstrate the novel applicability of electron beam irradiation for tailoring the physical properties of ZnO-based materials.
Introduction
ZnO, which has a wide band gap and high exciton binding energy, has been extensively studied in the context of the development of optoelectronic applications.1 These applications, which include LEDs, photo-detectors, and solar cells, are facilitated by the fact that the physical properties of ZnO can be customized by doping. For example, aluminum (Al) or gallium (Ga) dopants act as donors in ZnO system and increase the concentration of electron carriers, which is useful in the development of transparent electrodes.2 Transition-metal dopants such as cobalt (Co) or manganese (Mn) have been proposed to develop ZnO-based magnetic semiconductor.3,4 In addition to dopants, crystalline defects significantly influence the physical properties of ZnO; hydrogen, oxygen vacancies and zinc interstitial are known to be stable, naturally occurring defects of ZnO.4–12 These may increase the charge carrier concentration or optical absorption in the visible range. Such defects have also been considered necessary for ferromagnetism in transition metal-doped ZnO. Therefore, the development of sophisticated methods to control defects and hence the characteristics of ZnO have been an intriguing research topic. These methods typically make use of thermal annealing processes or plasma treatments.13–20Recently, electron beam (e-beam) irradiation has started to be used for defect and nano-structural engineering of advanced condensed matter.21–25 In general, e-beam irradiation has been employed at localized positions where specific thermal, mechanical and irradiation effects are required. It has also been used in various industrial applications, such as lithography and surface modification. Recently, e-beam irradiation has been used to create or cure defects in carbon-based materials.22,24,25 This enables to control their mechanical, electrical, and magnetic properties. Such efforts have been made for other material systems as well, including ZnO-based materials,26–28 but no one has reported the effect of e-beam irradiation on transition metal-doped ZnO systems. If specific defect is strongly correlated with the magnetic properties of transition metal-doped ZnO systems, and the e-beam irradiation stimulates such defects in this system, it would enable the control of magnetism-related properties of transition metal-doped ZnO.
In this study, we investigate the effect of e-beam irradiation on the physical properties of Co-doped ZnO (ZnCoO) and demonstrate that the magneto-transport characteristics of ZnCoO can be controlled by e-beam irradiation. This is due to the removal of shallow donor-type hydrogen impurities (Fig. 1). We show that e-beam irradiation can create oxygen vacancy defects in ZnCoO but cannot induce any significant change in the electrical and magnetic properties of highly resistive and paramagnetic ZnCoO. In contrast, in the case of conductive and ferromagnetic ZnCoO prepared by post hydrogen treatment, we observed that e-beam irradiation reduces the charge carrier concentration attributed to hydrogen impurities and increases magnetic scattering effects. This results in an increase in the positive magnetoresistance and a decrease in the anomalous Hall conductivity.
 | ||
| Fig. 1 Conceptual image of the effect of e-beam irradiation on ZnCoO. We demonstrate that e-beam irradiation can be used to remove shallow donor type hydrogen located in Zn(Co)–O bonding centers. | ||
Results and discussion
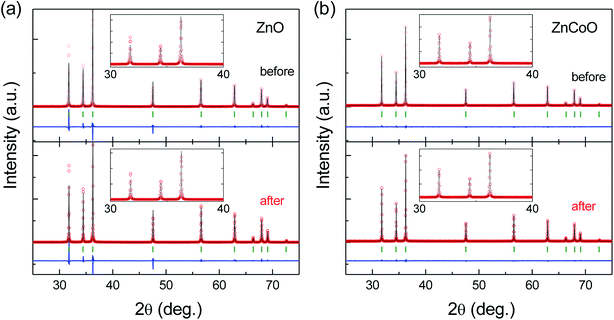
To investigate the effect of e-beam irradiation on the structural properties of ZnCoO, we prepared ZnCoO powder samples for precise structural analysis using Rietveld refinement and maximum entropy method (MEM). Further MEM analysis enables us to reduce errors in the Rietveld refinement analysis and thus obtain more reliable structural parameters.29–32 We also used ZnO sample as a reference sample to see the effect of e-beam irradiation depending on Co dopant. The details for the sample fabrication are described in the Experimental section. Fig. 2(a) shows experimental powder XRD patterns, and simulated patterns (using Rietveld refinement and MEM) for ZnO before and after e-beam irradiation where the energy and dose of e-beam were fixed to be 0.2 MeV and 1.8 × 1017 electron per cm2, respectively. We summarized the structural parameters obtained from the Rietveld refinement and MEM analysis in Table 1. Reliability factors based on Bragg intensities, RIs, of both ZnO samples before and after the e-beam irradiation were 1.209% and 1.123%, respectively, which indicates that the analysis is reliable. All peaks are consistent with the diffraction peaks corresponding to the ZnO wurtzite single phase, which indicates that the e-beam irradiation with 0.2 MeV does not create any impurity phases such as zinc blende-structured ZnO or secondary phases such as Zn metal, within the resolution limit. To find out whether e-beam irradiation induces crystal defects, we carried out occupancy fitting of Zn and O atoms, from which variations in the occupancy of Zn and O atoms could be determined. It was found that the occupancy of O atoms did not vary significantly after the e-beam irradiation, while that of Zn atoms was reduced within a measurable range: 97.174% before the e-beam irradiation and 96.407% after the e-beam irradiation. This implies that Zn vacancies were created in the ZnO powder by the e-beam irradiation.| E-beam irradiation | ZnO | ZnCoO | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| RI (%) | 1.209 | 1.123 | 1.630 | 1.189 |
| RF (%) | 0.829 | 0.722 | 1.050 | 0.841 |
| a (Å) | 3.25023 | 3.25014 | 3.25298 | 3.25273 |
| c (Å) | 5.20310 | 5.20310 | 5.20253 | 5.20197 |
| Zn(O) occupancy | 97.174 | 96.407 | (95.091) | (93.677) |
Fig. 2(b) shows XRD patterns and simulated patterns for ZnCoO powders before and after the e-beam irradiation with energy 0.2 MeV and dose 1.8 × 1017 electron per cm2. The RIs of ZnCoO before and after the e-beam irradiation were 1.630% and 1.189%, respectively. These values also suggest high reliability of this analysis. Any peaks corresponding to secondary phases were not observed within the detection limit, indicating that the e-beam irradiation also does not induce any creation of secondary species in ZnCoO. We also performed occupancy fitting for the ZnCoO powders and these data are represented in Table 1. In contrast to the results obtained for ZnO, the occupancy of oxygen was drastically reduced after the e-beam irradiation (95.091% before e-beam irradiation and 93.677% afterwards). This result indicates that e-beam irradiation can create oxygen vacancies in ZnCoO systems, and that the effect of e-beam irradiation may appear to be different depending on a kind of dopant.
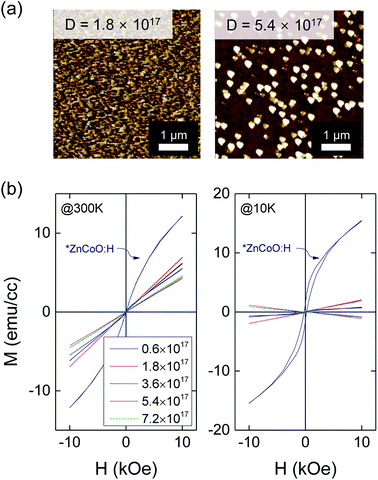
To study the effect of e-beam irradiation on the electrical and magnetic properties of ZnCoO, we prepared ZnCoO thin films using the sputtering method (the details are described in the Experimental section). We then performed e-beam irradiation varying doses of the irradiation for which the energy of e-beam irradiation was fixed to 0.2 MeV and the duration time was varied (doses: 0.6 to 7.2 × 1017 electron per cm2). It is not shown here but all ZnCoO films had strong diffraction peaks, corresponding to ZnO (002), indicating their c-axis oriented structure. Fig. 3(a) shows atomic force microscopy (AFM) images of ZnCoO thin films after the e-beam irradiation with the doses of 1.8 × 1017 and 5.4 × 1017 electron per cm2. At the highest irradiation dose (7.2 × 1017 electron per cm2), surface damage such as aggregation and clustering was observed. The damage may be attributed to an electron charging effect due to the highly insulating nature of the ZnCoO film; before the irradiation, the ZnCoO sample exhibited low conductivity comparable to an insulator (resistance ∼ 1 MΩ).33–35 We did not observe any significant changes in the electrical properties of the ZnCoO thin films after the e-beam irradiation; the high resistance of order MΩ, persisted in the ZnCoO film.
To see if there is any change in the magnetic properties of ZnCoO after the e-beam irradiation, we measured the field-dependent magnetization (M–H) of ZnCoO after the e-beam irradiation, and show the M–H curves of the ZnCoO films as a function of the radiation dose in Fig. 3(b). As shown in Fig. 3(b), ZnCoO samples exhibit paramagnetic behavior relative to the applied magnetic field after e-beam irradiation, regardless of the dose. There have been several reports suggesting that oxygen vacancies are the main source of the ferromagnetism of ZnCoO.8,10,15,36 If this scenario is correct, we could have seen the emergence of ferromagnetism in ZnCoO after the e-beam irradiation because we observed the creation of oxygen vacancy in ZnCoO after e-beam irradiation above. However, we did not observe any ferromagnetic features in the films after the e-beam irradiation. It suggests that the oxygen vacancy may not be a crucial factor for the ferromagnetism. We might not be able to observe a significant change because the e-beam irradiation may have affected the surface of the film due to the charge effect as we discussed above. The slopes of the M–H curves change slightly, which may be attributed to a change in Pauli paramagnetism as a result of creation of donor-type defects (i.e., oxygen vacancy) or secondary species due to the surface damage as seen in the AFM results (Fig. 3(a)).37 These results indicate that the e-beam irradiation cannot modify electrical and paramagnetic properties of pristine ZnCoO.
It has been suggested that hydrogen impurities are responsible for the ferromagnetism of ZnCoO.19,35,37–40 It is also known that hydrogen impurities act as shallow donors and increase the electron carrier concentration in ZnO. It should be noted that the physical status of hydrogen as a donor is different to that of hydrogen participating in ferromagnetism;6,41 the hydrogen acting as a shallow donor has +1 ionic state and preferentially occupies the positions between the Zn(Co)–O bonding centers. However, the hydrogen participating in ferromagnetism is expected to be neutral and located at a co-sharing antibonding site of two Co atoms in the basal plane of ZnO. This state is relatively more stable than that of hydrogen at shallow donor sites.41 We speculated that if hydrogen is introduced to the ZnCoO, the charging effect observed in ZnCoO can be suppressed, so it would be useful to study the effect of e-beam irradiation. Therefore, we intentionally introduced hydrogen impurities to ZnCoO thin films (ZnCoO:H) using post hydrogen plasma treatment (the details of this process are described in the Experimental section), and measured AFM and M–H curve of the films after the e-beam irradiation. We displayed the M–H curve of ZnCoO:H before the e-beam irradiation in Fig. 3(b) for comparison and a strong ferromagnetic hysteresis loop can be seen for ZnCoO:H at 2 K.
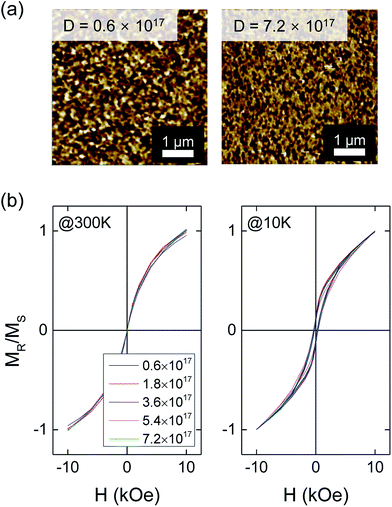
Fig. 4(a) shows AFM images of ZnCoO:H after e-beam irradiation. In contrast to the case of ZnCoO, no significant changes in surface morphology were observed after the irradiation. As ZnCoO becomes conductive after the hydrogen treatment (resistance ∼ 1 kΩ),33,34 the electron charging effect seems to be suppressed as expected. Fig. 4(b) shows M–H curves for ZnCoO:H after e-beam irradiation as a function of the irradiation dose; curves were normalized against the saturation magnetization of each sample to remove the paramagnetic contribution and to observe the changes in ferromagnetic hysteresis induced by the e-beam irradiation. As shown in Fig. 4(b), ferromagnetic hysteresis does not change significantly after the e-beam irradiation. This implies that the e-beam irradiation appears to have no influence on the ferromagnetism induced by post-hydrogen treatment and that the e-beam irradiation dose not affect the hydrogen atoms participating in ferromagnetism.
To find out whether e-beam irradiation alters the electrical properties of ZnCoO:H, we measured the temperature-dependent resistance of ZnCoO:H thin films before and after e-beam irradiation, where the dose of e-beam irradiation was fixed to be 1.8 × 1017 electrons per cm2. Fig. 5(a) shows temperature-dependent resistivity (ρxx) for ZnCoO:H before and after the irradiation. After the irradiation, the resistivities of the ZnCoO:H thin films increased significantly for the entire temperature region, which is totally different with the pristine ZnCoO case observed. To determine the origin of the increase in the resistivity, we measured the Hall resistance (ρxy) of the ZnCoO:H thin films before and after irradiation, at a temperature of 2 K (not shown here) to find the change in carrier concentration (n) and mobility (μ) of ZnCoO:H after the e-beam irradiation. As a result, the values of n = 9.02 × 1019 per cm3 and μ = 1.21 cm2 V−1 s−1 were obtained for the sample before the e-beam irradiation, and n = 3.47 × 1019 per cm3 and μ = 1.29 cm2 V−1 s−1 for the sample after the irradiation. The μ value remained approximately constant but n was clearly reduced after the e-beam irradiation. This indicates that the number of electron donors in ZnCoO:H is reduced by e-beam irradiation. In the case of the pristine ZnCoO thin films, we have seen no evidence of donor elimination after the e-beam irradiation; the oxygen vacancies we observed after the e-beam irradiation are known to be electron donors with a deep energy state,4 thus these do not account for the increase in resistivity observed in ZnCoO:H after the e-beam irradiation. Therefore, we conclude that the observed increase in the resistivity of ZnCoO:H thin films should be interpreted as arising from a reduction in the number of shallow donor Hs intentionally introduced. In fact, shallow donor type H is predicted to be energetically unstable compared to other possible defects in ZnO systems, and to migrate at very low activation energies such as those supplied by simple thermal treatments.4,6,42 Our observations support these predictions.
We have observed the positive magnetoresistance and anomalous Hall effect in ZnCoO:H.35 The magneto-transport characteristics are attributed to an s–d exchange interaction.8,43 In the s–d exchange interaction, s represents the Zn 4s orbital of the conduction band of ZnCoO. We hypothesized that if the e-beam irradiation affects the conduction band of ZnCoO:H, by reducing the charge carrier concentration, the magneto-transport characteristics will also change. Thus, to determine the effect of e-beam irradiation on magneto-transport properties, we measured the magnetic field-dependent resistance of ZnCoO:H before and after the e-beam irradiation. The results are presented in Fig. 5(b). The degree of the magnetoresistance (MR) was calculated by MR (%) = (R(H) − R(0))/R(0), where R(H) and R(0) are the resistances measured with and without an applied magnetic field, respectively. The inset of Fig. 5(b) shows the MR of ZnCoO:H before and after the e-beam irradiation as a function of applied magnetic field. Both samples exhibit clear positive magnetoresistance (i.e., increase in electrical resistance with increasing magnetic field) as a result of the s–d interaction. At lower temperature than 10 K, a small contribution of negative magnetoresistance has been observed in the low field, which was ascribed to the weak localization effect of the ZnO host.44,45 However, after e-beam irradiation, the magnitude of the MR increases and the positive behavior is more pronounced. In Fig. 5(b), it can be seen that positive magnetoresistance behavior increased after the e-beam irradiation below 50 K and the temperature is related to the onset of ferromagnetism in ZnCoO:H.20,35 Generally, the positive magnetoresistance attributed to the s–d exchange interaction is inversely proportional to the charge carrier concentration because the proportion of spin polarized carriers increases at low carrier concentrations.43,46–48 This suggests that the increase in positive magnetoresistance after the e-beam irradiation is attributed to the reduction in shallow donor Hs and corresponding carrier concentration.
We have also observed the anomalous Hall effect (AHE) in ZnCoO:H and described it in terms of the s–d exchange interaction based on the asymmetric scattering of carriers hopping between the localized states driven by the ferromagnetic Co–H–Co state.35 If the increase in positive magnetoresistance by the e-beam irradiation is truly related to the s–d exchange interaction based on the asymmetric scattering, the effect of the irradiation would be more apparent in AHE because the AHE term depends on the magnetization of materials and reflects the contribution of the spin-dependent scattering. Thus, we calculated the saturated anomalous Hall conductivity (σsAHE) of ZnCoO:H as a function of temperature before and after the e-beam irradiation and presented the results in Fig. 5(c). σsAHE was calculated by σsAHE = ρxy/(ρxx2 + ρxy2) where the values of ρxx and ρxy at 3 T were used because the resistivities were fully saturated at 3 T. After the e-beam irradiation, the value of σsAHE of ZnCoO:H decreases significantly, and almost zero above 20 K. This suggests that the e-beam irradiation can increase the asymmetric scattering of charge carriers hopping between localized states, driven by the hydrogen treatment.35
The increase in magnetoresistance and the decrease in anomalous Hall conductivity result from decreasing carrier concentrations due to elimination of shallow donor H after e-beam irradiation. As shown above, hydrogen impurities are necessary for inducing the ferromagnetism and the s–d interactions, which is responsible for the magneto-transport behavior of ZnCoO but e-beam irradiation can be used to manipulate the magneto-transport characteristics by only adjusting electron concentrations near the conduction band minimum without influencing ferromagnetism. In other words, the e-beam irradiation enables site-selective reduction in hydrogens in the ZnCoO lattice: the e-beam irradiation has no effect on the hydrogen responsible for the ferromagnetic spin ordering but it can only reduce the number of the hydrogen impurities that act as shallow donors in the ZnCoO lattice. It enables the control of the magneto-transport characteristics of ZnCoO.
Conclusions
Electron beam irradiation can create oxygen vacancy in ZnCoO and it does not induce any significant changes in the electrical and magnetic properties of ZnCoO due to charging effects, because of highly insulating nature of ZnCoO. However, we were able to suppress this insulating behavior and the charging effect using hydrogen treatment. After the hydrogen treatment, the ZnCoO became highly conductive and ferromagnetic. We did not observe changes in the ferromagnetic properties of hydrogen-treated ZnCoO thin films after e-beam irradiation but we did observe a decrease in the carrier concentration due to the reduction of shallow donor-type hydrogen atoms introduced intentionally. The reduction of shallow donor hydrogen achieved by e-beam irradiation modifies the conduction band of ZnCoO and the s–d exchange interaction, resulting in the change in the magneto-transport characteristics of ZnCoO. These results demonstrate the feasibility of using e-beam irradiation on ZnO-based magnetic semiconductors.Experimental section
Pure ZnO and 10 mol% Co-doped ZnO powder were fabricated using a sol–gel process,14,30,31,49 and ZnCoO thin films were fabricated on an Al2O3 substrate using an rf-sputtering method. The base pressure was ∼1 × 10−5 Torr and the thin films were grown at 300 °C in an atmosphere of argon (Ar) (99.999%). The working pressure was kept at 10 mTorr. E-beam irradiation was conducted at Korea Atomic Energy Research Institute (KAERI). E-beam irradiation was carried out at room temperature with Ar (99.999%) as the ambient gas and an acceleration of 0.2 MeV (200 kV, 1 mA), permitting doses in the range of 0.6 × 1017 to 7.2 × 1017 electron per cm2. For the intentional hydrogen injection (ZnCoO:H), hydrogen treatment was performed via plasma exposure with mixed Ar![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2 gas (9
H2 gas (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 vol%). X-ray diffraction (XRD) imaging was performed with θ/2θ geometry on a Panalytical Empyrean series2 with a Cu Kα source (40 kV, 30 mA). XRD patterns of ZnO and ZnCoO powder samples were collected within the range of 20° < 2θ < 80° with a 0.0167° step size and with a count time of 60 s per point for all cases. A Rietveld refinement with the maximum entropy method (MEM) was conducted using a program package ENIGMA at 66 × 66 × 104 pixels.50 Field-dependent magnetization was measured using a vibrating sample magnetometer installed in a physical-property measurement system (PPMS model 6000, Quantum Design, Inc.). Magneto-transport properties of the samples with a Hall-bar pattern were measured using a combination of a PPMS system, a function generator (HP 3325B), a lock-in amplifier (Stanford SR830 DSP), and source meter (Keithley 2425C).
1 vol%). X-ray diffraction (XRD) imaging was performed with θ/2θ geometry on a Panalytical Empyrean series2 with a Cu Kα source (40 kV, 30 mA). XRD patterns of ZnO and ZnCoO powder samples were collected within the range of 20° < 2θ < 80° with a 0.0167° step size and with a count time of 60 s per point for all cases. A Rietveld refinement with the maximum entropy method (MEM) was conducted using a program package ENIGMA at 66 × 66 × 104 pixels.50 Field-dependent magnetization was measured using a vibrating sample magnetometer installed in a physical-property measurement system (PPMS model 6000, Quantum Design, Inc.). Magneto-transport properties of the samples with a Hall-bar pattern were measured using a combination of a PPMS system, a function generator (HP 3325B), a lock-in amplifier (Stanford SR830 DSP), and source meter (Keithley 2425C).
Acknowledgements
This research was supported by the Converging Research Center Program (NRF-2014M3C1A8048820 and NRF-2014M3C1A8048818) and National Nuclear R&D Program (NRF-2012M2B2A4029513) through the Ministry of Science, ICT and Future Planning, Korea.Notes and references
- S. J. Pearton, D. P. Norton, K. Ip, Y. W. Heo and T. Steiner, Prog. Mater. Sci., 2005, 50, 293 CrossRef CAS.
- M. Gabás, A. Landa-Cánovas, J. Luis Costa-Krämer, F. Agulló-Rueda, A. R. González-Elipe, P. Díaz-Carrasco, J. Hernández-Moro, I. Lorite, P. Herrero, P. Castillero, A. Barranco and J. Ramón Ramos-Barrado, J. Appl. Phys., 2013, 113, 163709 CrossRef.
- K. Ueda, H. Tabata and T. Kawai, Appl. Phys. Lett., 2001, 79, 988 CrossRef CAS.
- A. Janotti and C. G. Van de Walle, Rep. Prog. Phys., 2009, 72, 126501 CrossRef.
- D. Look, G. C. Farlow, P. Reunchan, S. Limpijumnong, S. Zhang and K. Nordlund, Phys. Rev. Lett., 2005, 95, 225502 CrossRef CAS PubMed.
- C. G. Van de Walle, Phys. Rev. Lett., 2000, 85, 1012 CrossRef CAS PubMed.
- S. B. Ogale, Adv. Mater., 2010, 22, 3125 CrossRef CAS PubMed.
- J. M. D. Coey, M. Venkatesan and C. B. Fitzgerald, Nat. Mater., 2005, 4, 173 CrossRef CAS PubMed.
- J. M. D. Coey and S. A. Chambers, MRS Bull., 2008, 33, 1053 CrossRef CAS.
- G. Chen, C. Song, C. Chen, S. Gao, F. Zeng and F. Pan, Adv. Mater., 2012, 24, 3515 CrossRef CAS PubMed.
- N. Khare, M. J. Kappers, M. Wei, M. G. Blamire and J. L. MacManus-Driscoll, Adv. Mater., 2006, 18, 1449 CrossRef CAS.
- I. Lorite, J. Wasik, T. Michalsky, R. Schmidt-Grund and P. Esquinazi, Thin Solid Films, 2014, 556, 18 CrossRef CAS.
- W.-K. Kim, S. Lee, Y. C. Cho, H. Koinuma, S.-Y. Jeong, J.-M. Shin, C. R. Cho, J.-S. Bae, T.-Y. Kim and S. Park, Appl. Phys. Lett., 2011, 98, 122109 CrossRef.
- S. Lee, Y. C. Cho, S.-J. Kim, C. R. Cho, S.-Y. Jeong, S. J. Kim, J. P. Kim, Y. N. Choi and J. M. Sur, Appl. Phys. Lett., 2009, 94, 212507 CrossRef.
- H. S. Hsu, J. C. A. Huang, Y. H. Huang, Y. F. Liao, M. Z. Lin, C. H. Lee, J. F. Lee, S. F. Chen, L. Y. Lai and C. P. Liu, Appl. Phys. Lett., 2006, 88, 242507 CrossRef.
- N. Tahir, A. Karim, K. A. Persson, S. T. Hussain, A. G. Cruz, M. Usman, M. Naeem, R. Qiao, W. Yang, Y.-D. Chuang and Z. Hussain, J. Phys. Chem. C, 2013, 117, 8968 CAS.
- D. Rubi, J. Fontcuberta, A. Calleja, L. Aragonès, X. G. Capdevila and M. Segarra, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 75, 155322 CrossRef.
- B.-S. Kim, S. Lee, W.-K. Kim, J. H. Park, Y. C. Cho, J. Kim, C. R. Cho and S.-Y. Jeong, Nanoscale Res. Lett., 2014, 9, 1–7 CrossRef PubMed.
- H. Zhang, W. Hao, Y. Cao, X. Chang, M. Xu, X. Guo, K. Shen, D. Xiang and Q. Xu, RSC Adv., 2016, 6, 6761 RSC.
- J. M. Shin, H. S. Lee, S. Y. Cha, S. Lee, J. Y. Kim, N. Park, Y. C. Cho, S. J. Kim, S.-K. Kim, J.-S. Bae, S. Park, C. R. Cho, H. Koinuma and S.-Y. Jeong, Appl. Phys. Lett., 2012, 100, 172409 CrossRef.
- K. E. Jacobs and P. M. Ferreira, Adv. Funct. Mater., 2015, 25, 5261 CrossRef CAS.
- A. V. Krasheninnikov and F. Banhart, Nat. Mater., 2007, 6, 723 CrossRef CAS PubMed.
- H. J. Kim, H. B. Bae, Y. Park and S. H. Choi, RSC Adv., 2012, 2, 12670 RSC.
- W.-K. Lee, K. E. Whitener Jr, J. T. Robinson and P. E. Sheehan, Adv. Mater., 2015, 27, 1774 CrossRef CAS PubMed.
- B.-H. Kim, D. H. Lee, K. S. Yang, B. C. Lee, Y. A. Kim and M. Endo, ACS Appl. Mater. Interfaces, 2011, 3, 119 CAS.
- S. M. Evans, N. C. Giles, L. E. Halliburton and L. A. Kappers, J. Appl. Phys., 2008, 103, 043710 CrossRef.
- E.-J. Yun, J. W. Jung, Y. H. Han, M.-W. Kim and B. C. Lee, J. Appl. Phys., 2009, 105, 123509 CrossRef.
- E.-J. Yun, J. W. Jung, C. I. Cheon, J. S. Kim, Y. H. Han, M.-W. Kim and B. C. Lee, J. Mater. Res., 2011, 24, 1785 CrossRef.
- J. H. Park, Y. J. Lee, J.-S. Bae, B.-S. Kim, Y. C. Cho, C. Moriyoshi, Y. Kuroiwa, S. Lee and S.-Y. Jeong, Nanoscale Res. Lett., 2015, 10, 657 Search PubMed.
- S. J. Kim, S. Lee, Y. C. Cho, Y. N. Choi, S. Park, I. K. Jeong, Y. Kuroiwa, C. Moriyoshi and S.-Y. Jeong, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 81, 212408 CrossRef.
- S. J. Kim, S. Y. Cha, J. Y. Kim, J.-M. Shin, Y. C. Cho, S. Lee, W.-K. Kim, S.-Y. Jeong, Y. S. Yang, C. R. Cho, H. W. Choi, M.-H. Jung, B.-E. Jun, K.-Y. Kwon, Y. Kuroiwa and C. Moriyoshi, J. Phys. Chem. C, 2012, 116, 12196 CAS.
- R. Kitaura, S. Kitagawa, Y. Kubota, T. C. Kobayashi, K. Kindo, Y. Mita, A. Matsuo, M. Kobayashi, H.-C. Chang, T. C. Ozawa, M. Suzuki, M. Sakata and M. Takata, Science, 2002, 298, 2358 CrossRef CAS PubMed.
- Y. C. Cho, S.-J. Kim, S. Lee, S. J. Kim, C. R. Cho, H.-H. Nahm, C. H. Park, I.-K. Jeong, S. Park, T. E. Hong, S. Kuroda and S.-Y. Jeong, Appl. Phys. Lett., 2009, 95, 172514 CrossRef.
- Y. C. Cho, S. Lee, H. H. Nahm, S. J. Kim, C. H. Park, S. Y. Lee, S.-K. Kim, C. R. Cho, H. Koinuma and S.-Y. Jeong, Appl. Phys. Lett., 2012, 100, 112403 CrossRef.
- Y. C. Cho, S. Lee, J. H. Park, W. K. Kim, H.-H. Nahm, C. H. Park and S.-Y. Jeong, New J. Phys., 2014, 16, 073030 CrossRef.
- G. Ciatto, A. Di Trolio, E. Fonda, P. Alippi, A. M. Testa and A. A. Bonapasta, Phys. Rev. Lett., 2011, 107, 127206 CrossRef CAS PubMed.
- S. Lee, B.-S. Kim, J. H. Park, T.-W. Lee, Y. C. Cho and S.-Y. Jeong, RSC Adv., 2015, 5, 65840 RSC.
- J. H. Park, S. Lee, B.-S. Kim, W.-K. Kim, Y. C. Cho, M. W. Oh, C. R. Cho and S.-Y. Jeong, Appl. Phys. Lett., 2014, 104, 052412 CrossRef.
- S. Lee, W.-K. Kim, Y. C. Cho, B.-S. Kim, J. H. Park, C.-W. Lee, Y. Lee, S. Lee, S. Fackler, I. Takeuchi, C. R. Cho and S.-Y. Jeong, Appl. Phys. Lett., 2014, 104, 052405 CrossRef.
- L. Li, Y. Guo, X. Y. Cui, R. Zheng, K. Ohtani, C. Kong, A. V. Ceguerra, M. P. Moody, J. D. Ye, H. H. Tan, C. Jagadish, H. Liu, C. Stampfl, H. Ohno, S. P. Ringer and F. Matsukura, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 85, 174430 CrossRef.
- C. Park and D. Chadi, Phys. Rev. Lett., 2005, 94, 127204 CrossRef CAS PubMed.
- A. Janotti and C. G. Van de Walle, Nat. Mater., 2006, 6, 44 CrossRef PubMed.
- W. Liang, B. D. Yuhas and P. Yang, Nano Lett., 2009, 9, 892 CrossRef CAS PubMed.
- I. Lorite, P. Esquinazi, C. Zapata and S. P. Heluani, J. Mater. Res., 2013, 29, 78–83 CrossRef.
- T. Andrearczyk, J. Jaroszyński, G. Grabecki, T. Dietl, T. Fukumura and M. Kawasaki, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 72, 121309 CrossRef.
- Q. Xu, L. Hartmann, H. Schmidt, H. Hochmuth, M. Lorenz, R. Schmidt-Grund, C. Sturm, D. Spemann, M. Grundmann and Y. Liu, J. Appl. Phys., 2007, 101, 063918 CrossRef.
- Q. Xu, L. Hartmann, H. Schmidt, H. Hochmuth, M. Lorenz, R. Schmidt-Grund, C. Sturm, D. Spemann and M. Grundmann, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 73, 205342 CrossRef.
- A. J. Behan, A. Mokhtari, H. J. Blythe, M. Ziese, A. M. Fox and G. A. Gehring, J. Phys.: Condens. Matter, 2009, 21, 346001 CrossRef CAS PubMed.
- S. Lee, B.-S. Kim, S.-W. Seo, Y. C. Cho, S. K. Kim, J. P. Kim, I.-K. Jeong, C. R. Cho, C. U. Jung, H. Koinuma and S.-Y. Jeong, J. Appl. Phys., 2012, 111, 07C304 Search PubMed.
- Y. Kuroiwa, S. Aoyagi, A. Sawada, J. Harada, E. Nishibori, M. Takata and M. Sakata, Phys. Rev. Lett., 2001, 87, 217601 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |