Synthesis and characterization of novel sulfanilic acid–polyvinyl chloride–polysulfone blend membranes for metal ion rejection†
Vignesh Nayaka,
M. S. Jyothia,
R. Geetha Balakrishna*a,
Mahesh Padaki*a and
Arun M. Isloorb
aCentre for Nano and Material Sciences, Jain University, Ramanagaram, Bangalore-562112, India. E-mail: br.geetha@jainuniversity.ac.in; sp.mahesh@jainuniversity.ac.in; Tel: +91-80-27506270
bMembrane Technology Laboratory, Chemistry Department, National Institute of Technology-Karnataka, Surathkal, Mangalore 575 025, India
First published on 26th February 2016
Abstract
Near-complete removal of heavy metals, namely Cd(II), Cr(VI) and Pb(II), has been attempted by a membrane purification process using a blend of modified polyvinyl chloride (PVC) and polysulfone (PSf), prepared by the diffusion induced phase separation (DIPS) method. The prepared novel material was characterized by NMR, ATR-IR spectroscopy and DSC. The sulphonyl groups incorporated into PVC enhance the hydrophilicity and are substantiated by water uptake, contact angle (CA) and flux studies. The obtained properties of the blend membrane like increased surface roughness and porosity are observed from AFM and SEM analysis. An enhanced rejection of ∼95% which is about 1.15, 1.41 and 1.37 times better than the commercially available NF 270 membrane was observed, for Cd(II), Cr(VI) and Pb(II) respectively. The work was further extended to study the antifouling property and the interference of other existing metal ions on the performance. An improved antifouling property with 98.5% rejection for bovine serum albumin (BSA) and a 75.6% flux recovery ratio (FRR) was achieved. The study gains significance in exploring the incorporation of sulphonyl groups in to polymers, to enhance membrane performance.
1. Introduction
The concept of water salvaging has gained a tremendous boost in recent times due to the rise of contaminants being discharged into water resources.1 Among the various pollutants, heavy metals are abundantly present in polluted waters and are highly toxic. Human activities such as electroplating, wood processing, leather tanning, and anodizing produce wastewater containing heavy metals causing immense intimidation to mankind.2 Heavy metals such as lead (Pb), cadmium (Cd) and chromium (Cr) can cause various disorders in humans such as nephrotoxicity, neurotoxicity, effects on the cardiovascular system, and effects on cellular anti-toxicity defences, damage to oxidative DNA repair systems, and kidney and liver problems.3–5 Moreover, they are non-biodegradable and tend to accumulate in the tissues of living organisms6 when ingested even at very low concentrations. Hence, in the present era an effective removal method for such heavy metals is necessary.7 There are several methods such as chemical precipitation, ion-exchange, membrane filtration, the membrane filtration technique and adsorption8 for heavy metal removal.The membrane filtration technique has shown great promise towards water purification.9 The feasible operating conditions, energy savings and high efficiency have created new interest in heavy metal removal with membrane technology.10 Membrane technology is a servomechanism technique in separation and purification research. Several efforts from researchers are continuing for the betterment of membrane separation in respect to better productivity with minimal selectivity, ease of operational conditions and the ability to be scaled up at low costs,11 including the use of other membrane processes such as reverse osmosis (RO) and nanofiltration (NF).12,13 However, UF membranes are not suitable for metal ion rejection due to their larger pore size compared to the size of metal ions14 in comparison to nanofiltration membranes which have shown good results towards the removal of heavy metals.15 But this setback can be resolved by modifying the polymer chemically by incorporating functional groups which in turn induce a charge on the membrane surface.16 The charge on the membrane can considerably mitigate fouling and can append to improve the efficiency and life span of the membrane.17
Polymers show great promise for the production of membranes due to their good flexibility, toughness and separation capability.18 PVC is one of the most versatile polymers with good flexibility, chemical resistance, and film forming property, as well as being economical and easy to modify.19,20 PVC has been extensively used in industrial applications, namely as a packing material, cable insulation, and in pipes. However, its use as a membrane for water filtration is limited due to its hydrophobicity and membrane fouling property.21 The polymer properties can be changed by chemical modification. In this regard, chemical modification of PVC promises to render the necessary properties like membrane charge and hydrophilicity. The surface charge and hydrophilicity are very necessary tools for improvement in the selectivity of ions and productivity in membrane separation. There are reports already available in the literature on PVC modification, where Tooma modified PVC with ethyl acrylate monomer for vacuum membrane distillation (VMD) application.22 A methylimidazolium group-modified PVC doped with phosphoric acid membrane was used for proton exchange by Che.23 Conversely there are a few reports on PVC blends and composite membranes for salt rejection by Cui using blend PVC/P(DMA-co-MMA) precursors,24 a PVC–silica mixed-matrix membrane as a membrane bioreactor by Bilad25 and CO2 gas separation using a PVC/Pebax composite membrane by Ahmadpour.26 But none have studied the removal of metal ions. Hence, the aim of this study is to prepare a novel polymeric membrane by a novel approach to obtain a synergistic effect of optimum antifouling, selectivity and productivity.
PVC was modified using sulfanilic acid (SA) by a simple chemical reaction. The present paper reports a novel functional group as a substituent, a novel chemical modification method for PVC modification, and its use in a membrane for water purification. To the best of our literature knowledge, such work has not been reported yet. Modified PVC is further blended with PSf (with different compositions) to regain its lost strength after modification via the DIPS method. The chemical modification of PVC was confirmed by NMR, ATR-IR and DSC. A blend membrane has properties like enhanced surface roughness and porosity, which were observed from AFM and SEM analysis. The sulphonyl group present in the modified PVC enhances the hydrophilicity and it was confirmed by water uptake, contact angle measurements and flux studies. The prepared blend membranes were used for rejection of metal ions, as the blends were charged and charge can play a very important role in the rejection of heavy metals in the ultrafiltration range. To validate the applicability of the blend membranes to real samples, the rejection in the presence of interfering ions is also studied and the results were also compared with the commercially available membrane NF 270 under the same operating conditions. The antifouling properties of the prepared membrane were also examined and the effects of modification on the membrane performance were discussed.
2. Experimental
2.1. Materials and methods
Udel PSf with a molecular weight of 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Da, PVC with a high molecular weight and 1-sulfanilic acid (4-(H2N)C6H4SO3H, molecular weight: 173.19) were obtained from Sigma-Aldrich Co, India. Lead nitrate, cadmium nitrate and potassium dichromate were used as the source for Pb(II), Cd(II) and Cr(VI) respectively. Chemicals like dimethyl sulfoxide (DMSO), triethylamine and methyl-2-pyrrolidone (NMP) were of Merck brand. The commercial membrane NF 270 was purchased from Sterlitech, USA.
000 Da, PVC with a high molecular weight and 1-sulfanilic acid (4-(H2N)C6H4SO3H, molecular weight: 173.19) were obtained from Sigma-Aldrich Co, India. Lead nitrate, cadmium nitrate and potassium dichromate were used as the source for Pb(II), Cd(II) and Cr(VI) respectively. Chemicals like dimethyl sulfoxide (DMSO), triethylamine and methyl-2-pyrrolidone (NMP) were of Merck brand. The commercial membrane NF 270 was purchased from Sterlitech, USA.
2.2. Synthesis of PVC–SA
Modification of PVC is being studied by Herrero et al.27 using different functional groups. Similarly, Huang et al.28 reported aminoethanol substitution on PVC for Heck reactions. In our approach, the reaction was carried out using a two-neck round bottom flask mounted on a magnetic stirrer which is equipped with a hot plate. 1 g of SA was dissolved in 50 mL DMSO, together with 10 mL of triethylamine to abstract the proton from the SO3H group of SA. Subsequently, 1 g of PVC was added to the same solution and the reaction mixture was stirred for 24 hours at 60–65 °C at a constant rate of heating in nitrogen atmosphere. The reaction scheme is proposed in Fig. 1. The reaction was quenched by precipitating out in a mixture of cold water/methanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) after 24 hours. The precipitate was then washed with distilled water several times to remove the excess solvent and dried overnight in an oven.
2) after 24 hours. The precipitate was then washed with distilled water several times to remove the excess solvent and dried overnight in an oven.
The % of modification (% M) and % modifying efficiency (% ME) of the graft PVC polymer were calculated using the following equation as described elsewhere:29
 | (1) |
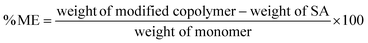 | (2) |
2.3. Preparation of the blend membrane
The membrane was prepared by the DIPS method. The DIPS process is based on the phenomenon of precipitation of the polymer in a controlled manner, when it comes in contact with water to form a solid porous film. 4 g of PVC was dissolved in 16 g (15.54 mL) of NMP and stirred for 24 hours at 60 °C to obtain a viscous solution. The obtained viscous solution was then casted on a glass plate using a glass rod. The thickness of the membrane was maintained using double-sided tape. The glass plate was dipped in a coagulation bath containing distilled water at room temperature.30 The membrane was peeled off by the phase inversion method and the obtained membrane was washed and stored in distilled water for 24 hours to remove any excess NMP and also to gain mechanical strength. Similarly, a neat PSf membrane was prepared by the same method by adding PSf instead of PVC. The same procedure was extended to prepare blend membranes; in this case both the polymers were added in different concentrations. A series of blends was prepared with different compositions as depicted in Table 1.| Sr. No. | Membrane | Composition (%) | ||
|---|---|---|---|---|
| PSf | PVC/SA–PVC | NMP | ||
| 1 | PSf | 20 | — | 80 |
| 2 | PVC | — | 20 | 80 |
| 3 | 3% PVC–SA/PSf | 19.4 | 0.6 | 80 |
| 4 | 5% PVC–SA/PSf | 19.0 | 1.0 | 80 |
| 5 | 8% PVC–SA/PSf | 18.4 | 1.6 | 80 |
| 6 | 10% PVC–SA/PSf | 18.0 | 2.0 | 80 |
2.4. Characterization of the membrane
1H NMR was recorded on a Perkin-Elmer EM 300 MHz spectrometer using tetramethylsilane (TMS) as the internal standard. The samples were dissolved in dimethyl sulfoxide (DMSO) at 60 °C before analysis, as the polymer was partially soluble at room temperature.
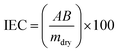 | (3) |
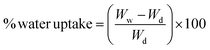 | (4) |
The surface hydrophilicity of the membrane was measured by standard goniometer Ramehart-200 F1 as per the sessile droplet method. To minimize the errors, the contact angle was observed at five different positions of the membrane and the average is reported.
The slope for the plot of water flux and the applied pressure gives the hydraulic permeability coefficient (Lp), with the intercept fixed at zero and calculated from the equation:34
| Lp (m s−1 Pa−1) = slope × 2.77 × 10−10 | (5) |
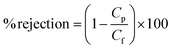 | (6) |
To investigate the competitive behavior of different metal ions towards rejection, the experiments were studied in the presence of the mixture of metal ions.
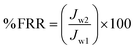 | (7) |
The % of BSA rejection was calculated as per the equation given in Section 2.4.8. The concentration of BSA was calculated using a UV-visible spectrophotometer at λmax = 280 nm.
In order to get a complete idea of fouling, the total fouling ratio (Rt), reversible fouling ratio (Rr) and irreversible fouling ratio (Rir) were calculated by the following equations:38
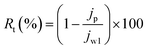 | (8) |
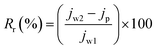 | (9) |
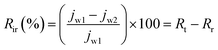 | (10) |
Furthermore, the best performing membrane (3% PVC–SA/PSf) was subjected to several cycles of filtration/rinsing as per the same procedure as that explained above, to find out the membrane life span and to validate the resistance towards fouling.
3. Results and discussion
Modification of PVC is vital to enhance the hydrophilicity and this can be done by incorporating hydrophilic groups into the polymer chain. This property improves the membrane performance in terms of flux and rejection, but may lead to severe loss of mechanical strength of the membrane which is not appropriate for its use. Hence, the blending of modified PVC with a much stronger polymer such as PSf regains its lost strength. The blend membrane will have the optimum properties like improved mechanical strength, surface charge and hydrophilicity for heavy metal ion rejection.3.1. Characterization
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching for the aromatic groups. All these peaks were absent in the PVC spectrum, and the peaks arising at 1240 and 1084 cm−1 may be attributed to C–C and C–H deformation and stretching respectively as shown in the spectra (ESI S.2†). C–H stretching was observed at 2934 and 2930 cm−1 in both the cases of PVC–SA and PVC respectively. Hence the incorporation of SA in the PVC chain is confirmed.
C stretching for the aromatic groups. All these peaks were absent in the PVC spectrum, and the peaks arising at 1240 and 1084 cm−1 may be attributed to C–C and C–H deformation and stretching respectively as shown in the spectra (ESI S.2†). C–H stretching was observed at 2934 and 2930 cm−1 in both the cases of PVC–SA and PVC respectively. Hence the incorporation of SA in the PVC chain is confirmed.The extent of modification as calculated from the equation given in the experimental section is shown in Table 2.
Weight ratio of PVC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) SA SA |
% modification (% M) | % modifying efficiency (% ME) |
|---|---|---|
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
31.2 | 31.2 |
PSf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PVC–SA membrane PVC–SA membrane |
Ion-exchange capacity (mequiv. g−1), IEC = AB/mdry |
|---|---|
| a An increase in the ion-exchange capacity of the membranes implies that the density of the functional groups is greater in the membranes containing a greater concentration of PVC–SA. | |
90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
0.1673 ± 0.006 |
92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 8 |
0.1348 ± 0.022 |
95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
0.1146 ± 0.020 |
97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
0.09421 ± 0.009 |
The PVC membrane shows a slightly higher water uptake due to the macroporous nature of the membrane as can be seen from the SEM image (Fig. 4a). The water uptake capacity of the blend membrane is much higher and increases proportionately with the increase in concentration of functional groups to enhance the hydrophilicity of the membranes. The contact angle decreases from 73.6 to 64.8 while the water uptake increases from 22.5% to 82%, proportionately for the corresponding increase in the PVC–SA concentration in the blends. The sulfanilic group being hydrophilic facilitates absorption of water and hence an increase in functional groups proportionately enhances the affinity towards water. This is because the presence of hydrophilic groups makes it easy for the water to get absorbed in the membrane and the greater the number of functional groups present, the greater the affinity towards water will be and the greater the absorption will be.
 | ||
| Fig. 3 3D images of AFM: (a) neat PVC, (b) 3% PVC–SA/PSf, (c) 5% PVC–SA/PSf, (d) 8% PVC–SA/PSf, and (e) 10% PVC–SA/PSf membranes. | ||
An increase in the content of PVC–SA increases the number of valleys (differentiated by light and dark areas) leading to an increased surface roughness from 6.9 nm to 48.4 nm. The trend observed is due to the increase in hydrophilic groups which enhances the rate of the phase inversion process, due to its mobility towards water surroundings. This observed phenomenon is in good agreement with the results obtained by Nazri et al.29 and Liu et al.42
 | ||
| Fig. 4 Cross section SEM images of (a) PSf, (b) PVC, (c) 3% PVC–SA/PSf, (d) 5% PVC–SA/PSf, (e) 8% PVC–SA/PSf, and (f) 10% PVC–SA/PSf membranes. The scale represents 100 μm in all the images. | ||
As it can be seen, the neat PVC membrane has long and more finger-like structures without much differentiation in the layers, whereas the neat PSf membrane depicts three distinct layers. The upper dense layer (skin layer), middle layer (finger-like projections) and bottom layer (sponge-like with big pores) are responsible for the selectivity, productivity and strength.43 In the PSf membrane, the bottom layer is very thick as compared to the other membranes. The addition of PVC–SA to PSf separates the three different structural layers with a substantial density of the skin layer being visible in comparison to neat PSf. An increase in the concentration of PVC–SA diminishes the appearance of sub-layers. The skin layer becomes less dense and they appear as a merge of the middle and bottom layers in 5% PVC–SA/PSf. The selective layer has completely merged with the middle layer to give one single layer with a broad finger-like structure in the 8% PVC–SA/PSf membrane. Whereas the big hollow finger-like projections (marked in red for better understanding) are randomly observed in the 10% PVC–SA/PSf membrane running from the top to the bottom of the membrane, where all three of the layers have merged to give one single layer. During the phase inversion process, the non-solvent penetrates at a faster rate and the solvent (NMP)–non solvent (water) interchange occurs very quickly to give big pores.44,45 This observation is in good accordance with the roughness values obtained in AFM analysis. Similarly as the composition of PVC–SA is lowered the interchange of the solvent and non-solvent takes place at a slow rate giving it the dense pore network.
3.2. Performance study
Whereas, if we compare the flux of the commercially available membrane NF 270 it is considerably high compared to that of the PVC–SA/PSf blend membranes which may be attributed to its hydrophilicity.
The flux graph is a model fit, and a trend line was drawn to study the hydraulic permeability. Table 4 depicts the Lp value of the membranes. From the table it is clear that Lp of the membrane decreases with a decrease in the concentration of functional groups present in the blend membrane. However, Lp of the neat PVC is near to that of the 5% PVC–SA/PSf membrane due to its high flux from its macro-size pores. Also Lp of the NF 270 membrane is the highest due to its pretty high flux compared to the other membranes.
PSf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PVC–SA membrane PVC–SA membrane |
Hydraulic permeability coefficient (Lp) (m s−1 Pa−1) |
|---|---|
| Neat PVC | 2.70 × 10−12 |
90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
12.44 × 10−12 |
92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 8 |
6.89 × 10−12 |
95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
3.12 × 10−12 |
97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
0.26 × 10−12 |
| NF 270 | 32.02 × 10−12 |
However, the 10% PVC–SA/PSf membrane showed the lowest rejection compared to all the other membranes even though it had the maximum density of functional groups in its vicinity. This can be because of the larger pore size (Fig. 4f) and flux of the membrane (Fig. 5). This indicates that even though the rejection depends on the charge, porosity plays a very important role. If the pores are large and more numerous, than the ions can easily pass through the pores of the membranes due to the applied pressure. Hence, there is no chance for charge interaction. Instead the 3% PVC–SA/PSf membrane results in maximum rejection. As explained in the SEM section, the hydrophilic component PVC–SA beyond a particular limit causes the loss of the selective skin layer. The same trend is observed in the 5% PVC–SA/PSf and 8% PVC–SA/PSf membranes. It also was confirmed by AFM images, where the surface roughness increased with the increasing concentration of PVC–SA. This is because of the hydrophilic functional group moving towards water molecules of the coagulation bath, which causes the greater membrane surface roughness. Furthermore the rejection performance was decreased with respect to the pressure increases from 100 to 500 kPa. The metal ions are forced out from the membrane, decreasing the rejection at high pressures. This suggests the importance of the morphology of the membranes in metal ion retention.
However, the metal ion rejection was almost similar because of the nearly similar size of 0.43 nm, 0.426 nm and 1.34 nm for Pb(II), Cd(II) and Cr(VI) metal ions respectively in their hydrated states.17,48 The trend of rejection follows the order of the 10% < 8% < 5% < 3% PVC–SA/PSf membrane. In the 10% PVC–SA/PSf membrane, the rejection was much less due to the greater charge and porosity. Hence, it is necessary to attain an optimum size and charge of the membrane to obtain good results. The obtained result is consistent with the reported value of 99.44% of lead removal (at pH 5.7) by Gherasim49 and 98% retention of iron(III) ions by Bernat et al.50 (at pH 2).
It is observed in the figure (ESI S.7†) that the rejection pattern for all three of the metal ions by the NF 270 membrane shows a maximum of 78% rejection for Cd(II) ions followed by Pb(II) (76%) and Cr(VI) (60%). Also there is variability in the rejection pattern over the increasing pressure range which is due to the fouling of the membrane over time. However the selectivity of the commercial membrane towards metal ions is not acceptable. Hence it can be concluded that the prepared membranes gave a better rejection efficiency compared to the NF 270 membrane, however it lacked the flux of the NF 270 membrane. The lack of flux in the blend membrane is overcome by its enhanced rejection, thus making it compatible with the existing membranes.
The flux recovery ratio (FRR) of the prepared membrane is shown in Fig. 6. 59.2% of the FRR and 74% of total fouling (Fig. 7) was observed in the PVC membrane. The modification of PVC increases the FRR up to 79.65% and decreases the total fouling up to 40%. Increasing the composition of PVC–SA in the membrane tends to increase the FRR and lower the fouling. This is because of the increasing hydrophilicity and membrane surface charge.
In studying complete fouling, the reversible and irreversible fouling parameters are very important. In reversible fouling, the foulant can be removed by washing because these are loosely bonded on the membrane surface. Whereas, in irreversible fouling, the foulant cannot be removed because these are stuck inside the membrane pores. The total fouling, reversible and irreversible fouling ratios were calculated and are presented in Fig. 7. Reversible fouling decreases from the 3% to 10% PVC–SA/PSf membrane. However, irreversible fouling increases from the 3% to 10% PVC–SA/PSf membrane. It is the effect of the hydrophilic membrane surface and pore size. BSA is hydrophobic in nature, which loosely adheres on the hydrophilic membrane surface.53 Hence, increasing the hydrophilicity of the membrane decreases the reversible fouling. The foulant can be easily removed from such membrane surfaces by washing. On the other hand, the pore size of the membrane goes on increasing with respect to the concentration of PVC–SA, which allows the BSA particles to go inside the membrane matrix and block the pores, which causes more irreversible fouling.
However during the fouling experiment, rejection of BSA was also observed. Fig. 6 presents the percent of BSA rejection from the membranes. The result obtained is similar to that of metal ion rejection with the highest of 98.5% of BSA being rejected by the 3% PVC–SA/PSf membrane. However, the rejection of BSA is greater in comparison to that of the metal ions due to the larger size of proteins, unlike the small sized metal ions.
The effect of counter ions on the rejection of a specific ion can be observed (ESI S.9†). The mixture concentration in the feed is the sum of 10 ppm of each metal ion, which is overall 30 ppm concentration of the feed. One more intention of this study is to know the performance of the membranes in a highly concentrated feed sample. The results reveal that there is a considerable decrease in the metal ion rejection. The rejection of Cr(VI) is about 68%, and for Cd(II) and Pb(II) it is about 62%. It can be pointed out that the amount of Cr(VI) rejected is more compared to the other two ions present. This is accounted for by the difference in the rejection mechanism of Cr(VI) ions (repulsion), whereas attraction for Pb(II) and Cd(II) ions causes the difference. The rejection was decreased in mixed ion rejection up to 10% in Cr(VI), 18% in Cd(II) and 13% of Pb(II) as compared to single ion rejection. In a higher concentration of the feed solution, accumulation of ions near the surface causes saturation of functional groups and allows some of the ions to pass through the membrane. Also the mobility of the ions decreases with an increase in concentration leading to reduced overall rejection, which is observed by Jyothi et al.46 However, a characteristic feature of charged membranes is less rejection at a high feed concentration and vice versa.54 An unexpectedly high rejection was observed at high pressure. This is because a high concentration of ions can lead to the deposition of metal ions on the membrane surface, which causes membrane fouling with time. Meanwhile, a decrease in the water flux rate (ESI S.9†) as compared to pure water flux, is evidences for the membrane fouling. A similar observation was found for the NF 270 membrane, where the rejection of metal ions varies with pressure unlike that of the blend membrane, where the rejection remains constant throughout the experiment. Besides, the NF membrane shows a greater affinity towards Pb(II) ion rejection over the Cr(VI) ions distinct from the blend membrane. Also the flux of the membrane has reduced to half of its initial pure water flux, which is due to the large amount of fouling that takes place on the membrane surface and also leads to the constant amount of rejection over the complete set of experiment.
The present study demonstrates an improved membrane for the removal of heavy metal ions as compared to the existing NF 270 membrane but with a lack of flux; however the study opens a lot of scope for improvement in the area of productivity in the near future.
4. Conclusions
A blend of modified PVC with the PSf membrane matrix showed a high impact on heavy metal removal. The modification was carried out by a newly developed method using sulfanilic acid. 1H NMR, ATR-IR spectroscopy and DSC confirmed the modification of PVC with sulfanilic acid. The blend membrane showed enhanced hydrophilicity and surface charge, which was confirmed by water uptake, water flux and IEC respectively. AFM and SEM analysis substantiated an improved surface roughness and porosity, due to the modified PVC. A good rejection of 90–95% was observed for the 3% PVC–SA/PSf membrane for Cd, Cr and Pb metal ions. The blend membranes show enhanced flux and antifouling property compared to the neat PVC and PSf membranes. The antifouling study demonstrated a good FRR value of 75.6% for the 10% PVC–SA/PSf membrane. The study gains significance in synthesizing an optimized and functionalized PVC–SA blend for good metal ion rejection and with a good antifouling property.Acknowledgements
The authors sincerely acknowledge the Indian Ministry of Drinking Water and Sanitation (no. 11017/33/2012-WQ) for financial support.References
- A. Vijayalakshmi, D. L. Arockiasamy, A. Nagendran and D. Mohan, Sep. Purif. Technol., 2008, 62, 32–38 CrossRef CAS.
- G. Arthanareeswaran, P. Thanikaivelan, J. A. Raguime, M. Raajenthiren and D. Mohan, Sep. Purif. Technol., 2007, 55, 8–15 CrossRef CAS.
- M. Hua, S. Zhang, B. Pan, W. Zhang, L. Lv and Q. Zhang, J. Hazard. Mater., 2012, 211–212, 317–331 CrossRef CAS PubMed.
- M. H. Ajmal Khan and S. Parvez, J. Trace Elem. Med. Biol., 2015, 31, 53–60 Search PubMed.
- F. Tan, X. Liu, X. Quan, J. Chen, X. Li and H. Zhao, Anal. Methods, 2011, 3, 343–347 RSC.
- C. H. Huang, T. H. Hsieh and W. Y. Chiu, Carbohydr. Polym., 2015, 116, 249–254 CrossRef CAS PubMed.
- Y. Mansourpanah, K. Alizadeh, S. S. Madaeni, A. Rahimpour and H. Soltani Afarani, Desalination, 2011, 271, 169–177 CrossRef CAS.
- F. Ferrero, C. Tonetti and M. Periolatto, Carbohydr. Polym., 2014, 110, 367–373 CrossRef CAS PubMed.
- A. Jayalakshmi, S. Rajesh, S. Senthilkumar and D. Mohan, Sep. Purif. Technol., 2012, 90, 120–132 CrossRef CAS.
- P. Daraei, S. S. Madaeni, N. Ghaemi, E. Salehi, M. A. Khadivi, R. Moradian and B. Astinchap, J. Membr. Sci., 2012, 415–416, 250–259 CrossRef CAS.
- R. Kumar, A. M. Isloor and A. F. Ismail, Desalination, 2014, 350, 102–108 CrossRef CAS.
- C. Hegde, A. M. Isloor, M. Padaki, P. Wanichapichart and Y. Liangdeng, Desalination, 2011, 265, 153–158 CrossRef CAS.
- Y. Zhang, J. Tian, H. Liang, J. Nan, Z. Chen and G. Li, J. Environ. Sci., 2011, 23, 529–536 CrossRef CAS.
- S. Liu, Z.-S. Li, C.-Y. Wang and A.-Y. Jiao, Sep. Purif. Technol., 2013, 116, 131–136 CrossRef CAS.
- P. Cañizares, A. Pérez, R. Camarillo and R. Mazarro, J. Membr. Sci., 2008, 320, 520–527 CrossRef.
- Z. Yu, Z. Sui and C. Tai-Shung, Environ. Sci. Technol., 2015, 49, 10235–10242 CrossRef PubMed.
- C. Mbareck, Q. T. Nguyen, O. T. Alaoui and D. Barillier, J. Hazard. Mater., 2009, 171, 93–101 CrossRef CAS PubMed.
- Y. Yang and P. Wang, Polymer, 2006, 47, 2683–2688 CrossRef CAS.
- H. Rabiee, M. H. D. A. Farahani and V. Vatanpour, J. Membr. Sci., 2014, 472, 185–193 CrossRef CAS.
- A. Gholami, A. R. Moghadassi, S. M. Hosseini, S. Shabani and F. Gholami, J. Ind. Eng. Chem., 2014, 20, 1517–1522 CrossRef CAS.
- S. Mei, C. Xiao, X. Hu and W. Shu, Desalination, 2011, 280, 378–383 CrossRef CAS.
- M. A. Tooma, T. S. Najim, Q. F. Alsalhy, T. Marino, A. Criscuoli, L. Giorno and A. Figoli, Desalination, 2015, 373, 58–70 CrossRef CAS.
- Q. Che, Z. Zhu, N. Chen and X. Zhai, Mater. Des., 2015, 87, 1047–1055 CrossRef CAS.
- Y. Cui, Z. K. Yao, K. Zheng, S. Y. Du, B. K. Zhu, L. P. Zhu and C. H. Du, J. Membr. Sci., 2015, 492, 187–196 CrossRef CAS.
- M. R. Bilad, L. Marbelia, C. Laine and I. F. J. Vankelecom, J. Membr. Sci., 2015, 493, 19–27 CrossRef CAS.
- E. Ahmadpour, A. A. Shamsabadi, R. M. Behbahani, M. Aghajani and A. Kargari, J. Nat. Gas Sci. Eng., 2014, 21, 518–523 CrossRef CAS.
- M. Herrero, P. Tiemblo, J. R. Labarta, C. Mijangos and H. Reinecke, Polymer, 2002, 43, 2631–2636 CrossRef CAS.
- X.-J. Huang, F. Dong, L. Chen and Y.-Q. Li, Monatshefte für Chemie - Chemical Monthly, 2008, 139, 1447–1451 CrossRef CAS.
- N. A. M. Nazri, W. J. Lau, M. Padaki and A. F. Ismail, J. Polym. Res., 2014, 21, 594 CrossRef.
- M. S. Jyothi, M. Padaki, R. Geetha Balakrishna and R. K. Pai, J. Mater. Res., 2014, 29, 1537–1545 CrossRef CAS.
- C. Klaysom, B. P. Ladewig, G. Q. M. Lu and L. Wang, J. Membr. Sci., 2011, 368, 48–53 CrossRef CAS.
- Z. Wang, H. Ni, M. Zhang, C. Zhao and H. Na, Desalination, 2009, 242, 236–244 CrossRef CAS.
- V. Nayak, M. S. Jyothi, R. G. Balakrishna, M. Padaki and A. F. Ismail, ChemistryOpen, 2015, 4, 278–287 CrossRef CAS PubMed.
- M. Padaki, A. M. Isloor and P. Wanichapichart, Desalination, 2011, 279, 409–414 CrossRef CAS.
- M. K. Aroua, F. M. Zuki and N. M. Sulaiman, J. Hazard. Mater., 2007, 147, 752–758 CrossRef CAS PubMed.
- M. Padaki, A. M. Isloor, P. Wanichapichart and A. F. Ismail, Desalination, 2012, 298, 42–48 CrossRef CAS.
- S. Zinadini, A. A. Zinatizadeh, M. Rahimi, V. Vatanpour and H. Zangeneh, J. Membr. Sci., 2014, 453, 292–301 CrossRef CAS.
- M. Padaki, D. Emadzadeh, T. Masturra and A. F. Ismail, Desalination, 2015, 362, 141–150 CrossRef CAS.
- L. P. Donald, G. M. Lampman, G. S. Kriz and J. R. Vyvyan, Introduction to Spectroscopy, Brooks/Cole Cengage Learning, Boston, 2008 Search PubMed.
- M. Padaki, A. M. Isloor, R. Kumar, A. Fauzi Ismail and T. Matsuura, J. Membr. Sci., 2013, 428, 489–497 CrossRef CAS.
- K. Aouachria and N. Belhaneche-Bensemra, Polym. Test., 2006, 25, 1101–1108 CrossRef CAS.
- B. Liu, C. Chen, T. Li, J. Crittenden and Y. Chen, J. Membr. Sci., 2013, 445, 66–75 CrossRef CAS.
- M. Padaki, A. M. Isloor, A. F. Ismail and M. S. Abdullah, Desalination, 2012, 295, 35–42 CrossRef CAS.
- V. Vatanpour, S. S. Madaeni, R. Moradian, S. Zinadini and B. Astinchap, J. Membr. Sci., 2011, 375, 284–294 CrossRef CAS.
- S. Zinadini, A. A. Zinatizadeh, M. Rahimi, V. Vatanpour, H. Zangeneh and M. Beygzadeh, Desalination, 2014, 349, 145–154 CrossRef CAS.
- M. S. Jyothi, V. Nayak, M. Padaki, R. Geetha Balakrishna and A. F. Ismail, Desalination, 2014, 354, 189–199 CrossRef CAS.
- K. Y. Wang and T.-S. Chung, J. Membr. Sci., 2006, 281, 307–315 CrossRef CAS.
- S. Habibi, A. Nematollahzadeh and S. A. Mousavi, Chem. Eng. J., 2015, 267, 306–316 CrossRef CAS.
- C.-V. Gherasim and P. Mikulášek, Desalination, 2014, 343, 67–74 CrossRef CAS.
- X. Bernat, A. Pihlajamäki, A. Fortuny, C. Bengoa, F. Stüber, A. Fabregat, M. Nyström and J. Font, J. Membr. Sci., 2009, 334, 129–137 CrossRef CAS.
- R. Kumar, A. M. Isloor, A. F. Ismail and T. Matsuura, J. Membr. Sci., 2013, 440, 140–147 CrossRef CAS.
- R. Kumar, A. M. Isloor, A. F. Ismail, S. A. Rashidc and T. Matsuura, RSC Adv., 2013, 3, 7855–7861 RSC.
- R. S. Hebbar, A. M. Isloor, K. Anandab and A. F. Ismail, J. Mater. Chem. A, 2016, 4, 764–774 CAS.
- L. M. Ortega, R. Lebrun, J.-F. Blais and R. Hausler, Desalination, 2008, 227, 204–216 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra02590k |
| This journal is © The Royal Society of Chemistry 2016 |






